Abstract
Background and Aims:
There are only few data on the influence of cinacalcet on the outcome of parathyroidectomy in patients with renal hyperparathyroidism. Indication and timing of surgery have changed since its introduction, especially with regard to kidney transplantation. Therefore, we retrospectively analyzed patients undergoing parathyroidectomy for renal hyperparathyroidism in our institution.
Material and methods:
Between 2008 and 2015, 196 consecutive operations in 191 patients were analyzed. About 80 operations (41%) were performed in patients receiving cinacalcet compared with 116 operations (59%) in patients without cinacalcet. Clinical data, preoperative medication, pre- and postoperative laboratory values, type and details of surgery including complications, as well as cardiovascular complications and kidney transplantation with graft function were recorded.
Results:
Demographical data were similar in patients with or without cinacalcet treatment. A total of 54% of patients received a kidney graft before or after parathyroidectomy. Pre- and postoperative parathormone levels were similar in both groups (preoperatively 755 vs 742 ng/L, postoperatively 50 vs 46 ng/L, p > 0.10), whereas patients with cinacalcet showed significantly lower calcium levels preoperatively (2.28 vs 2.41 mmol/L, p = 0.0002). There was no difference in recurrence or persistence of hyperparathyroidism, duration of surgery, hospital stay, or complication rate. Creatinine levels in patients with tertiary hyperparathyroidism were similar after 1-year follow-up.
Conclusion:
Cinacalcet did not influence outcome of patients with parathyroidectomy for renal hyperparathyroidism and can be safely offered to patients not responding to medical treatment.
Introduction
Since the introduction of cinacalcet in the treatment of renal hyperparathyroidism (rHPT) in 2004, clinical practice for indication and timing of surgery has changed following the Kidney Disease Improving Global Outcomes (KDIGO) International guideline group (1). Overall, the number of parathyroidectomies (PTxs) has declined (2) and the timepoint of surgery is delayed in patients with end-stage renal disease (ESRD) developing rHPT (3). But especially the long-term benefit of cinacalcet on patient morbidity and mortality has not been shown so far (4). In addition, the cost-effectiveness of surgery is better than that of cinacalcet treatment after 7.25 months of therapy (5), and cinacalcet is only cost-effective in patients not eligible for surgery or receiving a kidney-graft beforehand (6). On the contrary, a large cohort study showed a rather high 30-day morbidity and mortality (7) as well as an increased morbidity and rehospitalization rate in the first year for patients after PTx not receiving a kidney graft (8). Yet, the overall risk of death for dialysis patients is lower after PTx compared with patients with medical therapy alone (9), and PTx shows a trend in improving cardiovascular morbidity and mortality (10).
In 2015, Messa (11) highlighted the need for additional randomized controlled trials comparing conservative and surgical therapy. Another question of concern is the timing of surgery in relation to kidney transplantation (KTx). It is known that in more than 40% of patients, rHPT persists after KTx (12) and that especially long duration of dialysis prior to KTx negatively influences spontaneous normalization. Studies have shown that parathyroidectomy after KTx has no negative influence on overall graft survival (13), although a temporary decrease in glomerular filtration rate (GFR) was confirmed (14). On the contrary, a retrospective analysis reported that PTx before KTx was associated with a lower risk of graft failure (15). A very recent study documented that persistent hyperparathyroidism (pHPT) more than 1 year after KTx is a risk factor for graft failure (16). In addition, cinacalcet treatment in patients receiving a kidney graft was a risk factor for delayed graft function experimentally and in a single cohort study (17).
While in Germany cinacalcet cannot usually be prescribed for patients after KTx, it has been shown that its use after KTx corrects hypercalciuria and hyperparathyroidism (HPT) (18). In 2015, a randomized study concluded that patients with persistent HPT at 6 months after KTx should undergo PTx for normalization of parathyroid hormone (PTH) because of beneficial effects on bone mineral density compared with continuing cinacalcet therapy (19).
So far, there are only few data on the influence of cinacalcet on the short- and long-term outcome of parathyroidectomy in patients with rHPT. Therefore, we retrospectively analyzed all patients undergoing parathyroidectomy for rHPT from the introduction of cinacalcet into clinical practice up to 2015.
Material and Methods
Study Population
All patients undergoing parathyroidectomy for rHPT are prospectively collected in our database and were retrospectively analyzed. Between January 2004 and December 2007, only six patients had been treated with cinacalcet and were not considered for this retrospective analysis. From January 2008 until January 2015, 191 consecutive patients were included. All patients were followed until 1 May 2015 or death or loss of follow-up with a mean follow-up of 48.35 ± 25.2 months. Data were collected from medical charts including standard demographical data and from the kidney transplant database of our institution/European transplantation database (T-base) as well as from referring physicians, especially dialysis centers.
Data concerning duration of dialysis, type of kidney disease, preoperative medication, pre- and postoperative laboratory values (calcium, intact para-thyroid hormone (iPTH), phosphate, creatinine, alkaline phosphatase (AP)), type and details of surgery including complications, as well as cardiovascular complications and KTx with graft function were recorded.
Five patients who required redo surgery for recurrent or persistent HPT were calculated twice, because both operations were performed at our hospital within the study period. Thus, 196 operations in 191 patients were finally analyzed. About 80 operations (41%) were performed in patients receiving cinacalcet at the time of surgery (group A) compared with 116 operations (59%) in patients not receiving cinacalcet (group B).
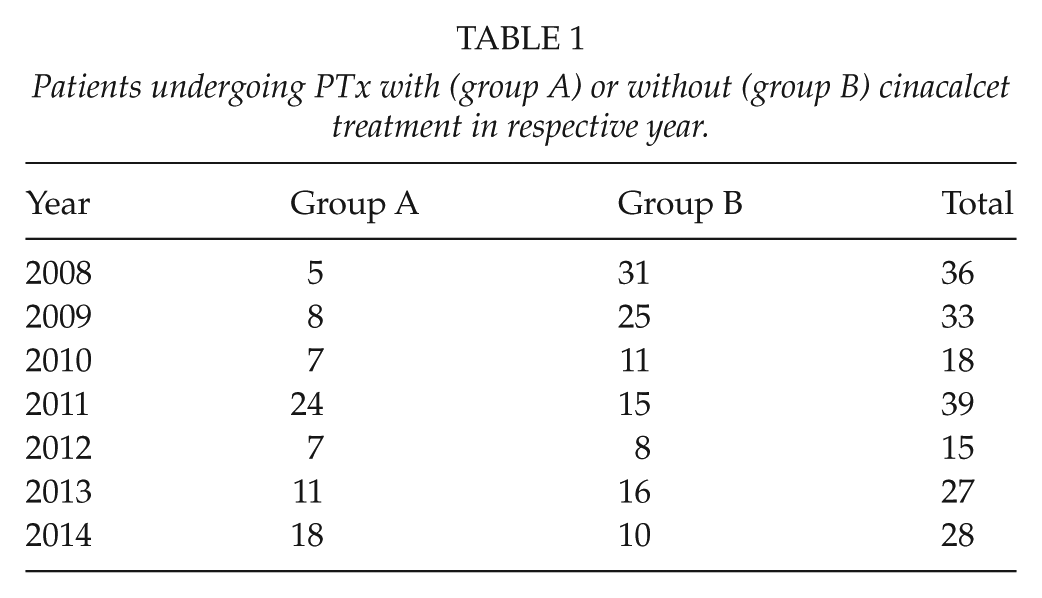
A total of 156 patients (82%) were treated with phosphate binders, and 164 (86%) with 1,25 (OH)-vitamin D. After 2008, there was an increasing number of patients with cinacalcet treatment with a maximum in 2011 (Table 1).
Patients undergoing PTx with (group A) or without (group B) cinacalcet treatment in respective year.
Our standard surgical procedure is a bilateral cervical exploration with subtotal PTx leaving a small nodular-free cervical remnant. A total PTx was performed only in patients with a high risk of recurrent HPT who were not listed for KTx. Cervical thymectomy was not routinely performed but carried out if less than four cervical glands were found. Neuromonitoring of the recurrent laryngeal nerve was routinely performed in all patients. In case of suspicious thyroid nodules or parathyroid glands with close relation to or inside the thyroid capsule, simultaneous thyroid surgery was done. In all patients, parathyroid tissue was cryopreserved.
Postoperatively, all patients had daily controls of calcium, starting on the first postoperative day (POD). In case of persistently high iPTH levels directly after operation, consecutive controls were carried out. In case of missing glands, intraoperative iPTH was measured. Calcium substitution was instituted orally with 1000 mg/day immediately after PTx and adjusted to individual needs including additional intravenous substitution of calcium gluconate and changes in dialysate composition by our in-house nephrologists, as needed. All patients received 0.5 µg calcitriol twice daily with adjustment if needed. Patients were discharged if they were asymptomatic and as soon as the calcium level was stable (at least 1.9 mmol/L) or rising on two consecutive days.
For measuring iPTH, an electrochemiluminescence immunoassay was performed by our laboratory (normal range: 15–65 ng/L). Calcium (normal range: 2.15–2.5 mmol/L), AP (35–105 U/L), creatinine (0.5–0.9 mg/dL), and phosphate (0.87–1.45 mmol/L) were measured routinely by our in-house nephrologists or referring dialysis centers later on.
In order to define persistent or recurrent HPT, both elevated iPTH and calcium may be used. For this analysis, iPTH > 100 ng/L and/or calcium > 2.5 mmol/L were defined as persistent or recurrent hyperparathyroidism.
All patients had an indirect laryngoscopy by an ear, nose, and throat specialist before and after the operation. In case of recurrent laryngeal nerve palsy, logopedic training was prescribed and patients were followed until vocal recovery in our outpatient clinic.
Statistical Analysis
Data were analyzed using the Statistica software 7.0 (StatSoft Inc., USA). All numerical results are presented as mean ± standard deviation (SD) independent from normal distribution. Categorial variables are presented as percentages. For testing normality of numerical variables, the Kolmogorov–Smirnov test (KS test) was used. Normally distributed data were tested with the unpaired t-test after comparing variances with the Levin test. For nonparametric variables with continuous distribution, the KS test was used for independent groups. Chi-square test with Yates’ correction was applied for nominal variables. Nonparametric data in dependent groups were analyzed with the Wilcoxon matched pairs test. Statistical significance was defined by p < 0.05.
Results
Demographic Data and Kidney Disease
The study population consisted of 191 patients with 196 operations distributed to group A (80 cases, 41%) and group B (116 cases, 59%). The mean age was comparable in both groups (group A: 51.2 ± 13.3 vs group B: 51.5 ± 12.7 years), and the male/female ratio was 50% for group A, whereas group B consisted of more male patients (66%). Most patients had chronic kidney disease stage V (CKD V; 91% vs 84%, respectively) at the time of parathyroidectomy, an overview of underlying diseases is given in Table 2.
Underlying diseases leading to renal failure of patients with renal hyperparathyroidism.
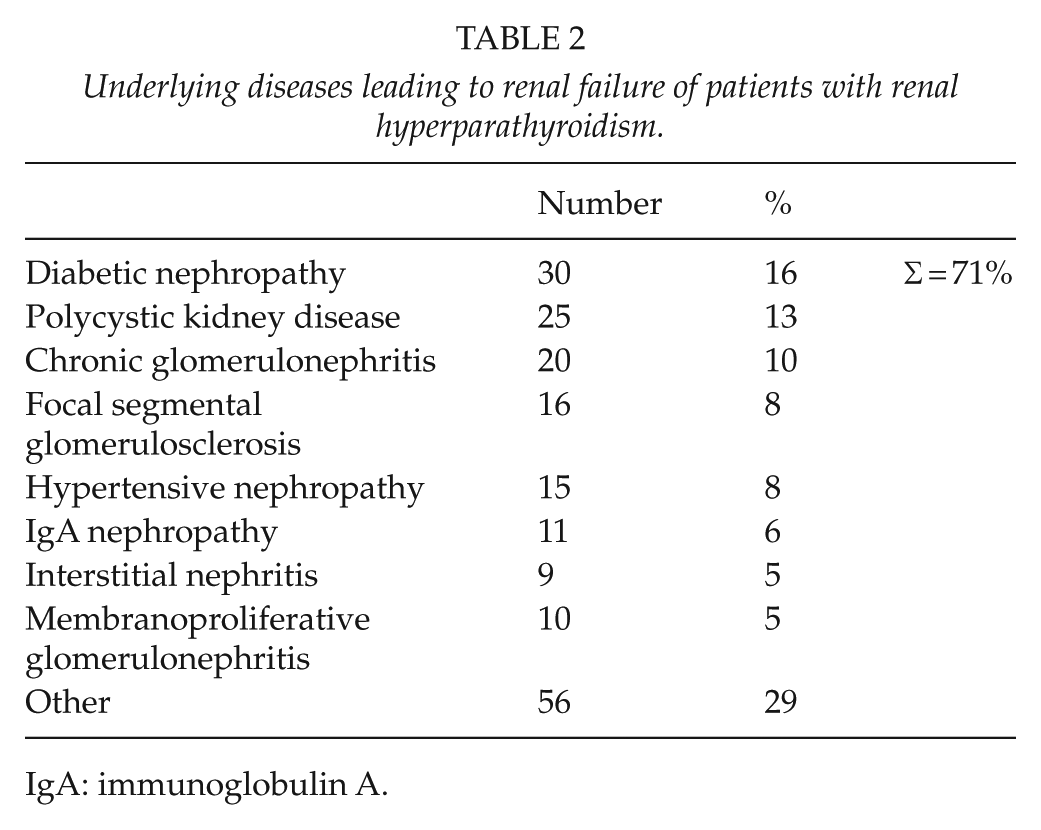
IgA: immunoglobulin A.
A small number of patients had a functioning kidney graft at the time of parathyroidectomy (group A: 6% vs group B: 11%). Rarely, patients with CKD III and IV were operated in preparation of a living donor KTx (3% vs 5%). All patients were presented with comorbidities, including arterial hypertension, diabetes mellitus, adipositas, and coronary artery disease. Follow-up for patients with cinacalcet (group A, 39.7 ± 22.6 months) was significantly shorter compared with patients without cinacalcet (group B, 54.4 ± 25.3 months, p < 0.001, KS test).
Kidney Transplantation
Altogether 53% of the patients (n = 101) underwent KTx before or after PTx (25 patients lost to follow-up). At the end of the follow-up period, more patients were transplanted in both groups (54% vs 64%), but graft function varied between the groups. About 90 patients had no KTx during the study period (Table 3).
Clinical features of patients undergoing parathyroidectomy with (group A) or without cinacalcet (group B) including consecutive or prior kidney transplantation, type of parathyroidectomy, duration of surgery and hospital stay as well as complications and outcome.
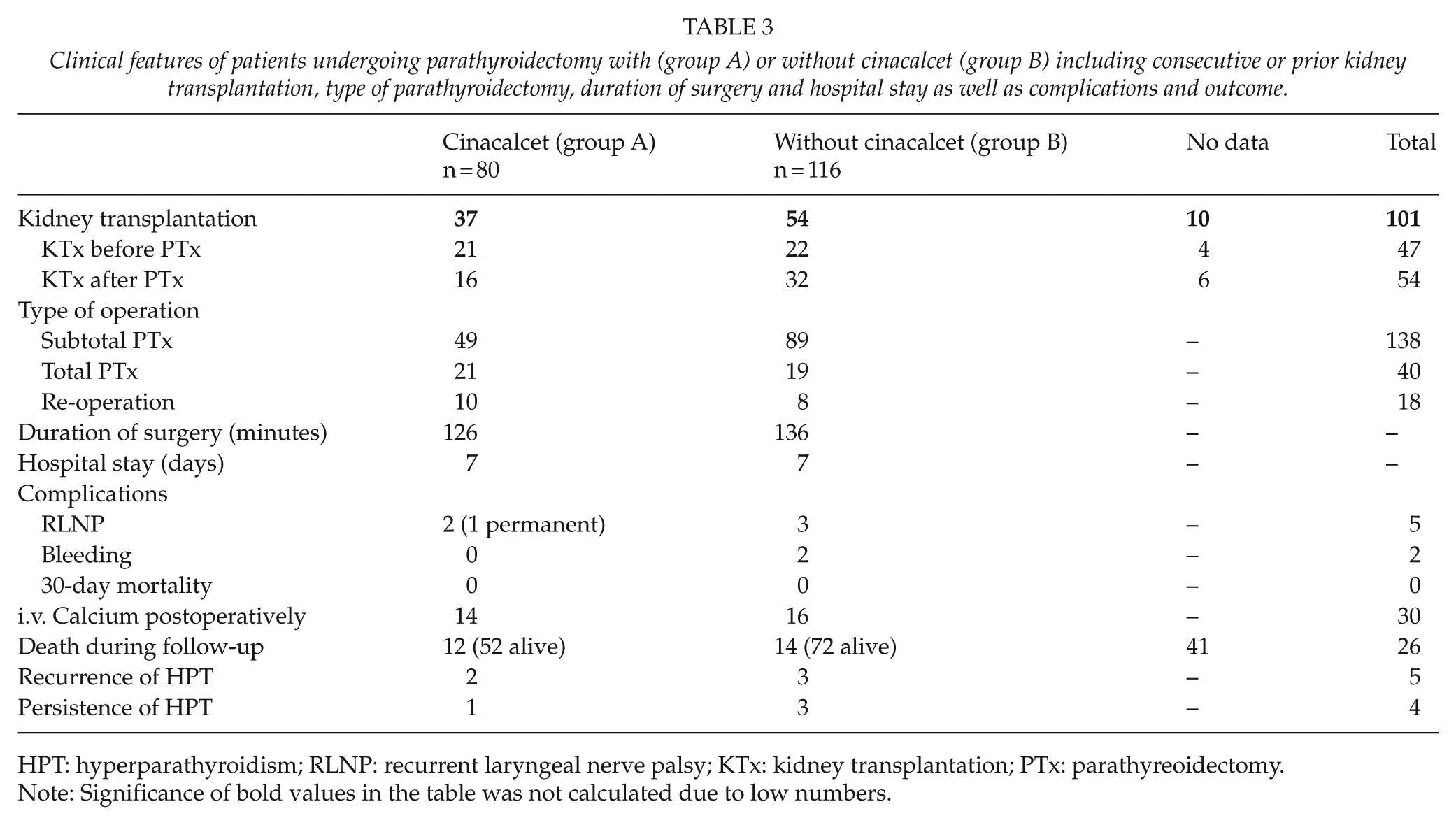
HPT: hyperparathyroidism; RLNP: recurrent laryngeal nerve palsy; KTx: kidney transplantation; PTx: parathyreoidectomy.
Note: Significance of bold values in the table was not calculated due to low numbers.
Of the 47 patients undergoing PTx after KTx for tertiary hyperparathyroidism (tHPT), 15 (32%) were on dialysis (CKD V) in group A, while 6 patients (13%) had functioning grafts (creatinine: 1.23 ± 0.41 mg/dL) at the end of follow-up. In group B, 11 patients (23%) had CKD V and 11 (23%) presented with functioning grafts (creatinine: 1.91 ± 1.02 mg/dL), while 4 patients had unknown graft function. KTx was performed at a mean of 93.5 ± 83.1 months before PTx for both groups.
In 54 patients with rHPT receiving their KTx after PTx, 42 had functioning grafts (group A: n = 13 (24%) vs group B: n = 29 (54%)), while six patients (11%) were back on dialysis (three patients in each group) and six had unknown graft function. Mean follow-up for these patients was 15.6 ± 10 months.
Six patients received more than one KTx, with the PTx taking place after loss of function of the first kidney graft. Four of these were retransplanted after PTx and presented with good graft function.
Creatinine levels in patients with tHPT (i.e. those with a functioning kidney transplant, defined as CKD I-III, n = 22) were analyzed before PTx and 1 year after operation. There were no significant differences neither in the whole group (p = 0.11), nor in the subgroups related to cinacalcet therapy (p = 0.14 with cinacalcet, p = 0.33 without cinacalcet; Fig. 1).
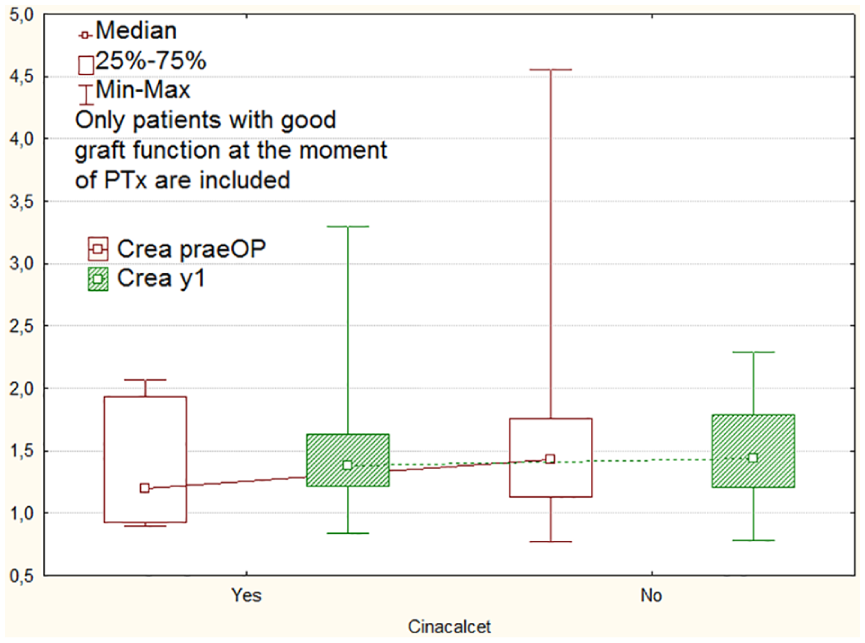
Creatinine levels (mg/dL) in patients with kidney graft and good graft function at the time of PTx and at 1-year follow-up related to cinacalcet preoperatively (yes/no). There was no significant difference in creatinine levels for both groups (p = 0.14 with cinacalcet, p = 0.33 without cinacalcet).
Perioperative Data
Operative technique
A majority of 78% of all primary operations (138/178) underwent subtotal PTx (49 vs 89 cases in both groups, respectively). In 40 patients, a total PTx without autotransplantation was done. About 18 operations were performed for recurrent HPT (Table 3). In 86 cases (48%), concomitant thyroid surgery was carried out. Indications for thyroid surgery were solitary autonomous adenoma (n = 1), diffuse autonomy (n = 2), multinodular goiter (n = 56), suspicious thyroid nodules (n = 18), or a missing parathyroid gland (n = 12). In seven patients, a differentiated thyroid carcinoma was histologically confirmed.
There was no difference in operative time between the groups (126 ± 44 min vs 136 ± 39 min with/without cinacalcet, p = 0.11, t-test). Length of in-hospital stay did not differ between the groups with a mean of 7.0 versus 7.1 days, respectively (p > 0.1, KS test). There is a trend toward shorter in-hospital stay in recent years, yet no significant difference could be seen (Table 3).
Complications
A total of five unilateral vocal cord palsies occurred (1.28% of 392 nerves at risk), two with and three without cinacalcet treatment. Four were temporary, while one patient after recurrent PTx revealed permanent unilateral vocal cord palsy. There were three reoperations for hematoma (n = 2) and infection (n = 1). One patient developed paroxysmal atrial fibrillation, but there was no perioperative mortality and all patients were discharged from hospital (Table 3).
Perioperative laboratory values
Preoperatively, calcium was significantly lower in group A (2.28 ± 0.23 mmol/L vs 2.41 ± 0.23 mmol/L, p = 0.0002, t-test), while neither postoperatively (1.89 ± 0.30 mmol/L vs 1.92 ± 0.30 mmol/L, p = 0.40, t-test) nor at discharge (2.12 ± 0.30 mmol/L vs 2.12 ± 0.27 mmol/L, p = 0.90, t-test) a statistically significant difference could be observed.
Lowest calcium levels measured postoperatively were 1.91 ± 0.30 mmol/L for all patients. The lowest postoperative serum Ca levels were not significantly different between the groups, 1.89 ± 0.30 versus 1.92 ± 0.30 (p = 0.40, t-test).
Altogether 30 patients received i.v. calcium postoperatively due to symptoms of hypocalcemia and/or calcium levels below 1.7 mmol/L (14 vs 16 patients).
Preoperative iPTH levels were comparable between the groups (755 ± 657 ng/L vs 742 ± 545 ng/L, p > 0.10, KS test). In both groups, there was a significant drop on POD 1 with no difference between the groups (50 ± 124 ng/L vs 46 ± 83 ng/L, p > 0.10, KS test).
More than 1/3 (36%) of the patients after KTx presented with good graft function at the moment of PTx. These patients showed no deterioration of creatinine levels at 1-year follow-up (1.42 mg/dL perioperatively vs 1.38 mg/dL after 1 year, p = 0.11, Wilcoxon test), and there was no difference between patients with or without cinacalcet preoperatively.
1-year surveillance data
Calcium levels 1 year after PTx were not significantly different between the groups (2.26 ± 0.25 mmol/L vs 2.24 ± 0.25 mmol/L, p = 0.67, t-test). Regarding iPTH levels, no significant difference could be detected between patients with or without cinacalcet preoperatively (26.9 vs 42.1 ng/L, p > 0.1, KS test).
Seven patients had extremely high iPTH (>2000 ng/L, n = 7) preoperatively, but none of these presented with persisting or recurrent hyperparathyroidism. Only two of these patients had been on cinacalcet before PTx. All of them had normalized iPTH at discharge. About 1 year after PTx, four of the patients presented with normal iPTH levels, for three of the patients iPTH was not available, but none of them was re-operated for HPT. Five of the patients had been transplanted before (n = 2) or after PTx (n = 3) and four presented with good graft function until the end of follow-up or death (n = 1).
After 1 year, four patients were presented with iPTH > 100 ng/L and calcium > 2.5 mmol/L, while five patients only had elevated iPTH with normal calcium. Five of these patients had recurrent HPT (two with cinacalcet, group A) and four persisting HPT (one with cinacalcet, group A).
Looking more closely at type of operation (subtotal PTx, total PTx, and recurrent PTx), there was no difference between postoperative iPTH and iPTH at 1 year. Patients with second PTx for recurrent or persisting HPT displayed a mean higher iPTH both postoperatively (45 ng/L) as well as at 1 year (92 ng/L). Patients with subtotal and total parathyroidectomy showed low levels of PTH both at discharge (11 and 8 ng/L) and at 1 year (8 and 7 ng/L). There was no significant difference between patients with or without cinacalcet.
Overall mortality and morbidity
Considering long-term follow-up, a considerable number of patients were lost to follow-up. There was no difference in mortality rate between the groups (p = 0.68, χ² test). At the end of follow-up, 12 of 77 patients in group A had died (16%), 52 (68%) were alive (no data in 16%, n = 13). In group B, 14 of 114 had died (12%), whereas 72 (63%) were alive (no data in 25%, n = 28).
The prevalence of cardiovascular events was not significantly different between the groups. Myocardial infarction occured in 13% (group A) versus 18% of patients (group B), and the need for coronary artery intervention or bypass occurred in 13% versus 22% of patients in both groups, respectively. Also the need for peripheral revascularization (9% vs 12%) and stroke was recorded with a similar prevalence (13% vs 10%) in patients after PTx with or without cinacalcet.
Discussion
This single-center study in patients undergoing parathyroidectomy for rHPT shows excellent outcome independent of pretreatment with cinacalcet. Since the introduction of cinacalcet as treatment modality for rHPT, decreasing numbers of patients are being referred to parathyroidectomy (20). Nevertheless, no reduction of all-cause or cardiovascular mortality compared with surgery has been shown for patients with ESRD (4).
A substantial part of these conservatively treated patients presents with persistently elevated iPTH, aggravation of complications, and consecutive referral to surgery (3). In our center, the number of patients referred to surgery was decreasing after the introduction of cinacalcet but is increasing again since 2012.
Timing of PTx
From our referring nephrology and transplant unit, a majority of patients on the waiting list for KTx is referred to parathyroidectomy, especially those with very high iPTH levels. This is due to the fact that only up to 56% of patients normalize PTH after KTx, mostly those with shorter waiting time on the transplant list (13).
A total of 53% of our patients underwent KTx, either before or after PTx. In those patients undergoing PTx before KTx for rHPT, a majority of 78% presents with functioning grafts during follow-up.
While it is clear that pHPT after KTx impairs long-term graft survival (17), optimal therapy still is under debate. An interesting report by Torregrosa et al. (21) has shown no decline in graft function 1 year after KTx for patients with rHPT that were treated with cinacalcet and also stable graft function for patients on cinacalcet during long-term follow-up. This was underlined by a meta-analysis by Cohen et al. (22) reporting that cinacalcet treatment for pHPT after KTx was not associated with inferior graft survival.
In our study, 55% of patients undergoing PTx after KTx were on dialysis at the end of follow-up, mostly for graft failure before PTx. But those patients undergoing PTx with functioning kidney grafts did not show deterioration of creatinine levels postoperatively or during 1-year follow-up, irrespective of cinacalcet treatment. Interestingly, six of the patients underwent second or even third KTx after PTx and presented with functioning grafts during follow-up. In contrast, 78% of patients with PTx and consecutive KTx showed functioning grafts, irrespective of treatment with cinacalcet prior to surgery.
Overall, these results favor parathyroidectomy before KTx, yet prospective trials will be necessary to evaluate optimal treatment for tHPT after KTx.
Cardiovascular Morbidity
Treatment with cinacalcet only showed limited effects on cardiovascular morbidity or mortality of patients with ESRD in the Evaluation of Cinacalcet HCl Therapy to Lower Cardiovascular Events (EVOLVE) trial (23), which evaluated patients on hemodialysis. Both a Cochrane analysis (4) and a meta-analysis by Palmer et al. (24) demonstrated that for patients with ESRD treatment with cinacalcet only reduced the risk for surgery without positively influencing all-cause or cardiovascular mortality.
A smaller trial by Trombetti et al. (25) showed that PTx, on the contrary, is associated with both reduced morbidity and mortality of these patients. In this cohort, 19 months after PTx, mortality of patients with ESRD was less than without PTx.
In our study, morbidity and mortality during follow-up were not different between patients with or without cinacalcet treatment before surgery. Both small sample size and patients lost to follow-up reduce the statistic power of our cohort.
Laboratory Values
Overall, we could see no significant differences in laboratory values perioperatively between the groups, only preoperative calcium levels were significantly lower in patients with cinacalcet. At 1 year after surgery, no difference could be detected in iPTH between the groups.
A study by Somnay et al. (26) focused on patients with tHPT and the effect of cinacalcet treatment on perioperative findings and outcome. Although they presented a small patient cohort with cinacalcet treatment, they interestingly had higher iPTH levels preoperatively in this cohort, while cure rate was not influenced by cinacalcet pretreatment. Compared with our analysis, patients without cinacalcet treatment had much lower preoperative iPTH levels in this study, possibly reflecting lower thresholds for PTx after KTx. Only seven patients with tHPT in our study received cinacalcet treatment with similar preoperative iPTH levels, as patients with rHPT.
A very recent study by Baker et al. (27) examined influence of cinacalcet on outcome in patients with rHPT and found greater iPTH levels in the untreated group before surgery with similar outcome.
Only the study of Wirowski et al. (28) demonstrated comparable laboratory findings in patients operated for rHPT. In their analysis, pre- and postoperative iPTH levels were equal in patients with or without cinacalcet and calcium was significantly lower in cinacalcet patients. This is in line with our findings, and they also concluded that cinacalcet did not alter perioperative course.
Looking more closely on recurrent, or persisting rHPT, only four patients were diagnosed during follow-up with both elevated iPTH and hypercalcemia, while five were normocalcemic with elevated iPTH. For this analysis, relatively strict values were applied as cut-off values for recurrent HPT. When increasing the cut-off values for iPTH, only two patients of the cohort would have been identified with recurrence of HPT. There was no significant difference in iPTH levels at 1 year between subtotal or total parathyroidectomy and also no difference related to cinacalcet treatment preoperatively.
So overall, surgical technique with mostly subtotal PTx in our cohort showed good results and low rates of recurrent or persisting rHPT, independent of preoperative treatment with cinacalcet. Several groups have shown that subtotal PTx or total PTx with autotransplantation of parathyroid tissue represent a good compromise between effective treatment of rHPT while avoiding unnecessary complications of hypocalcemia, adynamic bone disease, or reoperations (29).
Complications
Overall, the rate of surgical complications was low in both groups and no difference could be calculated for patients with or without cinacalcet treatment. There was no perioperative mortality in our group. Duration of surgery and hospital stay did not differ between the groups, and hospital-stay was reduced during the study period but similar in both groups.
Conclusion
Our study underlines the fact that PTx represents a safe and successful treatment option for patients with rHPT, even after KTx and irrespective of co-medication with cinacalcet. In view of the morbidity and mortality of long-term dialysis, patients will need individualized decisions taken by interdisciplinary transplant and tumor boards.
Surgery should at least be offered to patients not responding to cinacalcet treatment and especially to those planned for KTx. This not only should reduce complications of pHPT but also improve patient morbidity in view of a successful KTx.
Footnotes
Acknowledgements
Authors thank Prof. Dr. med. Peter E Goretzki for discussion and critical review of the manuscript.
Author Contributions
M.T.M., T.S., N.R. contributed to the study conception and design, analysis, and interpretation of data and writing of the manuscript. N.M.O., E.T., C.B. participated in the acquisition and analysis of data. P.R., N.R., J.P. contributed to the analysis and interpretation of data, the study conception, and critical review of the manuscript.
Compliance with Ethical Standards
This is a retrospective non-interventional study. All procedures performed were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval was given by the local ethics committee (EA4/086/19)
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
