Abstract
Background and Aims:
Patients with ulcerative colitis are often treated with multiple immunomodulative agents to achieve remission. In refractory disease, the next option is frequently proctocolectomy with ileal pouch–anal anastomosis. No consensus exists as to whether immunomodulatory therapy at the time of ileal pouch surgery leads to any increase in postoperative complications. Our aim was to assess, in ulcerative colitis patients with restorative proctocolectomy, the effect of preoperative anti-tumor necrosis factor therapy and corticosteroids on postoperative complications and pouch failure.
Materials and Methods:
A retrospective medical record review of 445 patients with ulcerative colitis who underwent proctocolectomy with ileal pouch–anal anastomosis in Helsinki University Hospital between January 2005 and June 2016.
Results:
Anti-tumor necrosis factor agents were not associated with postoperative complications. Only high-dose corticosteroids (prednisolone ⩾20 mg or equivalent) were associated with higher incidence of anastomotic leak (12.6% vs 2.5%, P = 0.002) and wound dehiscence (4.2% vs 0%, P = 0.019), but pouch failure rate was no higher (2.1% vs 0%, P = 0.141) than in patients without corticosteroid treatment. A lower dosage of corticosteroids had no effect on early postoperative complications, but pouch failure rate was increased (4.4% vs 0%, P = 0.015).
Conclusion:
Corticosteroids, but not anti-tumor necrosis factor therapy, were associated with postoperative complications. Preoperative use of corticosteroids may increase pouch failure rate, but the risk is still minor in high-volume centers performing ileal pouch surgery.
Keywords
Introduction
Ulcerative colitis is an idiopathic chronic inflammatory disease that usually starts in the rectum and extends proximally to the colon. First-line therapy for ulcerative colitis is 5-aminosalisylates and corticosteroids followed by thiopurines, methotrexate, anti-tumor necrosis factor (TNF) agents (infliximab, adalimumab, and golimumab), or adhesion molecule inhibitors (vedolizumab) (1). However, approximately 14% of patients need surgery during the first 10 years after their diagnosis (2) and as high as 20%–30% at some point during their disease course (3). Refractory disease not responding to medical therapy frequently leads to surgery. Surgery is also indicated when dysplasia or cancer is present and in patients with fulminant colitis (4).
In ulcerative colitis, the now-standard operation is restorative proctocolectomy with ileal pouch–anal anastomosis (IPAA) avoiding a permanent ileostomy. Furthermore, what seems to be safe is to omit a protective ileostomy in low-risk patients undergoing proctocolectomy, as we recently showed (5). Pelvic sepsis, including anastomotic leak, pouch abscess, and pouch-anal fistula, is a predictor for poor functional outcome and pouch failure (6), making it therefore important to identify possible risk factors for pelvic sepsis. Some evidence suggests that preoperative corticosteroids may promote the risk for early postoperative complications, especially infections (7, 8). Regarding biologics, the data remains conflicting (9). Anti-TNF agents and vedolizumab are currently approved for the treatment of moderate-to-severe ulcerative colitis, creating a challenge to distinguish whether the complications are due to disease severity or are associated with the immunomodulative therapy. We therefore performed this retrospective single center cohort study to evaluate, in patients with ulcerative colitis, the association of preoperative anti-TNF agents and corticosteroids on postoperative complications and pouch survival. We hypothesize that anti-TNF agents and corticosteroids might increase the complication rate.
Materials and Methods
We retrospectively reviewed the medical records of 460 consecutive patients undergoing restorative proctocolectomy and IPAA due to ulcerative colitis at Helsinki University Hospital between January 2005 and June 2016. The exclusion criteria were the use of other immunosuppressive medication than anti-TNF agents, corticosteroids, 6-mercaptopurine, azathioprine, or methotrexate. We excluded nine patients who had undergone liver transplantation and used tacrolimus to prevent rejection and three other were excluded because they were using vedolizumab. In addition, three patients were excluded because their corticosteroid dosage was unclear. In total, the final analysis included 445 patients.
Four specialized colorectal surgeons operated on all patients using an open technique with a low midline incision. We described the surgical technique in detail previously (5). Surgeons preferred a one-stage procedure, and the IPAA was mainly hand sewn (90.1%). Of all patients, those without a diverting ileostomy amounted to 71.9%. All 125 ileostomies were closed later.
Data recorded included preoperative anti-TNF agents administered within 12 weeks before primary surgery, total number of anti-TNF-agent infusions, preoperative use, and dosage of corticosteroids within 6 weeks before primary surgery, type of operation, other immunomodulatory therapy (6-mercaptopurine, azathioprine, or methotrexate), postoperative early and late complications, and need for relaparotomy. Furthermore, we recorded preoperative hemoglobin and albumin levels to analyze their effect on postoperative complications.
All complications within 3 months after the primary surgery were collected and graded according to the Clavien–Dindo Classification (10). If a patient had more than one complication, the higher grade was the choice for analysis. Computed tomography (CT) scan with per-anally administered contrast agent, pouchscopy, or surgery were used to diagnose anastomotic leaks when symptoms were alarming. We assessed dehydration as a disturbance of sodium and potassium levels and as a need for supplementary hydration postponing hospital discharge. Determination of bowel obstruction was based on excess vomiting, need for a nasogastric tube, or stoma malfunction necessitating one or more additional hospital days. Pouch failure was defined as excision of the ileoanal pouch or permanent loop ileostomy. All possible pouch excisions were performed in Helsinki University Hospital, therefore we had full data of failures.
Patients who had undergone anti-TNF-α infusions within 12 weeks before surgery were compared with all other patients to learn whether preoperative anti-TNF agents results in increased risk for postoperative complications. We also compared patients who underwent surgery while taking corticosteroids, but taking no anti-TNF agents, to patients without corticosteroid treatment. European Crohn’s and Colitis Organization (ECCO) Guidelines state that “prednisolone 20 mg daily or equivalent for more than 6 weeks prior to surgery is a risk factor for surgical complications” (11). With that in mind, we divided steroid-using patients into a low-dose group and high-dose group. The high-dose group included all patients using prednisolone 20 mg or more.
Categorical variables are expressed as number of patients and percentage of the total number of patients. Continuous variables are presented as mean and standard deviation (SD) or median and range. Statistical analyses were performed with the Mann–Whitney U test or Student’s t-test for continuous variables and the chi-square test or Fisher’s exact test for categorical variables. We used the Shapiro–Wilk test to test normality of continuous variables. We considered P values less than 0.05 to be statistically significant, and used a two-tailed test. To compare the no-corticosteroid group, low-dose group, and high-dose group, we used the Jonckheere–Terpstra Trend Test for continuous variables and the linear-by-linear association test for categorical variables. The Bonferroni correction served to account for multiple comparisons as stated in the tables. The Firth’s penalized-likelihood logistic regression was used to create age adjusted multivariate model for anastomotic leak. Missing data are stated in the text. All statistics were calculated using IBM SPSS Statistics software version 24 (IBM Corporation, Armonk, NY, USA) and SAS v 9.4 (SAS Institute Inc, Cary, NC, USA). The institutional review board granted study approval.
Results
Patient Characteristics
Our study population comprised 445 patients. Of those, 33 (7.4%) were treated with anti-TNF agents within 12 weeks prior to surgery. Median time between last infusion and surgery was 6 weeks (3–11 weeks). The most frequently used anti-TNF agent was infliximab (29/33), but two patients received injections of golimumab and two were treated with adalimumab. Median number of infusions was four (1–22). Comparing those 33 patients with all others, no statistical differences between the groups emerged with respect to age, gender, or number of covering ileostomies (Table 1). However, the median disease duration time was shorter in patients treated with anti-TNF agents (P = 0.039), and these were also on a higher dose of prednisolone (P = 0.001).
Characteristics of the patients treated with anti-TNF agents.
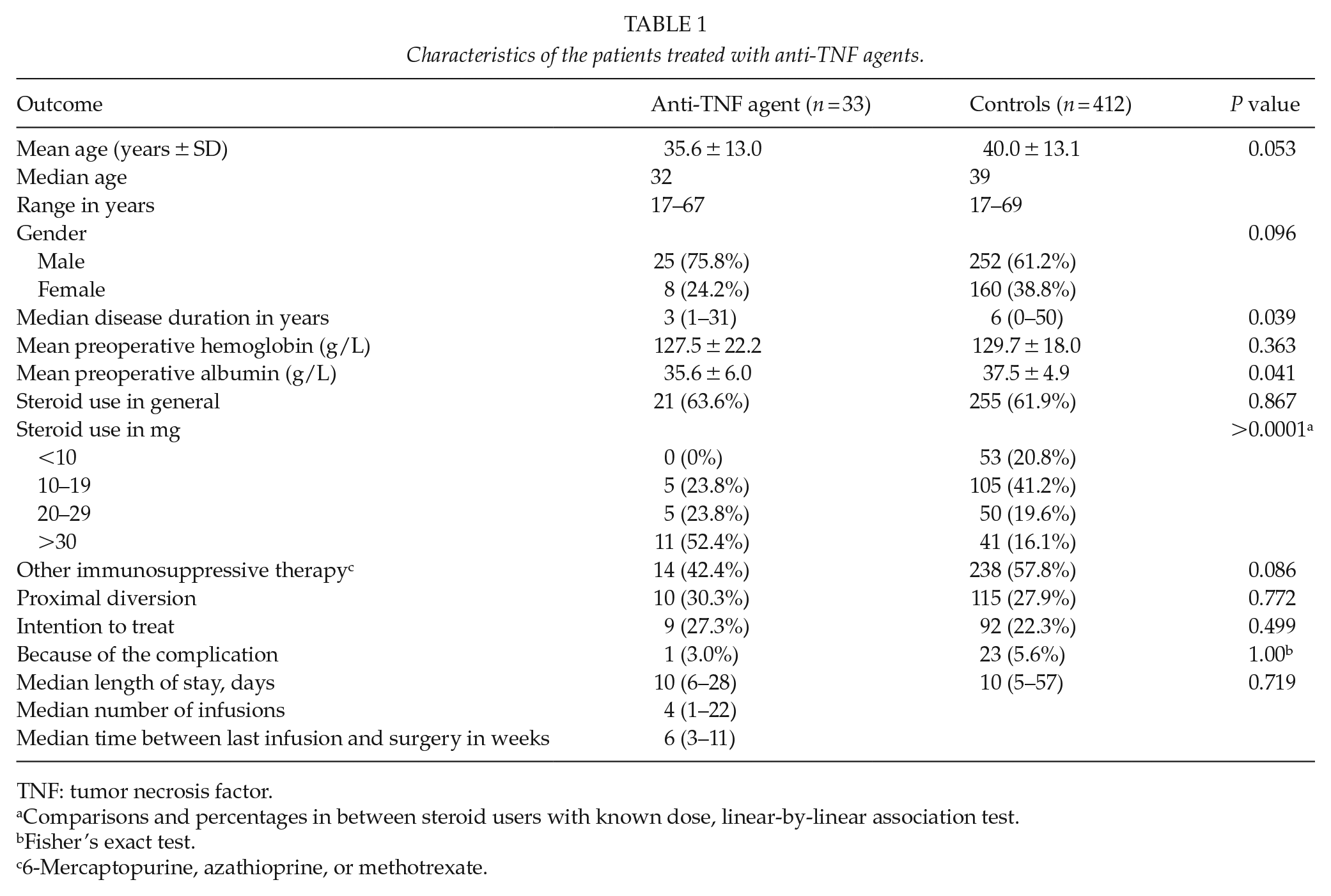
TNF: tumor necrosis factor.
Comparisons and percentages in between steroid users with known dose, linear-by-linear association test.
Fisher’s exact test.
6-Mercaptopurine, azathioprine, or methotrexate.
Among patients without anti-TNF-α therapy, 255 (56.0%) used corticosteroids, of whom 95 (37.3%) were on a dosage equivalent to or more than prednisolone 20 mg (high-dose group), and 160 (62.7%) were on less than 20 mg (low-dose group). Although we lack the exact dosage data from six patients, we were able to categorize them into the low- or high-dose group. Both groups were compared with the 157 patients who received no corticosteroids within 6 weeks prior to surgery (no-corticosteroid group). Median disease duration time was significantly longer in that group than in either corticosteroid groups (P < 0.0001, Table 2). The explanation for this difference may depend on the indication for surgical treatment. Nearly half the no-corticosteroid group had surgery because of dysplasia or cancer: 68 (43.3%), but the majority of those in steroid groups underwent surgery because of refractory disease: 232 (91.0%). Hemoglobin and albumin levels were lower in both steroid groups, indicating that those patients had more severe ulcerative colitis.
Characteristics of no-corticosteroid group, low-dose group, and high-dose group.
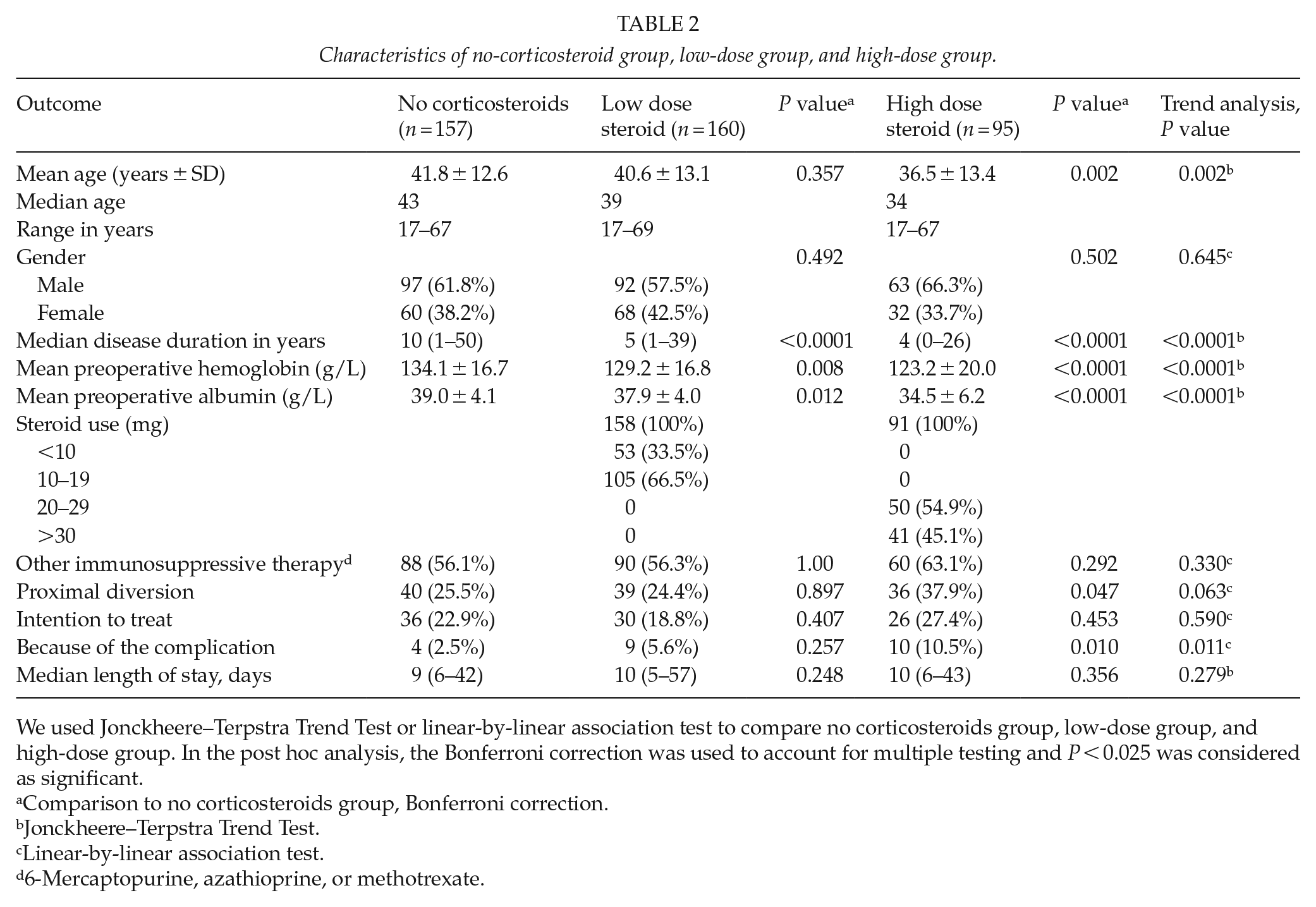
We used Jonckheere–Terpstra Trend Test or linear-by-linear association test to compare no corticosteroids group, low-dose group, and high-dose group. In the post hoc analysis, the Bonferroni correction was used to account for multiple testing and P < 0.025 was considered as significant.
Comparison to no corticosteroids group, Bonferroni correction.
Jonckheere–Terpstra Trend Test.
Linear-by-linear association test.
6-Mercaptopurine, azathioprine, or methotrexate.
In our study population, 69 patients were operated without any immunosuppressive therapy prior surgery. We compared other therapy groups to this group to further analyze the risk ratio of anastomotic leak with different immunomodulative therapies (Table 5).
Early Postoperative Complications
The overall leakage rate in our cohort of 445 was 5.4%, 24 patients. Of these, 21 patients needed operative treatment. Most of the leakages were diagnosed with CT scan or pouchscopy. During the relaparotomies, we diagnosed nine leaks. As in Table 3, comparison of the anti-TNF agent group to all others revealed no statistical difference in early postoperative complications.
Postoperative complications in patients treated with biologics.
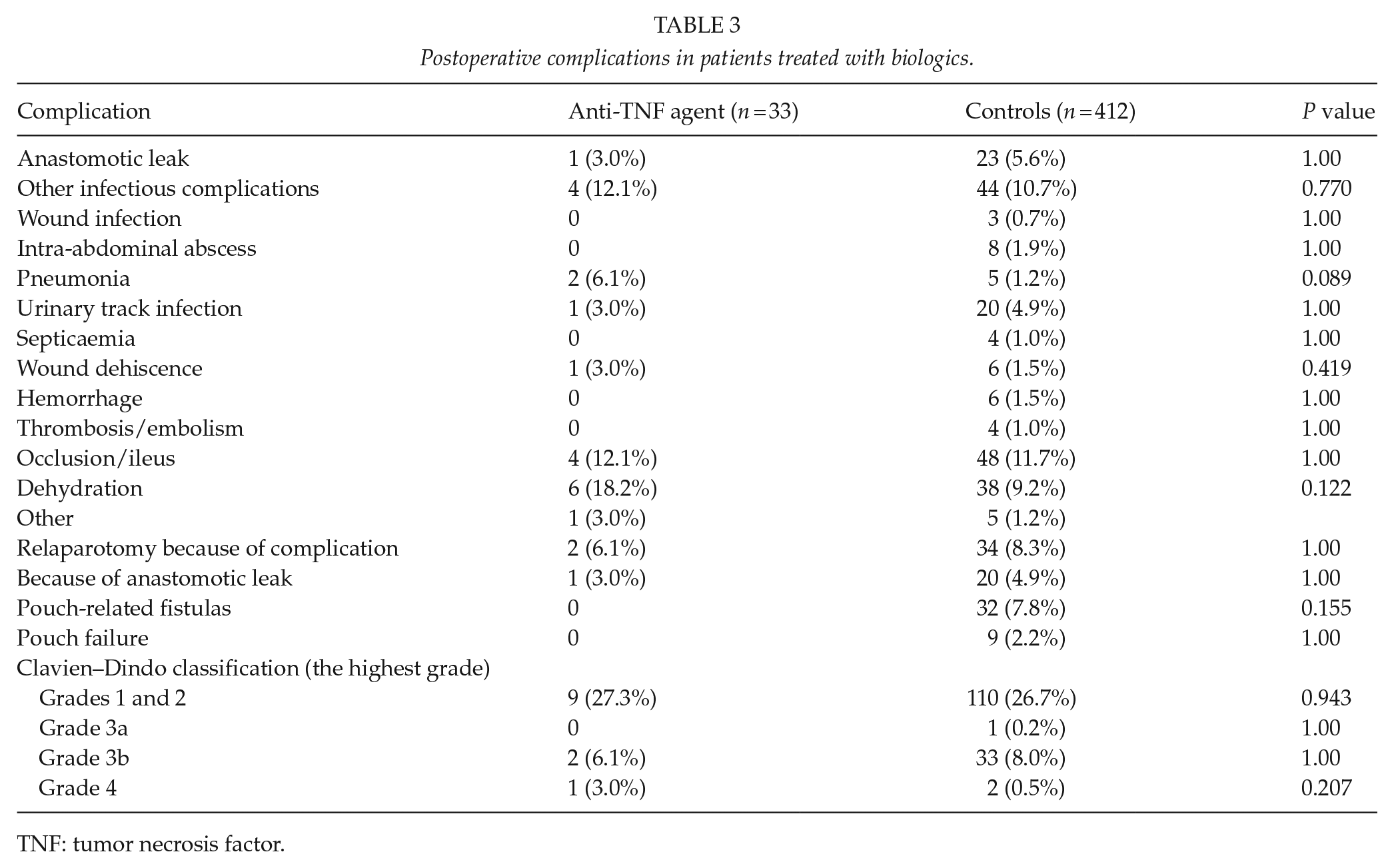
TNF: tumor necrosis factor.
Postoperative complications in patients on corticosteroids are presented in Table 4. High-dose corticosteroids was associated with anastomotic leak (P = 0.002, age adjusted odds ratio (OR) 5.7, 95% confidence interval (CI) 1.87–17.32, P = 0.024, Table 5) and wound dehiscence (P = 0.019), which led to a higher relaparotomy rate (12.6%, P = 0.025), but low dose (<20 mg/day) was not a risk factor for early complications. Albumin levels were lower in both steroid groups (low-dose group, P = 0.012; high-dose group, P < 0.0001; Table 2) but low albumin level itself (<25 g/L, Fisher’s exact test) was not associated neither with anastomotic leak (P = 0.570), wound dehiscence (P = 1.00), nor pouch failure (P = 1.00). Preoperative 6-mercaptopurine, azathioprine, or methotrexate led to no increase in the odds for anastomotic leak (age adjusted OR 1.03, 95% CI 0.49–2.33, P = 0.938, Table 5).
Postoperative complications in no-corticosteroid group, low-dose group, and high-dose group.
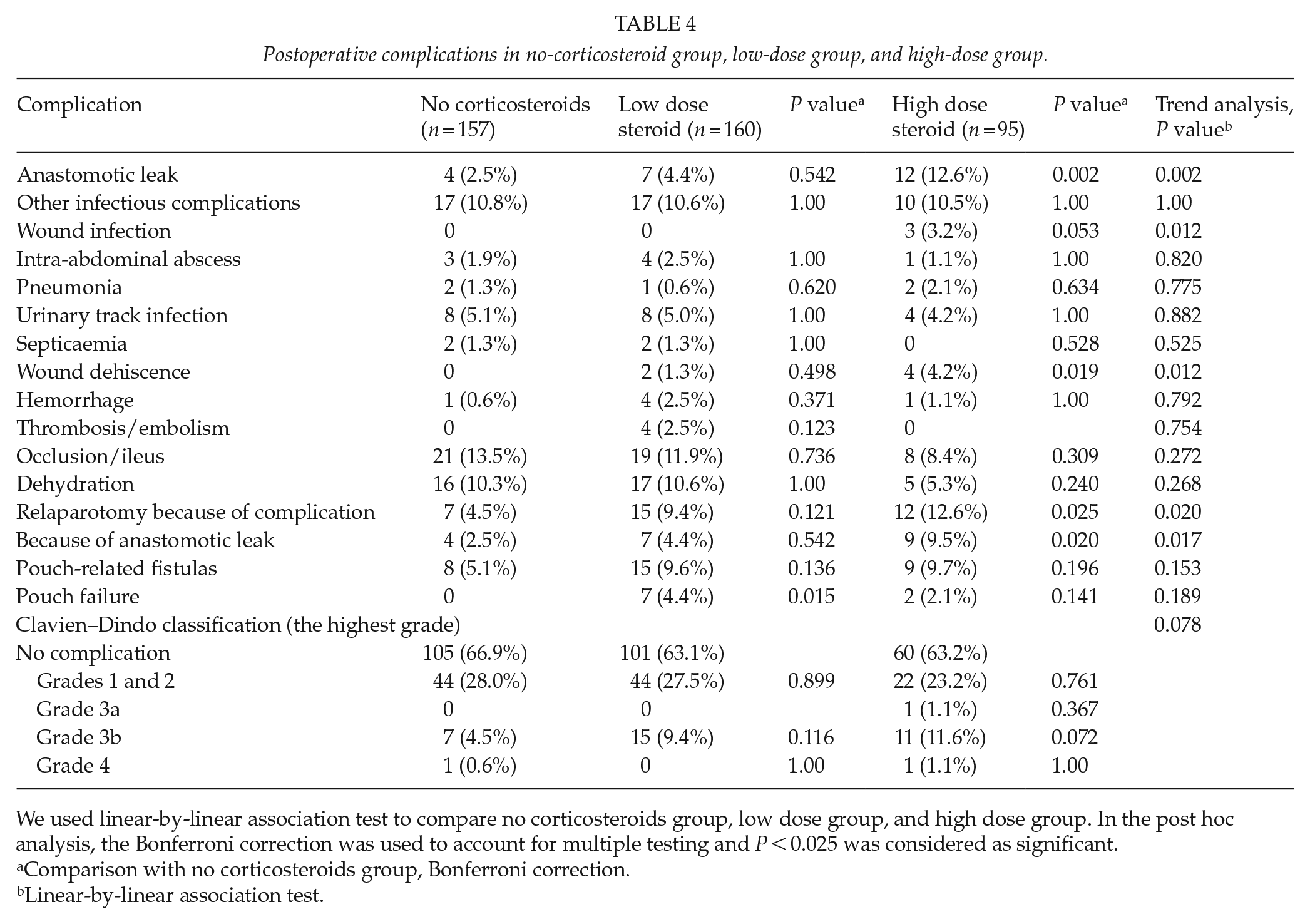
We used linear-by-linear association test to compare no corticosteroids group, low dose group, and high dose group. In the post hoc analysis, the Bonferroni correction was used to account for multiple testing and P < 0.025 was considered as significant.
Comparison with no corticosteroids group, Bonferroni correction.
Linear-by-linear association test.
Risk ratio of anastomotic leak in patients with different immunomodulative therapies compared with patients without any immunosuppressive medication.
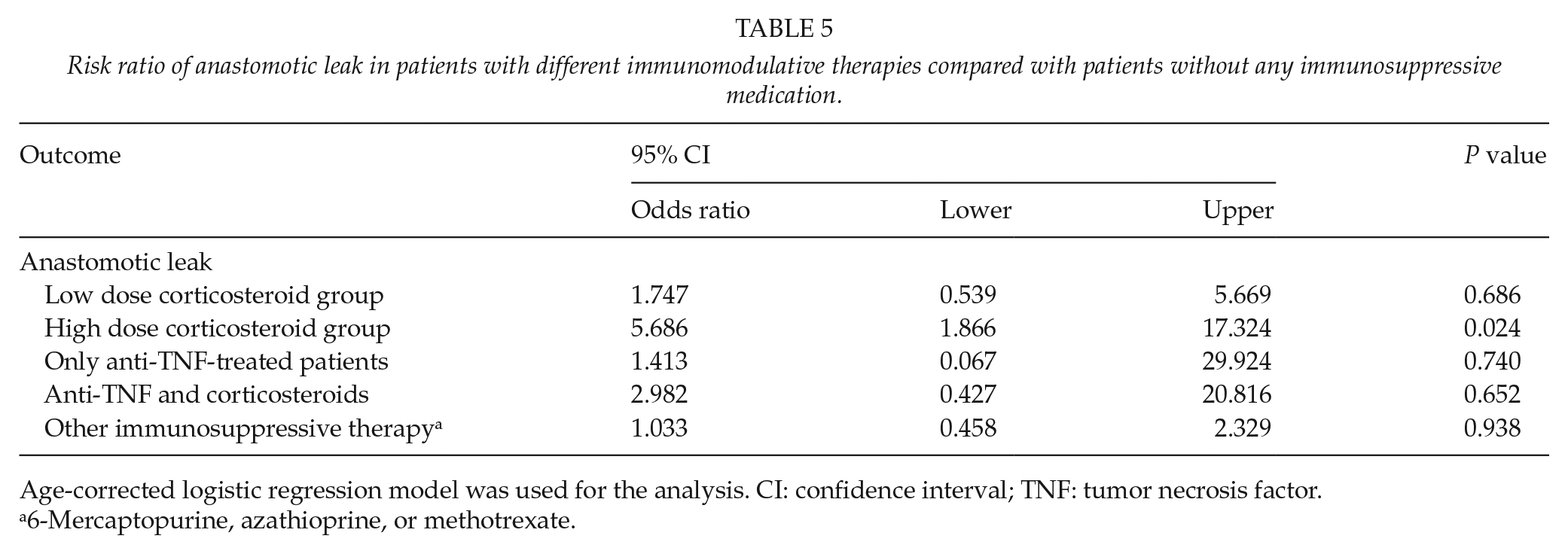
Age-corrected logistic regression model was used for the analysis. CI: confidence interval; TNF: tumor necrosis factor.
6-Mercaptopurine, azathioprine, or methotrexate.
Long-Term Pouch Survival
During the follow-up period, overall pouch survival was 98.0%. Median follow-up time was 6.9 years (1.2–12.6 years). We did not have any patients with permanent loop ileostomy.
In the anti-TNF group, pouch removal was not necessary for any of these patients, and none suffered any pouch-related fistulas (Table 3). Pouch failure was slightly more frequent in the low-dose steroid group (7 patients, 4.4%, P = 0.015) compared with the no-corticosteroid group with its absence of any failures (Table 4). Kaplan–Meier results also showed a difference in long-term survival of the pouch between the no-corticosteroid group and low-dose steroid group (P = 0.011, Fig. 1), however the number of events is relatively low. Between these groups, the rate of pouch-related fistulas did not differ.
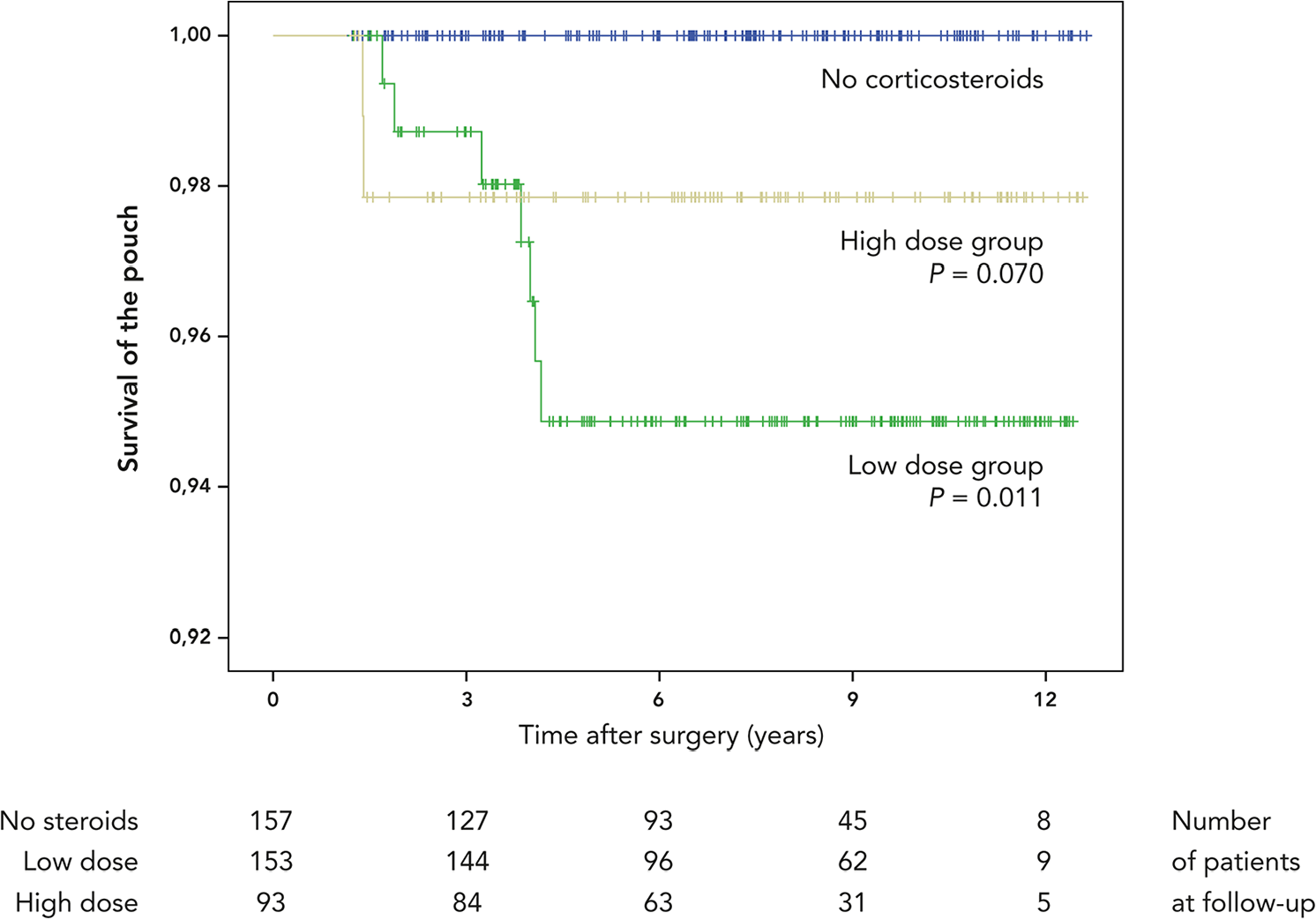
Kaplan–Meier curve for cumulative survival of the pouch in no-corticosteroid group, low-dose group, and high-dose group. P values presented are comparisons to no-corticosteroid group. Log-rank was used for the analysis.
Discussion
Debate is ongoing as to whether preoperative use of anti-TNF agents in ulcerative colitis is a risk factor for postoperative complications. European guidelines recommend avoiding single-stage proctocolectomy in anti-TNF-treated patients (11). Recently, Kulaylat et al. (12) in a retrospective study with one of the largest patient cohorts (1172 patients of whom 303 received anti-TNF agents) showed that anti-TNF agents were associated with higher postoperative complication rates among patients undergoing proctocolectomy and IPAA but not among patients who underwent colectomy. The data came from a database comprising only privately insured patients, which might have biased the results. However, in agreement with our results, retrospective data from Canada showed the opposite with 758 patients, of whom 196 received anti-TNF treatment (13). They also evaluated the association of serum infliximab levels and short-term complications but again found no correlation. Furthermore, the meta-analyses have reported conflicting results (14, 15).
Evidence is stronger regarding preoperative corticosteroids. Aberra et al. (7) demonstrated a higher risk for any or for major infectious complications in inflammatory bowel disease (IBD) patients taking steroids prior to any abdominal surgery. Another study from the United Kingdom suggested that steroids cause an increase in the pelvic sepsis in the early postoperative period but not later (16). Two studies from Japan concluded that a greater risk factor for postoperative infections was a total preoperative dosage of corticosteroids rather than a dose at the time of proctocolectomy (17, 18).
In our patient cohort, preoperative prednisolone at a 20 mg equivalent or more was associated with anastomotic leakage and wound dehiscence, and an anastomotic leak may lead to the pouch’s poor functional outcome and eventually to its failure (6, 19). In our study, however, pouch failure was not higher in the high-dose group perhaps because of our relatively small group size and short follow-up. Supporting this assumption, we found significantly more failures of the pouch in low-dose group than in patients without corticosteroids, although the failure rate was still low (4.4%).
Biologics are associated with antibody development. To reduce this risk, biologics are frequently combined with low-dose thiopurines or methotrexate, which, in their case, may elevate the risk for postoperative complications. Earlier, 6-mercaptopurine or azathioprine alone was not associated with postoperative complications (7, 20). Less data are available regarding methotrexate. One retrospective study found, in IBD patients, no association between preoperative methotrexate and postoperative complications (21). One multicenter retrospective study suggested that concurrent anti-TNF and steroids may elevate risk for anastomotic leakage (22). In Crohn’s disease, a combination of corticosteroid, immunomodulator, and biological treatment has been significantly associated with intra-abdominal sepsis (23). For ulcerative colitis, however, another recent study found no association between multiple immunosuppressive agents and surgical-site infections including pelvic sepsis and anastomotic leakage (24). In our study, anti-TNF agents caused no increase in early postoperative complications or pouch failure rate. Nor did we find any additive effect when combining anti-TNF agents and steroids (Table 5). In addition, thiopurines or methotrexate alone did not seem to be associated with increased risk for anastomotic leakage, but our low occurrence of anastomotic complications weakens the statistical power of the analysis.
Overall, the occurrence of pouch failures in our study population was 2.0%. A systematic review and meta-analysis of de Zeeuw et al. (25) showed a pouch failure rate of 4.7%, along with an incidence of pelvic sepsis of 7.5%. Thus, our pouch failure rates, even in those patients using corticosteroids, compared favorably with rates in other studies. In our center, the covering ileostomy is performed only for high-risk patients. We had a higher incidence of relaparotomies due to complications in our high-dose group (10.5%), mainly because of anastomotic leaks (9.5%). The covering ileostomy should be considered for all patients using high doses of corticosteroids before surgery, especially in the presence of other risk factors for anastomotic leakage such as tension of the anastomosis or suboptimal visibility during suturing of the anastomosis.
Our study is retrospective single-center cohort study. However, it is population-based which we consider to be a strength. The limitations of our study are its retrospective design and the small number of patients treated with anti-tumor necrosis factor therapy. Presumably, anti-TNF agents and higher dosage of corticosteroids were more frequent for patients with more severe ulcerative colitis, and this may have caused selection bias.
In conclusion, preoperative anti-tumor necrosis factor therapy was not associated with early postoperative complications or pouch failure. High-dose corticosteroids seem to elevate the risk for anastomotic leak and wound dehiscence. Although corticosteroids possibly lead to a slight increase in risk for later pouch failure, the risk is still small in high-volume centers performing restorative proctocolectomy with IPAA.
Footnotes
Author Contributions
E.K.K., L.R.-S., and A.L. designed the study. E.K.K. collected the data. E.K.K. and H.K.M. performed the analysis. E.K.K. drafted the manuscript while L.R.-S., M.F., and A.H.L. provided supervision. All authors have critically commented the manuscript and approved the final version.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
