Abstract
Background and Aims:
This randomized controlled trial was conducted to examine whether prophylactic active drainage decreases morbidity associated with pancreati-coduodenectomy.
Material and Methods:
A prospective, randomized controlled trial was conducted between April 2010 and May 2015 when 160 consecutive patients following elective pancreaticoduodenectomy were randomized intraoperatively to either prophylactic active drainage group or conventional passive drainage group. The main objectives were the incidence of postoperative pancreatic fistula and the associated clinical outcomes.
Results:
There were 82 patients in the active drain group and 78 patients in the passive drain group. The overall pancreatic fistula rate occurred similarly in the two groups (11.0% (9/82) vs 14.1% (11/78), p = 0.360). Grade C pancreatic fistula in active drain group was significantly less than that in passive drain group (0% (0/82) vs 6.4% (5/78), p = 0.026). The mean postoperative hospital stay and parenteral nutrition support time in active drainage group were shorter than those in passive drainage group (12.6 days vs 14.5 days, p = 0.037; 6.9 days vs 8.6 days, p = 0.047, respectively).
Conclusion:
Prophylactic active drainage reveals significant reduction in severity of complications associated with pancreatic fistula and might be recommended as an alternative for patients with high risk of developing serious pancreatic fistula after pancreaticoduodenectomy.
Introduction
Pancreaticoduodenectomy (PD) is one of the most complicated abdominal operations and it has been performed increasingly in recent years as a safe and appropriate option for selected patients with malignant and benign lesions of pancreatic head, duodenum, distal part of common bile duct, and peri-ampullary region. Although this complex operation can be performed successfully in many specialized centers with relatively lower mortality rate below 5% (1, 2), the overall postoperative complications remain quite high, usually averaging 30%–50% (3, 4). One of the most notable complications is leakage of pancreatic juice from the pancreatic-enteric anastomosis after PD, so-called postoperative pancreatic fistula (POPF) (5), a major trigger of other complications such as dehiscence of the pancreaticojejunostomy or pancreaticogastrostomy, intraperitoneal hemorrhage, intra-abdominal sepsis or abscess, multiple-organ dysfunction and failure, and even death.
Although surgical techniques have undergone significant improvement and innovations in recent years, still nearly 10%–20% of the patients develop pancreatic fistula after PD (6, 7). For those patients who have experienced a pancreatic fistula, the ensuing complications greatly augment healthcare costs due to prolonged hospital stay, need for reoperation to address complications of the primary procedure, and need for intravenous nutrition, among other indirect costs to family and friends. It is therefore important to conduct research to explore effective strategies to decrease pancreatic leakage rate after PD (8).
Until recently, effective methods to reduce the rate of pancreatic fistula are yet to be established. Possible factors that are likely to prevent complications associated with pancreatic fistula may include directly anastomosing pancreatic duct to intestinal mucosa, putting an indwelling stent into the pancreatic duct to provide external or internal drainage to decrease the pressure of the pancreaticojejunostomy, adopting pancreaticogastrostomy reconstruction (9, 10), topical application of biologic fibrin glue around the pancreatic-enteric anastomosis, and utilizing somatostatin or octreotide to inhibit pancreatic exocrine secretion (11 –13).
However, these methods mainly focus on dealing with the pancreatic-enteric anastomosis and cannot completely prevent the occurrence of pancreatic fistula. One important principle to manage digestive tract fistula is to provide the accumulated collections within peritoneal cavity of an unimpeded drainage at the earliest stage to prevent development of complications and reduce the severity associated with pancreatic fluid collection. We therefore initiated a single-institution, prospective randomized trial to evaluate the value of prophylactic active drainage in treating pancreatic fistula as well as the associated clinical outcomes following PD.
Materials And Methods
Patients and Methods
This study was approved by the ethics committee of Affiliated Hospital of Qingdao University. Information on the proposed study was provided to all potential participants scheduled to undergo PD and informed consent was obtained preoperatively from every willing candidate before randomization. In all, 185 patients were enrolled between April 2010 and May 2015. Twenty-five patients were excluded from this study due to explorative laparotomy or distal pancreatectomy. The remaining 160 eligible patients were randomized intraoperatively to either active drainage group (n = 82) or passive drainage group (n = 78). The protocol is summarized in Fig. 1. The active drainage was a dual-lumen suction tube with an outer diameter of 8 mm and inner diameter of 6 mm, which is designed by our own department (Fig. 2). A conventional silicon drainage of 8 mm diameter was used in the passive drainage group. The drains were placed at the vicinity of pancreaticojejunostomy and hepatojejunostomy via the foramen of Winslow before closing the incision.
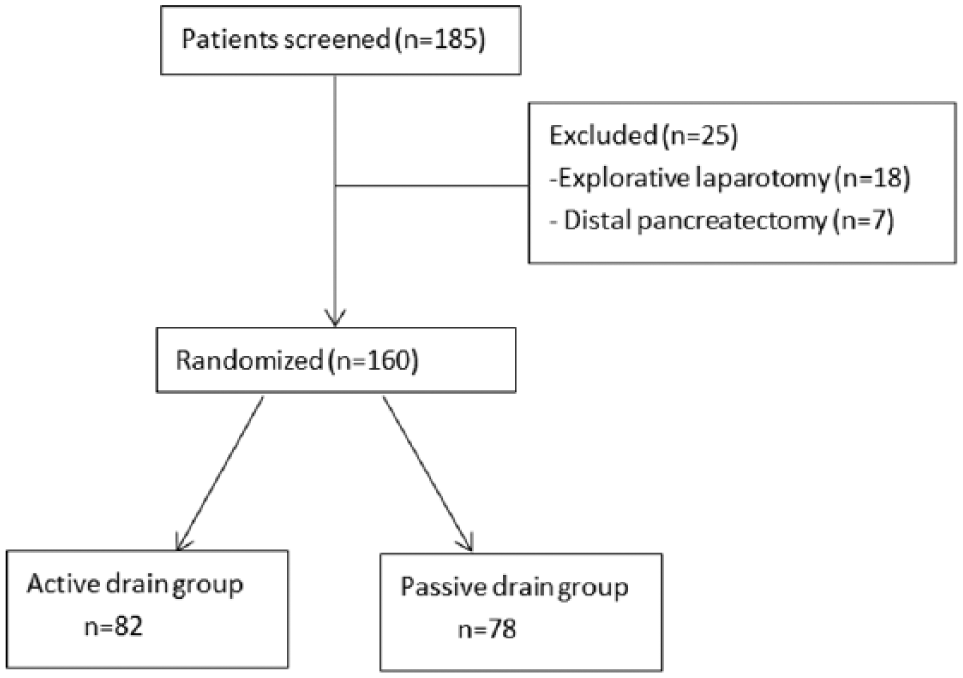
Flow consort diagram to show enrollment and randomization process.

Active drainage (dual-lumen tube suction drain, with diameter of 8 mm). A) Efferent pipe (inner tube with diameter of 6 mm) connecting to suction. B) Afferent pipe (with diameter of 3 mm) connecting to irrigating water.
Surgical Procedure
All surgical procedures were accomplished by a team of experienced surgeons specialized in hepatobiliary and pancreatic surgery, and reconstruction of pancreaticojejunostomy was performed by a single experienced senior surgeon. Standard PD was performed for all eligible patients with lymph nodes around the pancreatic head and hepatoduodenal ligament routinely dissected for malignant cases. Segmental resection of the superior mesenteric vein or portal vein was performed when a pancreatic head tumor was inseparable from the vein. After en bloc resection of head, neck, and uncinate process of pancreas, gallbladder, distal part of stomach (except for pylorus preservation), and distal common bile duct, a retrocolic jejunal limb was utilized for anastomosing with the pancreatic stump and hepatic bile duct. An end-to-side pancreaticojejunostomy was performed. Using a 3-0 polyglactin (Ethicon) suture, continuous anastomosis of the posterior wall of the pancreatic capsule parenchyma to the seromuscular layer at anti-mesenteric side of jejunum and a small incision with appropriate diameter were made at the jejunum. An internal duct to mucosa reconstruction was created with interrupted 5-0 polypropylene (Ethicon) suture.
Concerning different pancreatic texture and the diameter of pancreatic duct, an indwelling stent tube was inserted into the main pancreatic duct via a purse string suture of the duct to brace reconstructed anastomosis (if the pancreatic stump is hard, fibrotic, or the diameter of pancreatic duct is greater than 3 mm, no indwelling stent tube was placed into the duct-to-mucosa anastomosis). An outer row of continuous 3-0 polyglactin (Ethicon) suture was used to connect the anterior side of pancreatic capsule parenchyma to the seromuscular layer of jejunum. Next, an end-to-side hepaticojejunostomy was performed about 10 cm downstream from the pancreaticojejunostomy using a single layer of 4-0 or 5-0 polyglactin (Ethicon). Finally, an antecolic limb of jejunum was brought for gastrojejunostomy or duodenojejunostomy about 45 cm downstream from the hepaticojejunostomy. Before closing the incision, we randomized our patients using a computer-generated random number into either an active drainage group or conventional silicon passive drainage group. Accordingly, the respective drainage was placed at the vicinity of pancreatic and biliary anastomosis via the foramen of Winslow.
Postoperative Management
All patients were treated in intensive care unit (ICU) for the first 24–48 h postoperatively unless further monitoring and special treatment was required. Antibiotics were routinely discontinued after 48 h, and histamine H2-receptor antagonists were administered during the postoperative hospital stay as prophylactic medication against stress ulcer. A liquid diet was often commenced after removing the nasogastric tube when bowel movement returned (usually 3 or 4 days postoperatively).
The efferent pipe of active drainage was connected to continuous suction (0.02–0.04 MPa) and the afferent pipe was irrigated with 3-L normal saline every 24 h for the first 3 days postoperatively and kept on intermittent suction for another 2 days. The passive drainage tube was connected to the drainage pack. The amylase level in the two kinds of drainage was examined on days 3, 5, and 7 postoperatively, and pancreatic fistula was defined as any measurement of total amylase activity in the intra-abdominal collection that exceeded the upper normal limit of serum amylase activity by three-fold on or after 3 days postoperatively according to the definition of POPF by International Study Group of Pancreatic Fistula (ISGPF). The drainage was routinely removed at the discretion of the attending surgeon between postoperative days 5 and 7 in the absence of evidence of any pancreatic fistula. In patients who had amylase level of intra-abdominal collection three-fold or greater than the serum upper limit amylase level on postoperative day 5 or 7, the drainage was maintained in situ until the fistula was resolved or required further intervention.
Study Endpoints
The primary study endpoint was the number of patients with POPF, which was diagnosed as the drainage of abdominal fluid with amylase concentration that was three times higher than the serum concentration, and was further classified into three different grades (A: has no clinical impact on patients, B: require changes in management or adjustment in the clinical pathway, and C: a major change in clinical management or deviation from the normal clinical pathway occurs) in accordance with the definition by ISGPF.
Second study endpoints included overall morbidity rate, hospital mortality rate, rate of surgical intervention (reoperations or percutaneous drain for collections), length of hospital stay, number of days on total parenteral nutrition, and duration to resumption of oral diet. Postoperative complications were classified as depicted in Clavien’s classification (14).
Data Collection
Patients were prospectively assigned a code and data were recorded in a database by two nurses. Data included the preoperative demographic and clinical data, details of surgical procedure, pathological results, and relevant clinical information concerning the postoperative course, postoperative morbidities, and mortality. Study statisticians were blinded to the two allocation groups and did not participate in the management of patients for all analyses.
Statistical Analysis
A statistical software package (SPSS Version 17.0, Chicago, IL, USA) was used for statistical analysis. Qualitative variables between the two groups were compared by chi-square test (or Fisher’s exact test where necessary) while quantitative data were expressed as mean ± standard deviation (SD) and were assessed using a Student’s t-test. p < 0.05 was considered statistically significant.
The study size was calculated by the premise of detecting a 20% difference in complication rate between the two groups, with alpha set at 0.05 and beta set at 0.2 yielding a statistical power of 80% and estimating a sample size of 160 subjects (80 patients in each arm of the study). We enrolled 185 patients to take into account the possibility that about 15% patients would not be able to accomplish PD.
Results
Patient Characteristics
Of 185 patients enrolled for this study, 25 patients were excluded because they underwent either explorative laparotomy or distal pancreatectomy. The remaining eligible 160 patients successfully underwent elective PD and were randomized intraoperatively to either active drainage group (n = 82) or conventional passive drainage group (n = 78). The two groups were similar in preoperative characteristics and there were no significant differences with regard to age, gender, general clinical state, concomitant diseases, and preoperative blood results of biochemical analyses (hepatic and renal function) (Table 1).
Patient’s characteristics and preoperative factors.
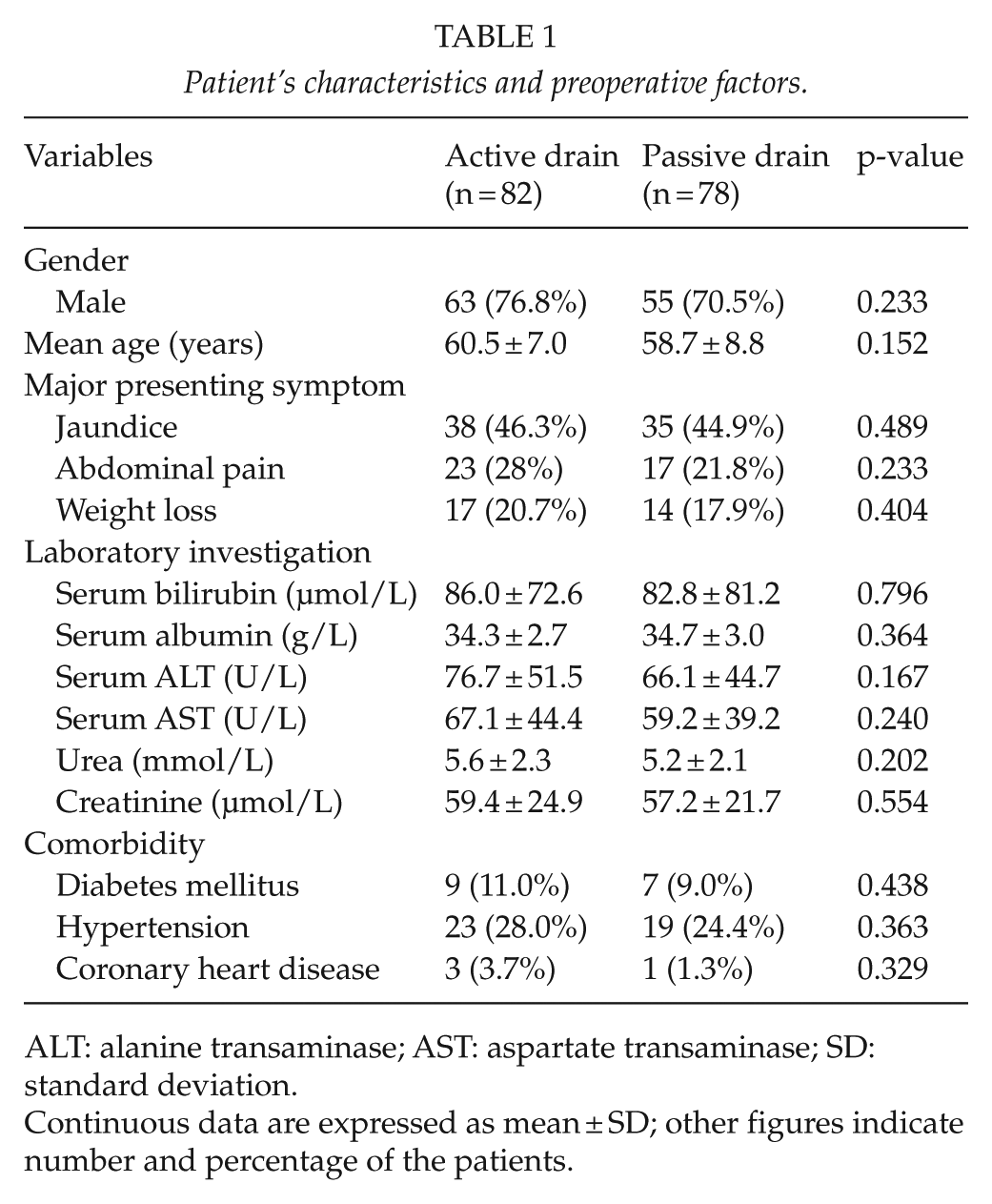
ALT: alanine transaminase; AST: aspartate transaminase; SD: standard deviation.
Continuous data are expressed as mean ± SD; other figures indicate number and percentage of the patients.
Surgical Details and Pathologic Diagnosis
The main indications for surgical details and pathological diagnosis are depicted in Table 2. There was no significant difference in terms of multiple intraoperative parameters between the two groups. Although some surgeries involved pylorus preservation and superior mesenteric-portal venous resection, most of the pancreatic-enteric reconstructions were performed as direct duct-to-mucosa anastomosis with only a small proportion of which soft pancreatic parenchyma was performed with indwelling stent to brace duct-to-mucosa anastomosis. Pancreatic duct diameter and consistency were similar between the two groups and there were no statistically significant differences between the two groups with respect to mean operative time (4.5 vs 4.3 h, p = 0.161), intraoperative blood loss (395 vs 381 mL, p = 0.325), requirement for intraoperative blood transfusion (median blood transfusion in both groups was 0 unit of packed red cells), and other associated procedures. The two groups were comparable with regard to the type of pathology and carcinoma of the pancreas was the most common diagnosis in both groups.
Surgical details and pathological diagnosis.
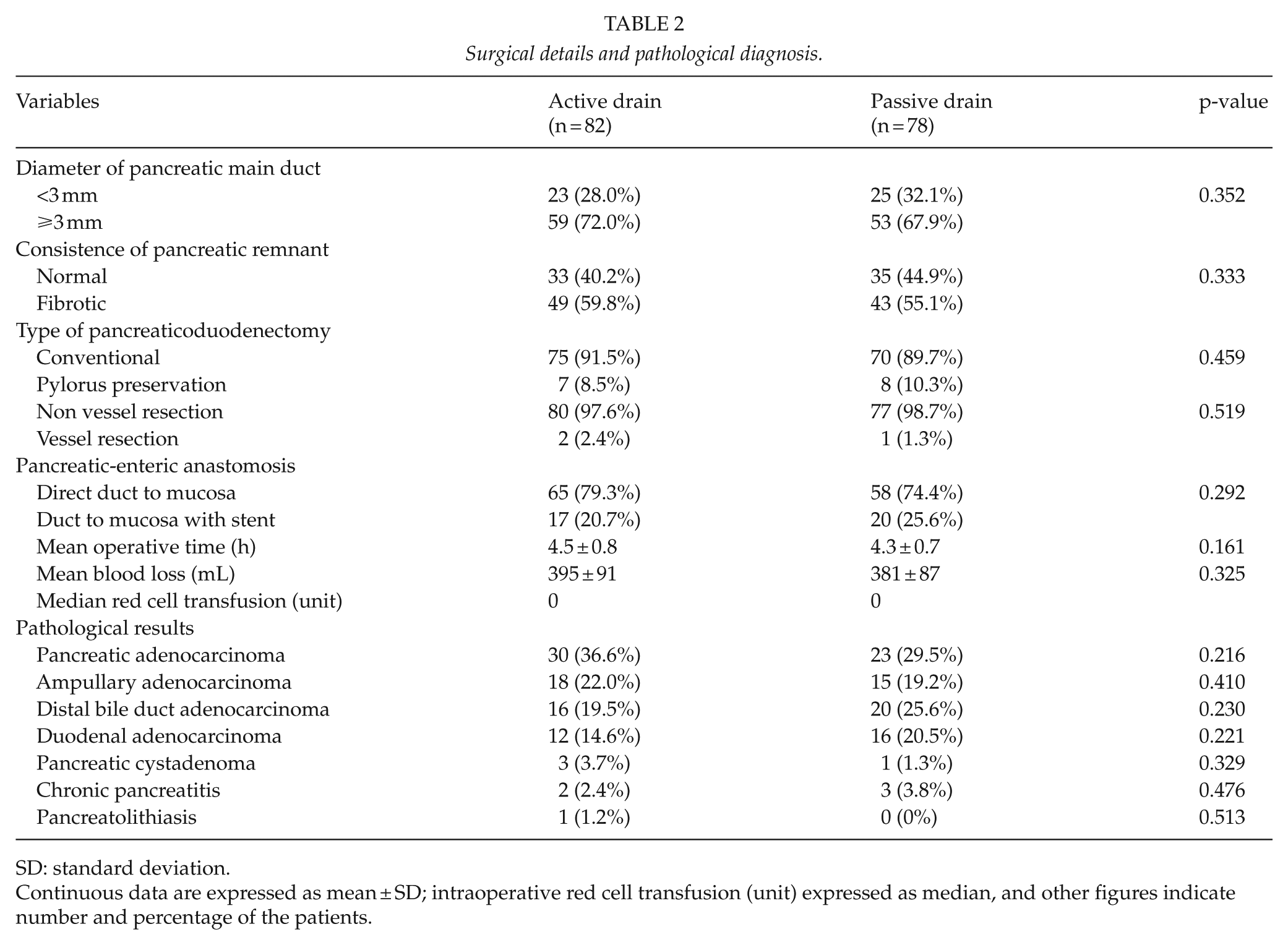
SD: standard deviation.
Continuous data are expressed as mean ± SD; intraoperative red cell transfusion (unit) expressed as median, and other figures indicate number and percentage of the patients.
Main Postoperative Outcomes
The major postoperative outcomes, complications, and perioperative deaths are depicted in Table 3. The overall morbidity rate was 22.0% in the active drain group and 32.1% in the passive drain group (p = 0.103). One death occurred in the active drain group 6 days postoperatively as a result of pulmonary artery embolism, but no death was reported in the passive drain group during the duration of hospitalization.
Major postoperative outcomes.
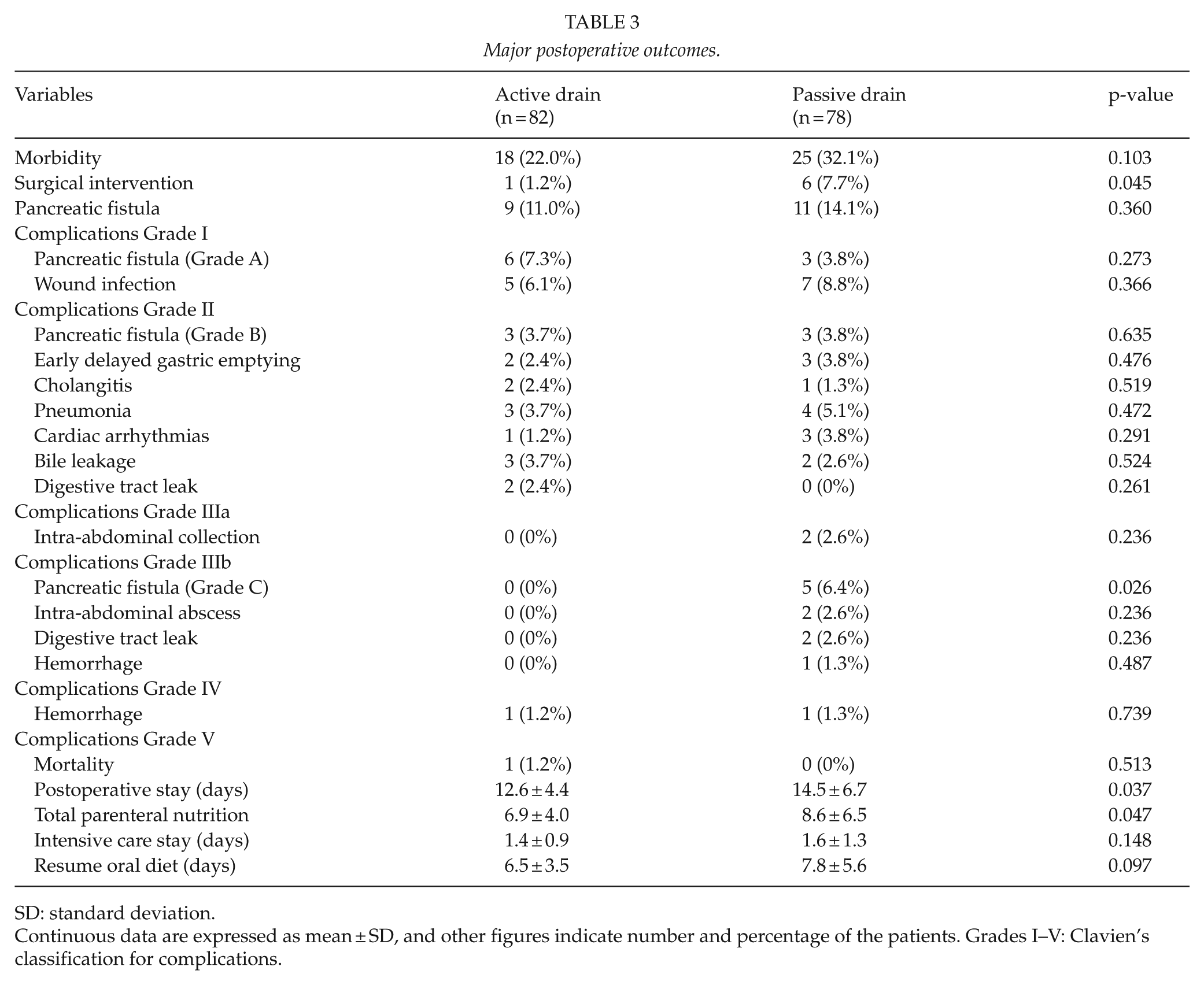
SD: standard deviation.
Continuous data are expressed as mean ± SD, and other figures indicate number and percentage of the patients. Grades I–V: Clavien’s classification for complications.
POPF was the most common complications in both groups with an incidence of 9 (11.0%) in active drain group and 11 (14.1%) in passive drain group (p = 0.360). In the active drain group, POPF was either Grade A or B, with no Grade C fistula, while five cases in the passive drain group were of Grade C, with the difference in POPF of Grade C occurrence significant between the two groups (0% (0/82) vs 6.4% (5/78), p = 0.026). Of the five cases with Grade C POPF, two cases developed serious abdominal abscess, two cases were complicated with digestive tract fistula, and one case was concomitant with hemorrhage. These patients were kept in ICU longer, requiring longer time of total parenteral or enteral nutrition, intravenous antibiotics, and somatostatin analogs. Furthermore, aggressive surgical interventions were performed for them to recovery. Two of them underwent reoperation to put active drainage to funnel egress of the abscess with ensuing percutaneous drainage insertion, one patient underwent re-laparotomy to manage the hemorrhage after failure of artery embolization, and the other two patients of Grade C POPF were re-operated to manage the complicated digestive fistula. Occurrence of other complications, including wound infection, delayed gastric emptying, bile leakage, and cholangitis, was similar between the two groups (Table 3).
Each group had one patient needing reoperation to manage the postoperative hemorrhage, which was not associated with POPF. The total surgical interventions for complications in active drain group were less than those in passive drain group (1.2% (1/82) vs 7.7% (6/78), p = 0.045). The mean postoperative hospitalization was 12.6 days in the active drain group, significantly shorter than the 14.5 days in the passive drain group (p = 0.037). The total parenteral nutrition support period was also shorter in active drain group than that in passive drain group (6.9 vs 8.6 days, p = 0.047).
Discussion
The practice of placing routine drainage in the field of pancreatic resections especially following PD has recently become a controversial issue. Successful management of pancreatic fistula often depends on early identification of the characteristics of drainage fluids and collections. The rationale behind placement of drainage after PD procedure may contribute to effective evacuation of blood, bile, pancreatic juice, and chyle that may accumulate within abdomen postoperatively and might serve as an early warning sign of anastomotic leakage and the associated hemorrhage (15, 16). However, more and more evidences argue that elimination of routine intraperitoneal drainages in elective pancreatic resection was not associated with increased morbidity or severity of POPF (17). In fact, drainage placement may cause more harm. The drainage itself might serve as the portal of entry for bacteria, which may change a relatively clean intra-abdominal collection into an infection and abscess, and they might even erode into the anastomosis to increase the occurrence of a fistula (18), and early removal of drains might reduce the risk of intra-abdominal infections (19). Recent prospective randomized trials demonstrated that intra-abdominal drain can be safely removed early after surgery for patients with low risk of pancreatic fistula, and prolonged duration of drain insertion is associated with higher rate of postoperative morbidity and leads to a longer hospitalization with more economic resource expense (20 –22).
This has prompted a recent multicenter randomized trial of drain versus no drain in patients who underwent PD by Van Buren et al. (23). The trial had to be terminated early due to increased severity and rate of morbidity, need for postoperative percutaneous drainage placement, and prolonged length of hospital stay as well as a nearly four-fold increase in mortality in the no drain group. Furthermore, two recent meta-analysis articles indicated that it is still premature to conclude that abandoning drainage after pancreatic resection leads to a decrease in complications (24, 25). A systemic review involving 4194 patients undergoing pancreatic resection showed that there is still insufficient evidence to determine which type of drain is more suitable following pancreatic surgery and trials to compare different types of drains after pancreatic resection are still needed (26).
Active drainage (dual-lumen tube suction drainage) was invented by Prof. Li to manage enterocutaneous fistula and self-healing rate of intestinal fistula was improved significantly with active drainage (27). This novel active drainage has an efferent suction pipe inside an outer silicon tube with side holes, and the end of the outer tube was connected to a slim afferent pipe for supplying continuous circulating sterilized water to keep the tube unimpeded. When the efferent pipe was connected to the negative pressure suction and the afferent pipe was connected to continuous thorough irrigated water, the active drainage becomes a continuous suction device to drain the collection within the peritoneal cavity fairly rapidly and effectively. This active suction drainage was placed through the foramen of Winslow in the vicinity of both the pancreatic and biliary anastomosis. Compared with the conventional passive drainage, the active drainage can effectively remove collected blood, bile, chyle, or pancreatic juice within the peritoneal cavity or between intestinal loops and can greatly reduce the incidence of intra-abdominal infection, formation of abdominal abscess, and subsequent systemic inflammation or massive hemorrhage due to enzymatic erosion of intra-abdominal tissue and fistula orifice ensuing pancreatic fistula.
One fundamental principal to improve outcome in patients with digestive tract fistula is the unblocked drainage of collections within the abdominal cavity. In this study, this single-institution, randomized controlled trial was designed to evaluate the hypothesis that active drainage can minimize postoperative complications associated with pancreatic fistula. In this study, the two groups were similar for preoperative patient and disease characteristics, the type and site of the underlying tumor, resection, and anastomotic technique. Some patients were also characterized by soft remnant pancreatic tissue and the main duct diameter less or larger than 3 mm. The only management difference was adopting different drainages during operation.
This randomized trial showed that compared with passive drainage, active drainage had significant potential in ameliorating development into serious POPF. According to the aforementioned criteria of definition for pancreatic fistula by ISGPF (5), the overall POPF rate was 12.5% (20/160), with 11% in the active drain group and 14.1% in the passive drainage group, which is comparative to the reported 14% POPF rate (16 of 114) in a recent prospective controlled trial by Bassi et al. (20). Our incidence of POPF is also similar to another recently published article of early drain removal after PD by Ven Fong et al. (22). In this study, Grade C POPF occurrence in the active drain group was markedly less than that in the passive drain group (0/82 vs 5/78, p = 0.026). The potential mechanism of active drainage in alleviating severity of POPF may mostly be explained by its ability to drain collected pancreatic juice much more effectively, greatly reducing pancreatic juice accumulation within peritoneal cavity or diluting the accumulated collection by irrigating water to minimize its erosion and impairment to other tissues, thus preventing the mild pancreatic leakage from developing a serious pancreatic fistula.
Occurrence of pancreatic fistula is usually associated with a higher incidence of reoperation, wound infection, intra-abdominal abscess formation, and increased length of hospital stay (7, 28). Only one patient in this cohort with POPF required surgical intervention because of intra-abdominal hemorrhage in the active drain group. Meanwhile, six patients in passive drain group underwent surgical management for anastomosis dehiscence, intra-abdominal abscess, and hemorrhage. Since all these surgically intervened complications inevitably extended the length of hospital stay, it was expected that a significantly shorter hospital stay was observed in active drainage group (mean 12.6 days) compared to passive drainage group (mean 14.5 days). The reduced rate of complications and surgical management contributed to a good recovery of the nutritional status and a shorter time of total parenteral nutritional support period in active drain group compared with that in the passive drain group, which may indirectly favor the use of prophylactic active drainage.
Our study is a single-institutional cohort analysis and external validity of our results might be limited. Single-institution studies may be confounded by the evolution of a surgeon’s techniques and an institutional learning curve over time. Our institutional overall POPF and postoperative mortality rate was comparable to the recently published literatures. The overall other morbidity rate in our cohort was also consistent with the results of recent studies (10, 29). Another limitation of this trial is the potential detrimental effect of vacuum exerted by active drain, possibly eroding or sucking the pancreatojejunostomy anastomosis and causing a pancreatic fistula. However, such phenomenon was not observed by using relatively lower negative pressure suction in our study. Other limitations include relatively small number of patients, the fact that pancreatic-jejunum anastomosis was performed by a single surgeon, and the variation in relationships between drains and pancreatic and biliary anastomosis. A multi-institutional, prospective randomized controlled trial may still be needed for further investigation.
In conclusion, we found that active drainage can significantly reduce the severity of pancreatic fistula and their associated complications. Active drainage may be recommended as a safe and effective alternative for patient with high risk of developing serious pancreatic fistula following PD.
Footnotes
Acknowledgements
This study would have not been possible without the support of the staff from surgical department and nurses of the Affiliated Hospital of Qingdao University. Their dedication to the care of our patients was critical for our accomplishments. We also express our sincere thanks to Prof. Jiapeng Huang (Jewish Hospital and St. Mary’s Healthcare of Louisville University, USA) for his careful revision of this manuscript. N.L. is the first co-author and contributed equally to this manuscript. The trial was authorized by the ethics committee of Affiliated Hospital of Qingdao University (QYFYKYLL-2010-017).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
