Abstract
The current application of robotic surgery is evolving at a high pace in the current years. The technical advantages enable several abdominal surgical procedures to be performed minimally invasive instead of open surgery. Furthermore, procedures previously performed successfully using standard laparoscopy are now performed with a robotic approach, with conflicting results. The present narrative review reports the current literature on the robotic surgical procedures typically performed in a typical Scandinavian surgical department: colorectal, hernia, hepato-biliary, and esophagogastric surgery.
Introduction
The early robotic surgical systems were developed in collaboration with the American army and NASA with a vision of performing surgery on soldiers or astronauts on the battlefield or in space remotely. However, the actual application of the robotic technology ended up in a very different field—minimally invasive surgery (MIS). In MIS, the robotic technology showed a potential to overcome the intrinsic limitations of conventional laparoscopy (e.g. limited degrees of movement, the fulcrum effect, and tremor from the surgeon). 1
The most widely used robotic system is currently the da Vinci system (Intuitive Surgical, Inc., Sunnyvale, CA, USA), and the term robotic surgery has until now been used synonymously with surgery with the da Vinci system. However, in recent years and the years to follow, other systems are becoming commercially available, which potentially can alter the mechanisms of the market. 2 The overall principle is the same in all the available robotic systems; the robotic platform, is placed next to the patient, and a separate surgeon console is used for manipulating the robotic arms installed with surgical instruments and a camera. The basic principles of MIS are identical between robotic and laparoscopic approaches (including pneumoperitoneum, placements of surgical ports, prolonged instruments, etc.), and the surgical procedures available are the same. However, the most significant difference is the ability to control the articulated tip of the robotic instrument, which increases the degrees of movement (Fig. 1). This is presumed to facilitate difficult surgical tasks (e.g. intracorporeal suturing, precise dissection, or surgery in narrow spaces such as the pelvis or posterior mediastinum). Regardless of these theoretical advantages, it is still controversial if the robotic approach conveys any clinical benefits that can justify the increased economic costs compared to the laparoscopic approach.3,4 Nonetheless, the number of robotic systems is increasing drastically worldwide.
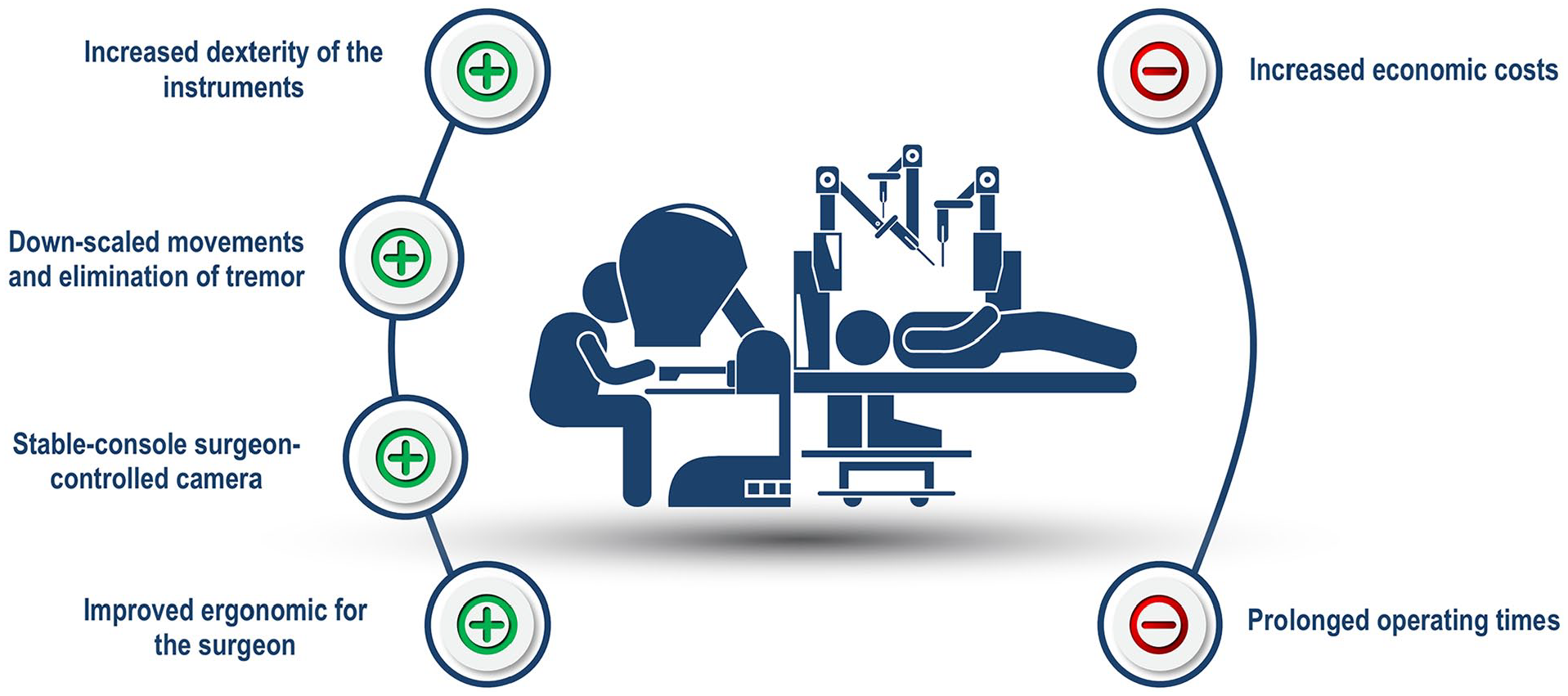
Illustration of the da Vinci system and the possible advantages and disadvantages.
The purpose of this article was to describe the current use and the available high-quality evidence in the subspecialties of abdominal surgery faced in most surgical departments.
Colorectal surgery
High-quality evidence shows that a laparoscopic approach conveys clinical benefits such as faster recovery and lower morbidity while still maintaining similar oncological outcomes, compared to an open approach in colorectal cancer (CRC) surgery.5 –7 Overall, the minimally invasive technique is on the rise worldwide, but the degree of implementation of a minimally invasive approach has probably been susceptible to differences in socioeconomics and preferred clinical practices around the world. In some countries, more than 90% of CRC cases are now performed with a laparoscopic approach and, consequently, a high number of surgeons are expert laparoscopists. 8 In such cases, robotic surgery should be compared to conventional laparoscopy; however, an important notion is that if conventional laparoscopy has not been implemented in a specific center, the technical benefits from the robotic platform may facilitate a transition to a minimally invasive approach from open surgery. The surgical resections of colon and rectal cancer are very distinctive and involve different reasonings and potential benefits for the application of a robotic approach.
In rectal cancer resections, surgery is performed in the narrow surgical field restricted by the pelvis, and the increased maneuverability provided by the robotic instruments and improved vision is hypothesized to facilitate precise oncological dissection and reduce the risks of conversion to open surgery and damaging urogenital nerves compared to conventional laparoscopy. There is an abundance of preliminary studies (retrospective cohort and non-randomized prospective) affirming the theoretical benefits;9 –11 however, there are only a limited number of randomized controlled trials (RCTs), with contradicting results, available on the subject.4,12 –14 One of the trials is the early pragmatic multinational ROLARR trial 4 involving patients (n = 471) undergoing rectal resections operated with either a robotic or a laparoscopic approach. ROLARR found no statistically significant difference in the primary outcome formulated as conversion to open surgery (robotic 8% vs conventional laparoscopic 12%, P = 0.16) or in any of the secondary outcomes regarding surgical specimen quality (e.g. circumferential resection margins (CRMs), postoperative complications, urogenital function, or late functional outcomes).4,15 However, later sensitivity analyses have indicated that there was an ongoing learning curve in the robotic 16 which besides questioning the internal validity of the results, also stresses a significant challenge in surgical research in general. Another key trial on the subject is the most recent multicenter REAL trial from China, 14 involving patients (n = 1171) with middle or low rectal cancer. The primary outcome with long-term results is still unavailable, but in contrast to ROLARR, the short-term results suggest that a robotic approach provides better oncological specimen quality with improved CRM (4% vs 7%, P = 0.02), fewer postoperative complications (16% vs 23%, P = 0.003), and fewer conversions to open surgery (2% vs 4% P = 0.02) compared to conventional laparoscopy. 14 However, a major limitation to these trials is a lack of masking of the treatment allocation, resulting in an inevitable risk of bias.
In colonic cancer resections, the robotic approach is assumed to facilitate a precise oncological dissection (central mesocolic excision (CME) and central vascular ligation) while also enabling the surgeons to perform right colectomy with intracorporeal anastomosis safely, 17 because intracorporeal suturing is essential when performing an intracorporeal anastomosis (Fig. 2). The intestines are not eviscerated during the creation of the anastomosis. Therefore, intracorporeal anastomosis is thought to cause less manipulating and traction of the intestines and mesentery with a resulting reduced surgical stress and improved postoperative recovery compared to the extracorporeal anastomosis. However, the actual benefits of intracorporeal anastomosis are still a matter of debate.18,19 The available high-quality evidence comparing robotic with laparoscopic colectomy is sparse and currently limited to only one small RCT comparing robotic with laparoscopic right colectomy.3,20 Apart from increased economic costs and longer operative times from robotic surgery, the study did not find any differences.
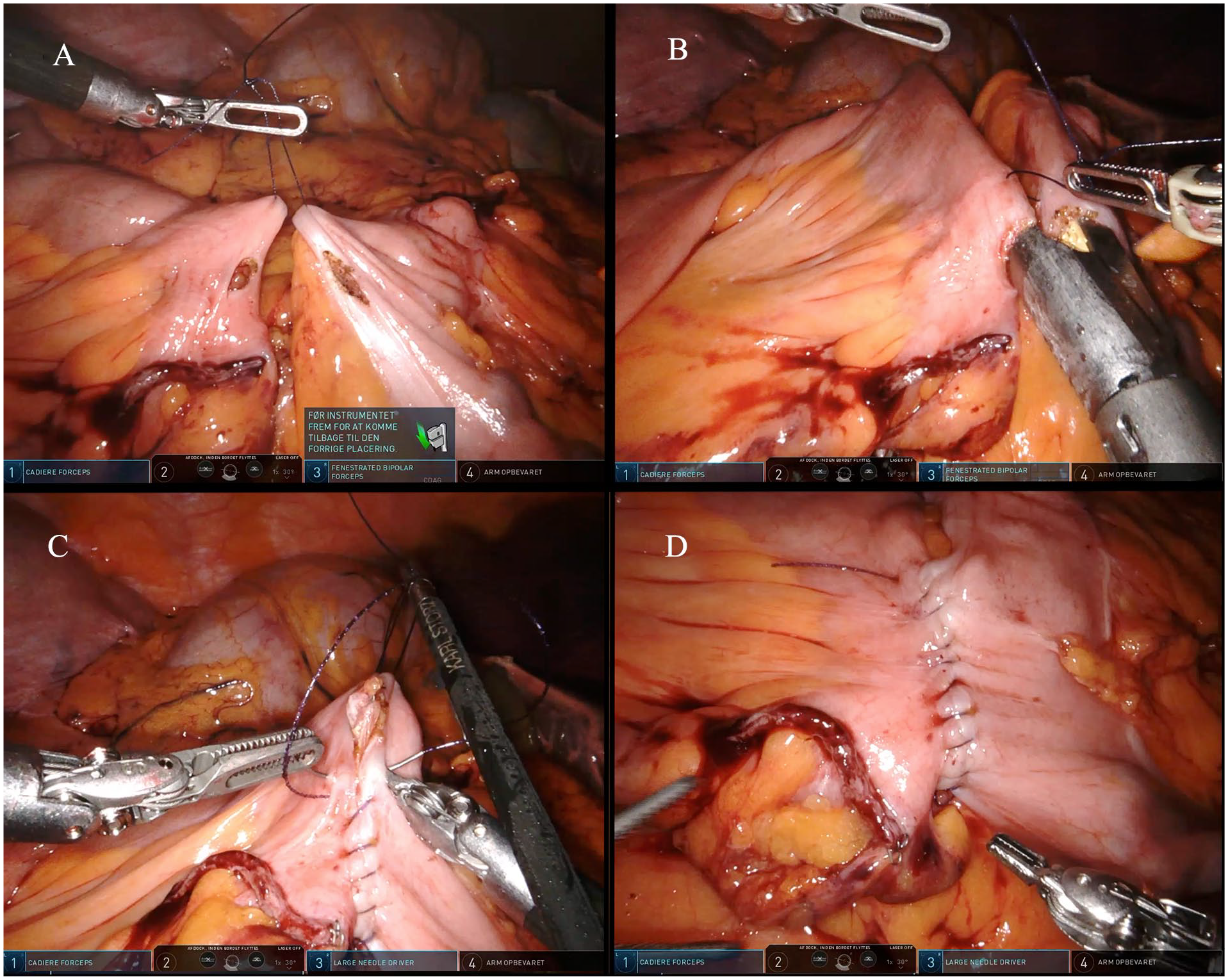
Intracorporeal ileocolic anastomosis. (A) Small intestine and transverse colon enterotomy, (B) mechanical stapler is applied with a jaw in each enterotomy and a common enterotomy is created, (C) intracorporeal suturing, and (D) final anastomosis.
In benign colorectal surgery, the robotic approach is assumed to offer some benefits when performing ventral mesh rectopexy procedures for rectal prolapse compared to conventional laparoscopy because the procedure necessitates intracorporeal suturing. Although there is currently no high-quality evidence available, the robotic and laparoscopic approaches do not seem to offer any considerable differences. 21
Hernia surgery
Robotic abdominal wall surgery is rapidly spreading among centers across Europe. Common for all types of hernia surgery is that the evidence behind the introduction of the robotic procedure is limited. 22 The vast majority of robotic hernia repairs in Scandinavia is inguinal hernia repair, which in many ways is similar to the traditional laparoscopic transabdominal preperitoneal prosthetic repair (TAPP) procedure. In the literature, there is currently only one RCT comparing laparoscopic to robotic inguinal hernia repair. This trial found no difference between the two approaches in postoperative pain or quality of life, whereas robotic inguinal hernia repair had an increased mean duration of surgery (76 vs 41 min), was more expensive, and increased frustrations for the surgeon. 23 Two-year follow-up on this trial further found no differences in regard to long-term outcomes. 24 On the contrary, retrospective cohort studies reported fewer complications after robotic inguinal hernia repair compared to laparoscopic inguinal hernia repair. 22
The major advantage of robot-assisted compared to laparoscopic abdominal wall surgery is anticipated to be in patients undergoing surgery for complex ventral hernias. This is due to the articulation in the robotic instruments, allowing for much easier intracorporeal suturing and dissection in the abdominal wall (Fig. 3). These possibilities allow for advanced reconstructions which were traditionally reserved for open surgery. Two RCTs from North America compared laparoscopic and robotic approach for ventral hernia repair. Both studies found no significant differences in postoperative complication rates or patient-reported quality of life.25,26 A common point of critique for both studies is that they were comparing simple intraperitoneal onlay mesh repair using either a laparoscopic or robot-assisted approach, thus failing to take advantage of the technical possibilities offered by the robot. 27 A recent large register-based study from Denmark found robotic retromuscular hernia repair of small- to medium-sized hernias to be associated with less postoperative length of stay and incidence of readmission compared to traditional laparoscopic intraperitoneal onlay mesh (IPOM) repair. 28 Smaller retrospective cohort studies have shown promising results after the repair of large incisional hernias, where patients undergoing bilateral component separation were discharged after median one compared to 4 days after open repair. 29 In addition, patients undergoing robotic repair reported less postoperative pain and elicited a reduced incidence of postoperative wound complications. The quality of life after robotic versus laparoscopic ventral hernia repair has only been examined in few studies, finding no significant differences between the two modalities. 30
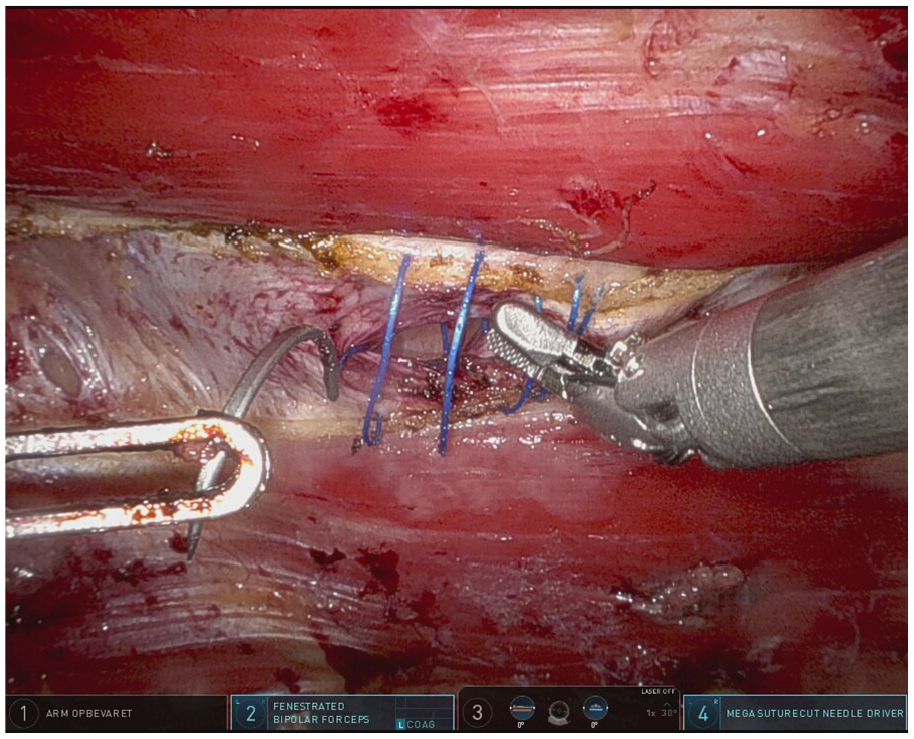
Robotic suturing of the fascial defect in a ventral hernia repair.
In conclusion, it seems that robotic hernia repair holds great potential regarding complex ventral hernias; however, adequately powered and well-designed RCTs are currently lacking.
Hepato-pancreato-biliary surgery
During the last decade, MIS has also been implemented in hepato-pancreato-biliary (HPB) surgery. The technical benefits from the robotic approach offer advantages in HPB surgery because the liver and—especially—the pancreas are fixated organs. With the implementation of the robot, more complex minimally invasive procedures are performed, and the surgical development will continue with the robotic technology and complexity of the instruments.
In general, minimally invasive pancreatic surgery (MIPS) has been shown to reduce morbidity without compromising the oncological results compared to open surgery. 31 However, due to the finding of increased mortality after laparoscopic pancreaticoduodenectomy (Whipple’s procedure) in a single RCT, it is now the recommendation only to perform this highly complex procedure as MIS if it can be done with a robotic approach or in a high-volume center.32,33 The context for this is that the technical advantages from the robotic platform are assumed to make the essential anastomoses (hepato-jejunal and pancreatico-jejunal) less complex compared to conventional laparoscopy and because surgical volume is imperative for the morbidity and mortality following the procedure.
The optimal approach for the surgical management of lesions in the pancreatic body or tail is MIPS because high-quality evidence has shown an improved postoperative recovery compared to open surgery. 34 However, the robotic approach has not shown substantial benefits other than reduced intraoperative blood loss, lower conversion rate, and a reduced risk of additional splenectomy (in benign surgery) compared to conventional laparoscopy. 35
There are multiple studies suggesting that minimally invasive liver surgery (MILS) is the optimal approach, as it has been shown to reduce intraoperative blood loss, reduce morbidity, and fasten postoperative recovery, while still showing similar long-term oncological results compared to open liver surgery. 36 In addition, multiple studies have shown that a robotic approach to MILS results in lower mortality and faster recovery compared to open liver surgery. It is thought that the technical advantages of the robot with increased maneuverability facilitate the more complex liver surgeries such as posterior resections in close relation to the hilus. 37 The current downside of robotic hepatic resection seems to be the economic burden, as the literature on costs shows increased expenses associated with robotic compared to both open and laparoscopic hepatectomy. 38
Esophagus-cardia-ventricle surgery
A minimally invasive resection of the cardia or a gastrectomy with radical lymphadenectomy, in general, combined with new adjuvant treatment, is the preferred treatment for localized esophagus and gastric cancer.39,40
Different surgical modalities have been examined, each with its own specific technical advantages and disadvantages. In controlled studies from high-volume centers, MIS has shown reduced postoperative mortality compared with open surgery, especially by reducing the rate of postoperative pulmonary complications. 41 In reality and everyday practice, MIS is much more complex than open due to technical difficulties, introducing a higher risk of operative reintervention as well as an increased risk of anastomotic leak, which has both been documented in several registered studies.42,43 The current literature has failed to show an improvement in long-term survival after minimally invasive compared to open upper GI surgery.44,45
Throughout the years, the use of the robotic approach in upper GI surgery has increased, and there are multiple ongoing multicenter RCTs comparing a robotic with a conventional laparoscopic approach.46,47 Early results have shown advantages for robotic surgery with reduced duration of surgery as well as an increased lymph node yield. 48
One trend which seems to be receiving an increasing amount of attention is that robotic surgery is better for the surgeon, as opposed to what is better for the patient. However, the available data pointing at a reduced duration of surgery, improved ergonomics for the surgeon as well as a steeper learning curve are hard to argue against. 49 Considering the often very long duration of upper GI resections, these arguments weigh heavily.
In patients undergoing bariatric surgery only one RCT exists, whereas a high number of both unrandomized prospective and retrospective studies have been published. Summarized, it seems that there is a longer duration of surgery using a robotic compared to laparoscopic approach, which has been consistently demonstrated. 50 Similarly, no advantages for a robotic approach in revisional bariatric surgery have been demonstrated, although the literature on this is rather sparse. 51
Discussion
Overall, there is a lack of high-quality evidence on robotic versus conventional laparoscopy in general surgery. Most of the available evidence is retrospective cohort studies or prospective non-randomized and non-blinded trials. 52
The introduction of advanced computer and robotic technology to the operation table provides endless prospects for modern surgery (e.g. augmented reality, three-dimensional (3D) modeling, automatization, and artificial intelligence (AI) with constant assessment of tissue vitality). The use of AI, patient data, and the integration of patient imaging (cross-sectional scans, fluorescence angiographies, or other biometric tests) unlock new possibilities for precision surgery while also allowing for machine learning.
Evidence-based implementation of new treatments is a general problem in surgery where clinical practice often has gone ahead of the evidence. Issues with randomization, blinding, and standardization of the surgical intervention may be some of the problems in the surgical literature.16,53,54 An evidence-based approach with protocolized trials with focus on completion of the learning curves and relevant outcomes during an implementation phase should be pursued. 53 However, surgeons are also faced with the clinical reality, where proficiency in a new surgical technique must be acquired on simple procedures whereas the more complex procedures (with possibly most to gain from the new technique) can be added later. The current lack of convincing long-term upsides to most types of robotic surgery is thus a constant cause of debate.
Common for most robotic approaches to surgical procedures is that it in many ways enables surgeons to perform minimally invasive instead of open surgery. One example of this is pancreaticoduodenectomy which in skilled hands can be performed using traditional laparoscopy; however, in centers with insufficient procedural volume, the learning curve may be too long for a minimally invasive program to be efficient. 55 Similarly, a few reports of laparoscopic bilateral transverse abdominis release for large incisional hernia have been published, but for most surgeons, the advantages from a robotic approach are required to perform this procedure in an acceptable timeframe. 56
In general, the robotic approach seems to yield a clinically important upside in incisional hernia repair, as this approach enables conversion from open to MIS. This is especially important in incisional hernia repair, as the significantly reduced risk of surgical site infections leads to a reduced risk of mesh infection and hernia recurrence. 57 Thus, in repair of large hernia defects, the long-term recurrence rate is expected to be reduced after robotic compared to open approach; however, these data have not surfaced yet. In colorectal surgery, the future upsides of robotic approach may be found in extended rectal resections, by some authors referred to as beyond-TME, for instance, in the form of pelvic sidewall lymph node resection. 58
The status of the use of the robotic approach in general surgery may thus be somewhat compared to the introduction of laparoscopic surgery in the 1980s and 1990s, in which lack of prospective trials did not stop innovative surgeons from developing their techniques with this new approach. 59 Nevertheless, it took until the late 2000s before several RCTs confirmed a reduction in short-term complications as well as the long-term incidence of incisional hernia after laparoscopic compared to open colorectal resection. 60 Considering that many of the described procedures in the current review can already be performed using standard laparoscopy, which features upsides similar to robotic surgery in comparison with open (reduced length of stay, fewer wound complications, and incisional hernias), it may take more than a decade before long-term oncological outcomes from well-designed RCTs can show improved outcomes of robotic compared to laparoscopic surgery. In the meantime, it seems that centers worldwide must rely on internal quality assessment as well as the emerging short-term follow-up studies for an indication of whether the robotic approach is indeed superior to standard laparoscopy for any given procedure. Consequently, there is an urgent need for well-designed multicenter RCTs investigating the role of a robotic approach in various subspecialties.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
