Abstract
Background and Aims:
To report the experience of a tertiary vascular surgery center using Omniflow II® biosynthetic vascular grafts for treatment of prosthetic aortic graft infection.
Materials and methods:
Retrospective analysis of all patients with prosthetic graft infections who underwent in situ aortic reconstruction using Omniflow II® grafts or other conduits between March 2015 and May 2017. Early and late mortality, perioperative complications, and reinfection rate were analyzed.
Results:
Sixteen patients (14 males, median age 68.5, range 57–89) with prosthetic aortic graft infection were treated at our center. Eight patients received an Omniflow II® biosynthetic graft, two patients silver-triclosan coated grafts, three patients bovine pericardial tube grafts, and three patients composite bovine pericardial tube grafts with Omniflow II® graft extensions. Perioperative complications occurred in seven patients (43.8%). Early mortality rate was 18.7% (n = 3). In addition, four patients died during follow-up after a median of 11 months (range 0–34 months). We did not observe any reinfections. Bypass grafts were patent in all patients. No major limb amputations were performed during follow-up.
Conclusion:
Treatment of prosthetic aortic graft infection with Omniflow II® vascular grafts is feasible. Graft material seems to have an excellent resistance to infection and might be a valuable alternative to traditional replacement materials. Especially long-term durability has to be continuously monitored and documented.
Introduction
Despite the improvements in perioperative management, surgical techniques, and vascular grafts, there are still quite a number of infected arterial grafts in the aorto-iliac position (1, 2) facing vascular surgeons with difficult decisions. One of them is which replacement material might be the best in terms of reinfection rate, durability, accessibility, and surgical trauma (3). There are several replacement materials including coated synthetic grafts, arterial allografts, and femoral vein grafts as well as ex-situ and in situ replacement strategies (4, 5). Recently, different groups reported encouraging results with the use of xenografts as pericardial patches or biosynthetic grafts to replace infected prostheses in different arterial positions (6–9). In this case series, we discuss the use of the Omniflow II® biosynthetic graft to treat aortic prosthetic graft infection especially concerning feasibility and technical aspects of handling and application.
Material and Methods
Using the database of our institution, we identified all patients with cavitary infections of prosthetic abdominal aortic grafts (Szilagyi 3) (10) between March 2015 and May 2017. All patients who underwent total graft excision and replacement by Omniflow II® grafts (LeMaitre Vascular, Sulzbach, Germany) were analyzed (group 1). Patients with prosthetic aortic graft infection who received other conduits were also examined (group 2). Other conduits used were silver-triclosan coated grafts (Intergard Synergy®, Maquet, France), xenografts (bovine pericardial tube grafts, Vascu-Guard®, Lamed, Germany), or composite xenografts with Omniflow II® graft extensions (LeMaitre Vascular, Sulzbach, Germany).
Diagnosis of aortic graft infection was made by clinical, sonographic, and radiologic means in combination with intraoperative findings and microbiologic culture results. If required, an additional PET CT-scan was performed to confirm diagnosis. Patients with suspected aortoenteric graft erosion received endoscopy (11).
Treatment modality was determined on a case by case basis. All patients received duplex ultrasound to determine the suitability of the femoral vein. Femoral veins with a diameter smaller than 6 mm or a history of deep vein thrombosis were not used as graft material. If an appropriate femoral vein was not available, we used biosynthetic grafts, xenografts, or silver-triclosan coated grafts. Patients with septicemia, aortoenteric fistula, or extensive intra-abdominal abscess or perigraft purulence received biosynthetic grafts or xenografts. The other patients received silver-triclosan coated grafts.
Surgical treatment was done in two teams. One team performed the preparation and the removal of the infected graft, the other team tailored the biosynthetic prosthesis back table. After a full length abdominal midline-incision, aorta was dissected above the site of the proximal anastomosis to achieve bleeding control. Then distal anastomoses of the infected graft were exposed. Patients were heparinized and cross clamps were placed at the proximal and distal anastomotic sites. All infected graft material was removed carefully and a radical debridement of the surrounding tissue was performed followed by an extensive irrigation with HOCl solution (Granudaycn®, Sastumed). Prior to cross-clamping, Omniflow II® grafts (8 mm diameter, length 20–45 cm; LeMaitre Vascular, Sulzbach, Germany) were prepared according to the instructions for use. For the bifurcated grafts, two Omniflow-II®-Tubes were connected together proximally by running polypropylene sutures (5.0 Optilene, Braun, Melsungen, Germany). Using an end-to-end anastomosis, a cuff also made out of Omniflow II® was sewn in to eliminate the problem of size mismatch at the proximal anastomotic site. Proximal anastomosis was performed end-to-end or end-to-side to the infrarenal aorta by continuous polypropylene sutures (4.0 Optilene, Braun, Melsungen, Germany). After completion of the central anastomosis, grafts were passed retroperitoneally to the distal iliac or femoral anastomotic sites. Grafts were cut to an appropriate length and anastomosed by running polypropylene sutures (5.0 Optilene, Braun, Melsungen). Following the instructions for use, grafts were not clamped but only occluded with occlusion balloons.
In patients with an aorto- (prosthetic-) duodenal fistula, the duodenal defects were sparingly resected as a tube. The proximal jejunum was mobilized and the duodenojejunostomy was performed side-to-side with the descending part of the duodenum just distal to the ampulla of Vater. Whenever possible, an omental flap was used to cover the abdominal reconstruction with viable autologous tissue. Patients with groin infection received sartorius muscle flaps and a negative wound pressure therapy was administered. After 4–10 days, femoral incisions were closed secondarily or a split skin graft transplantation was done.
With suspected diagnosis, serial aerobic and anaerobic blood culture samples were taken even if the patient was already on antibiotic treatment prior to admission. Calculated antibiotic treatment with broad-spectrum antibiotics was started intraoperatively after collection of culture samples from the infected prosthesis and the surrounding tissue according to our hospital guidelines for vascular graft infections. As soon as possible, treatment regimens were adapted to the cultural findings and the resistance screening. Intravenous therapy was changed to oral after 2 weeks and continued for 6 weeks. Prior to discontinuation, a clinical assessment and laboratory tests for c-reactive protein and leucocyte-count were done. Patients were seen in an outpatient clinic on a regular basis every 3–6 months. All patients received permanently a single platelet aggregation inhibition with acetylsalicyclic acid.
Data acquisition was performed retrospectively from patient medical records including clinical symptoms at presentation, comorbidities, and perioperative management. Study endpoints included postoperative complications, survival, reinfection rate, and graft-related complications (e.g. rupture, graft occlusion, graft degeneration). Due to the small number of patients, no statistical analysis was conducted. Early mortality was defined as death ⩽30 days postoperatively and late mortality as death ⩾30 days postoperatively. The study was approved by the local ethics committee. All patients gave informed consent.
Results
Between March 2015 and May 2017, 16 patients (14 males, median age 68.5, range 57–89) with prosthetic aortic graft infection were treated at our center. Eight patients received an Omniflow II® biosynthetic graft, two patients silver-triclosan coated grafts, three patients bovine pericardial tube grafts, and three patients composite bovine pericardial tube grafts with Omniflow II® graft extensions. Indication for index open aortic repair was arteriosclerotic occlusive disease in 10 cases and aneurysmal disease in 6 cases. Median time between index operation and presentation was 4.2 years (range 0–19.6 years). There were 5 patients with early-onset infection (<4 months after index procedure) and 11 patients with late-onset infection (>4 months after index procedure). Patient characteristics and comorbidities are shown in Table 1.
Patient characteristics and comorbidities.
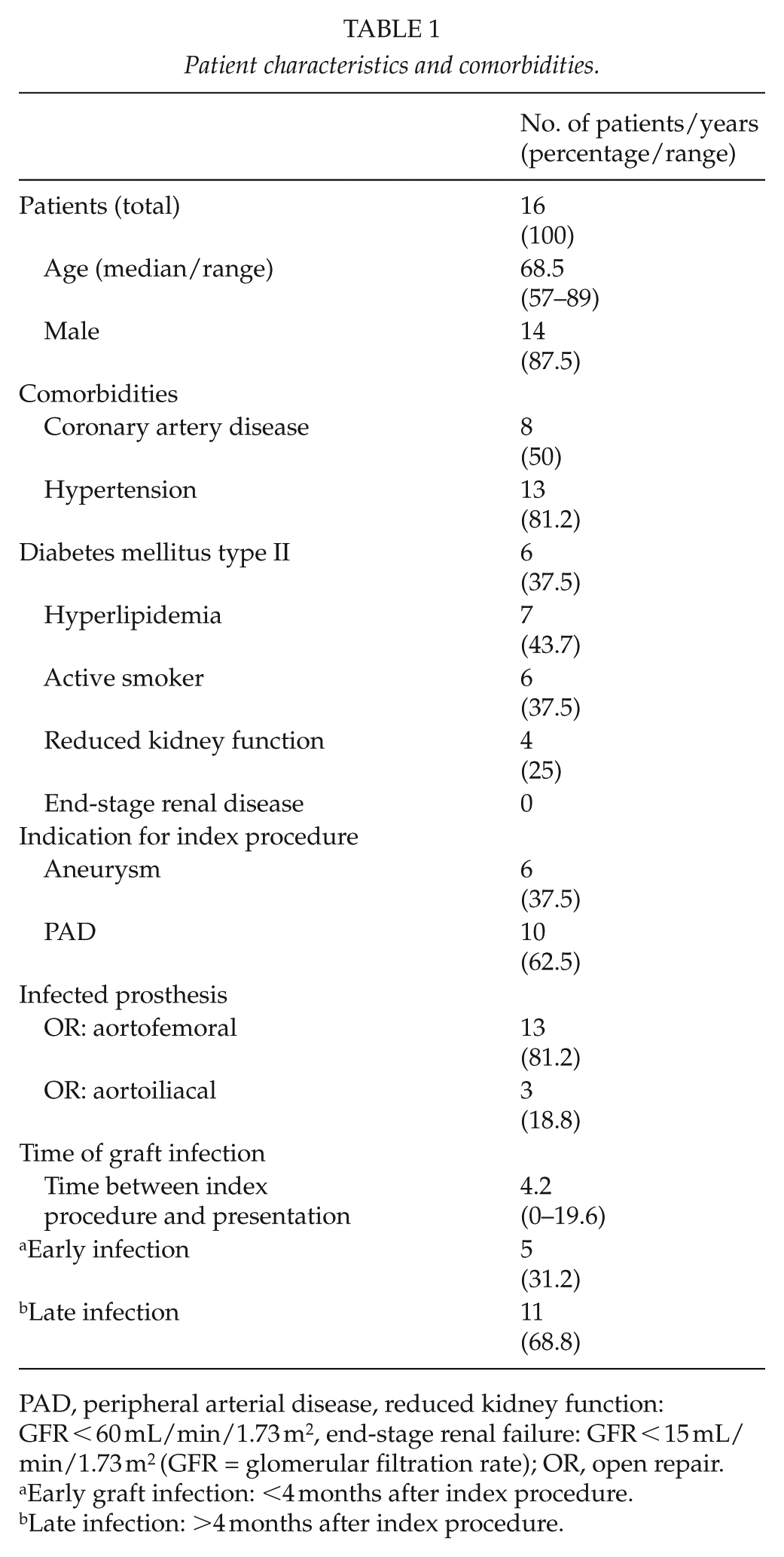
PAD, peripheral arterial disease, reduced kidney function: GFR < 60 mL/min/1.73 m2, end-stage renal failure: GFR < 15 mL/min/1.73 m2 (GFR = glomerular filtration rate); OR, open repair.
Early graft infection: <4 months after index procedure.
Late infection: >4 months after index procedure.
Fourteen patients presented with clinical signs of systemic illness. In 12 cases, CT-A showed perigraft fluid and/or presence of periaortic air. In 3 cases, diagnosis was uncertain and patients received PET-CT to confirm diagnosis. Three patients presented with a wound infection in the groin. In 4 cases, CT-A showed a loss of fat plane between the bowel and the prosthetic graft. In these cases, an upper gastrointestinal endoscopy was performed. In one case, aortoenteric fistula was assumed preoperatively; in the other cases, endoscopy was unremarkable and diagnosis was made intraoperatively.
Aerobic and anaerobic blood culture samples were negative in all patients. Culture samples of intraoperative specimens were positive in 13 patients. Among these, 10 specimens were monomicrobial and 3 specimens were polymicrobial culture samples. Isolated organisms included Staphylokokkus aureus, Staphylokokkus hominis, Staphylokokkus epidermidis, E. coli, Enterococcus faecalis, Klebsiella pneumoniae, Klebsiella oxytoca, Enterobacter cloacae, Citrobacter coseri, Propionibacterium acnes, Lactobacillus rhamnosus, and Bacteriodes fragilis. There were 3 patients where no causative organisms could be identified (Table 2). No intraoperative deaths were observed. Median length of hospital stay was 29 days (range 10–78 days). Median length of stay on the intensive care unit was 9 days (range 1–60 days).
Patient presentation, treatments, and outcomes.
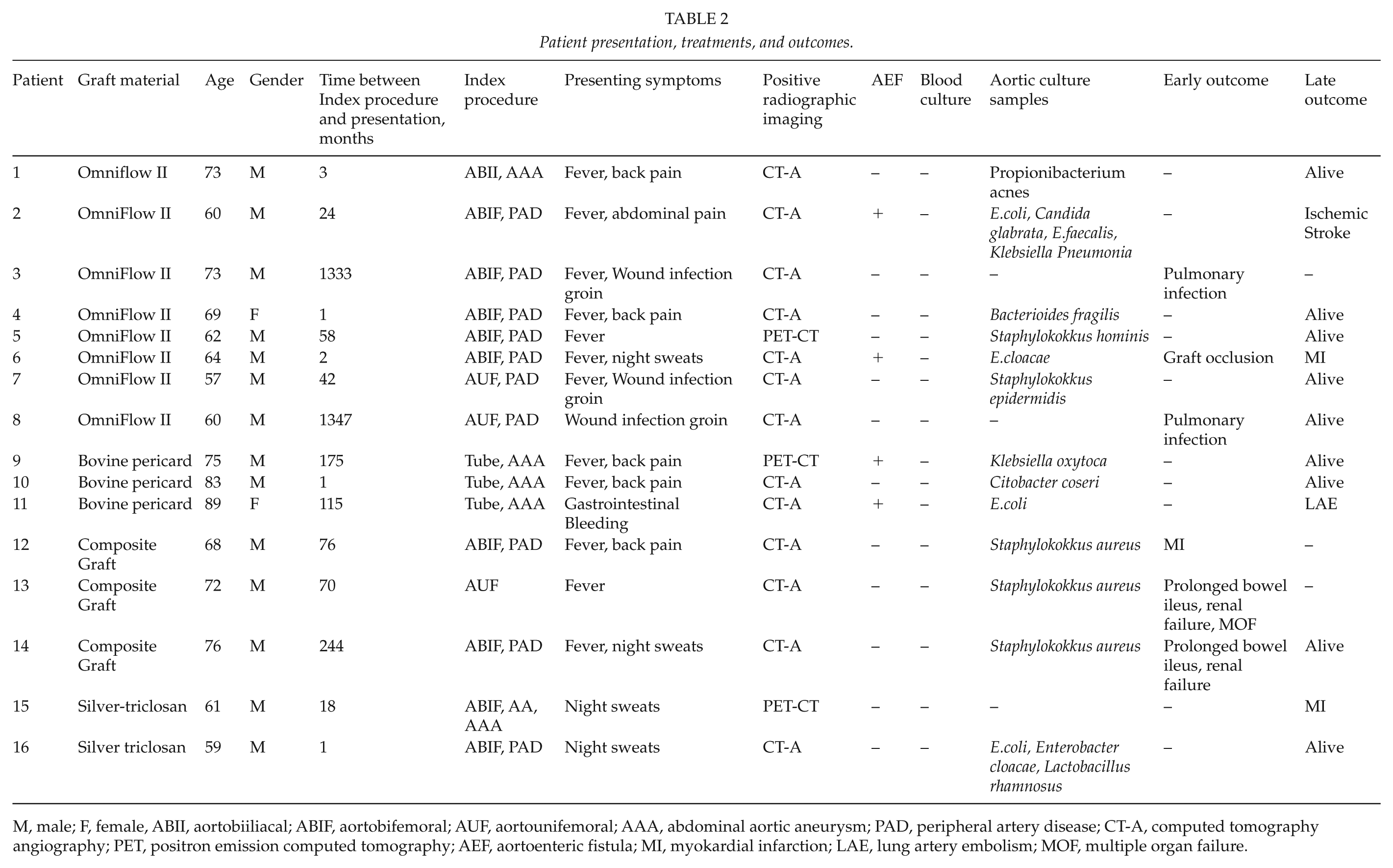
M, male; F, female, ABII, aortobiiliacal; ABIF, aortobifemoral; AUF, aortounifemoral; AAA, abdominal aortic aneurysm; PAD, peripheral artery disease; CT-A, computed tomography angiography; PET, positron emission computed tomography; AEF, aortoenteric fistula; MI, myokardial infarction; LAE, lung artery embolism; MOF, multiple organ failure.
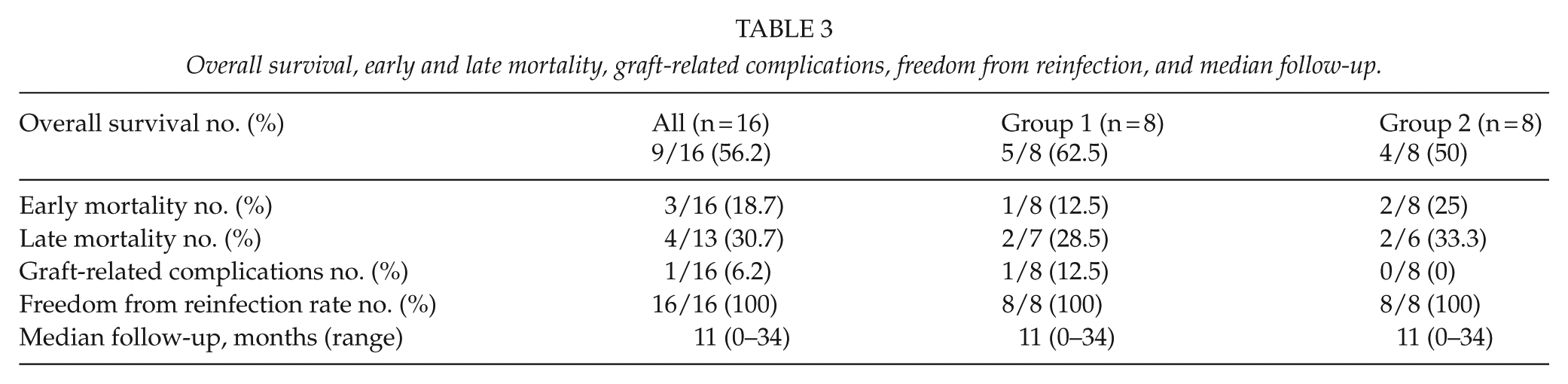
For the whole cohort of patients, the perioperative mortality rate was 18.7% (3/16). One patient from group 1 and two patients from group 2 died. The causes of death were myocardial infarction (n = 1), pulmonary infection (n = 1), and multiple organ failure due to persistent septicemia (n = 1). Postoperative complications occurred in seven patients (43.8%). Two perioperative complications occurred in group 1 (12.5%) and five perioperative complications (62.5%) in group 2. Perioperative complications were pulmonary infection (n = 2), prolonged bowel ileus (n = 2), renal failure (n = 2), and acute graft occlusion (n = 1).
Median follow-up was 11 months (range 0–34 months). No patients were lost to follow-up. Four additional patients (two patients per group) died during follow-up. The causes of late death were ischemic stroke (n = 1), myocardial infarction (n = 2), and pulmonary embolism (n = 1). During follow-up, no graft rupture or graft degeneration occurred. We did not observe any reinfections. Bypass grafts were patent in all patients. No major limb amputations were performed during follow-up. Overall survival, early and late mortality, graft-related complications, freedom from reinfection, and median follow-up are depicted in Table 3.
Overall survival, early and late mortality, graft-related complications, freedom from reinfection, and median follow-up.
Discussion
Aortic graft infection following arterial reconstructive surgery is a rare but serious complication. Depending on treatment strategy, authors reported an amputation rate between 10% and 40% and a mortality rate between 30% and 60%, respectively (12). Surgical treatment depends on patient clinical conditions, the virulence of the causative organism and the occurrence of an aortoenteric graft erosion (13). Several conduits including arterial allografts, autologous veins, or prosthetic grafts coated with silver or antibiotics as well as ex-situ and in situ replacement strategies are used to prevent reinfection. To date, all studies that have been published were retrospective and observational. So, it is currently not possible to draw firm conclusions about the optimal graft material in terms of reinfection rate, durability, accessibility, and surgical trauma (3). Despite these problems, a previous systematic review and meta-analysis by O’Connor et al. showed that extra-anatomic bypass has several disadvantages in comparison to any in situ reconstruction. Authors questioned whether an extra-anatomic approach should remain the gold standard for treatment of infected vascular grafts (14). Furthermore, authors demonstrated the lowest reinfection rate for biological conduits in comparison to rifampicin bonded grafts and extra-anatomic bypass grafts (14). These findings were consistent with a recent meta-analysis from Batt et al. comparing biological conduits with Standard polytetrafluoroethylene (PTFE)/polyester, rifampicin bonded grafts and silver impregnated grafts. However, reinfection rate was not significantly different between each type of conduit, except for Standard PTFE/polyester, which had the poorest results (5). In our cohort, early mortality was 12.5%. Batt et al. (5) reported operative mortality rates of 12%, 10%, and 11% for rifampicin, autologous veins, and standard polyester/PTFE treatments, whereas early mortality rates of cryopreserved allografts and silver-coated prostheses were significantly higher. Unfortunately, our late mortality rate of 28.5% was not satisfactory. However, we observed no procedure-related deaths. Furthermore, no graft degeneration or graft rupture was observed in our cohort during short time follow-up and graft thrombosis rate was 12.5%. In literature, rupture rates of biological conduits, especially of arterial allografts, vary between 2% and 7% with a 5%–25% risk for aneurysmatic degeneration (15, 16) and graft occlusion rate ranges from 2%–13% depending on the graft material used (5). On the contrary, biological conduits show low reinfection rates and autologous vein for in situ reconstructions of infected grafts is claimed to be the most effective means of avoiding reinfection (5, 14). Previous studies demonstrated excellent results with the use of femoral vein grafts with no incidence for reinfection during follow-up (17, 18). However, there might be several disadvantages associated with the use of autologous veins, including lengthier operative time, extensive surgical trauma as well as technical limitations (14). Furthermore, a recent study described a venous morbidity due to the vein harvesting itself (19).
Several studies have demonstrated that biosynthethic grafts might be a valuable conduit to treat prosthetic graft infections (20). A recent study by Töpel et al. (21) showed no reinfection during a follow-up of 24 months using biosynthetic Omniflow II® grafts to replace infected infrainguinal synthetic vascular grafts. Similar findings were reported by Wiltberger et al. (7). However, the number of studies about biosynthetic grafts in aorto-iliac position is currently limited. Krasznai et al. presented first results after replacement of infected infrarenal prosthesis using an Omniflow II® graft alone or in combination with an arterial allograft in three patients. During follow-up, one reinfection, but no graft degeneration, occurred (22). In our cohort, no composite grafts were used. We could achieve freedom from reinfection in all cases during follow-up. By contrast, a recent animal study reported a higher susceptibility to infection in a biosynthetic graft compared to PTFE (23). However, in this experimental study, authors used a bacterial strain of methicillin resistant Staphylokokkus aureus. To date, no study has evaluated the role of multidrug resistant pathogens in treatment of graft infections using biosynthetic prosthesis in human beings. In our study, no multidrug resistant pathogen was isolated. Therefore, further studies are needed to supply reliable evidence. We think that besides the removal of the infected graft and the radical debridement followed by in situ reconstruction also an appropriate antibiotic therapy is the key to successful treatment of aortic graft infection. According to the literature and to the regular microbiological spectrum found at our hospital facility, our initial calculated antibiotic therapy consisted of a betalactam in combination with a glykopeptid followed by a test-appropriate or calculated long-term antibiotic therapy (24). This study is the first case series using biosynthetic OmniFlow II® prosthesis for replacement of infected aortic grafts. Although there are limitations in interpretation due to the small number of patients and the short follow-up period, reinfection rates seem to be excellent and early morbidity and mortality rates are in the expected range to those observed with other grafts materials. Because they can be held in stock, used without time-delay or harvesting trauma, and handled easily during vascular procedures, they might be a valuable alternative to traditional replacement materials.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
