Abstract
Background and Aims:
Enterococcus has emerged as a virulent species; Enterococcus faecium especially has arisen as a source of nosocomial infections. Furthermore, specific Enterococcus faecalis species are significantly associated with anastomotic leakage in rodent studies. The objective of this study was to investigate whether the occurrence of Enterococci (E. faecium and E. faecalis) obtained from drain samples was associated with leakage in humans undergoing pancreaticoduodenectomy.
Materials and Methods:
All patients undergoing pancreaticoduodenectomy had a peritoneal drain sample sent for culturing between postoperative days 3 and 10. Postoperative pancreatic fistulas were defined and classified according to the International Study Group of Pancreatic Fistula. Bile leakage was radiologically verified. Postoperative complications were classified according to the Dindo–Clavien classification.
Results:
A total of 70 patients were eligible and enrolled in this study. Anastomosis leakage was observed in 19 patients; 1 leakage corresponding to the hepaticojejunostomy and 18 pancreatic fistulas were identified. In total, 10 patients (53%) with leakage had Enterococci-positive drain samples versus 12 patients (24%) without leakage [odds ratio (OR) = 5.1, 95% confidence interval (CI) = 1.4-19.4, p = 0.02]. Preoperative biliary drainage with either endoscopic stenting or a percutaneous transhepatic cholangiography catheter was associated with the occurrence of Enterococci in drain samples (OR = 5.67, 95% CI = 1.8-12.9, p = 0.003), but preoperative biliary drainage was not associated with leakage (OR = 0.45, 95% CI = 0.1-1.7, p = 0.23).
Conclusion:
Enterococci in drain sample cultures in patients undergoing pancreaticoduodenectomy occurs significantly more among patients with anastomotic leakage compared to patients without leakage.
Introduction
The incidence of pancreatic adenocarcinomas has remained stable in recent decades, with an incidence of 17 per 100,000 in Denmark in 2014 (1). In Denmark, the overall five-year survival rate is 5% and 26% for patients who have not and have undergone a resection, respectively (2).
The primary curative therapy is a surgical resection of the tumor, and a pancreaticoduodenectomy (PD) is the approach of choice if the tumor is localized in the head of the pancreas. A small number of patients undergoing PD are due to benign reasons, for example, intraductal papillary mucinous neoplasm. Anastomotic leakage and the subsequent pancreatic fistula are the most common surgical complications after a PD (3). Known risk factors of leakage have previously been identified including the diameter of the pancreatic duct, a soft pancreas texture, ischemia, perioperative bleeding, and body mass index (BMI) (3, 4). In addition, a prospectively validated clinical score that accurately predicts pancreatic fistulas after PD has been published (5). In colorectal surgery, the presence of Enterococcus faecalis has hypothetically been related to anastomotic leakage due to the collagen-degrading activity of the bacteria (6). Enterococcus species normally reside in the upper gastrointestinal tract (GI tract) and are among the most common bacteria (7). Over the last few decades, Enterococcus has been observed as an increasingly virulent species that is often resistant to commonly used antibiotics and sometimes has the ability to produce collagenase (8–11). Therefore, it is hypothesized that Enterococci play an important role in the development of anastomotic leakage by preventing a newly created anastomosis from healing properly.
The aim of this study was to investigate the occurrence of the microbial species present in drain fluid from patients who had a PD with or without postoperative anastomotic leakage.
Material and Methods
Study Design
This prospective study included patients who had a PD due to malignant or non-malignant diseases of the pancreatic head, the periampullary area, or the duodenum. All patients were operated on with a modified Whipple’s operation, which included a resection of the antral part of the stomach, the duodenum, the pancreatic head, the gall bladder, and the common bile duct. The reconstruction was performed with a pancreaticojejunostomy, a hepaticojejunostomy, and a gastrojejunostomy. An intraperitoneal drain to an external bag was positioned dorsal to the hepaticojejunostomy and the pancreaticojejunostomy. The drain was connected with active suction until postoperative day (POD) 1. Hereafter, passive collection and removal on the fifth POD in case the drain fluid was free of bile, chyle, and amylase (less than 1000 IU/L) and if the drain volume output was below 100 mL for 24 h. Otherwise, the drain was kept in situ. In the case of postoperative bile leakage or a pancreatic fistula grade B or C, the drain remained and a computed tomography was scheduled to exclude fluid collections. In this study, fluid from the peroperatively placed drain was sent for culture and amylase level testing once daily at the third and fourth POD. Further samples were collected if medical conditions required continuous samples for analysis. The time interval between the appearance of Enterococci in the drain fluids and the clinical symptoms of leakage was registered. However, no patients with leakage had clinical symptoms prior to POD 10. Hence, leakage was only verified with a significantly elevated amylase in drain output. Ultrasonographically placed drains to evacuate intra-abdominal collections were excluded from the study. Bacterial cultures were done according to standard methods: drain fluids were cultured on a 5% blood agar plate and a blue plate (selective for Gram-negative rods), and each sample was cultivated for 2 days, followed by the identification of resistance patterns. The majority of the bacteria in the drain samples were excluded due to infrequent presence.
All patients received preoperative intravenous antibiotic prophylaxis with 3 g Cefuroxim (Fresenius Kabi, Bad Homburg, Germany) and 1.5 g metronidazole (SAD, Copenhagen, Denmark). All patients received postoperative 1.5 g Cefuroxim three times daily and 1.5 g metronidazole once a day, for three days.
Inclusion Criteria
Patients were included based on the following criteria: (1) >18 years of age; (2) tumor in the periampullary region, head of pancreas, or duodenum; (3) surgical approach in the form of a PD; (4) consecutive drain samples, and (5) at least one drain fluid sample routinely sent for culture from POD 3 to 10. Patients with drain samples after POD 10 had symptomatic leakage, but as the purpose was to identify asymptomatic patients with positive microbial drain samples as an indicator of leakage, patients with drain samples after POD 10 were excluded.
Patients
Comorbidity was assessed according to the American Society of Anesthesiologists score (12). Patients were followed until death or 3 months postoperatively. Due to the short follow-up time, the cancer recurrence rate was not relevant or applicable in our study.
Postoperative Complications
Anastomotic leakage corresponding to the pancreaticojejunostomy was defined and classified according to the International Study Group of Pancreatic Fistula (13), but an updated definition of pancreatic fistula has been published during the preparation of the final manuscript (14). The updated version defines a grade A leakage as a biochemical leakage, thus not a leakage. However, our aim was to identify whether appearance of bacteria in drain samples was related to leak or excretion of amylase intra-abdominal regardless of the patient was symptomatic. We therefore chose to proceed with the former definition. Anastomotic leakage corresponding to the hepaticojejunostomy was suspected if the drain fluid contained visible bile. It was verified by a percutaneous transhepatic cholangiography (PTC) followed by the placement of a percutaneous biliary catheter.
Postoperative complications were classified according to Dindo–Clavien (15). To merge the complication scores into our analyses, we grouped scores 0–2 and 3–5 due to the fact that only a score ≥3 resulted in a significant intervention and a significant deviation from the normal postoperative course.
Statistics
Statistical analyses were performed with IBM SPSS Statistics for Windows (version 22.0; IBM Corp., Armonk, NY, USA). Non-parametric statistics were applied throughout due to the inability to meet requirements for parametric statistics. Categorical variables were compared with a chi-square test or with Fisher’s exact test. The Mann–Whitney test was used to compare continuous variables when appropriate. A p value below 0.05, two-sided, was considered significant. For risk context, we performed a univariate or multivariate binary logistic analysis. The results from the binary logistic analyses are shown as an odds ratio (OR), 95% confidence interval (95% CI), and p value.
Results
A total of 153 patients underwent PDs from January 2014 to May 2015 at the Department of Surgical Gastroenterology, Rigshospitalet, Denmark. Of these, 70 patients (45.8%) were eligible for inclusion in the study according to the inclusion criteria (Fig. 1). Demographic data are shown in Table 1.
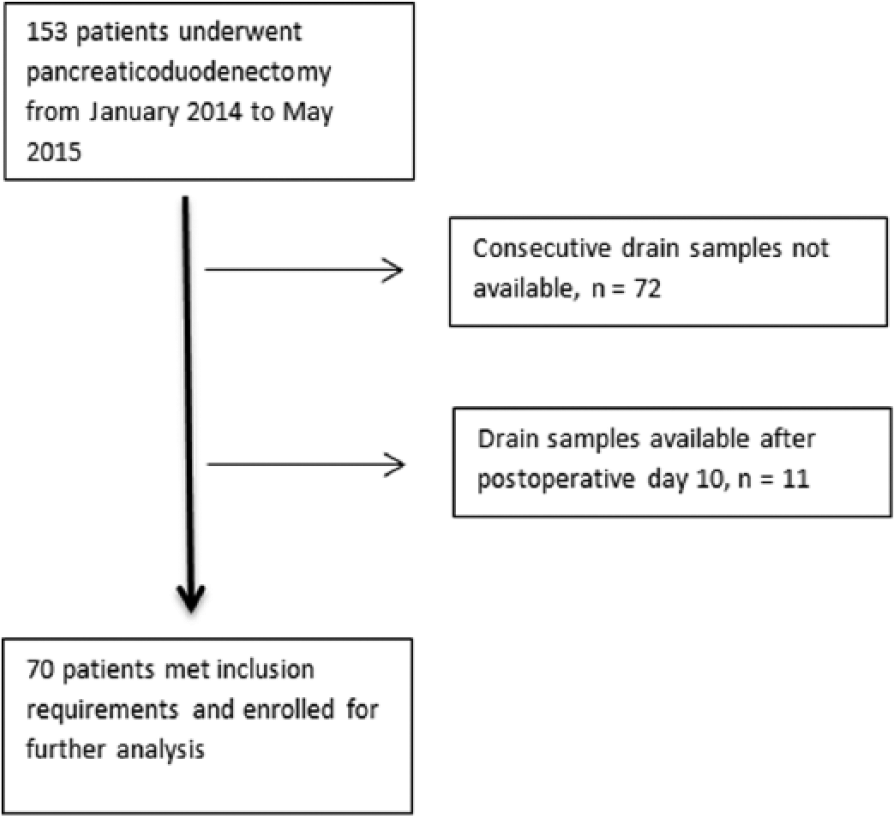
Inclusion flowchart.
Demographic characteristics—n (%) is applicable unless otherwise listed.
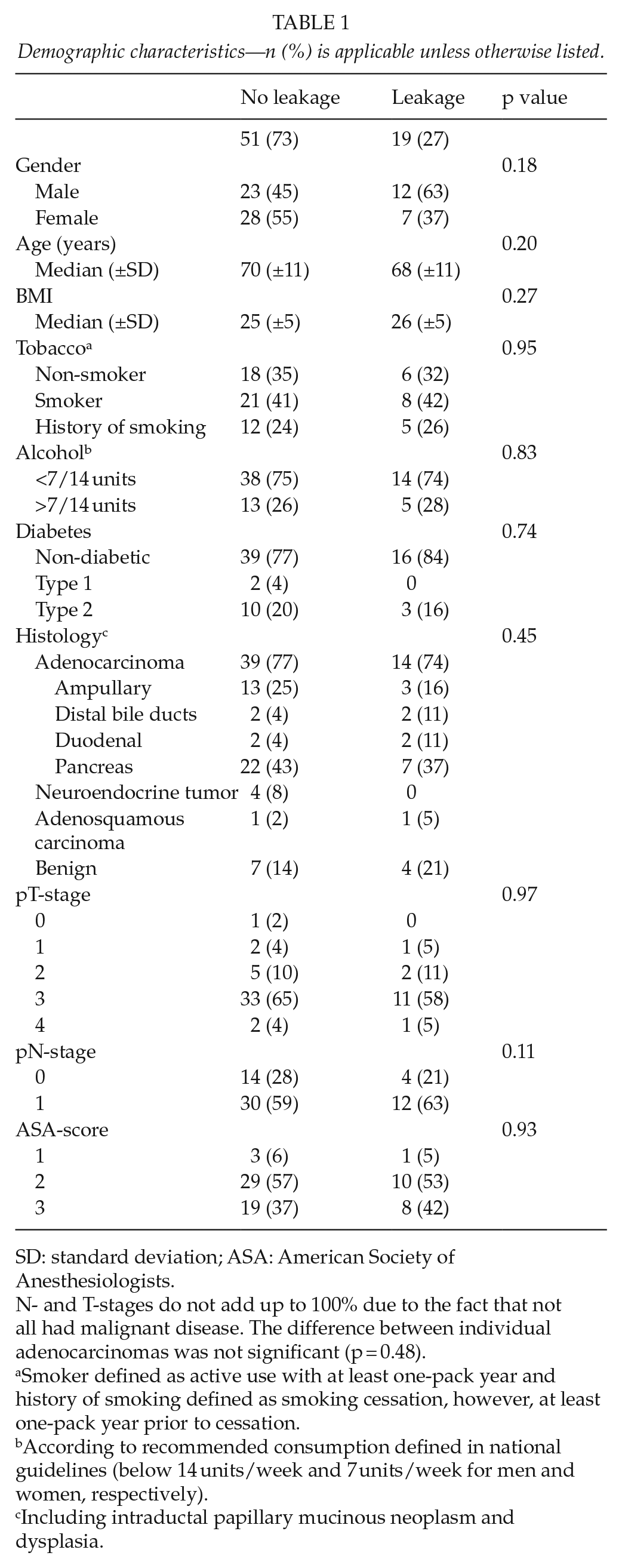
SD: standard deviation; ASA: American Society of Anesthesiologists.
N- and T-stages do not add up to 100% due to the fact that not all had malignant disease. The difference between individual adenocarcinomas was not significant (p = 0.48).
Smoker defined as active use with at least one-pack year and history of smoking defined as smoking cessation, however, at least one-pack year prior to cessation.
According to recommended consumption defined in national guidelines (below 14 units/week and 7 units/week for men and women, respectively).
Including intraductal papillary mucinous neoplasm and dysplasia.
Anastomosis leakage was observed in 19 patients. One leakage corresponding to the hepaticojejunostomy and 18 corresponding to postoperative pancreatic fistulas were identified. Of the 18 postoperative pancreatic fistulas, 8 patients (44%) were graded as A and 10 (56%) as B.
Postoperative Drain Fluid Cultures
Drain samples were collected from POD 3 to 10. Enterococci were observed in 10 patients (53%) with leakage and in 12 (24%) without leakage (OR = 3.6, 95% CI = 1.2-10.9, p = 0.02).
Monomicrobial infections were observed in 23 patients (32.9%), 16 without leakage versus 7 with leakage (p = 0.78). Polymicrobial infections were observed in 18 patients (25.7%), 10 without leakage versus 8 with leakage (p = 0.07). The occurrence of Enterococci was found in 10 samples as monomicrobial infections and in 12 samples as polymicrobial infections. The effect of monomicrobial and polymicrobial infections on anastomosis leakage including other bacteria was evaluated and no association was found. In addition, when Enterococcus infections were excluded, no association with leakage was found. Candida albicans was observed in 10 samples and only with other bacteria.
A total of 35 patients were treated for obstruction in the biliary tree by preoperative biliary drainage (PBD), with either endoscopic stenting or a catheter. When analyzing PBD for association with leakage, no correlation was found (OR = 0.87, 95% CI = 0.3-2.5, p = 0.80). However, a significantly higher occurrence of Enterococci was found in the PBD group (OR = 5.67, 95% CI = 1.8-12.9, p = 0.003).
Bacterial analyses of drain samples were compared with postoperative complications. The presence of Enterococci had no influence on the severity of postoperative complications (Dindo–Clavien score ≥ 3) or the severity of postoperative pancreatic fistulas (Table 2).
Association between enterococci and postoperative complications or postoperative pancreatic fistula grading —n (%) is applicable unless otherwise listed.
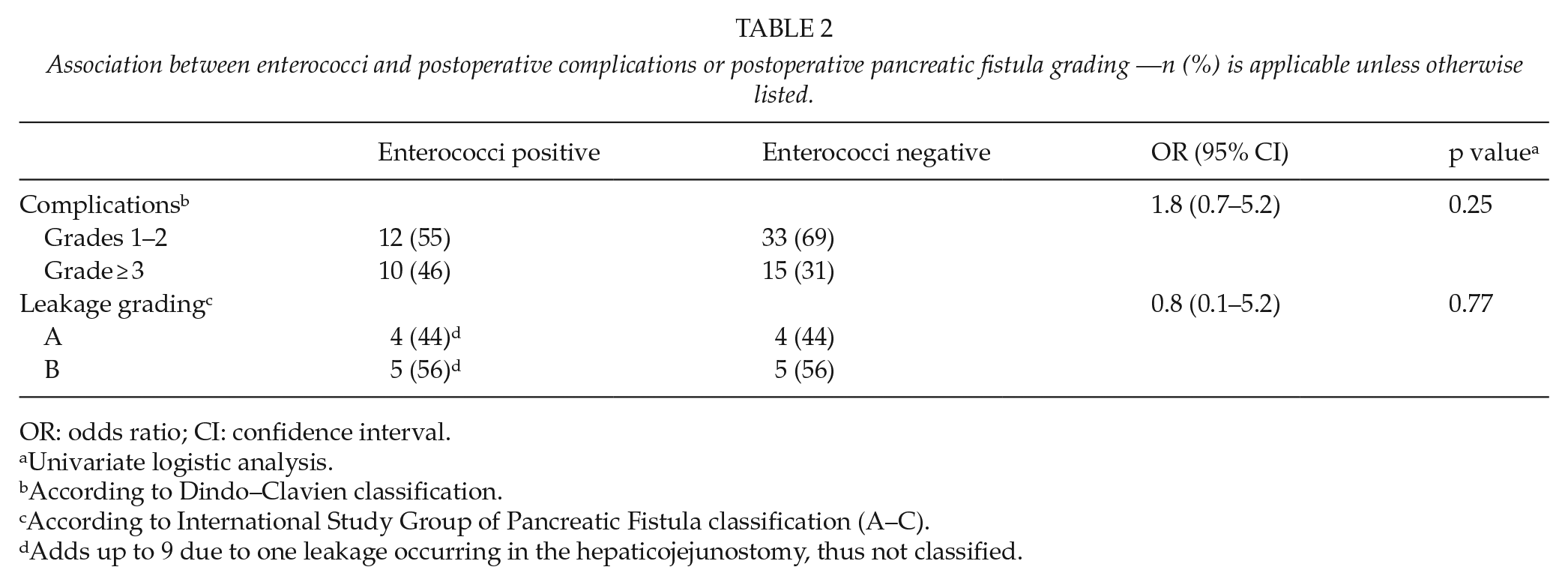
OR: odds ratio; CI: confidence interval.
Univariate logistic analysis.
According to Dindo–Clavien classification.
According to International Study Group of Pancreatic Fistula classification (A–C).
Adds up to 9 due to one leakage occurring in the hepaticojejunostomy, thus not classified.
The time to the appearance of Enterococci in the drain fluids was not different in the two groups, leakage versus no leakage, median time = 4 versus 4 days (p = 0.8). In all cases, positive Enterococci cultures were obtained before any clinical symptoms of leakage occurred. A leak was detected (median time = 3 days).
A multivariate binary logistic regression analysis of leakage was performed including the risk factors, Enterococci, PBD, and C. albicans. When adjusting for PBD and C. albicans, the occurrence of Enterococci was still found to be significant, with a higher OR of 5.10 in the leakage group (Table 3).
Multivariate binary logistic analysis of anastomotic leakage.
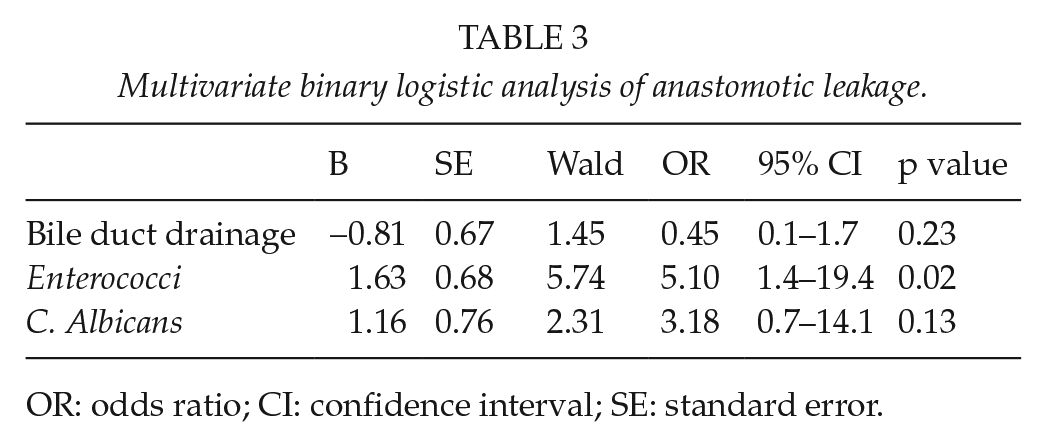
OR: odds ratio; CI: confidence interval; SE: standard error.
Discussion
The results from our study identified Enterococci (both monomicrobial and polymicrobial cultures of E. faecalis/faecium) as the most frequent commensal bacteria in drain samples from humans undergoing PDs (Appendix 1). In addition, Enterococci coexisted significantly more often among patients with anastomotic leakage (53% with leakage and 24% without leakage, p = 0.02). All cultures were obtained before the clinical symptoms of leakage had occurred. The presence of Enterococci was associated with neither a more serious grade of postoperative pancreatic fistulas nor a higher complication grade (Dindo–Clavien). We could not identify other factors correlated with leakage, except for an association of Enterococci with PBD in the univariate analyses. However, in the multivariate analyses, PBD was not an independent factor as it was not associated with leakage.
The finding of an increased frequency of positive Enterococci drain cultures in PD patients with leakage has not been demonstrated in humans before. While this study succeeded in showing the frequency of bacteria in drain samples, the causality between the bacteria and anastomotic leakage remains unknown. However, an association between bacteria and anastomotic leakage in the GI tract has been proposed previously, although actual studies concerning bacterial contamination and the association with leakage are scarce (6).
The association was first demonstrated in a study that showed that the leakage rate in dogs subjected to a colon anastomosis and occlusion of the mesenteric artery decreased if antibiotics were applied directly onto the anastomosis (16). The experiments were performed on unprepared dogs, thus not preceded with mechanical bowel preparation.
Almost 40 years later in 1994, a study group identified Pseudomonas aeruginosa as a possible microbial agent associated with leakage after a gastrectomy in rats (17). In this study, rats were allocated into three groups: the first group was inoculated with P. aeruginosa, the second was a control group, and the third was decontaminated with antibiotics. The leakage rate was significantly lower in the decontaminated group compared to the group inoculated with P. aeruginosa. On the basis of this, the authors initiated a randomized double-blinded multicenter study, finished in 1997, in which they found that oral antibiotics in addition to standard antibiotic prophylaxis significantly decreased the leakage rate in humans undergoing gastrectomies (18).
The ability of bacteria to degrade an anastomosis was recently studied (19). In this study, P. aeruginosa and Enterobacter cloacae in drain cultures from patients with a pancreatic fistula after a PD were shown to be associated with proteases capable of activating trypsinogen into trypsin. Trypsin was speculated to activate pancreatic juice and thereby cause autolysis leading to tissue damage around the pancreaticojejunostomy. In 2015, a study found that a specific strand of E. faecalis, both collagenase producing and matrix metallopeptidase 9 activating, was significantly associated with leakage among rats that underwent a colon resection with a primary anastomosis (9). Although Enterococci reside as part of the normal flora and are the most abundant bacteria in the upper GI tract, recent evidence of their increasing virulence, especially with E. faecium and E. faecalis, has emerged (7).
The increased virulence of E. faecium and E. faecalis accompanies a production of adhesion proteins, autolysin, and collagenases (9, 20–22). Adhesion protein production occurs in E. faecium and E. faecalis expressing Acm and Ace, respectively. Acm and Ace are genes enabling the production of wall-anchored adhesion proteins, which facilitate attachment to extracellular collagen I and IV (20, 23). Autolysins are proteins enabling cell wall degradation and biofilm formation. In addition, for E. faecium, specific adhesion proteins and autolysins have been identified that are associated particularly with biofilm formation and its stabilization (21, 22). The ability to produce these proteins may explain how Enterococci can degrade collagen in the extracellular matrix in the newly formed anastomosis. Biofilm formation adjacent to the anastomosis or on the sutures may be the starting point from which Enterococci colonize the anastomosis. Secured by the biofilm and by their resistance to commonly used antibiotics, Enterococci may colonize the anastomosis by attaching to the suture material and degrading the extracellular matrix around it, thereby weakening the structure. Thus, the increased virulence and antibiotic resistance could theoretically result in the events leading to leakage.
Our results suggest that PBD could explain an increased occurrence of Enterococci in the drains. We hypothesized that a stent or PTC catheter might function as the platform responsible for Enterococci colonization in the bile and pancreatic ducts. However, the association between PBD and leakage was not significant despite the increased presence of Enterococci. Subsequently, a newly published meta-analysis showed that PBD was not associated with leakage in patients undergoing PDs (24).
In our study, a standardized antibiotic prophylaxis regimen consisting of metronidazole and a second- and third-generation cephalosporin, administered intravenously, was given to the patients prior to and after surgery. However, the available data suggest that these antibiotics may have a decreased efficacy toward Enterococci in the intestines (8–11). Thus, the perioperative regimen eradicates the normal, non-pathogen bacterial flora in the intestines and lays the foundation for pathogen bacteria to flourish. A high incidence of C. albicans was also observed, which could be due to our rather broad spectrum antibiotic approach. Two studies supporting this theory showed that the abundance of Enterococci significantly increased following colon surgery after receiving preoperative antibiotics (6, 25). In agreement with this, two new studies have shown that mechanical bowel preparation and oral antibiotics in addition to standard antibiotic prophylaxis reduce leakage rates among colon resected patients with a primary anastomosis afterward (26, 27). Additionally, a meta-analysis, including eight randomized clinical trials, that examined the association between leakage and antibiotic regimens (oral + intravenous versus intravenous) has shown that the addition of oral antibiotics significantly decreases leakage rates (28). Two studies of upper GI tract surgery were included in the meta-analysis. The first study, with a large study group (n = 205), showed a decrease in the frequency of leakage after a gastrectomy (p = 0.049) (18). Contrary to this, the second study did not find a decrease in the frequency of leakage. However, the second study only included a small patient group (n = 91) that comprised both gastrectomies and gastroesophageal junction resections (29). Furthermore, the first study included peroral vancomycin, which targets Enterococci, and the second study did not.
A limitation of our study is the retrospective design and highly selected study group. Approximately 54% of all patients undergoing a PD during the investigation period were unable to meet our inclusion criteria, leaving only 70 patients for enrollment in the study. Logistical problems concerning the collection day were the primary cause of exclusion. This was due to the collection of drain fluid samples on different days than the days stipulated in the project description, within POD 10. This increased the probability for type II errors and made correlation to other risk factors difficult. Typically, patients with leakage had their drain tube retained after POD 5; hence, bacteria found after this day most likely occurred in leakage cases. However, we found no difference in time to occurrence of Enterococci in the leakage and no leakage groups. All cultures were consecutively obtained on POD 3 to 10, but this span may have reduced our chances of identifying whether or not Enterococci were present prior to leakage. Furthermore, analyses to identify collagenase and cell adhesion protein production were not performed; hence, the Enterococci virulence in our study is unknown. Enterococci were observed in patients without leakage, suggesting that leakage is multifactorial.
From our study, we conclude that the occurrence of Enterococci in drain cultures was significantly higher in patients with anastomotic leakage after a PD compared with patients without leakage. Oral antibiotics targeted against pathogenic bacteria in patients undergoing upper GI tract surgery may therefore be favorable in regards to a reduction in leakage. However, further randomized controlled trials are needed to establish the causality between bacteria and anastomotic leakage.
Footnotes
Appendix 1
Culture data.
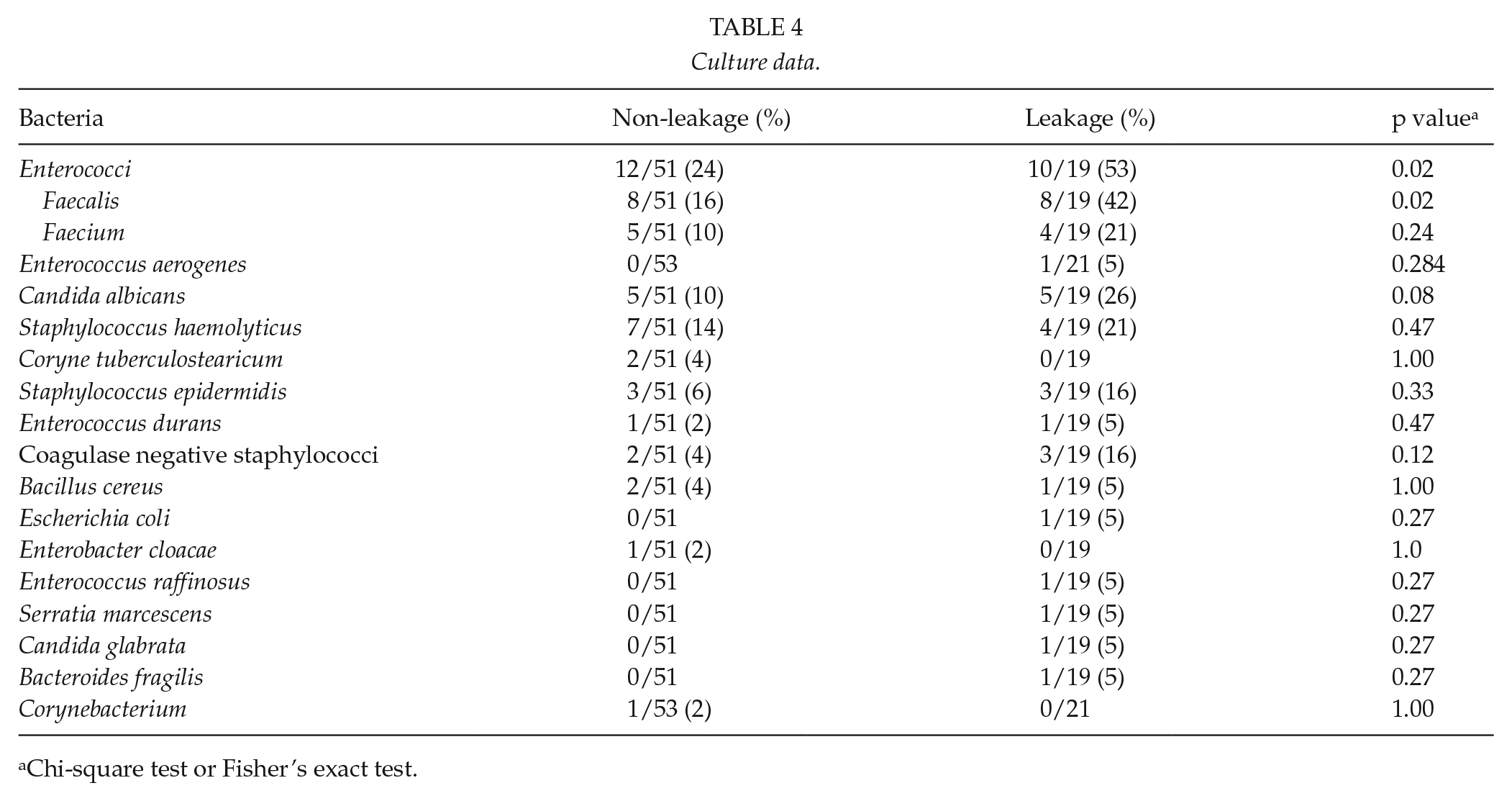
| Bacteria | Non-leakage (%) | Leakage (%) | p value a |
|---|---|---|---|
| Enterococci | 12/51 (24) | 10/19 (53) | 0.02 |
| Faecalis | 8/51 (16) | 8/19 (42) | 0.02 |
| Faecium | 5/51 (10) | 4/19 (21) | 0.24 |
| Enterococcus aerogenes | 0/53 | 1/21 (5) | 0.284 |
| Candida albicans | 5/51 (10) | 5/19 (26) | 0.08 |
| Staphylococcus haemolyticus | 7/51 (14) | 4/19 (21) | 0.47 |
| Coryne tuberculostearicum | 2/51 (4) | 0/19 | 1.00 |
| Staphylococcus epidermidis | 3/51 (6) | 3/19 (16) | 0.33 |
| Enterococcus durans | 1/51 (2) | 1/19 (5) | 0.47 |
| Coagulase negative staphylococci | 2/51 (4) | 3/19 (16) | 0.12 |
| Bacillus cereus | 2/51 (4) | 1/19 (5) | 1.00 |
| Escherichia coli | 0/51 | 1/19 (5) | 0.27 |
| Enterobacter cloacae | 1/51 (2) | 0/19 | 1.0 |
| Enterococcus raffinosus | 0/51 | 1/19 (5) | 0.27 |
| Serratia marcescens | 0/51 | 1/19 (5) | 0.27 |
| Candida glabrata | 0/51 | 1/19 (5) | 0.27 |
| Bacteroides fragilis | 0/51 | 1/19 (5) | 0.27 |
| Corynebacterium | 1/53 (2) | 0/21 | 1.00 |
Chi-square test or Fisher’s exact test.
Acknowledgements
The authors acknowledge the Danish Cancer Society for the grant support allotted to this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
A grant was provided by the Danish Cancer Society (to Mohamed Belmouhand, B.Sc.; No. R136-A8486-15-S7).
