Abstract
Background and aims:
There is no consensus regarding the routine placement of intra-abdominal drains after pancreaticoduodenectomy. We aim to determine the effects of intraperitoneal drain placement during pancreaticoduodenectomy on 30-day postoperative morbidity and mortality.
Methods:
Patients who underwent pancreaticoduodenectomy for pancreatic tumors were identified from the 2014–2015 American College of Surgeons—National Surgical Quality Improvement Program Database. Univariate and multivariate analyses adjusting for known prognostic variables were performed. A subgroup analysis was performed based on the risk for development of postoperative pancreatic leak determined by the pancreatic duct caliber, parenchymal texture, and body mass index.
Results:
A total of 6858 patients with pancreatic tumors who underwent pancreaticoduodenectomy were identified in the 2014–2015 American College of Surgeons—National Surgical Quality Improvement Program Database dataset. In all, 87.4% of patients had intraperitoneal drains placed. A 30-day mortality rate was higher in the no-drain group (2.9% vs. 1.7%, P = 0.003). Patients in the drain group had a higher incidence of overall morbidity (49.5% vs. 41.2%, P = 0.0008), delayed gastric emptying (18.1% vs. 13.7%, P = 0.004), pancreatic fistulae (19.4% vs. 9.9%, P ⩽ 0.0001), and prolonged length of hospital stay over 10 days (43.7% vs. 34.9%, P < 0.0001). Subgroup analysis based on risk categories revealed a higher 30-day mortality rate in the no-drain group among patients with high-risk features (3.1% vs. 1.6%, P = 0.02). Delayed gastric emptying and pancreatic fistula development remained significantly higher in the drain group only in the high-risk category. Prolonged length of hospital stay and composite morbidity remained higher in the drain group regardless of the risk category.
Conclusion:
To our knowledge, this is the largest study to date that aims at clarifying the pros and cons of the intraperitoneal drain placement during pancreaticoduodenectomy for pancreatic tumors. We showed a higher 30-day mortality rate if drain insertion was omitted during pancreaticoduodenectomy in patients with softer pancreatic textures, smaller pancreatic duct caliber, and body mass index over 25. Postoperative 30-day morbidity rate was higher if a drain was inserted regardless of the risk category. Further randomized controlled trials with prospective evaluation of stratification factors for fistula risk are needed to establish a clear recommendation.
Introduction
Pancreaticoduodenectomy (PD) is the surgical procedure of choice for the resection of periampullary and pancreatic tumors (1). PD carries a list of potential complications, of which pancreatic leaks and fistulae are the most serious and can lead to major morbidity and mortality (2, 3, 4, 5). The routine placement of drains in the vicinity of the pancreatic anastomoses after PD is a common practice to allow early detection and drainage of potential anastomotic/pancreatic leaks (6). However, the reliability and safety of using such drains has been recently questioned, making this issue a subject of controversy. Some authors still advocate for the use of drains after PD in all cases, while others advise against their placement (7, 8, 9, 10).
Currently, no consensus exists for routine post-PD drain placement. The utility of drains after PD has been explored in several studies with conflicting results. Some authors demonstrated that the use of drains reduces mortality (11), whereas others showed that drains were associated with an increased postoperative complication rate, mainly pancreatic fistulae formation among other problems (10). There are other studies that failed to demonstrate any effect of the placement of post-PD drains on mortality or morbidity (9).
The aim of this study was to investigate the impact of placing an intraperitoneal drain during PD on the postoperative morbidity and mortality. The study was carried out using the American College of Surgeons’ National Surgical Quality Improvement Program (ACS-NSQIP) Database.
Methods
Data Source
The ACS-NSQIP is a multi-institutional prospectively collected database. It captures demographic characteristics, comorbidities, and preoperative, intraoperative, and 30-day postoperative variables from patients in more than 600 urban and rural hospitals across the United States. Patient consent or Institutional Review Board (IRB) approval was not required for this study due to the anonymous nature of the data collected primarily for quality improvement purposes.
Study Population
We undertook a secondary analysis of the prospectively collected ACS-NSQIP database for all patients who underwent PD (CPT codes: 48150, 48151, 48152, 48153, 48154) between 2014 and 2015. Patients with missing information on intraperitoneal drains were excluded from the analysis. In addition, patients were categorized based on risk score, where patients who had pancreatic duct size smaller than 3 mm and/or soft gland texture and/or body mass index greater than 25 kg/m2 were considered as high-risk patients.
Variables
The following preoperative variables were included in the analysis: age, race, gender, history of diabetes mellitus, current smoking, hypertension, history of chronic obstructive pulmonary disease, body mass index and weight loss, American Society of Anesthesiologists (ASA) classification, preoperative blood transfusion, preoperative total bilirubin, white blood count, hematocrit, platelet count and international normalized ratio (INR) of prothrombin time (PT) values, pancreatic duct size, and preoperative chemotherapy and/or radiotherapy.
Operative variables including operative time, wound classification, pancreatic gland texture, specimen histopathology, intraperitoneal drain location (or its absence), pancreatic reconstruction, and gastrojejunostomy construction were analyzed. Outcome variables included 30-day mortality; length of hospital stay; bleeding; delayed gastric emptying; pancreatic fistula; respiratory, cardiac, and urinary complications; sepsis; thromboembolism; related readmission; and related 30-day reoperation.
Statistical Analysis
Characteristics of participants were described and compared across the two groups (drain and no-drain groups). Categorical variables were described using frequency and percentage, whereas continuous variables were described using mean and standard deviation. The chi-square and Fisher exact test were used to compare categorical variables between the two exposure groups. The independent t test was used to compare means of continuous variables. The association between the main exposure and 30-day outcomes was assessed using simple and multiple logistic regressions, from which unadjusted and adjusted odds ratios (ORs) with corresponding 95% confidence interval (CI) were estimated. Confounders were entered into multivariate analyses based on statistical and/or clinical significance, and included age, ASA Class, surgical wound class, total operative time, preoperative blood transfusion, bleeding disorder, disseminated cancer, body mass index, open wound (with or without infection), steroid use for chronic condition, systemic sepsis in the previous 48 h, platelet count, INR, serum albumin, functional health status prior to surgery, ascites, bilirubin, SGOT, hematocrit, preoperative obstructive jaundice, chemotherapy within 90 days, pancreatic duct size, pancreatic gland texture, pancreatic reconstruction, gastrojejunostomy or duodenojejunostomy, and vascular resection. Statistical analyses were performed using Statistical Analysis Software version 9.4 (SAS 9.4). Statistical significance was set at a two-sided P value of <0.05.
Results
Between 2014 and 2015, 6858 patients who underwent PD were included in the ACS-NSQIP database. Of the 6858, 5997 (87.4%) patients had a drain inserted during surgery (drain group). A drain was not placed in 861 (12.6%) patients (no-drain group) (see Table 1).
NSQIP data of demographics and comorbidities of patients, preoperative workup, intraoperative findings, and histology, in both arms drain verses no drain.
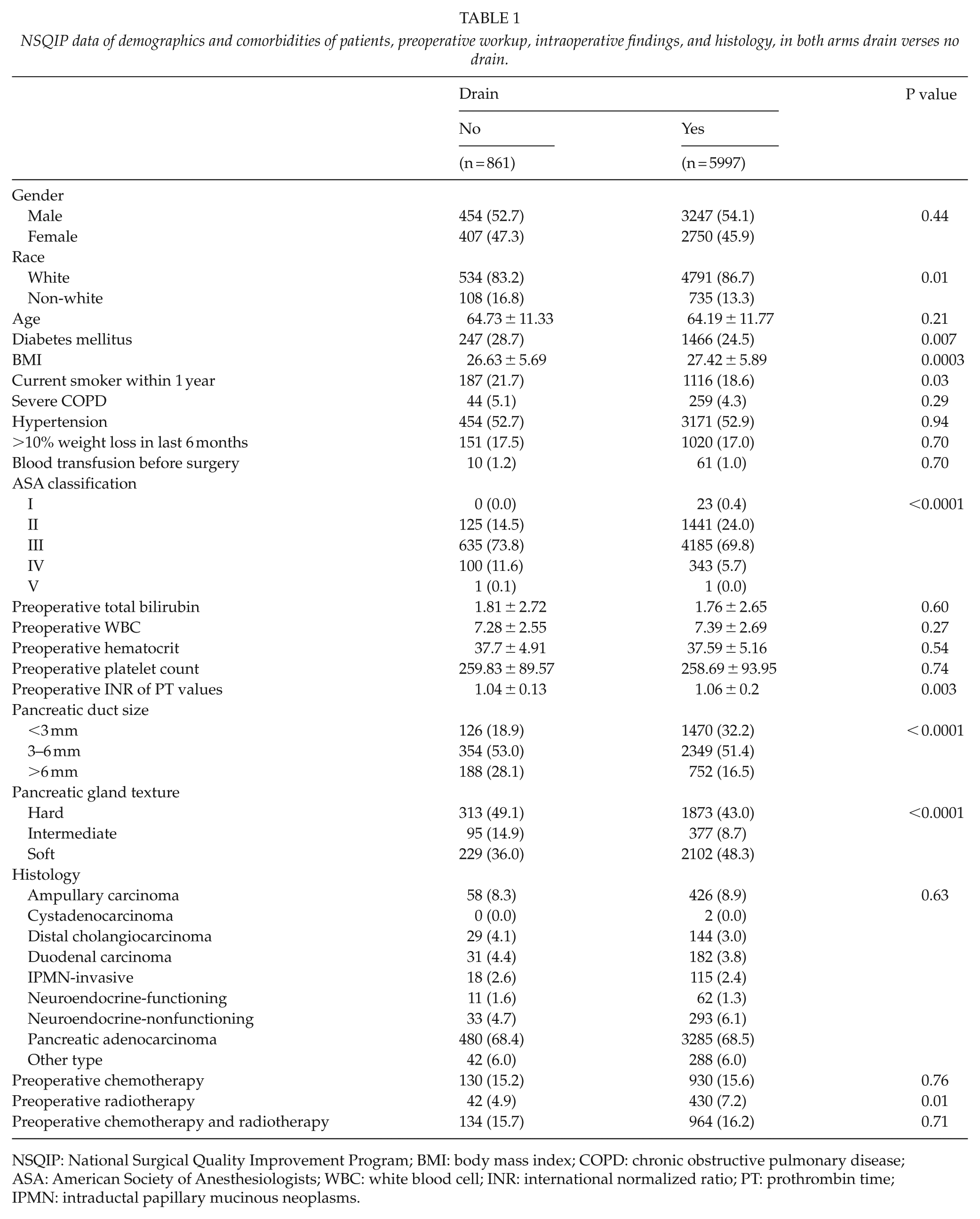
NSQIP: National Surgical Quality Improvement Program; BMI: body mass index; COPD: chronic obstructive pulmonary disease; ASA: American Society of Anesthesiologists; WBC: white blood cell; INR: international normalized ratio; PT: prothrombin time; IPMN: intraductal papillary mucinous neoplasms.
Demographic characteristics including gender, age, and smoking status were comparable between the two groups and outlined in Table 1. Patients were predominately of the white race in both groups; however, the ratio of white:non-white ethnicity was higher in the drain group (P = 0.01). There were no statistically significant differences in the associated comorbid conditions between the two groups. There was also no statistically significant difference in the rate of blood transfusion before surgery between the two groups. More patients in the no-drain group were diabetic as compared to the drain group, 247 (28.7%) versus 1466 (24.5%), P = 0.007. The mean body mass index was significantly higher in the drain group (27.42 + 5.89 vs. 26.63 + 5.69 kg/m2, P = 0.0003). Patients in the no-drain group had significantly higher ASA scores as compared to patients in the drain group; the difference was statistically significant (P < 0.0001). There were no statistically significant differences between the two groups in regard to preoperative total bilirubin, white blood count, hematocrit, platelet count, and INR (Table 1).
More patients in the drain group received preoperative radiotherapy, 430 (7.2%) versus 42 (4.9%), P = 0.01; however, there were no statistically significant differences in the rate of preoperative chemotherapy and combined chemo-radiotherapy between the two groups: 130 (15.2%) versus 930 (15.6%), P = 0.76, and 134 (15.7%) versus 964 (16.2%), respectively, P = 0.71 (Table 1).
Patients in the drain group had significantly smaller pancreatic duct diameters as compared to patients in the no-drain group. In all, 1470 (32.2%), 2349 (51.4%), and 752 (16.5%) in the drain group versus 126 (18.9%), 354 (53.0%), and 188 (28.1%) in the no-drain group had a pancreatic duct diameter of <3 mm, 3–6 mm, and >6 mm, respectively, P < 0.0001 (Table 1).
Regarding the operative findings, patients in the drain group had softer pancreatic gland texture; the difference was statistically significant. In all, 1873 (43.0%), 377 (8.7%), and 2102 (48.3%) in the drain group versus 313 (49.1%), 95 (14.9%), and 229 (36.0%) in the no-drain group were found to have hard, intermediate, and soft pancreas during surgery, respectively, P < 0.0001. Total operative time was longer in the drain group (374.62 + 127.55 min vs. 338.83 + 134.91 min), P < 0.0001 (Table 1).
Most of the patients in the drain group had a drain placed in the vicinity of the pancreatic and biliary anastomoses (68.3%), followed by the pancreatic anastomosis (16.7%), then the biliary anastomosis (2.9%), and followed by the pancreatic parenchyma (2.4%). The location of the drain was not specified in 9.7% of the patients (Table 2).
NSQIP data of surgical techniques, wound classification, and total operative time and wound classification in both arms drain and no drain.
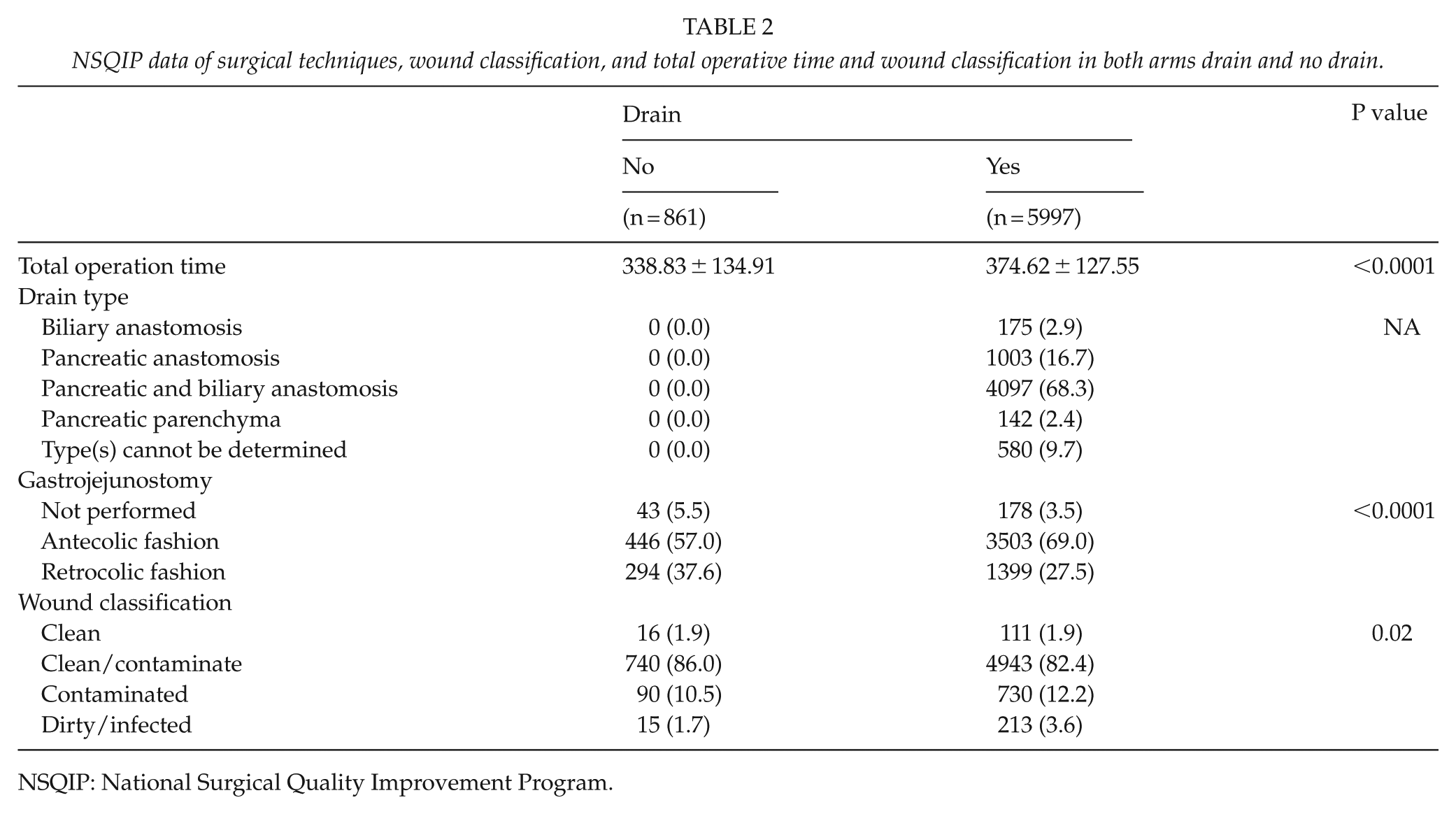
NSQIP: National Surgical Quality Improvement Program.
Histopathologic diagnoses of the resected specimens varied between ampullary carcinoma, cystadenocarcinoma, distal cholangiocarcinoma, duodenal carcinoma, invasive intraductal papillary mucinous neoplasm, functional neuroendocrine tumor, non-functional neuroendocrine tumor, and pancreatic adenocarcinoma. There was no significant difference in the histopathologic diagnoses between the two groups (Table 2).
There was a statistically significant difference in the 30-day mortality rate between the two groups in favor of the drain group, 25 (2.9%) versus 104 (1.7%) (OR = 0.59; 95% CI = 0.38–0.92), P = 0.002; the difference remained significant after risk adjustment (OR = 0.51; 95% CI = 0.32–0.80), P = 0.003 (Table 3).
NSQIP data of all the postoperative complications, composite morbidity, and 30-day mortality in both arms drain and no drain.
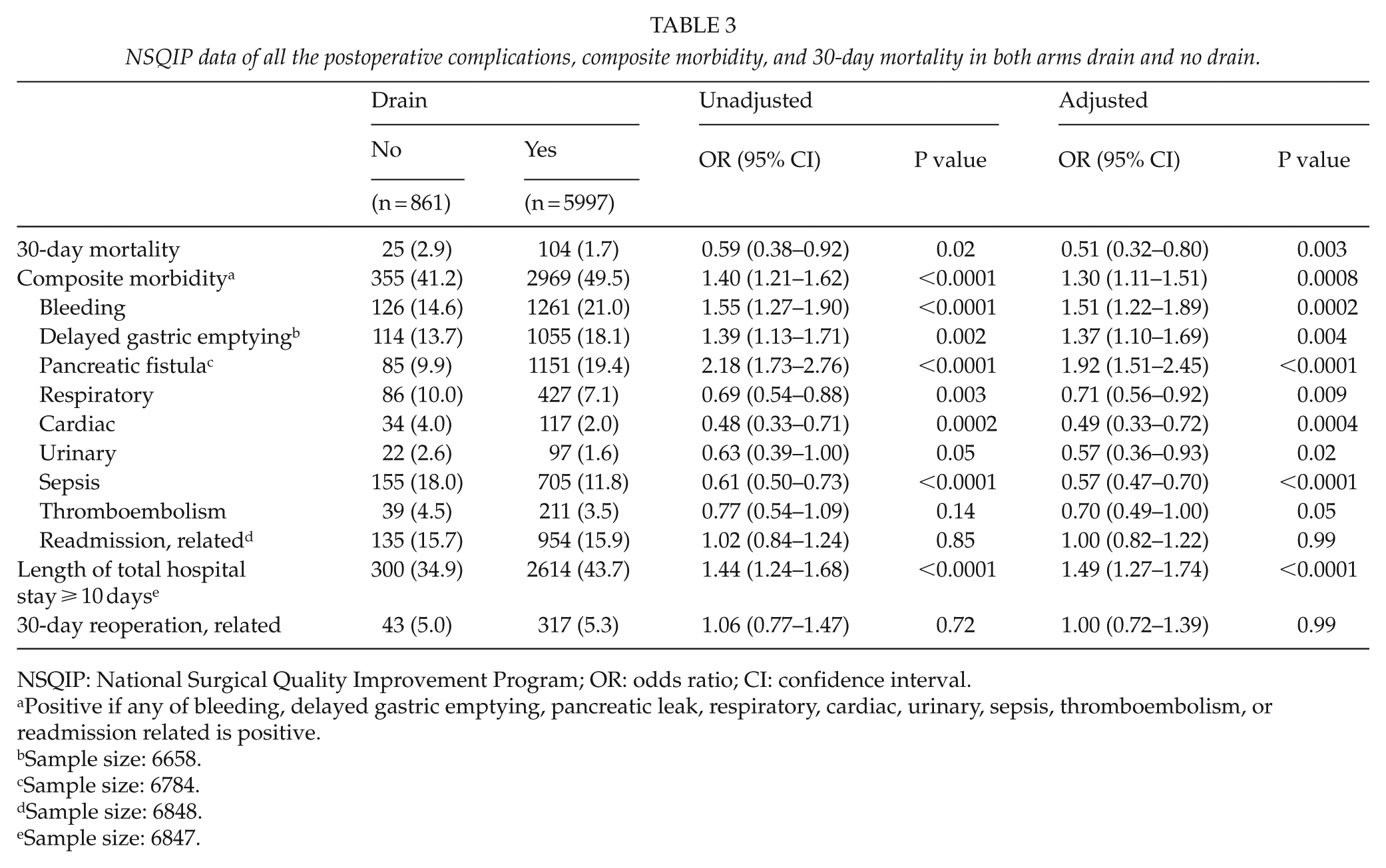
NSQIP: National Surgical Quality Improvement Program; OR: odds ratio; CI: confidence interval.
Positive if any of bleeding, delayed gastric emptying, pancreatic leak, respiratory, cardiac, urinary, sepsis, thromboembolism, or readmission related is positive.
Sample size: 6658.
Sample size: 6784.
Sample size: 6848.
Sample size: 6847.
Overall morbidity rate was higher in the drain group, 2969 (49.5%) versus 355 (41.2%) (OR = 1.40; 95% CI = 1.21–1.62), P < 0.0001. The difference remained statistically significant after adjustment for pertinent risk factors (OR = 1.30; 95% CI = 1.11–1.51), P = 0.0008 (Table 3).
There were statistically significant differences in the rate of blood loss, postoperative delayed gastric emptying, and pancreatic fistula development in favor of the no-drain group: the statistically significant after risk adjustment (OR = 1.51; 95% CI = 1.22–1.89), P = 0.0002, and difference remained (OR = 1.37; 95% CI = 1.10–1.69), P = 0.004, and (OR = 1.92; 95% CI = 1.51–2.45), P < 0.0001, respectively (Table 3).
There was a statistically significant difference in the rate of postoperative cardiac complications, urinary complications, respiratory complications, and sepsis in favor of the drain group: 117 (2.0%) versus 34 (4.0%) (OR = 0.48; 95% CI = 0.33–0.71), P = 0.0002, for cardiac complications; 97 (1.6%) versus 22 (2.6%) (OR = 0.63; 95% CI = 0.39–1.00), P = 0.05, for urinary complications; 427 (7.1%) versus 86 (10.0%) (OR = 0.69; 95% CI = 0.54–0.88), P = 0.003, for respiratory complications; and 705 (11.8%) versus 155 (18.0%) (OR = 0.61; 95% CI = 0.50–0.73), P < 0.0001, for septic complications, respectively. The differences remained significant after risk adjustment except for respiratory complications (Table 3).
Length of total hospital stay over 10 days was more common among the patients in the drain group: 2614 (43.7%) versus 300 (34.9%) (OR = 1.44; 95% CI = 1.24–1.68), P < 0.0001; difference remained statistically significant after adjustment: (OR = 1.49; 95% CI = 1.27–1.74), P < 0.0001 (Table 3).
The difference in postoperative thromboembolic complications did not show a statistical significance between the two groups in the unadjusted analysis; however, the difference approached borderline statistical significance after risk adjustment in favor of the drain group: 211 (3.5%) versus 39 (4.5%) (OR = 0.77; 95% CI = 0.54–1.09), P = 0.14, on the unadjusted analysis and (OR = 0.70; 95% CI = 0.49–1.00), P = 0.05, after adjustment (Table 3).
Statistical significance was not reached in the comparison between the two groups in regard to the rates of readmission: 135 (15.7%) versus 954 (15.9%) (OR = 1.02; 95% CI = 0.84–1.24), P = 0.85, on the unadjusted analysis and (OR = 1.00; 95% CI = 0.82–1.22), P = 0.99, after adjustment. Similarly, there was no statistically significant difference in the related 30-day reoperation rate between the two groups: 43 (5.0%) versus 317 (5.3%) (OR = 1.06; 95% CI = 0.77–1.47), P = 0.72, before adjustment and (OR = 1.00; 95% CI = 0.72–1.39), P = 0.99, after adjustment (Table 3).
We also categorized the patients based on the known risk factors to develop postoperative pancreatic fistulae. Those who had pancreatic duct caliber smaller than 3 mm and/or soft gland texture and/or body mass index greater than 25 were classified as high-risk patients. In all, 826 patients were low risk with mean age 65.78 ± 12.13 years and 3957 patients were high risk with mean age 63.38 ± 11.51 years (Table 4). The postoperative 30-day mortality and overall morbidity in low and high-risk groups are outlined in Table 5.
NSQIP data of demographics and comorbidities of patients in high-risk versus low-risk categories.
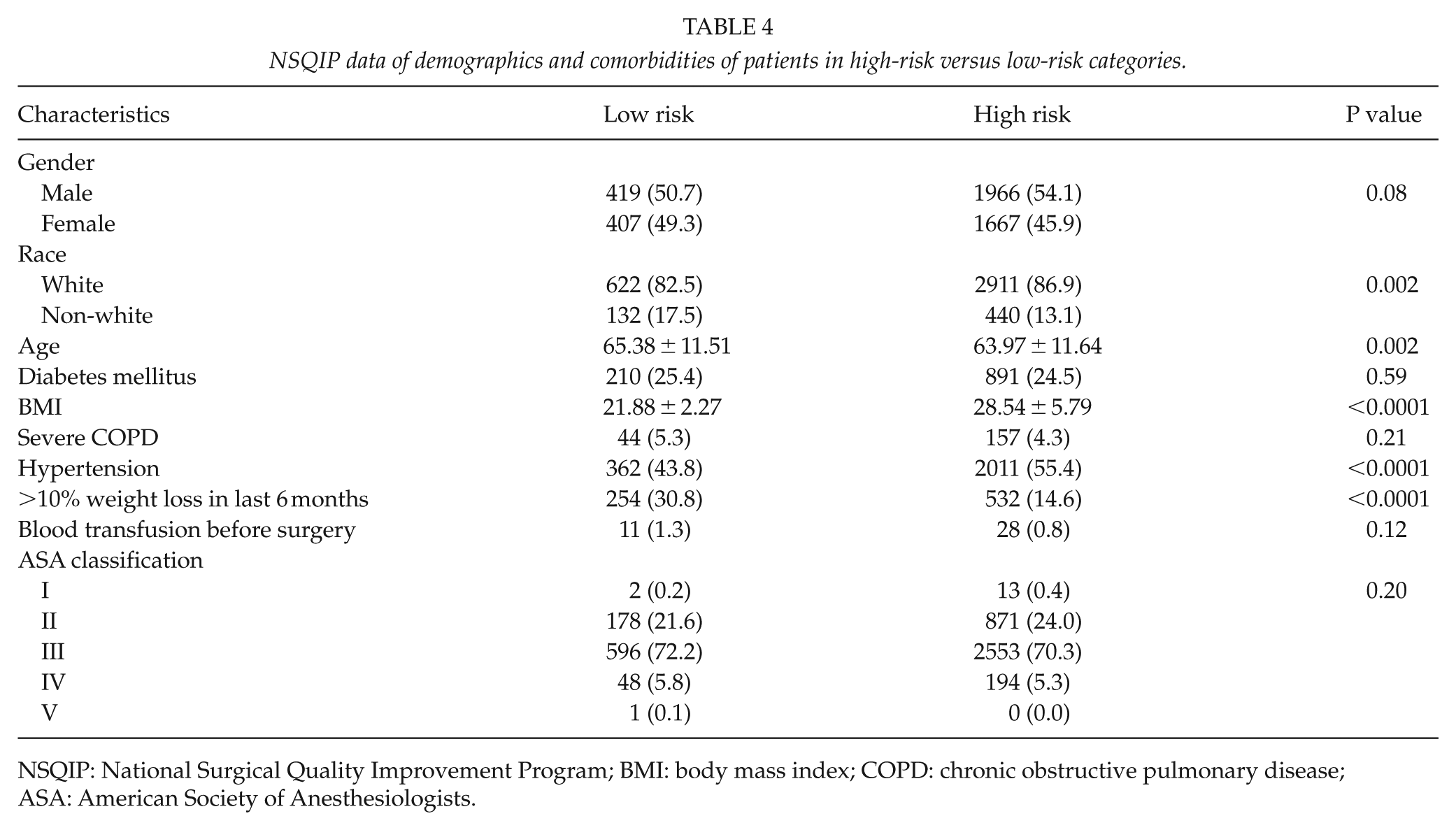
NSQIP: National Surgical Quality Improvement Program; BMI: body mass index; COPD: chronic obstructive pulmonary disease; ASA: American Society of Anesthesiologists.
NSQIP data of all the postoperative complications, composite morbidity, and 30-day mortality in drain versus no-drain groups both in high- and low-risk categories.
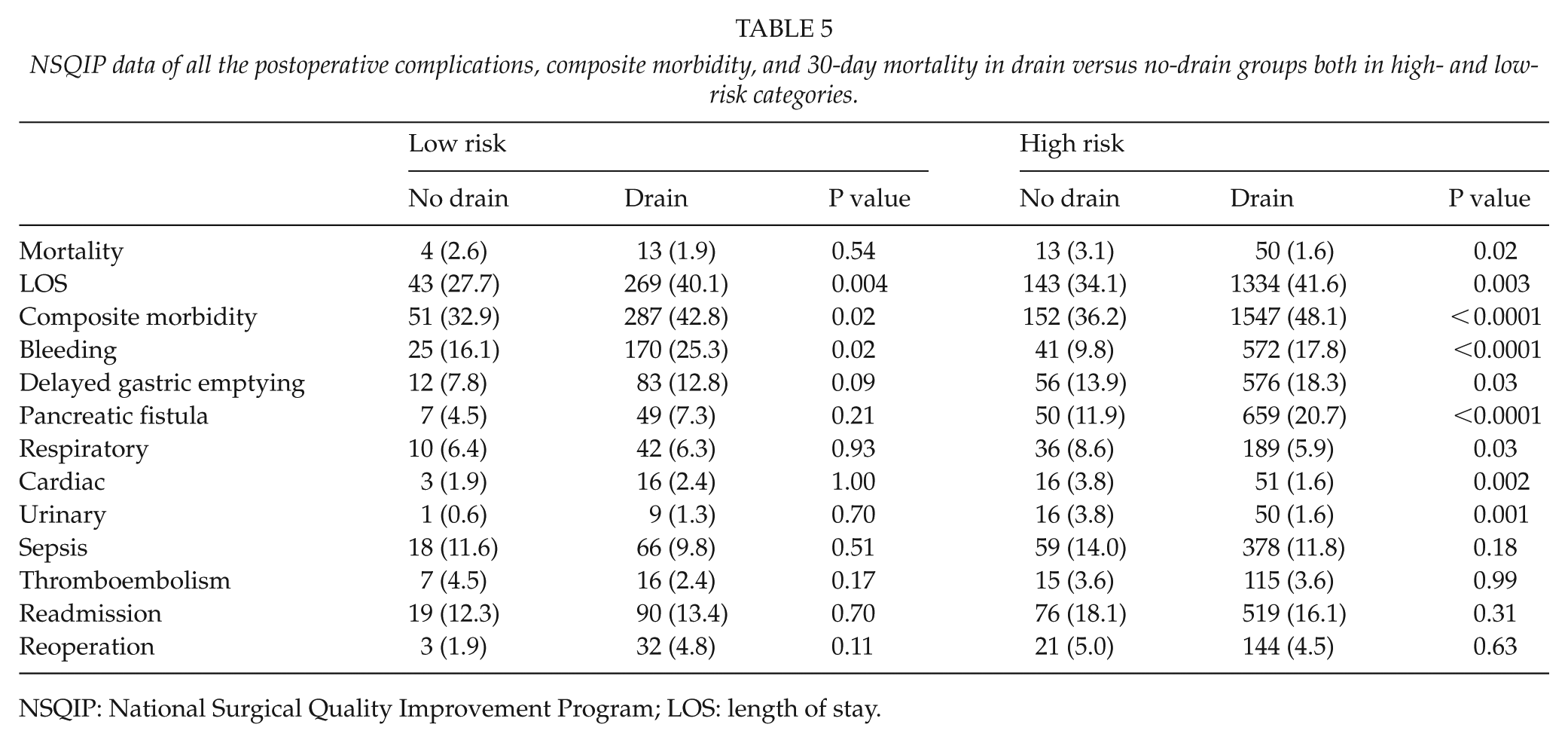
NSQIP: National Surgical Quality Improvement Program; LOS: length of stay.
Discussion
Due to the relatively common occurrence of potentially serious post-PD complications such as pancreatic fistulae, biliary and enteric leaks, and intra-abdominal collections, the placement of intraperitoneal drains during the time of pancreatic resections has been widely practiced for many decades by the majority of surgeons to allow for an early detection of pancreatic fistulae or biliary leaks. The placement of drain can facilitate the evacuation of any postoperative fluid collections and allows for controlled drainage of fistulae if they occur.
There has been a recent trend toward a more selective use of drains during surgical procedures in general; this trend has evolved as a result of data demonstrating a lack of benefit, and even potential harmful consequences of routine drain placement. Adverse consequences of drain placement include acting as a nidus for infection or exerting excessive pressure in close proximity to, and hence endangering, the integrity of the anastomotic site (12, 13).
Given a lack of definitive data specifically in the post-PD setting, pancreatic surgeons have been slow in embracing the new concept of avoiding the routine use of drains after pancreatic resections. This may partially be due to the fact that PD for periampullary tumors is extremely complex, and technically demanding surgical procedure, with a high-risk profile. Data from this study, for instance, show that almost half of all post-PD patients had at least one perioperative complication or morbidity.
Of the 6858 patients who underwent a periampullary tumor resection, 87.4% had an intraperitoneal drain placed during surgery. In this study, we were able to demonstrate a higher overall postoperative morbidity rate, and a lower 30-day mortality rate after PD if an intraperitoneal drain was placed during PD.
The overall postoperative pancreatic fistula rate was 9.9% in the no-drain group and 19.4% in the drain group (P ⩽ 0.0001). Soft pancreas and a small pancreatic duct size are well known risk factors for the development of post-PD pancreatic fistulae (14). Body mass index over 25 kg/m2 as well as intraoperative blood loss in excess of 10,000 mL have also been reported as significant risk factors for the development of pancreatic fistulae (14, 15).
In this cohort of patients, the distribution of the risk factors for development of pancreatic fistulae was different between the two groups. Patients in the drain group had significantly smaller pancreatic duct sizes (32.2% vs. 18.9% had pancreatic duct diameter <3 mm, P < 0.0001), softer pancreas (48.3% vs. 36% had soft pancreas, P < 0.0001), and higher body mass index as compared to the no-drain group. We could not compare the severity of intraoperative blood loss between the two groups because this information is not available in the NSQIP database.
We categorized the patients into two risk groups based on the pancreatic duct caliber, the texture of the pancreatic parenchyma, and body mass index. We then repeated the statistical analysis on each of these two categories separately. As expected, patients in the high-risk category had significantly higher rates of postoperative pancreatic fistulae development whether or not a drain was placed during surgery (Table 5). The overall 30-day morbidity, postoperative bleeding, and length of hospital stay in excess of 10 days remained significantly higher in the drain group in both risk categories. On the contrary, delayed gastric emptying and postoperative pancreatic fistula development were significantly higher in the drain group only in the high-risk category. The mortality rate remained higher in the no-drain group in both risk categories; however, the difference was statistically significant only in the high risk (P = 0.02) and not in the low-risk category (P = 0.54). Hence, only patients with high-risk features may potentially benefit from drain placement during PD.
In their prospective validation study, Ven Fong et al. (16) concluded that the risk of postoperative pancreatic fistula development is less than 1% if the drain-fluid amylase (DFA) level was under 600 I/U on postoperative day 1. Our results showed a significantly higher drain amylase level on postoperative day 1 in the patients who later developed pancreatic fistulae in both risk categories (Table 6). In the low-risk category, 1.7% of patients with drain amylase level under 600 I/U developed pancreatic fistula, whereas in the risk group, 6% of patients with drain amylase level under 600 I/U developed a pancreatic fistula (Table 7). Hence, we agree with Ven Fong et al.’s statement that on postoperative day 1, patients with drain amylase levels under 600 I/U are very unlikely to develop a pancreatic fistula and the drain may be safely removed; however, this applies only to the patients who have low-risk features (pancreatic duct caliber larger than 3 mm, non-soft pancreatic parenchyma, and body mass index under 25 kg/m2).
NSQIP data, drain amylase level on postoperative day 1 and postoperative days 2–30 versus postoperative pancreatic fistula development.
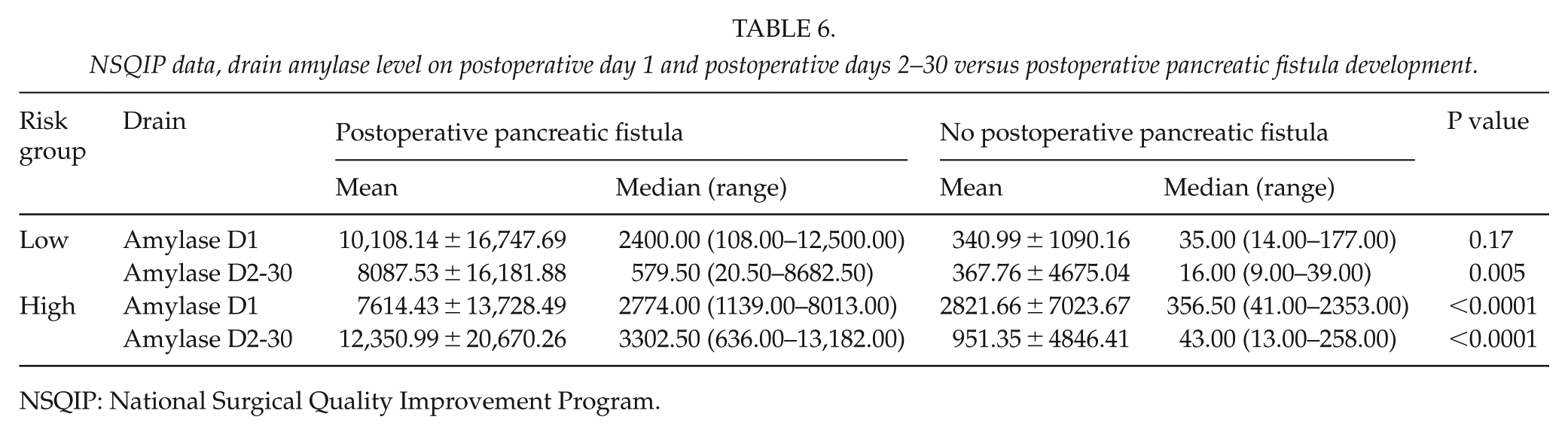
NSQIP: National Surgical Quality Improvement Program.
NSQIP data, incidence of postoperative pancreatic fistula in patients with drain amylase level under 600 I/U in high- and low-risk patients.
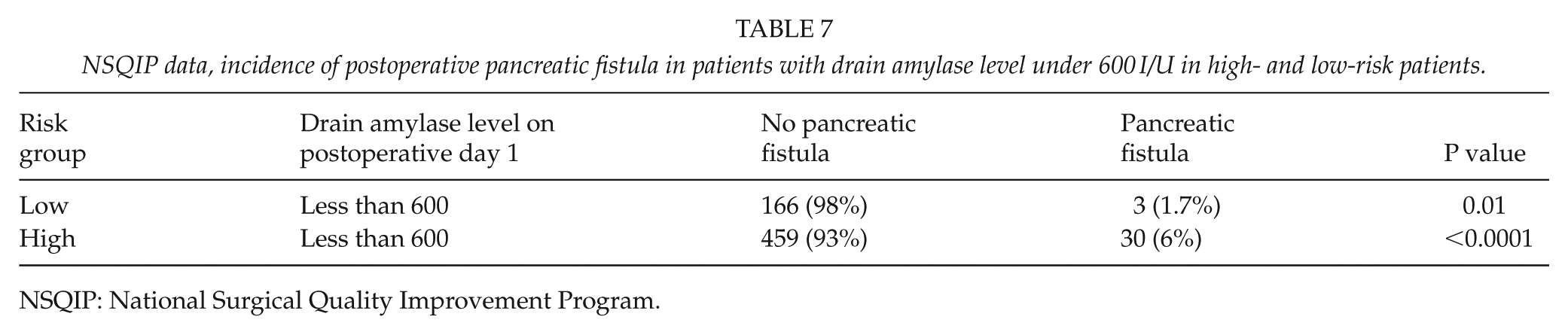
NSQIP: National Surgical Quality Improvement Program.
Over the last few years, there has been a great increase in the number of studies trying to establish the safety and the effects of drain placement during pancreatic surgery on postoperative morbidity and mortality. In 2001, Conlon et al. performed a randomized prospective clinic trial; their cohort included 179 pancreatic resections, with 88 patients randomized to have drains placed during surgery. This trial failed to show a reduction in mortality or postoperative complications with the addition of intraperitoneal closed suction drains after pancreatic resection; the authors concluded that drainage should not be considered mandatory after pancreatic resections (17). In line with the above study is a recent randomized controlled trial from Germany evaluating drain placement after pancreatic resections, where 395 patients were randomized into two groups: drain group (n = 202) and a no-drain group (n = 193). The authors in this study demonstrated noninferiority of the no-drain group in terms of re-intervention rate. Mortality rate and overall perioperative morbidity were comparable between the two groups; however, clinically significant pancreatic fistulae, delayed gastric emptying, hemorrhage, and intra-abdominal abscesses all occurred significantly more frequently in the drain group (10).
On the contrary, in 2014, Van Buren et al. performed a randomized multicenter prospective trial in which they randomized 137 patients to either PD with an intraperitoneal drain (n = 68) or PD without a drain (n = 69). The authors of this study showed that omission of drains results in an increased frequency and severity of postoperative complications. This trial was terminated early by the Data Safety Monitoring Board because of a four-fold increase in the mortality rate in the no-drain group (3% vs. 12%); hence the authors strongly advised against omitting drains after PD (18). Fistula risk score (FRS) is a prospectively validated 10-point clinical risk score that predicts the risk of clinically relevant postoperative pancreatic fistulae using four risk factors: gland texture, pathology, pancreatic duct diameter, and intraoperative blood loss (19).
The value of drains as a fistula mitigation strategy for PD was assessed in a randomized prospective multi-institutional study. In this trial, the authors from nine different institutions performed 137 PDs over a 15-month period; 68 patients were randomized to intraperitoneal drainage and 69 to no drainage. They demonstrated that drains diminish the rate and severity of clinically relevant postoperative pancreatic fistulae in patients with moderate/high risk (based on the FRS), whereas the use of drains increases the rate of CR-ROPF in patients with negligible/low risk. They also demonstrated that 90-day mortality in patients with moderate/high risk who developed clinically relevant postoperative pancreatic fistulae dropped from 42.9% to 22.2% when a drain was used; hence, they concluded that selective use of drains may improve outcomes in patients after PD (20). In addition to the selective drain placement, many authors have centered their research studies on the value of early drain removal after PD aiming to reduce the potential harm caused by the placement of drain, without giving up the sense of security provided by the drain as an “early warning system” (20, 21). In their recent multicenter prospective trial, McMillan et al. suggested a drain management protocol in which drains are not placed in negligible/low-risk patients, and DFA is measured on the first day post-PD for moderate/high-risk patients, then drains are removed on the third postoperative day in patients with DFA < 5000 U/L. In this study, the authors applied their protocol to 260 consecutive PDs performed at two institutions over 17 months. None of their negligible/low-risk patients developed a clinically relevant postoperative pancreatic fistula. Upon comparison with a historical cohort (557 patients over 3 years), the protocol-cohort demonstrated lower rates of severe complication, any complication, reoperation, as well as reduced hospital stay. The authors of this study concluded that drains can be safely omitted for one-quarter of PDs (22). In a meta-analysis by Nitsche et al., the authors included two randomized trails and six retrospective studies comparing the outcomes of post-PD patients with and without intraperitoneal drain placement. In this study, a subgroup analysis for post-PD patients was performed (370 patients with a drain vs. 565 patients without a drain). The authors showed that drain placement made no difference in terms of postoperative morbidity, pancreatic fistulae formation, need for interventional radiology procedures, or length of hospital stay. However, the omission of drains was associated with a slight increase in mortality and intra-abdominal abscesses formation. As such, the authors concluded that the routine omission of drains cannot be advocated (8). Similarly, Dou et al. and Van Der Wilt et al. also performed meta-analyses to address the same query; they both concluded that intraperitoneal drains are still needed after PD (23, 24). On the contrary, a Cochrane review was performed in 2016 involving 711 patients randomized to a drainage group (n = 358) and a no drainage group (n = 353) after pancreatic surgery. This review failed to demonstrate adequate evidence to establish the effect of drains on 30-day mortality, 90-day mortality, intra-abdominal infections, wound infection, morbidity, length of hospital stay, and reoperation rate (25, 26).
Limitations
Our study has several limitations inherent to the fact that these data are not randomized. The higher overall morbidity observed in the drain group might be explained by the higher risk profile of the patients in this group rather than by the presence of the drain per se; this is despite the fact that we have adjusted for the known risk factors for the development of pancreatic fistulae. NSQIP database captures potential complications that may occur during the first 30 days after the procedure; hence, we might have missed some of the patients who have developed a complication, namely, pancreatic fistulae, later than 1 month from the procedure. Despite these limitations, the large sample size of patients on whom data have been prospectively collected for the purposes of monitoring surgical quality gives us important insights in to this issue which has not been resolved following insufficiently powered randomized studies.
Conclusion
To our knowledge, this is the largest study to date investigating the advantages and disadvantages of intraperitoneal drain placement after PD. We were able to demonstrate higher overall postoperative 30-day morbidity and prolonged length of hospital stay in patients who had an intraperitoneal drain placed during PD regardless of the risk group. On the contrary, omission of drain placement was associated with a statistically significant higher mortality rate in patients with high-risk features. Hence, patients in the high-risk category may potentially benefit from drain placement during PD. At this point in time, we are not able to advice for or against drain placement post-PD. Further appropriately powered randomized controlled trials with prospective evaluation of stratification factors for fistula risk are needed to establish a clear recommendation.
Footnotes
Author Contributions
FW proposed the study. FW, ZA, HS, MD, and KM performed the research and wrote the first draft. TH, JR, MA, and ZA collected and analyzed the data. All authors contributed to the design and interpretation of the study and to further drafts. FW is the guarantor.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
