Abstract
Background and Aims:
A positive resection margin is considered to be a factor associated with poor prognosis after pancreatic ductal adenocarcinoma resection. However, analysis of the resection margin is dependent on the pathological slicing technique. The aim of this systematic review and meta-analysis was to study the impact of resection margin on the survival of pancreatic ductal adenocarcinoma patients whose specimens were analyzed using the axial slicing technique.
Material and Methods:
A systematic search in the PubMed, Cochrane, and Embase datasets covering the time period from November 2006 to January 2019 was performed. Only studies with axial slicing technique (Leeds Pathology Protocol or Royal College of Pathology Protocol) were included in the final database. Meta-analysis between the marginal distance and survival was performed with the Inverse Variance Method in RevMan.
Results:
The systematic search resulted in nine studies meeting the inclusion criteria. The median survival for a resection margin 0 mm ranged from 12.3 to 23.4 months, for resection margin <0.5 mm 16 months, for resection margin <1 mm ranged from 11 to 27.5 months, for resection margin <1.5 mm ranged from 16.9 to 21.2 months, and for resection margin >2 mm ranged from 53.9 to 63.1 months. Five studies were eligible for meta-analysis. The pooled multivariable hazard ratio favored resection margin ⩾1 mm (hazard ratio: 1.32 and 95% confidence interval: 1.03–1.68, p = 0.03).
Conclusion:
Resection margins ⩾1 mm seem to lead to better survival in pancreatic ductal adenocarcinoma patients than resection margin <1 mm. However, there is not enough data to evaluate the effect of oncologic therapy or to analyze the impact of other resection margin distances on survival.
Keywords
Introduction
A study of 28 European Union (EU) countries predicts that pancreatic cancer may become the third leading cause of cancer-related death in the EU after lung and colorectal cancers, in contrast to stable or even declining trends for most cancer types (1). At the moment, the best prognosis can be achieved by combining surgery and oncological therapy. Resection margin (RM) status is one of the most important factors influencing survival. Despite this, controversy exists as to what should be considered to be complete clearance of a pancreatic tumor. The International Study Group of Pancreatic Surgery (ISGPS) (2), Royal College of Pathologists (3), and the College of American Pathologists (CAP) (4) suggested an RM of at least 1 mm, while the International Union Against Cancer (UICC) (5) claims that an RM of 0 mm would suffice. In contrast to this, some authors even suggest that extending this cutoff to 1.5 or 2 mm (6–8) could significantly improve survival. Earlier studies (9, 10) reporting the effect of pancreatic cancer tumor-free RM have included a variety of different pathologic techniques. To study the impact of RM on survival among pancreatic ductal adenocarcinoma (PDAC) patients, we conducted a systematic review and a meta-analysis including only studies analyzing pathological samples with axial slicing techniques: Leeds Pathology Protocol (LEEPP) or Royal College of Pathology Protocol (RCP).
Materials and Methods
Data Sources and Search Strategy
A systematic search in MEDLINE, Embase, and the Cochrane library was conducted in collaboration with a medical librarian (search strategy in Supplemental Material 1). An additional search was performed by conducting a review of the references in the articles retrieved. The search included studies published between November 2006 and January 2019. Studies describing RM length and its relation to the survival of patients with pancreatic cancer were included. Both MeSH terms (pancreatic neoplasms, pancreaticoduodenectomy, pancreatectomy, pancreaticojejunostomy) and free text words concerning pancreatic tumors and resections were used as search words. We specifically avoided adding a search term “margin” to harvest all studies in which RM length could have been one of the outcomes. This systematic review followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) (11).
Study Selection
Search results were independently reviewed by two authors (R.A. and B.K.). Any disagreement over eligibility was resolved through discussion. Inclusion criteria were curative surgery for PDAC performed, length of tumor-free margin reported, patients older than 18 years, follow-up of at least 1 year available, survival data, and axial slicing technique used for pathological report (LEEPP or RCP) included. To minimize heterogeneity between the studies and to ensure more clinically significant results, we also used the following exclusion criteria: studies with combined results for other periampullary cancers (ampullary, distal bile duct, and duodenal), unclear pathological assessment of margins, reviews or meta-analyses, small case series (⩽6 patients), published as abstracts only, and non-English-language studies. The corresponding authors of the studies included were sent an email to receive additional results or data for this meta-analysis.
Risk for Bias and Publication Bias Evaluation
The Newcastle-Ottawa Scale (NOS, Supplemental Material 2) was used to assess the quality of the studies included. The evaluation was performed by three authors (R.A., B.K., E.Z.).
Statistical Analysis
Multivariate hazard ratio (HR) together with its 95% confidence interval (CI) was directly extracted from the text. HR pooled together according to different width of tumor-free margin was calculated using the Inverse Variance Random Effects Model and presented with Forest plots. A p-value less than 0.05 was considered statistically significant. A chi-square statistic was used to test the statistical heterogeneity. Heterogeneity between trials was evaluated with I2. All statistical analyses were performed in Review Manager 5.3 program (Cochrane Collaboration, Copenhagen, Denmark).
Results
Study Selection
The search strategy yielded 1629 studies for further analysis. The studies represented a wide spectrum of published data on pancreatic resections of which most of did not focus on the issues analyzed in this study. Full texts of 166 studies were assessed, and 9 studies were included in the qualitative analysis. The most common exclusion criterion was unclear pathological assessment protocol (Fig. 1).
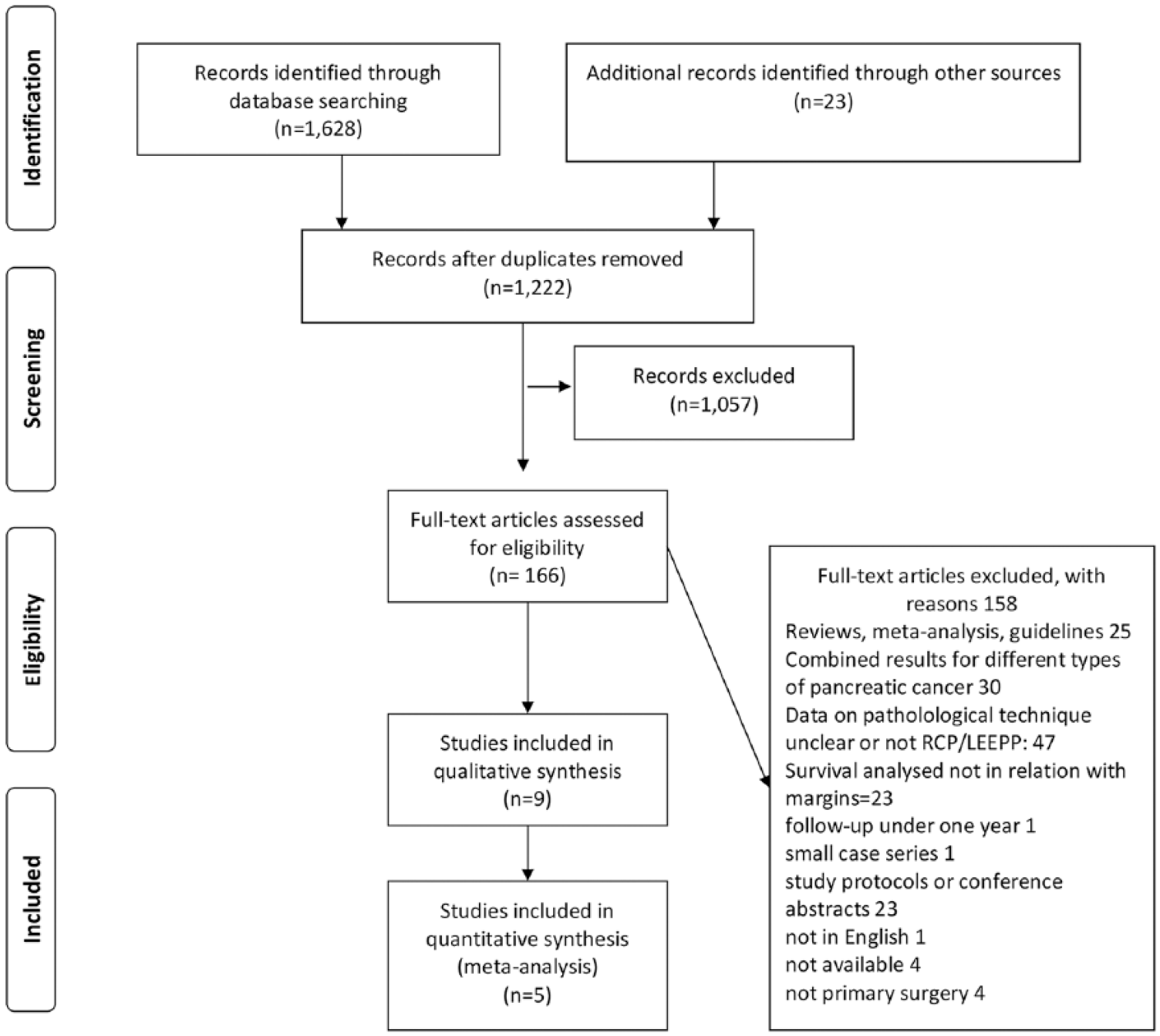
The PRISMA flow chart of the data search.
Study Characteristics
Six of the studies included had used LEEPP for the evaluation of surgical specimens and three studies reported using RCP. All studies originated in Europe. Two studies (Delpero et al. (12) and Ghaneh et al. (6)) were based on a multicentre database. Most of the patients had T3 tumor and 18%–37% of the tumors were poorly differentiated but information of those were also lacking in some of the included articles. The proportion of adjuvant therapy varied between the studies from 27% to 96%. The use of neoadjuvant therapy (NAT) varied from 0% to 21%. Data on adjuvant therapy or NAT were not reported in all studies. All studies reported univariate analyses for survival, and one study (Ghaneh et al. (6)) also reported disease-free survival in relation to margin lengths. In addition, Delpero et al. (12) reported disease-free survival of the whole cohort. Five studies reported multivariable HRs for survival in relation with at least one margin width and were included in the quantitative meta-analysis (Fig. 2).
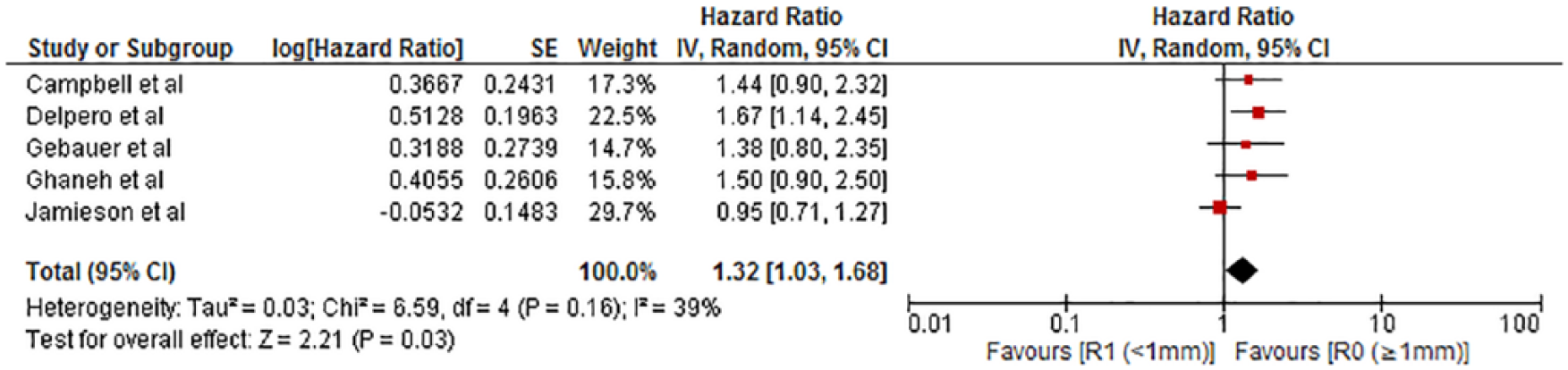
Meta-analysis of the eligible hazard ratios on 1-mm resection margin (RM).
Survival
Median survival varied 12.3–23.4 months for patients having a microscopic tumor at the margin (0-mm margin). One study reported a median survival of 16 months for patients with a margin under 0.5 mm. Eight studies demonstrated a median survival of 11–27.5 months for patients having a tumor-free margin under 1 mm. Nine studies reported median survivals of 17.3–53.9 months for over 1-mm margin (Table 1). Two studies reported median survivals of 16.9–21.2 months for a margin under 1.5 mm, and the same studies reported median survival of 53.9 and 63.1 months for patients having a RM over 1.5 mm. Two studies reported a median survival 15.1–63.6 months for a RM over 2 mm. An overview of the effect of margin length is shown in Fig. 3. Reported multivariate HRs are shown in Table 2. One study (18) reported multivariate HRs for RM ⩾1 mm versus RM 0 mm (HR = 0.73, CI: 0.54–098, p = 0.036) and RM ⩽1 versus 0 mm (HR: 0.71, CI: 0.54–0.95, p = 0.019). The most of the HRs (five studies) were reported for the RM ⩾1 mm. The pooled multivariable HR favored RM ⩾1 mm (HR: 1.32 and 95% CI: 1.03–1.68, p = 0.03).
List of included studies and study characteristics.
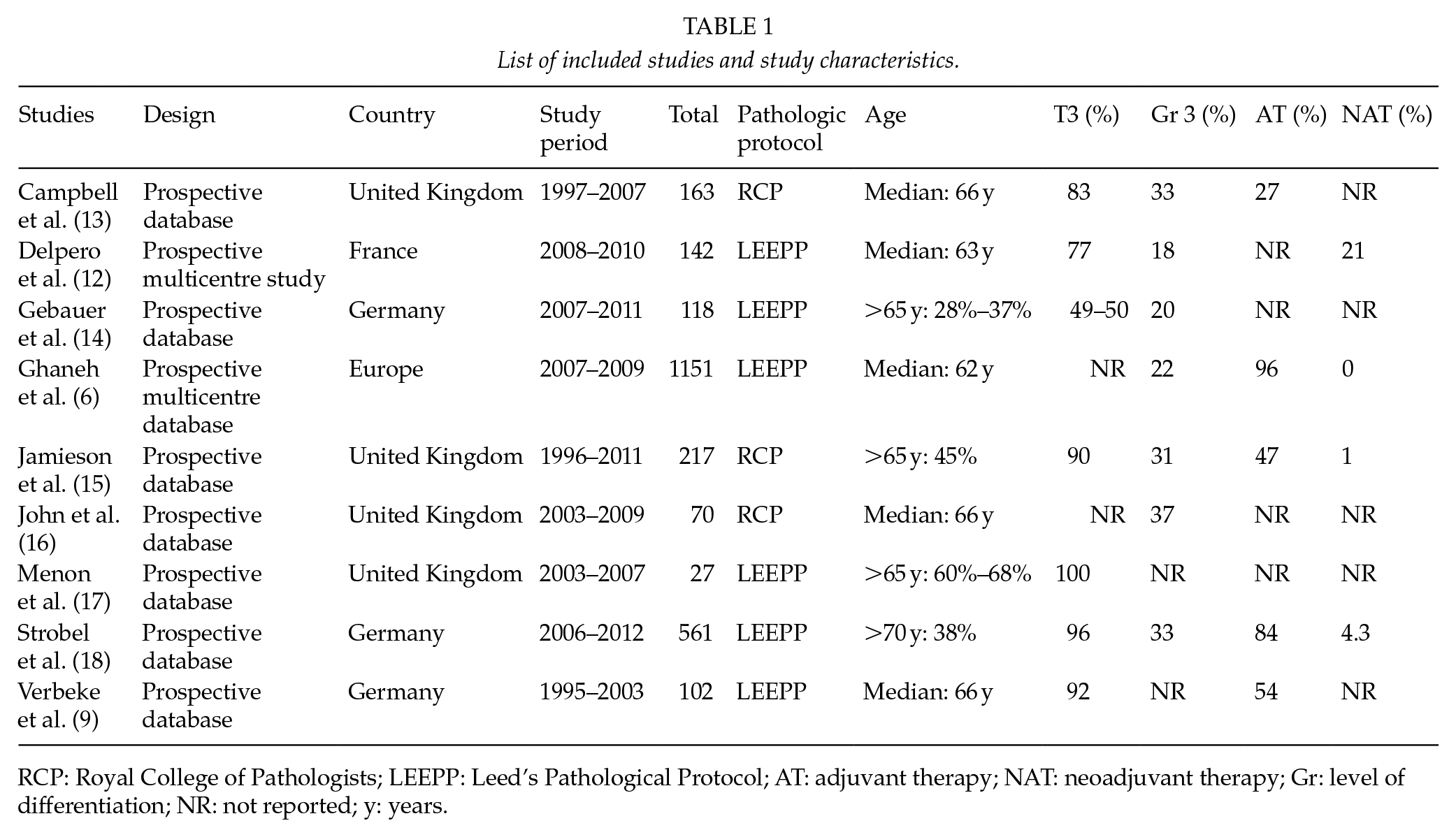
RCP: Royal College of Pathologists; LEEPP: Leed’s Pathological Protocol; AT: adjuvant therapy; NAT: neoadjuvant therapy; Gr: level of differentiation; NR: not reported; y: years.
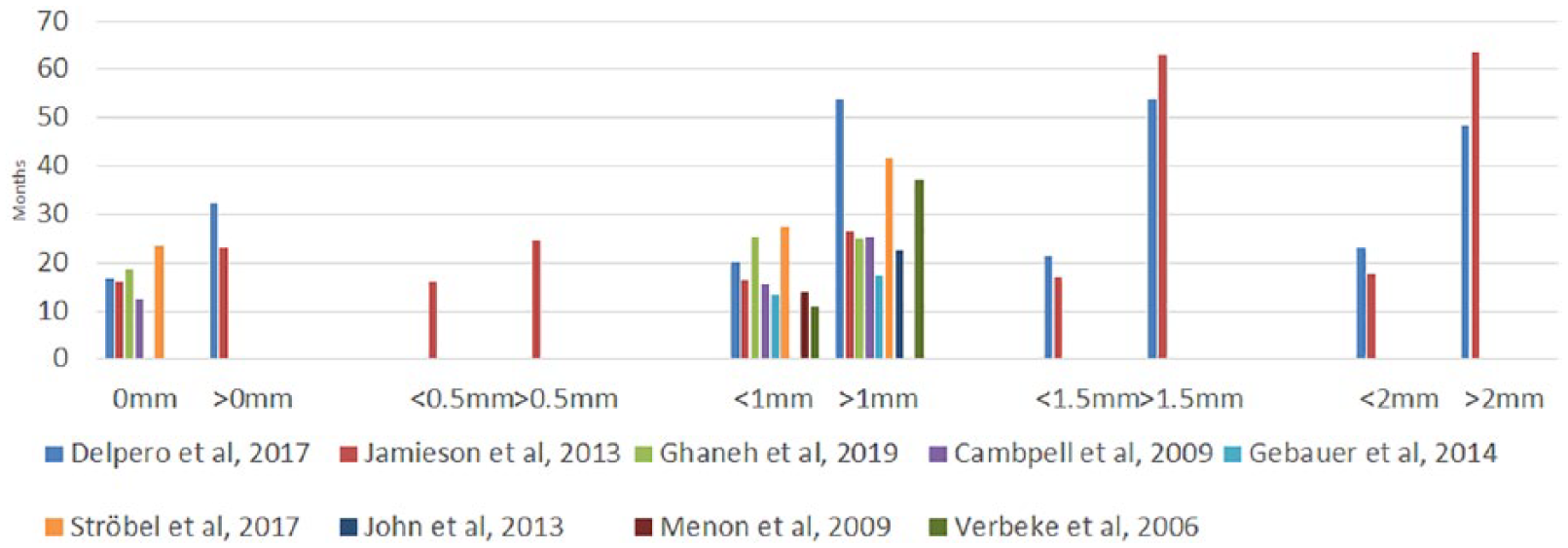
The reported median survivals in relation to different resection margin (RM) cutoffs.
Reported multivariate hazard ratios and p-values.
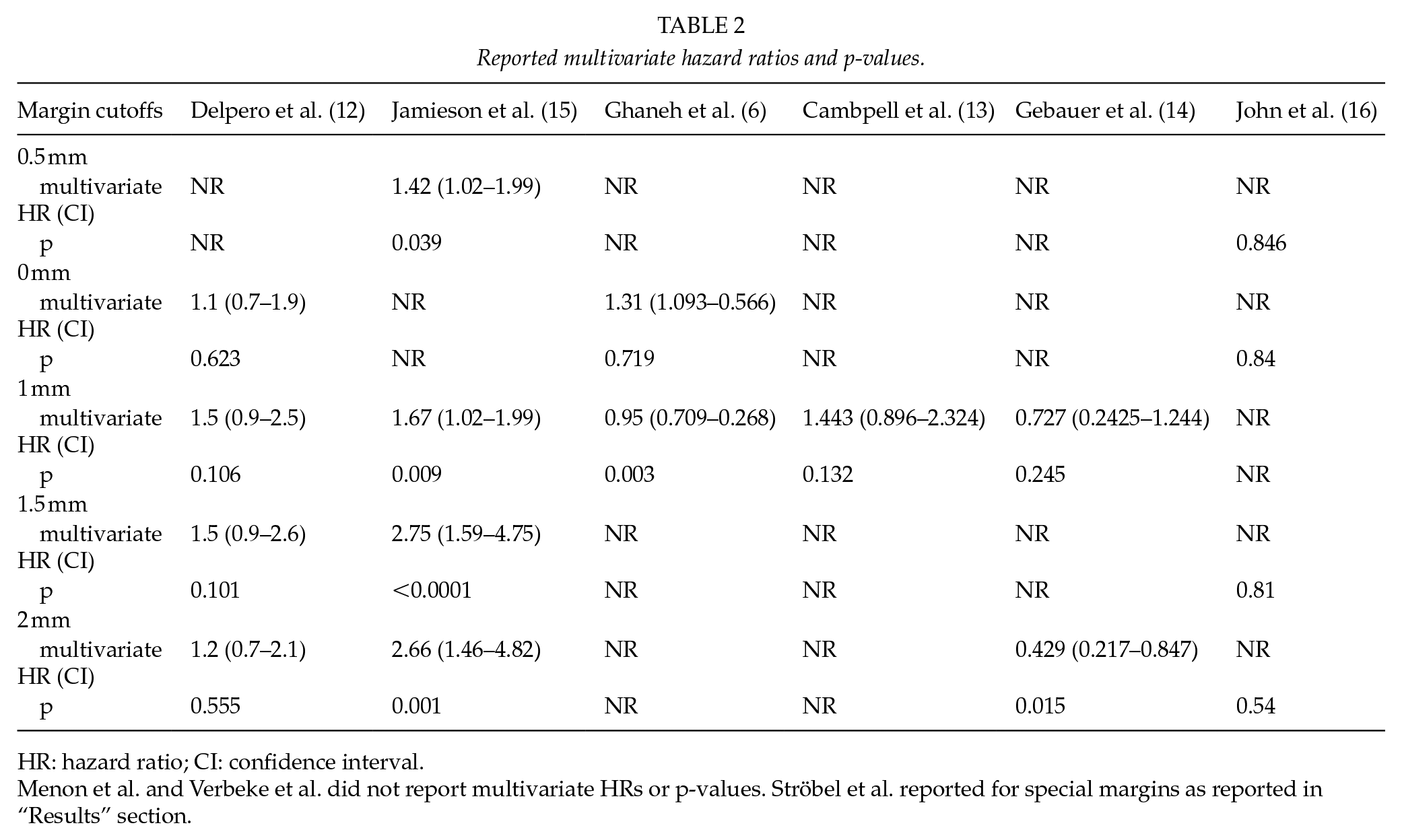
HR: hazard ratio; CI: confidence interval.
Menon et al. and Verbeke et al. did not report multivariate HRs or p-values. Ströbel et al. reported for special margins as reported in “Results” section.
Disease-Free Survival
Ghaneh et al. (6) reported recurrence-free survival in relation to margin widths. They reported a median recurrence-free survival of 15.7 (14.3–17) months for tumor clearance over 1 mm, 14.5 (12.8–17.1) months for margin clearance under 1 mm, and 11.9 (10.9–13.3) months for 0-mm margin clearance.
Discussion
RM status after curative surgery for PDAC is considered as one of the most crucial factors, influencing survival and in some centers serves as a selection criterion for different treatment modalities. In clinical practice, a tumor-free RM ⩾1 mm is considered to be an R0 resection. Yet, controversy exists what would be the ideal margin clearance. The ratio of R0 and R1 resections in studies differs significantly from 20% to 85% R1 resections (7, 8, 15, 16, 19, 20), due to the different pathology protocols used for RM evaluation. So far, none of the published meta-analyses have considered this important aspect (21, 22). Therefore, we decided to include only studies evaluating pathological samples with the axial slicing technique (LEEPP or RCP) in our meta-analysis (9). Here, we could demonstrate that a RM >1 mm leads to a better prognosis compared to <1 mm, but the barely significant CI and the small amount of evidence on other margins hamper a comprehensive analysis on the role of the best RM.
Earlier studies demonstrated a wide range of R1 resection rates after PDAC surgery (7, 8, 19). More interestingly, these studies reported that their patients had R1 resection rates of less than 20%, but demonstrated local recurrence rates from 60% to 80%. These observations reveal that the true R1 proportion seems to be underestimated. Keeping this in mind, Verbeke et al. published in 2006 (9) an article describing the development of a new pathology protocol for the examination of pancreatoduodenectomy specimens (9). This involved multicolour margin staining, axial slicing, and extensive tissue sampling, which later came to be known as the LEEPP. This led to a higher overall R1 rate (85%) and a significant correlation of RM status with survival. At about the same time, the Royal College of Pathologists also published their protocol (3). These protocols are to the best of our knowledge essentially identical. Later, studies using these protocols all concluded higher rates of R1, ranging from 76% to 81% (13, 17, 20). Considering the diffuse growth pattern of PDAC and the high rates of local recurrence, these findings seem to be more accurate and reflect the aggressive biological behavior of PDAC.
A meta-analysis by Butturini et al. (23) did not identify RM status as a significant factor for survival and doubted the 1-mm cutoff value. Further trials showed no major differences in outcomes between resection with involved margin and resections in which the tumor was found within 1 mm of the RM (10, 13). This led to the “1 mm principle” for radical PDAC surgery, which was originally adopted from rectal cancer surgery, where the distance of tumor-free circumferential margin was considered to have a significant association with reduced local recurrence rates (24, 25).
Verbeke et al. (26) compared the growth patterns of rectal and pancreatic tumors. They concluded that in PDAC the tumor is more dispersed, particularly on the periphery, and doubted that the 1-mm cutoff value would suffice. This was corroborated by other studies (14, 15, 27), which have shown that increasing RM could significantly improve survival. Chang et al. (27) contended with data from 1990 to 2007 that optimal long-term survival can only be achieved when the RM is more than 1.5 mm. Unfortunately, they did not define the pathological technique they used in the analysis which challenges the interpretation of the results. Considering these findings, it seems that dispersed cancer cells are very likely to remain after resection even though no tumor is present within 1 mm of the RM. This suggests that the R status might be a surrogate marker for PDAC aggressiveness, instead of indicating success of surgery. In addition, some of patients present metastasis early after pancreatic resection. This indicates that 1-mm tumor clearance might not be essential for PDAC.
In addition to RM and resection success, other factors are also associated with the prognosis. These factors include those detectable in pathological analysis (nodal status, tumor differentiation, size, vascular and perineural invasion), factors associated with the treatment process (operation volume, amount of blood lost, peri- and post-operative complications), oncological treatment given, and factors associated with the tumor biology of each individual patient (18, 20, 28–41). The studies included in our analysis concentrated on reporting histopathological and demographic features, and no data on patient’s individual tumor biology or treatment processes were available. The ESPAC trials 1, 3, and 4 (39–41) have proven the benefits of adjuvant therapy for PDAC patients. However, the proportion adjuvant therapy or NAT administered varied in the studies included or was not reported. This as well as the fact that also patients after NAT were included in some of the studies may bias the results.
As pointed out, a uniform slicing technique of a pancreatic specimen is crucial for a standardized comparison of the surgical RMs between different studies. The time span of the data search in our study included all the years after the LEEPPs technique (after 2006), and searches were conducted in three different databases. Only studies which reported using a standardized axial slicing technique for pathological analysis (LEEPP or RCP) were included which is a strength in our study. However, only nine studies were eligible for the systematic review and five studies for the meta-analysis. Other studies did not use standardized axial slicing technique or did not report the method used for pathological analysis. Furthermore, we could not find studies evaluating patients’ pancreatic RM after 2012 which would use one of these pathological methods. Imaging capabilities, surgical techniques, and oncological treatment have dramatically evolved since. All of this challenges the evaluation of the effect of margins on survival. Nevertheless, we consider a uniform slicing technique to be the most important factor, if the effect of RM on survival is studied. Moreover, the studies reported a variety of background data, and especially the role of oncologic therapy stays unresolved in this analysis. Despite the careful inclusion of the studies, part of the data included patients also from the era before 2006. However, it was possible to pool HR for the most commonly used RM of 1 mm, but in total the evidence for the role of margins stays weak.
In conclusion, over 1-mm RM results in better survival than <1-mm margin. However, the impact of the RM, and moreover, that of other margin distances remain uncertain because of the variability of the patients characteristics in the studies. Especially, the role of adjuvant therapy stays unclear. Further studies are needed to evaluate the true effect of the margin clearance.
Supplemental Material
Cochrane_Library – Supplemental material for In the Era of the Leeds Protocol: A Systematic Review and A Meta-Analysis on the Effect of Resection Margins on Survival Among Pancreatic Ductal Adenocarcinoma Patients
Supplemental material, Cochrane_Library for In the Era of the Leeds Protocol: A Systematic Review and A Meta-Analysis on the Effect of Resection Margins on Survival Among Pancreatic Ductal Adenocarcinoma Patients by B. Kurlinkus, R. Ahola, E. Zwart, A. Halimi, B. S. Yilmaz, G. O. Ceyhan and J. Laukkarinen in Scandinavian Journal of Surgery
Supplemental Material
Newcastle – Supplemental material for In the Era of the Leeds Protocol: A Systematic Review and A Meta-Analysis on the Effect of Resection Margins on Survival Among Pancreatic Ductal Adenocarcinoma Patients
Supplemental material, Newcastle for In the Era of the Leeds Protocol: A Systematic Review and A Meta-Analysis on the Effect of Resection Margins on Survival Among Pancreatic Ductal Adenocarcinoma Patients by B. Kurlinkus, R. Ahola, E. Zwart, A. Halimi, B. S. Yilmaz, G. O. Ceyhan and J. Laukkarinen in Scandinavian Journal of Surgery
Footnotes
Acknowledgements
This study was conducted as a project of the 9th Pancreas 2000 education and research program funded by the Acibadem Mehmet Ali Aydinlar University, the Karolinska Institute, and the European Pancreatic Club/EPC. The authors thank Patrick Masonneuve for his professional guidance in statistics and Virginia Mattila for proofreading the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors appreciate the support and funding from Cancer Center Amsterdam Foundation, The Netherlands (CCA); the Lithuanian Gastroenterology Society, Lithuania; Mylan, The Netherlands; State Research Funding (VTR), Finland; and the Sigrid Jusélius Foundation, Finland. Funding had no involvement in the study design, data collection, data analysis, manuscript preparation, or publication decisions.
Supplemental Material
Supplemental material for this article is available online.
