Abstract
Background
Individuals with a very high lifetime risk of developing pancreatic ductal adenocarcinoma; for example, hereditary pancreatitis and main-duct or mixed-type intraductal papillary mucinous neoplasm, may wish to discuss prophylactic total pancreatectomy but strategies to do so are lacking.
Objective
To develop a shared decision-making programme for prophylactic total pancreatectomy using decision tables.
Methods
Focus group meetings with patients were used to identify relevant questions. Systematic reviews were performed to answer these questions.
Results
The first tables included hereditary pancreatitis and main-duct or mixed-type intraductal papillary mucinous neoplasm. No studies focused on prophylactic total pancreatectomy in these groups. In 52 studies (3570 patients), major morbidity after total pancreatectomy was 25% and 30-day mortality was 6%. After minimally invasive total pancreatectomy (seven studies, 35 patients) this was, respectively, 13% and 0%. Exocrine insufficiency-related symptoms occurred in 33%. Quality of life after total pancreatectomy was slightly lower compared with the general population.
Conclusion
The decision tables can be helpful for discussing prophylactic total pancreatectomy with individuals at high risk of pancreatic ductal adenocarcinoma.
Keywords
Introduction
Pancreatic ductal adenocarcinoma (PDAC) is a major cause of cancer-related deaths worldwide. Globally, PDAC was responsible for 432,242 new deaths in 2018. 1 Overall survival is poor, even in the 17% of patients who can undergo resection 5-year survival is only 15–20%. 2 Therefore, prevention of PDAC may be preferred over early detection and treatment.
There are some individuals with a high (15–62%) lifetime risk of developing PDAC.3–5 This high-risk population can be divided into three main groups: known hereditary conditions (e.g. hereditary pancreatitis), familial clustering and patients with specific clinical predisposition factors (e.g. main-duct or mixed-type intraductal papillary mucinous neoplasm (IPMN), involving the entire pancreatic duct).6–8 Once diagnosed, these patients have to live with the knowledge that PDAC may develop. This knowledge is a considerable burden for most, especially when considering the very poor survival of PDAC. 2
In some countries, individuals with a high risk of developing PDAC may enter a dedicated screening programme in a research setting. 9 , 10 Such a programme has also been introduced in The Netherlands. The goal of surveillance is to detect asymptomatic high-risk lesions such as high-grade dysplastic IPMN or early cancer in order to improve survival. The 3-year psychological burden of such an approach appears acceptable. 11 International guidelines currently do not consider prophylactic total pancreatectomy as a treatment option in these patients, due to the relatively high mortality and morbidity associated with this procedure and reduced quality of life due to ‘brittle’ diabetes. 6 ,12–14 However, surgical outcomes and diabetes treatment have improved in recent years. 15 , 16
For such a preference-sensitive treatment dilemma as total pancreatectomy both the individual and the physician need to be well informed. A shared decision-making approach with decision tables may facilitate this process as it provides a structured and easily accessible evidence-based data overview. 17 Decision tables, also called ‘option grids’, have been developed for multiple conditions. Currently more than 40 are available on the option grid website (https://health.ebsco.com/products/option-grid). A decision table consists of a one-page matrix of evidence-based answers to the most frequently asked questions by individuals who are facing the choice between multiple treatment options.
Based on a request by the Dutch patient organisation, we developed a shared decision-making programme for prophylactic total pancreatectomy (PROPAN) using decision tables for high-risk individuals with either hereditary pancreatitis or main-duct or mixed-type IPMN (based on the European evidence-based guidelines) who wish to discuss prophylactic total pancreatectomy. 6 This study describes the development, including four systematic reviews and the design of this programme.
Methods
The PROPAN shared decision-making programme for individuals who wish to discuss the option of prophylactic total pancreatectomy in the case of a very high risk of developing PDAC was developed by the Dutch Pancreatic Cancer Group in collaboration with the pancreatic cancer patient organisation, Living with Hope (www.livingwithhope.nl).
Developing the PROPAN programme
A meeting was organised involving relevant stakeholders, specifically the patient organisation, a shared decision-making expert, clinical geneticist, gastroenterologists, surgeons and a diabetologist. During the initial meeting the possibility of shared decision-making was discussed regarding two possible (treatment) options for high-risk individuals (i.e. observation or prophylactic surgery). It was decided to take further steps to facilitate shared decision-making within specific groups (starting with main-duct or mixed-type IPNM or hereditary pancreatitis). Several steps were identified to develop the programme:
Step 1
Round table focus group meeting, composed of patients after total pancreatectomy and individuals at high risk of developing PDAC to ask for their input and opinions regarding which information they would seek in the decision-making process (e.g. the frequently asked questions).
Step 2
Identify other stakeholders, such as the familial pancreatic cancer risk screening programme (chair: Professor Marco Bruno, ErasmusMC Rotterdam), and professional organisations such as the Dutch Pancreatic Cancer Group.
Step 3
Start a nationwide, retrospective study to collect quality of life and clinical outcome data on patients after a total pancreatectomy.
Step 4
Perform a systematic literature review about the outcomes after total pancreatectomy to acquire the most up-to-date information.
Step 5
Create a concept ‘decision table’ for prophylactic total pancreatectomy, following the rules of the option grid collaborative association, based on steps 1 to 4.
Step 6
Organise a second, larger multidisciplinary meeting with all stakeholders to discuss the content of the PROPAN programme, (e.g. the points that need to be discussed when an individual with a high risk of developing PDAC comes to an outpatient department).
Step 7
Finalise and launch the PROPAN programme.
Systematic reviews
Four systematic reviews consistent with the preferred reporting items for systematic reviews and meta-analyses (PRISMA) guidelines were performed. 18 We searched PubMed, Embase and the Cochrane Library for studies published in the past 25 years (1992–2017), restricted to the English language. The following exclusion criteria were applied: paediatric patients, familial pancreatic cancer, total pancreatectomy with islet auto transplantation, no total pancreatectomy-specific outcomes, case reports (only in the minimally invasive review), conference abstracts, or reviews. The complete search strategies are presented in Supplementary Appendices 1–4. The following four systematic reviews were performed:
Systematic review 1
Studies on prophylactic total pancreatectomy programmes: all clinical and comparative studies reporting on or mentioning prophylactic total pancreatectomy were included. 5 , 19
Systematic review 2
Studies on patient groups with a greater than 10% lifetime risk of PDAC. All studies reporting on high-risk populations for PDAC were included.3–5,19–29
Systematic review 3
Studies addressing the outcomes of total pancreatectomy. All studies reporting on total pancreatectomy outcome were included. 16 ,30–80
Systematic review 4
Studies on minimally invasive total pancreatectomy. 34 ,81–86
Results
Decision tables
After two meetings with all stakeholders, it was decided to start by developing decision tables for high-risk patients in whom the pancreas is already affected by disease. This included individuals with main-duct or mixed-type IPMN and hereditary pancreatitis. The European evidence-based guideline on pancreatic cystic neoplasms was used for the definition of main-duct and mixed-type IPMN and the indication for total pancreatectomy in these patients. 6 For hereditary pancreatitis a PRSS-1 mutation was considered mandatory. 7 For high-risk individuals without pancreatic abnormalities on imaging (e.g. Peutz–Jeghers and p16-Leiden), it was decided to include them in a second round of the PROPAN programme. Individuals with familial clustering were not included in the PROPAN programme because of the unclear risk of PDAC (wide confidence intervals). 87 , 88
After receiving and implementing input from both stakeholders and individuals in the focus group, two decision tables were created (Table 1 and Table 2). The decision tables can be used when an individual visits the outpatient clinic rather than making the decision table available online for all individuals. This creates the opportunity to discuss whether a prophylactic total pancreatectomy could be an option for that patient’s individual circumstances and preferences.
Decision table for patients to discuss prophylactic total pancreatectomy in main-duct/mixed-type IPMN.
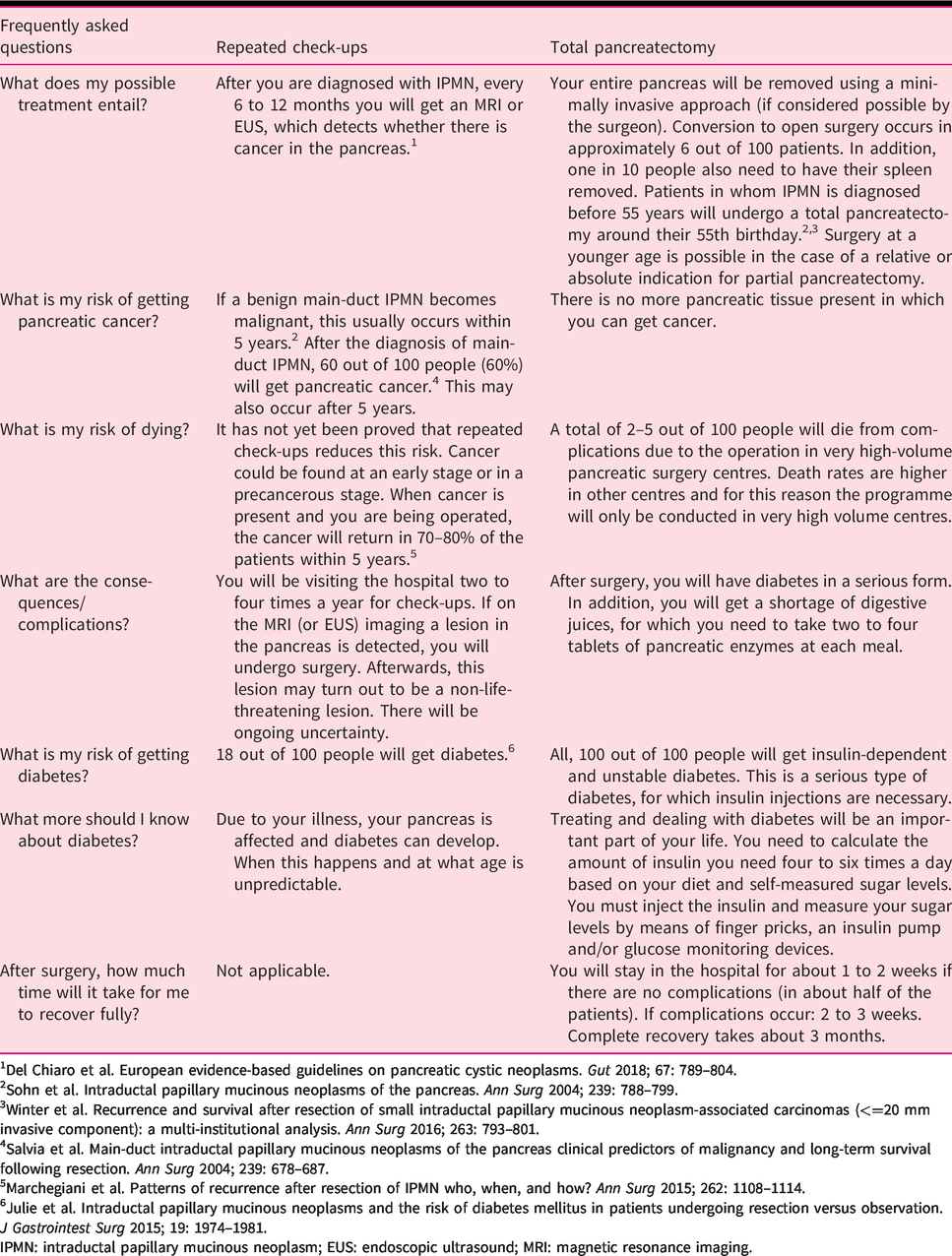
1Del Chiaro et al. European evidence-based guidelines on pancreatic cystic neoplasms. Gut 2018; 67: 789–804.
2Sohn et al. Intraductal papillary mucinous neoplasms of the pancreas. Ann Surg 2004; 239: 788–799.
3Winter et al. Recurrence and survival after resection of small intraductal papillary mucinous neoplasm-associated carcinomas (<=20 mm invasive component): a multi-institutional analysis. Ann Surg 2016; 263: 793–801.
4Salvia et al. Main-duct intraductal papillary mucinous neoplasms of the pancreas clinical predictors of malignancy and long-term survival following resection. Ann Surg 2004; 239: 678–687.
5Marchegiani et al. Patterns of recurrence after resection of IPMN who, when, and how? Ann Surg 2015; 262: 1108–1114.
6Julie et al. Intraductal papillary mucinous neoplasms and the risk of diabetes mellitus in patients undergoing resection versus observation. J Gastrointest Surg 2015; 19: 1974–1981.
IPMN: intraductal papillary mucinous neoplasm; EUS: endoscopic ultrasound; MRI: magnetic resonance imaging.
Decision table for patients to discuss prophylactic total pancreatectomy in hereditary pancreatitis.

1Rebours et al. The natural history of hereditary pancreatitis: a national series. Gut 2009; 58: 97.
2Lowenfels et al. Hereditary pancreatitis and the risk of pancreatic cancer. J Natl Cancer Inst 1997; 89: 442–446.
3Rebours et al. Risk of pancreatic adenocarcinoma in patients with hereditary pancreatitis: a national exhaustive series. Am J Gastroenterol 2008; 103: 111–119.
4Lowenfels et al. Cigarette smoking as a risk factor for pancreatic cancer in patients with hereditary pancreatitis. JAMA 2001; 286: 169–170.
5Neoptolemos et al. Adjuvant therapy in pancreatic cancer: historical and current perspectives. Ann Oncol 2003; 14: 675–692.
6Howes et al. Clinical and genetic characteristics of hereditary pancreatitis in Europe. Clin Gastroenterol Hepatol 2004; 2: 252–261.
CT: computed tomography; MRI: magnetic resonance imaging.
Systematic reviews
To answer the identified questions in the decision tables, four systematic reviews were performed. The first systematic review revealed two studies on prophylactic total pancreatectomy, but none on existing formal programmes for prophylactic total pancreatectomy. 5 , 19 The second systematic review identified four patient groups with a greater than 10% lifetime risk of PDAC: hereditary pancreatitis, main-duct or mixed-type IPMN, Peutz–Jeghers syndrome and p16-Leiden (CDKN2A mutation), see Table 3.3–5,19–29 The peak age of onset of cancer is 40 years in Peutz–Jeghers disease and 60 years in the other groups. 3 , 4 , 20 , 23 , 26 , 28 , 29 , 89 , 90 Although it is strongly recommended to perform surgery for main-duct or mixed-type IPMN, this mostly involves partial pancreatectomy with life-long follow-up and not total pancreatectomy. 6 In main-duct IPMN, a main pancreatic duct diameter of more than 10 mm is considered an absolute indication for surgery, whereas in a main pancreatic duct of 5–9 mm surgery is considered a relative indication for surgery. It is recommended to perform standardised surveillance. 6 , 91 The third systematic review identified 52 studies with 3570 patients who underwent total pancreatectomy with a pooled 30-day mortality of 6% (95% confidence interval (CI) 4–8%). 16 ,30–80 Of the four studies reporting on quality of life the mean global health status was 75% (on a scale of 0–100%) based on the EORTC QLQ-C30 questionnaire (102 patients) and moderately lower compared with the general population. 32 , 35 , 48 , 61 During a median follow-up of 38 months, 49/273 patients (18%) were re-admitted for endocrine-related morbidity, with hypoglycemia-related mortality in 10/568 patients (1.8%). Exocrine insufficiency-related symptoms were reported by 178/547 patients (33%) during a median follow-up of 33 months.
Estimated life-time risk of PDAC in high-risk patients and individuals.
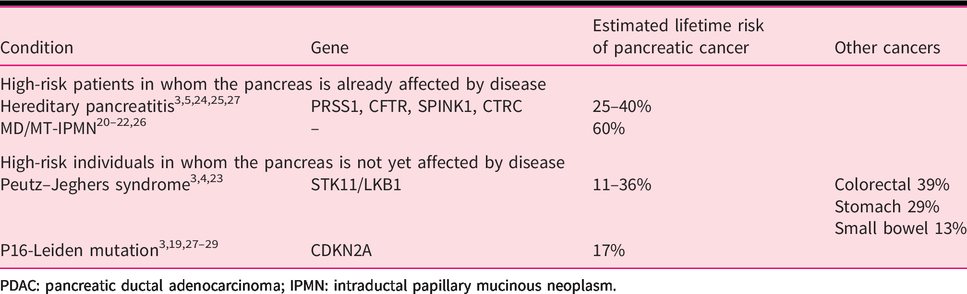
PDAC: pancreatic ductal adenocarcinoma; IPMN: intraductal papillary mucinous neoplasm.
The fourth systematic review showed that minimally invasive total pancreatectomy is poorly described in the literature, with seven studies, including a total of 35 patients. 34 ,81–86 Morbidity and mortality were, respectively, 13% and 0% after minimally invasive total pancreatectomy (Table 4). One report compared minimally invasive total pancreatectomy to open total pancreatectomy and found similar complication rates, although the mean operation time was longer in the robot-assisted group than in the open surgery group: mean 600 minutes (range 400–800) versus 469 minutes (range 300–660), P = 0.014. 34
Minimally invasive total pancreatectomy.
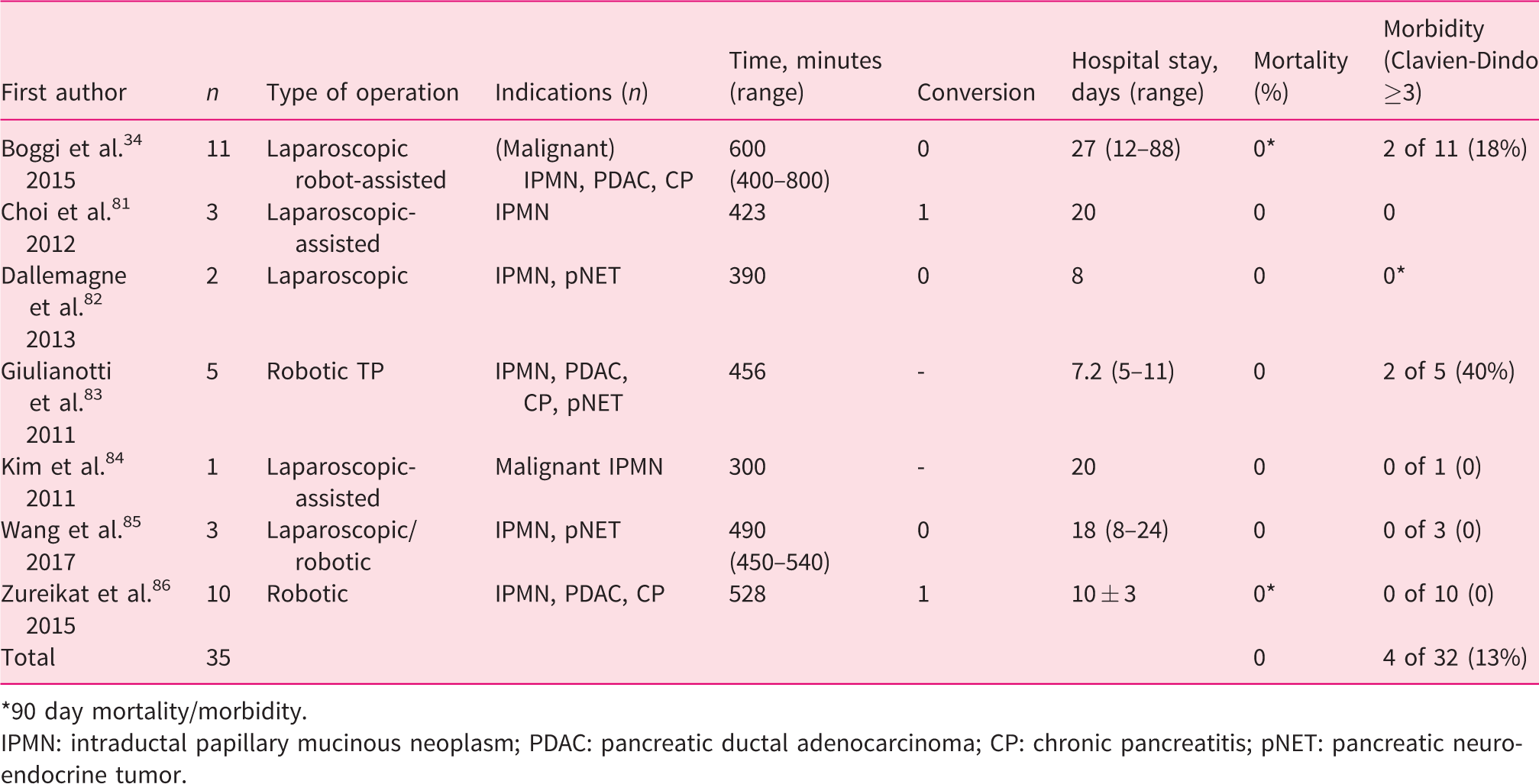
*90 day mortality/morbidity.
IPMN: intraductal papillary mucinous neoplasm; PDAC: pancreatic ductal adenocarcinoma; CP: chronic pancreatitis; pNET: pancreatic neuroendocrine tumor.
PROPAN programme
The design of the PROPAN programme is shown in Figure 1. Participants have several consultations during the programme. First, a combined consult with a gastroenterologist and pancreatic surgeon will take place in order to answer questions and fully inform the participant and relatives. Subsequently, a consultation with a diabetes nurse and a patient after total pancreatectomy will be organised. The surgeon will inform the participant about the procedure, expected postoperative course and the risk of complications. If the patient still wants to proceed, the specific case will be discussed in the PROPAN expert panel, consisting of a clinical geneticist, surgeon, gastroenterologist, internist/diabetologist and psychologist. The expert panel will check if the individual fulfils the inclusion criteria and give final approval. Next, the local pancreatic multidisciplinary team will be informed and asked for agreement on the decision to perform prophylactic (minimally invasive) total pancreatectomy based on the available imaging and reports of all completed consultations. Individuals will be advised to undergo this procedure at the age of 50 years, 10 years before the peak incidence of PDAC development.
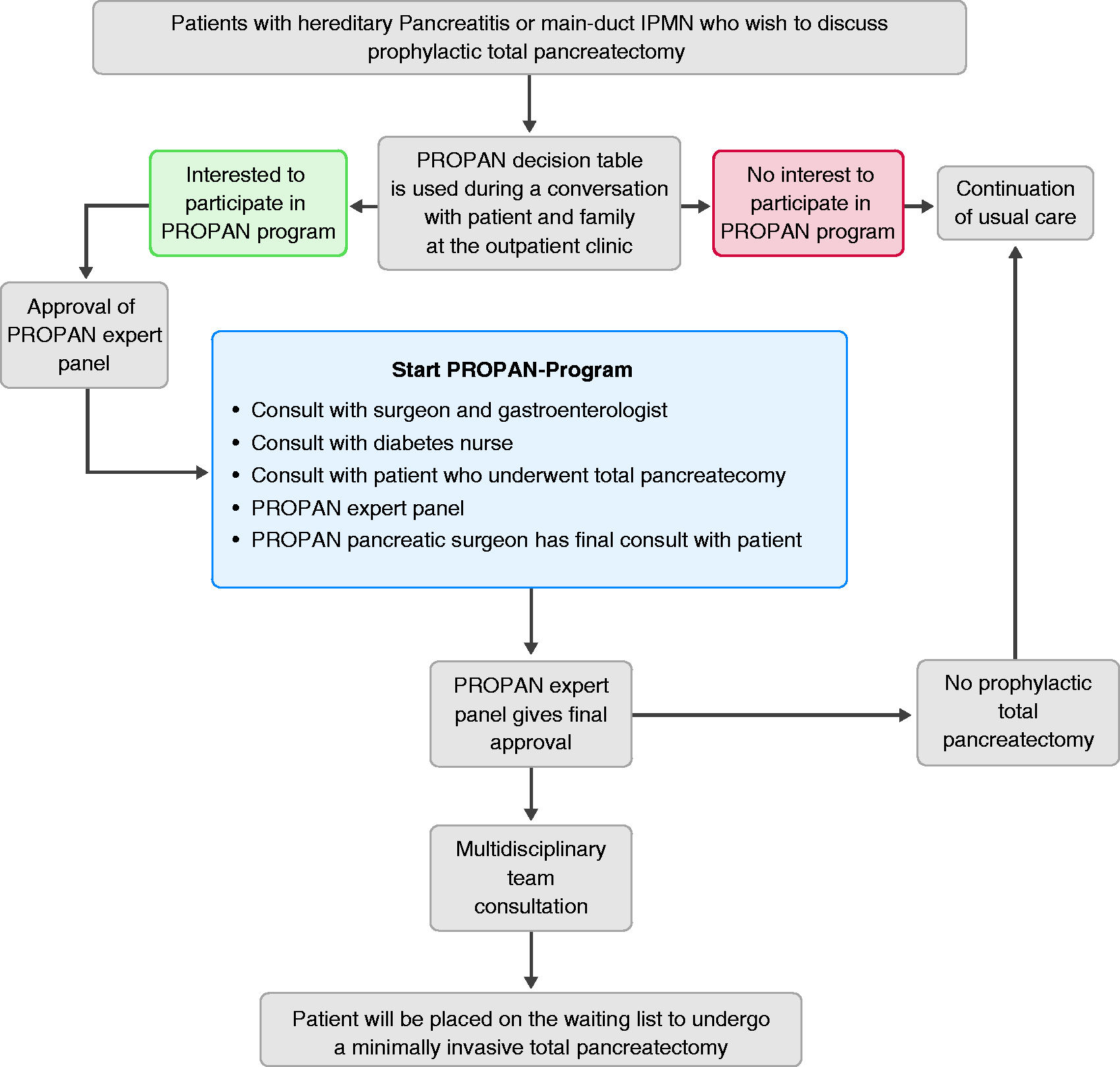
Flow chart of the PROPAN programme.
Implementation
It is expected that the number of Dutch patients eligible for participation in this programme is limited and that initially only a few will opt for minimally invasive total pancreatectomy. Therefore, the programme will only start in very high volume pancreatic centres which perform at least 80 pancreatoduodenectomies annually and have extensive experience with minimally invasive pancreatoduodenectomy and who can organise the programme as stated. 92 Experience with minimally invasive pancreatectomy was guaranteed by participation in the LEALAPS, LAELAPS-2, and LAELAPS-3 structured nationwide training programmes in, respectively, laparoscopic distal pancreatectomy, laparoscopic pancreatoduodenectomy and robotic pancreatoduodenectomy, based on the IDEAL framework.93–95 An evaluation meeting with all involved stakeholders will be planned after five individuals have completed the full protocol as well as surgery.
Discussion
Together, the Dutch Pancreatic Cancer Group and the Dutch patient organisation for pancreatic cancer developed a shared decision-making programme for prophylactic total pancreatectomy using decision tables, to assist individuals at high risk of developing PDAC who wish to discuss the pros and cons of (minimally invasive) prophylactic total pancreatectomy.
The PROPAN programme addresses an important dilemma. Prophylactic total pancreatectomy for high-risk patients is not mentioned in current guidelines and no results are available in the literature. Traditionally, surgeons have been reluctant to perform total pancreatectomy because of postoperative exocrine and endocrine insufficiency, which both worsen quality of life. 32 However, taking into account the improved surgical outcome of total pancreatectomy and the improved treatment of exocrine and endocrine insufficiency, the option to remove the pancreas prophylactically has been brought up by high-risk individuals and their family members. 6 ,12–14 The PROPAN programme offers a conceptual and informative framework for shared decision-making in this treatment dilemma. It should be noted that this programme does not aim to lower the threshold for prophylactic total pancreatectomy. This programme does offer a framework for discussion between patients and caregivers in the situation that the European evidence-based guideline on pancreatic cystic neoplasms advises to perform total pancreatectomy because of main-duct or mixed-type IPMN. Clearly, there is room for discussion based on this advice. For instance, an earlier series showed that in elderly patients with main-duct IPMN with worrisome features or high-risk stigmata, disease-specific 5-year survival was 81%, which was comparable with patients after resection. 96 , 97 In particular, a main pancreatic duct of 5–9 mm did not affect disease-specific survival. 96 Furthermore, in an international expert survey and case vignette study, 97 experts disagreed on the indication for pancreatectomy in patients with main-duct or mixed-type IPMN in the entire pancreas with a nodule or tumour: 41% advised surveillance whereas 59% advised operative intervention. 98 Of those who advised operative intervention, 46% would perform a total pancreatectomy and 31% pancreatoduodenectomy with follow-up. This again shows that there is room for shared decision-making.
Prophylactic surgery with removal of an entire target organ is already used in several genetic syndromes, such as hereditary breast and ovarian cancers, gynaecological cancers in Lynch syndrome, and has proved to improve survival. 8 In these patients the risks of cancer are 40–70% and similar to the 60% risk in main-duct IPMN and 40% in hereditary pancreatitis. 99 , 100 For hereditary pancreatitis, only patients with a PRSS1 mutation are included, because these patients have the highest risk of developing pancreatic adenocarcinoma. The relative roles of CFTR or SPINK1 mutations in carcinogenesis are not as well established compared with PRSS1 mutations and are therefore excluded in this first decision-making programme. 101 In individuals with familial pancreatic cancer the exact pancreatic cancer risk is not known and, as concluded by Kekis et al. in 2001, prophylactic total pancreatectomy should only be used in the presence of high-grade dysplasia. 102 The problem is how to diagnose or exclude high-grade dysplasia in these patients.
The diagnosis of hereditary pancreatitis is based on genetic testing and is therefore expected to be highly accurate. However, for patients with IPMN it has been demonstrated that there could be a considerable risk of misdiagnosis (approximately 20%). 37 , 103 , 104 Naturally, a total pancreatectomy is not advised in patients without main-duct IPMN or with only main-duct IPMN in the pancreatic head. This emphasises the importance of the PROPAN expert panel, which is clearly aware of this risk of misdiagnosis. Diagnosis should be confirmed in a multidisciplinary team meeting, after careful discussion of imaging potentially in combination with endoscopic ultrasound according to current guidelines. 6 , 91 In case of doubt about the diagnosis, patients are not eligible for the PROPAN programme.
It is yet unclear whether screening programmes improve the survival of individuals at high risk of developing PDAC. No randomised controlled trials are available and the only data available are from prospective cohort studies. Verna et al. showed, in a high-risk population of 51 patients enrolled in a screening programme, that six patients (12%) had neoplastic lesions in the pancreas. 105 They concluded that screening is effective and identifies curable neoplasms that can be resected. Vasen et al. prospectively collected screening outcomes in patients with familial pancreatic cancer or families with a gene defect that predisposes to PDAC. 28 Among 178 p16-Leiden mutation carriers, PDAC was detected in 13 patients (7.3%), whereas the resection rate was 75% and the 5-year survival rate was 24%. Two individuals (0.9%) in the familial pancreatic cancer cohort had a pancreatic tumour, including one with advanced PDAC and one with an early grade 2 neuroendocrine tumour. Four patients (1.9%) had high-risk lesions, out of 13 individuals with familial pancreatic cancer (6.1%) that underwent surgical resection for a suspected precursor lesion. The authors concluded that surveillance in this study of p16-Leiden mutation carriers is relatively successful, detecting most PDACs at a resectable stage, while the benefit of surveillance in families with familial pancreatic cancer is less evident.
All patients undergoing total pancreatectomy will instantly develop insulin-dependent diabetes. Glucose control in these patients may be challenging due to the complete loss of pancreatic endocrine parenchyma, secreting insulin and glucagon, which predisposes for difficult to control postoperative hypo and hyperglycemia. This will affect a patients’ life substantially and will therefore be emphasised by the surgeon and gastroenterologists, but also extensively discussed during the consult with the diabetes nurse and patient who underwent a total pancreatectomy. A recent systematic review on diabetes mellitus-related outcomes revealed a need for further improvement of diabetes management after total pancreatectomy, especially regarding the prevention of hypoglycaemia-related morbidity and even mortality. 13 This study also showed that quality of life seems moderately affected by total pancreatectomy. One of the largest studies on this topic to date reported that quality of life with new-onset diabetes mellitus after total pancreatectomy is similar to that of patients with type 1 diabetes. 14 , 106
Evidence about minimally invasive total pancreatectomy from large prospective studies is lacking. In a systematic literature review, Kuesters et al. included five studies with a total of 21 patients. They found 0% mortality after a short follow-up period ranging from 3 to 38 months and concluded that in this small group of selected patients this procedure is feasible, if carried out in centres with large expertise in minimally invasive and pancreatic surgery (see Table 4). 107 Our group has recently initiated a European study to assess outcomes after minimally invasive total pancreatectomy within the European Consortium on Minimally Invasive Pancreatic Surgery.
In conclusion, the PROPAN programme provides a conceptual and informative framework with decision tables for both high-risk individuals and physicians who wish to discuss prophylactic total pancreatectomy. The programme includes preoperative counselling, weighing the pros and cons between the reduction in PDAC risk and the risks and long-term consequences of total pancreatectomy, as well as the uncertainty regarding lifelong surveillance as an alternative management approach.
Supplemental Material
sj-pdf-1-ueg-10.1177_2050640620945534 - Supplemental material for Prophylactic total pancreatectomy in individuals at high risk of pancreatic ductal adenocarcinoma (PROPAN): systematic review and shared decision-making programme using decision tables
Supplemental material, sj-pdf-1-ueg-10.1177_2050640620945534 for Prophylactic total pancreatectomy in individuals at high risk of pancreatic ductal adenocarcinoma (PROPAN): systematic review and shared decision-making programme using decision tables by Lianne Scholten, Anouk EJ Latenstein, Cora M Aalfs, Marco J Bruno, Olivier R Busch, Bert A. Bonsing, Bas Groot Koerkamp, I Quintus Molenaar, Dirk T Ubbink, Jeanin E van Hooft, Paul Fockens, Jolanda Glas, J Hans DeVries, Marc G Besselink and for the Dutch Pancreatic Cancer Group in United European Gastroenterology Journal
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
Not applicable
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Lianne Scholten received a grant from Zealand Pharma A/S for studies on postpancreatectomy diabetes management. Marc G Besselink received a grant from the Dutch Cancer Society (grant number UVA2013-5842) for studies on pancreatic cancer.
Informed consent
Not applicable
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
