Abstract
End-stage renal disease is often complicated by the occurrence of secondary and eventually tertiary hyperparathyroidism, characterized by increased parathormone, calcium, and phosphate concentrations. Related symptoms include pruritus and osteodynia, concentration difficulties, and feelings of depression may be present. In the long-term, end-stage renal disease patients with hyperparathyroidism have an increased risk of all-cause and cardiovascular mortality. Among treatment options are vitamin D supplements, phosphate binders, calcimimetics, and surgical parathyroidectomy. Determining the optimal treatment for the individual patient is challenging for nephrologists and endocrine surgeons. This review resumes the pathogenesis of hyperparathyroidism, clinical presentation, required diagnostic work-up, and discusses indications for the available treatment options for patients with secondary and tertiary hyperparathyroidism.
Introduction
Hyperparathyroidism (HPT) is a condition in which the parathyroid glands produce too much parathormone (PTH). End-stage renal disease (ESRD) is the most common cause of secondary HPT (1, 2). In contrast to primary HPT, the hormonal disturbance in secondary and tertiary HPT is caused by an external stimulus.
The worldwide prevalence of ESRD is 0.1% (3). The incidence of patients with ESRD requiring renal replacement therapy (RRT) has remained fairly stable over the last decade (4, 5). More than 80% of the patients with a glomerular filtration rate (GFR) rate below 20 mL/min/1.73 m2, present with PTH levels above the upper limit of normal (PTH > 6.8 pmol/L) (6). Reported prevalence of HPT (defined as PTH levels >32 pmol/L) in ESRD patients is described between 12% and 54% (7, 8).
Pathophysiology
In early stages of kidney failure, phosphate retention is the driving force for the overstimulation of the parathyroid glands. Hyperphosphatemia occurs due to excretion failure in the proximal tubule because of a decreased renal mass, and failure of the kidneys to synthesize 1,25-dihydroxyvitamin D (1,25(OH)2D), the physiologically active form of vitamin D (9, 10). Continuous exposure to hyperphosphatemia, in addition to a decreased serum concentration of calcium and increased levels of fibroblast growth factor (FGF)-23 due to an impaired kidney function, causes the parathyroid glands to produce and excrete more PTH (10). The chronic exposure to these electrolyte imbalances leads to polyclonal, and eventually monoclonal hyperplastic parathyroid tissue (11).
PTH has numerous target organs, including bones, kidneys, brain, and the intestinal tract (Fig. 1). By binding to its receptors on osteoclasts, PTH stimulates bone resorption, which in turn leads to the release of calcium into the bloodstream (12). In the proximal renal tubule, PTH inhibits the reabsorption of phosphate, and at the same time, stimulates the reabsorption of calcium in the distal tubule, leading to an increase in serum calcium and a decrease in serum phosphate levels. PTH has no direct effect on the intestinal tract. However, via stimulation of 1,25(OH)2D production it indirectly increases intestinal calcium and phosphorus absorption.
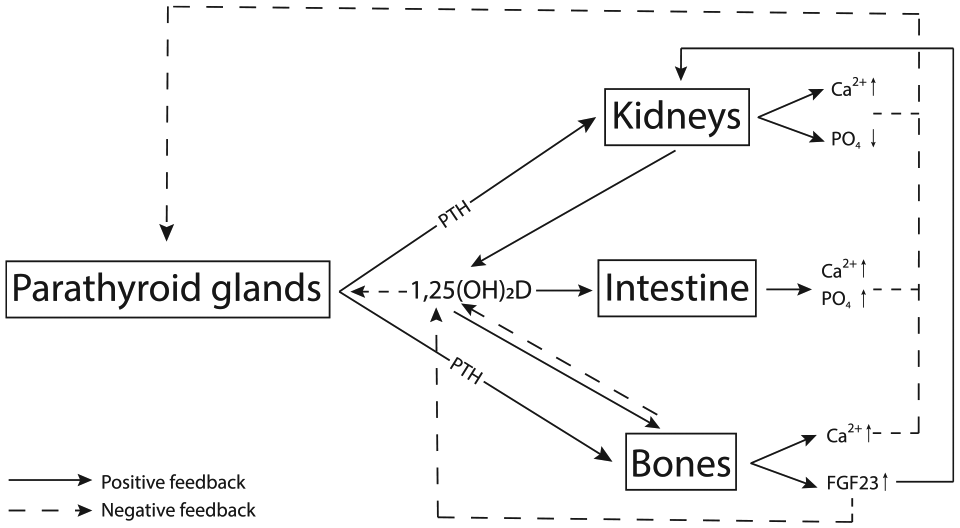
Parathyroid–kidney–bone–intestine feedback loop.
Ultimately, the process of the chronical exposure to the aforementioned electrolyte disturbance leads to autonomous PTH synthetization and secretion, regardless of serum calcium concentrations. The parathyroid–bone–kidney feedback loop becomes irreversibly distorted. This prompts to severe hypercalcemia, hyperphosphatemia, and HPT, also known as tertiary HPT. By definition, tertiary HPT is preceded by a prolonged period of secondary HPT and can be distinguished from secondary HPT by the presence of hypercalcemia, in contrast to hypocalcemia in secondary HPT. With a successful kidney transplantation (KTx), metabolic and biochemical disturbances will be eliminated, thereby removing the external stimulus triggering the parathyroid glands, leading to spontaneous resolution of HPT in almost 60% of the patients undergoing KTx (13). The exact role and timing of KTx in the treatment of secondary and tertiary HPT will be discussed below.
Clinical Manifestations
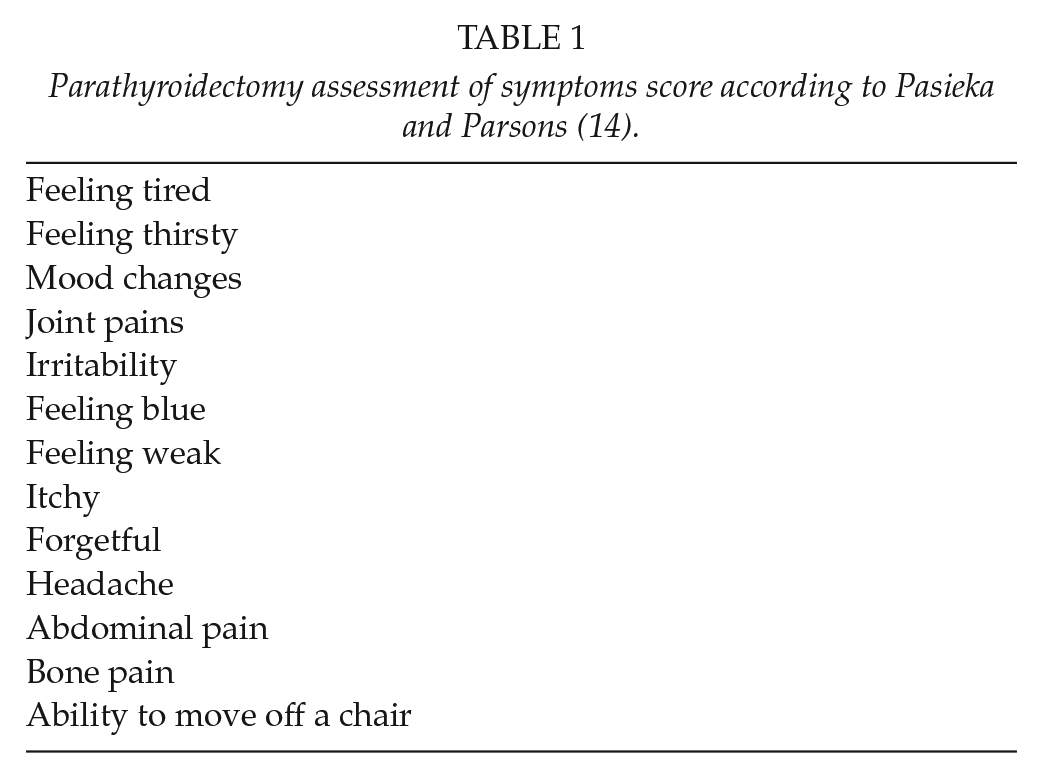
The clinical presentation of elevated calcium levels ranges from no symptoms at all, to severe osteodynia, pruritus, pathologic fractures, and muscle weakness, but also more vague symptoms such as memory loss, concentration difficulties, or feeling “blue”. The parathyroidectomy assessment of symptoms (PAS) score summarizes all relevant clinical features accompanying HPT, by which the severity of symptoms can be quantified (Table 1) (14). A high PAS score has been associated with a decreased quality of life in patients with secondary and tertiary HPT (15). In the long-term, HPT leads to progressive bone loss (also known as renal osteopathy) and an increased risk of cardiovascular disease and mortality (16, 17).
Parathyroidectomy assessment of symptoms score according to Pasieka and Parsons (14).
As this patient population is regularly seen in the outpatient clinic of the nephrologists, PTH, calcium, phosphate, and vitamin D levels are frequently monitored and the diagnosis is made by the treating nephrologist. Calcium binds to albumin in the serum, which makes it physiologically inactive. Therefore, serum calcium levels should always be corrected for the concentration of albumin.
Therapeutic Pharmacological Options
The Kidney Disease—Improving Global Outcomes (KDIGO) Chronic Kidney Disease—Mineral Bone Disease (CKD-MBD) guideline is considered leading in recommendations for the treatment of secondary and tertiary HPT (18). Worldwide, however, no guidelines or consensus have been established between nephrologists and endocrine surgeons. One of the first steps in the prevention and treatment of secondary HPT is to supplement active vitamin D. In 90% of pre-dialysis patients, vitamin D deficiency is already present, despite (excessive) sun exposure (19). Vitamin D supplements will obviate and treat a-dynamic bone diseases and prevent fractures, particularly in postmenopausal women (20, 21). However, studies with high-quality scientific evidence evaluating the effect of vitamin D supplementation in the ESRD population on mortality, cardiovascular events, quality of life, but also fracture rate, are lacking. The impact of vitamin D administration in the ESRD population comprises a decrease in serum concentration of PTH, a reduction of bone turnover, and reduced proteinuria (22–25). A sum dosage of 50,000–100,000 IU per month seems adequate to normalize serum 1,25(OH)2D levels in most patients, with a maximum dosage of 4000 IU per day (26, 27).
The aforementioned KDIGO CKD-MBD guideline recommends lowering elevated serum phosphate concentrations toward the normal range (18). Hyperphosphatemia has been associated with increased mortality; however, there is no scientific evidence that lowering serum phosphate levels decreases the risk of all-cause mortality or cardiovascular events, or improves quality of life (17, 28). Hyperphosphatemia can be controlled through dietary restrictions (max. 800–1000 mg per day), but most patients find it difficult to adhere to a low phosphorus-diet. Therefore, almost all ESRD patients receive phosphate binders. Phosphate binders can be initiated as soon as phosphate levels start to increase, since one of the earliest manifestations of renal excretion failure and secondary HPT is phosphate retention (18).
Unfortunately, in almost all patients, vitamin D supplements combined with phosphate binders do not sufficiently restore the calcium–phosphate homeostasis. Therefore, the KDIGO guideline recommends to add calcimimetics to the first-line therapy (18). Calcimimetics, which are allosteric modulators of the calcium sensing receptor (CaSR), mimic the binding of calcium and activate the CaSR. Furthermore, they increase the sensitivity of the CaSR to extracellular calcium by the conformational change of the receptor molecule. Altogether, PTH production by the parathyroid glands is reduced through these mechanisms (29). Currently, calcimimetics can be administered orally (cinacalcet) and intravenously (etelcalcetide). Cinacalcet was approved for the treatment of secondary HPT in 2004. Initial studies showed that treatment with cinacalcet is associated with a decrease of serum PTH, calcium, and phosphate levels, although moderately (30). A meta-analysis including over ten thousand ESRD patients found that cinacalcet significantly reduces PTH concentrations (weighted mean difference −22 pmol/L), calcium levels (weighted mean difference −0.7 mmol/L), and phosphate levels (weighted mean difference of −0.4 mmol/L) (31). Circulating FGF-23 levels also decrease significantly with cinacalcet treatment compared to vitamin D therapy alone (median change −40% (−63% to 16%), p < 0.001) (32). However, compared to placebo, the use of cinacalcet gives an eightfold increased risk of developing hypocalcaemia and other side effects such as vomiting and diarrhea (31). Compliance is a significant problem in the ESRD population due to polypharmacy, side effects, and economic factors (33, 34). Brunaud et al. (35) found in an observational cohort study that cinacalcet did not affect mean PTH levels compared to patients not using calcimimetics. Contradicting results have been reported on the effect of cinacalcet on bone resorption and formation markers, mineral bone disease, and fracture risk (31). On top of this, the Evaluation of Cinacalcet Hydrochloride Therapy to Lower Cardiovascular Events (EVOLVE) trial did not find that cinacalcet significantly reduces the risk on all-cause and cardiovascular mortality (31, 36). Interestingly, despite these findings, the KDIGO CKD-MBD guideline still recommends the administration of calcimimetics and/or calcitriol or vitamin D analogues for ESRD patients requiring PTH-lowering therapy (18). Garside et al. (37) calculated the additional costs per quality-adjusted life year (QALY) when cinacalcet is added to a therapy with vitamin D and phosphate binders. They estimated an incremental cost-effectiveness ratio of £61,890 per QALY. It should be noted that cinacalcet is only registered for hemodialysis patients and not for kidney transplant patients with persistent HPT, although frequently off-label prescribed nowadays (38).
In 2016, etelcalcetide was introduced for the treatment of secondary HPT. The peptide etelcalcetide directly agonizes the CaSR, thereby reducing circulating levels of PTH and subsequently calcium (39). By means of a triweekly bolus injection of the drug, a sufficient plasma level of the drug is attained, enabling the administration during dialysis. This makes drug adherence easier for the ESRD patient. Etelcalcetide has been compared to both placebo as to its precursor cinacalcet (40, 41). Almost half of the patients who received etelcalcetide showed a decrease of PTH levels to ⩽32 pmol/L. Etelcalcetide was expected to give less side effects as vomiting and diarrhea since it is administrated intravenously and thus bypasses the gastrointestinal tract. Unfortunately, no significant difference in side effects was found when comparing etelcalcetide with cinacalcet, probably due to PTH-receptors in the intestinal tract (40). A 30% decrease of FGF-23 levels was seen in 80% of the patients using etelcalcetide after 27 weeks (41). Studies investigating the long-term effects of etelcalcetide on mineral bone disease, overall survival, and cardiovascular events are not available yet. The costs of etelcalcetide are significantly higher than its precursor cinacalcet (42). Therefore, etelcalcetide should be used as a second in line treatment option, in patients unresponsive to oral calcimimetics or for those patients who fail to adhere properly. Etelcalcetide, like cinacalcet, is only registered for the treatment of dialysis patients, not for those who received a kidney transplant.
Surgical Management
Traditionally, patients with tertiary HPT were managed surgically by means of parathyroidectomy (PTx) when vitamin D analogues and/or phosphate binders were insufficient to control HPT. Twenty years ago, the European Dialysis and Transplant Association (EDTA) reported the performance of PTx in up to 40% of the patients receiving dialysis longer than 15 years (43). Currently, after the introduction of calcimimetics such as cinacalcet, PTx rates have declined significantly and the majority of ESRD related HPT patients is managed medically (44, 45).
Indications for Parathyroidectomy
PTx is only indicated in case of tertiary HPT, which is defined as hypercalcemia and elevated PTH levels. The KDIGO CKD-MBD guideline, a nephrologist guideline, states that PTx is indicated in patients with ESRD and severe HPT who fail to respond to pharmacological treatment (18). The definition of failure to respond is not specified in this guideline. The European Society of Endocrine Surgeons (ESES) established a consensus report regarding the surgical management of HPT in ESRD patients, stating that PTx is indicated when medical treatment fails to correct metabolic parameters (PTH > 85 pmol/L, hypercalcemia, and hyperphosphatemia) (46).
In addition, PTx is valuable in those with severe symptoms such as pruritus, bone pain, and calciphylaxis. However, a clear consensus between nephrologists and surgeons regarding the exact indications for PTx in patients on dialysis does not exist. The role of PTx in patients with no clinical manifestations is unclear. Tentori et al. (47) showed an association between increased PTH levels (>64 pmol/L) and the increased risk of all-cause mortality, cardiovascular mortality, and all-cause and cardiovascular hospitalization. Therefore, it could be argued that patients with elevated PTH levels, although asymptomatic, might benefit from PTx in the long-term.
In conclusion, the selection of dialysis patients with tertiary HPT who will benefit from PTx should be individualized. Different factors should be taken into consideration such as, biochemical values over time, comorbidity, age, chance of prompt kidney transplantation, clinical presentation, and patients’ preference. The goal of surgery for ESRD-related HPT should focus on long-term balance of biochemical values, improvement of symptoms, an increased survival, and a decreased cardiovascular and fracture risk (46).
As calcimimetics are not registered for patients with persistent HPT after KTx, PTx is the only adequate treatment option in these patients.
Surgical Anatomy
To safely perform a successful PTx, the surgeon should be well aware of the anatomy of the parathyroid glands and its surrounding tissue (Fig. 2). The (usually) four parathyroid glands are in close anatomical relation with the thyroid gland, despite having no functional relationship whatsoever. Embryologically, the parathyroid glands descend from the third and the fourth pharyngeal pouches. The inferior two parathyroid glands originate from the third pharyngeal pouch together with the thymus and need to descend over a larger course, which explains the wider anatomical localization compared to the two superior parathyroid glands deriving from the fourth pharyngeal pouch (48). Healthy parathyroid glands are leaf-like shaped, brown to purple colored, and measure less than 10 mm in all dimensions. The parathyroid gland can be recognized and distinguished from thyroid tissue by the fatty tissue surrounding the little glands. In general, all parathyroid glands are vascularized by the left and right inferior thyroid arteries (49). Preoperatively, the surgeon should be aware of the 13% chance on supernumerary parathyroid glands (more than four) and should know the anatomical relationship with the recurrent laryngeal nerve (RLN): the inferior parathyroid glands lie ventral (superficial) to the RLN, whereas the superior glands are located dorsally (deep) with respect to the RLN (48, 50). In 1%–3% of humans less than four parathyroid glands are found (51).
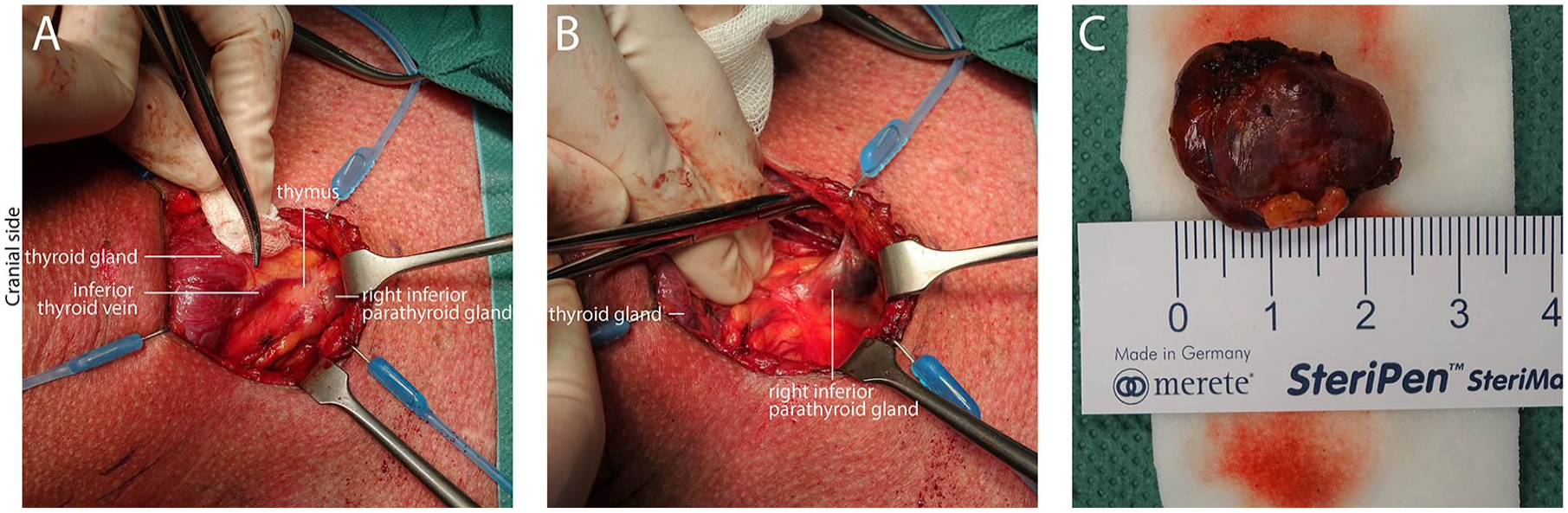
Intraoperative images of parathyroid gland of a patient with tertiary hyperparathyroidism: (A) intrathymic right inferior parathyroid gland, (B) right inferior parathyroid gland dissected from thymus, and (C) parathyroid gland ex vivo.
Preoperative Parathyroid Imaging
In secondary or tertiary HPT imaging modalities, such as Technetium (99mTc)-sestamibi scan, ultrasonography (US) or computed tomography (CT), aiming to detect enlarged parathyroid glands is not routinely required. The diagnosis of secondary or tertiary HPT is established by biochemical tests only, not by imaging.
A meta-analysis by Caldarella et al. (52) showed that the sensitivity of 99mTc-sestamibi scan for detecting hyperplastic glands in patients with secondary HPT and diffuse or nodular hyperplasia was 58%. They concluded that 99mTc-sestamibi should not be considered as a first-line diagnostic imaging method in the pre-surgical detection of hyperplastic parathyroid glands. Sensitivity of US for detection of enlarged parathyroid glands is 46%–81% in patients with secondary HPT (53–55). The combination of US with 99mTc-sestamibi SPECT/CT had a higher sensitivity than US or 99mTc-sestamibi SPECT/CT alone (55). The role of preoperative imaging in patients that will undergo a four-gland exploration due to four-gland hyperplasia is questionable, since it may not have any intraoperative surgical consequences. In the detection of potential ectopic glands or in re-operative setting, preoperative imaging might be useful.
Ptx Strategies
In secondary and tertiary HPT, the driving force of the hyperplastic proliferation of parathyroid tissue lies externally. This explains why usually not just one, but all four parathyroid glands are frequently enlarged and overactive in HPT related to ESRD. Consequently, when PTx is indicated, an ESRD patient should undergo a bilateral neck exploration. Different surgical approaches have been described and widely investigated: total PTx (tPTx), total PTx with autotransplantation (tPTx with AT), and subtotal PTx (sPTx) (46).
In tPTx, all four glands are resected, resulting in a hypoparathyroid state. This technique has been frequently performed in the past, when kidney transplantation was rare and the risk of recurrent disease was high. However, transient and persistent hypocalcaemia are commonly seen in these patients, leading to hungry bone syndrome, which is also potentially unbeneficial for a future kidney graft (56). Therefore, this procedure is not preferred anymore.
To avoid permanent hypoparathyroidism, one-half of resected parathyroid gland can be reimplanted (tPTx with AT). Frequently used locations of AT include transplanting parathyroid tissue in the sternocleidomastoid muscle (SCM) in the neck, or in the brachioradial muscle of the forearm. When using the SCM, no additional incision is required. On the other hand, when using the arm, in case of persistent or recurrent disease, a reoperation can be performed under local anesthesia of the arm only. It is advised to mark the transplanted tissue with a non-absorbable silk suture and/or a hemoclip (57). There is no definitive general guideline recommending to which anatomical location. This is usually decided on the basis of the surgeons’ preference. A commonly used technique of parathyroid tissue graft preparation is fragmentation of the gland by scissors or scalpel in a saline solution, followed by direct reimplantation by injecting the parathyroid fragments using a syringe with a wide needle. When performing sPTx, half of the most-normal appearing gland (30–50 mg) is left in situ, leaving vascularization intact. Finally, cryopreservation of parathyroid tissue has been described, creating the possibility to auto-transplant parathyroid tissue in a later stage, in case persisted hypoparathyroidism has developed (58, 59). In clinical practice, this technique is not widely used.
Surgical Procedure
Usually, general anesthesia is required for a formal parathyroid gland exploration. To gain optimal access to the neck, the patient’s neck should be extended placing a head ring under the patient’s head. A transverse Kocher incision, named after Dr. Theodor Kocher, one of the leading thyroid surgeons of the 19th century, is made. Afterwards, platysma is divided and the linea alba colli between the strap muscles is opened to expose the thyroid gland and parathyroid glands. To mobilize the thyroid, the middle thyroid vein can be divided. Standard practice is to first search for the parathyroid glands in the suspected superior and inferior regions, posteriorly to the thyroid gland by mobilizing and anteromedially retracting the thyroid gland and by using blunt dissection. If not found in these locations, further exploration is required. Additional thymectomy may be performed, since ectopic parathyroid glands are widely found in the thymus (60).
Surgical Adjuncts
As the RLN is at risk, it should ideally always be identified and carefully handled during parathyroid surgery. Surgical adjuncts such as RLN monitoring may be used to assist the surgeon in preservation of the RLN (61). The clinical benefit of RLN monitoring during PTx for secondary or tertiary HPT is not well established. Snyder et al. found that the use of RLN monitoring for thyroid and parathyroid tissue did lead to a decline in injury rates of the RLN, but other studies could not replicate this finding (61–63). RLN monitoring might especially be of assistance during difficult cases such as re-operative setting. Intraoperative (IO) PTH monitoring is possible due to the short half-life of PTH and can be of value when the surgeon is not sure whether enough parathyroid tissue is resected (64). Several applications are available to measure PTH intraoperatively, in the lab or even in the operating theater (65).
Outcomes of Ptx
tPTx without AT leaves the patient with complete hypoparathyroidism which is not desirable considering the negative consequences of chronic hypoparathyroidism and hypocalcemia.
tPTx with AT is very effective in decreasing PTH, calcium, and phosphate levels (66, 67). Schneider et al. (66) showed that tPTx with AT decreased calcium levels from 2.76 ± 0.05 to 1.91 ± 0.02 postoperatively. In addition, the majority of patients experienced a significant improvement of symptoms and quality of life after sPTx and tPTx with AT: Cheng et al. (15) found that the physical component score of the SF-36 quality-of-life questionnaire improved (40 ± 17 to 59 ± 15, p < 0.001) as well as the mental component score (48 ± 17 to 64 ± 13, p < 0.001).
Similar results are found evaluating sPTx performed in patients with ESRD-related HPT. In 606 ESRD patients with HPT undergoing sPTx, calcium levels of 2.70 ± 0.07 decreased to 2.09 ± 0.05 mmol/L. PTH levels already dropped with 85%–95% during surgery with sPTx, as well as with tPTX with AT (68). In sPTx, it is important that the remnant parathyroid tissue is still well vascularized after surgery. This appears sometimes difficult; in approximately, 2% of patients undergoing sPTx, hypoparathyroidism occurs (46). To determine the quality of vascularization of the remnant parathyroid tissue, near-infrared fluorescence imaging using indocyanine green (ICG) could be of extra value but is not yet universally applied (69).
Observational studies have shown that PTx reduces mortality, the risk of cardiovascular events, and fracture risk significantly (70–74).
Chen et al. (75) recently published a systematic review with a meta-analysis investigating the outcomes of sPTx and tPTx with AT. The authors evaluated a total of 1589 patients included in 13 studies comparing both surgical strategies. They found no significant difference in the rate of persistent or recurrent disease (OR 1.31; 95% confidence interval (CI): 0.02–1.56, p = 0.90) between sPTx and tPTx with AT. In addition, no significant difference in the reoperation rate was found (OR: 1.55; 95% CI: 0.62–3.86; p = 0.35). The authors did not compare postoperative permanent hypoparathyroidism rates, as none of the studies reported long-term hypocalcemia requiring calcium supplementation.
Reoperation is considered profoundly more complex due to scar tissue, which increases the risk of iatrogenic damage to the RLN from <1% in primary surgery up to 7% in the re-operative setting (57, 76).
Role of Ktx and Timing of Ptx
The ultimate treatment for patients with ESRD related HPT is a kidney transplantation (KTx), as this removes the driving force of HPT. This explains the reduction in parathyroid functional weight found in patients with HPT who underwent KTx (77). Unfortunately, not all patients have a spontaneous regression of HPT after KTx. A study investigating 1690 patients with HPT undergoing KTx, showed that one year after KTx, 70% of patients have persistent tertiary HPT, which decreases to 43% of the patients after 2 years (13). These patients will still require definite treatment for their HPT. The optimal timing of PTx with respect to KTx is still under debate, as a persistent deterioration of the calcium–phosphate homeostasis has been found as an independent risk factor for renal graft loss, and all-cause mortality in transplant recipients (68, 78).
Whether and when a HPT patient with a functional kidney transplant should undergo PTx should be carefully evaluated for every individual patient, taking biochemical values, severity of symptoms, likelihood of future KTx, and patient’s preference into account. We advocate a guideline established by nephrologists as well as endocrine surgeons. In the future, a multidisciplinary consensus should assist in clinical decision-making, leading to improved care for this complex, multimorbid patient population.
Footnotes
Declaration Of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
