Abstract
Background and Aims:
Managing infrainguinal vascular graft infections (VGI) in bypass reconstruction is complex. Although an autologous vein is the preferred graft material, alternative conduits are required when autologous veins are unavailable. This study aimed to compare the efficacy of cryopreserved arterial homografts and biosynthetic glutaraldehydemodifiedovine collagen grafts (Omniflow II) for the treatment of infrainguinal VGI.
Methods:
The study was retrospective across two centers and included patients with infrainguinal VGI treated with cryopreserved arterial homografts or Omniflow II grafts between 2009 and 2019. Freedom from reinfection, primary patency rates, amputation-free survival, and overall survival were analyzed using 5-year Kaplan-Meier estimates.
Results:
Overall, 63 patients with infrainguinal VGI were treated with either Omniflow II grafts (n = 34) or cryopreserved arterial homografts (n = 29). At 5 years, freedom from reinfection was 97.1% for Omniflow II grafts and 93.1% for cryopreserved arterial homografts (p = .4). Primary patency was 50% for Omniflow II grafts and 55.2% for the cryopreserved arterial homografts (p = .5). Amputation-free survival was 52.9% for Omniflow II grafts and 55.2% for cryopreserved arterial homografts (p = .7). No graft degeneration or graft ruptures were observed during the follow-up.
Conclusions:
This study suggests that biosynthetic grafts are viable and accessible alternatives for traditional graft materials, offering similar efficacy and ease of use. Although autologous vein grafts remain the gold standard for VGI management, biosynthetic grafts may serve as a viable alternatives to cryopreserved arterial homografts in the treatment of infrainguinal VGIs.
Keywords
Context and Relevance
Sixty-three patients with infrainguinal vascular graft infections were treated with either Omniflow II grafts (n = 34) or cryopreserved arterial homografts (n = 29).
At 5 years, freedom from reinfection was 97.1% for Omniflow II grafts and 93.1% for cryopreserved arterial homografts (p = .4).
Primary patency was 50% for Omniflow II grafts and 55.2% for the cryopreserved arterial homografts (p = .5).
Amputation-free survival was 52.9% for Omniflow II grafts and 55.2% for cryopreserved arterial homografts (p = .7).
Introduction
Vascular graft infections (VGIs) after peripheral bypass surgery are rare but serious complications. The reported perioperative amputation and mortality rates are 52% and 58%, respectively. 1 Moreover, VGIs substantially increase healthcare expenditures owing to the necessity for further interventions and prolonged hospitalization periods. 2 Surgical therapy consists of several key steps, including removal of the infected graft, debridement of the adjacent tissue, in situ reconstruction (ISR) of the affected vessel using an autologous vein, and subsequent coverage of the reconstruction with viable biological tissue. 3 However, autologous veins for ISR are not always available, particularly in cases following prior vascular or cardiac surgery. Currently, alternative graft materials, for example, cryopreserved arterial homografts or silver-coated prosthetic grafts, are used for ISR. Cryopreserved arterial homografts have demonstrated a favorable track record, with a low incidence of reinfection and a reduced rate of major amputations. 3 Nonetheless, it is important to recognize that the availability of cryopreserved homografts may not always be guaranteed, and these grafts can potentially experience known degenerative issues, leading to significant long-term complications. 4 In contrast, silver-coated prosthetic grafts are readily available but have higher rates of reinfection, occasionally reaching approximately 19%. 5 Recently, several authors reported promising results in the treatment of vascular graft infections using biosynthetic grafts.6,7 However, the number of studies comparing biosynthetic grafts with other graft materials for the treatment of infrainguinal VGI is limited. Therefore, this study aimed to evaluate the outcomes of Omniflow II grafts compared to those of cryopreserved human homografts in patients with infrainguinal VGI and no autologous vein.
Methods
Ethics
This study was approved by the local ethics committee of the University Medical Center Regensburg before data collection (Approval Number 14-101-0017). Written informed consent was obtained from all participants included in this study.
Study population
A retrospective analysis of all patients with infrainguinal VGI between January 2009 and March 2019 at two high-volume tertiary vascular centers was performed. Data were obtained from a prospective clinical database. Analytical procedures were performed by a vascular surgeon and a clinical research nurse, with validation by an additional vascular surgeon to ensure data integrity. The following information was recorded: patient characteristics, operative details, postoperative complications, and follow-up information. Patients were divided in two groups based on the graft material: those with Omniflow II biosynthetic grafts (group 1) and those with cryopreserved arterial homografts (group 2).
Diagnosis of VGI and surgical technique
The diagnosis of VGI was based on clinical symptoms, laboratory findings, and radiological evidence obtained from computed tomography angiography. When required, 18F-fluorodeoxyglucose positron emission computed tomography was performed for further confirmation. 8 A multidisciplinary team (MDT), including vascular surgeons, radiologists, and infectious disease specialists, comprehensively reviewed each clinical scenario.
Upon admission, patients were expeditiously triaged and scheduled for surgical intervention, ensuring that operative procedures were initiated within a 24 h timeframe of admission. The uniform use of a specific graft type across each center contributed to the development of substantial expertise in its deployment. The surgical regimen included the removal of the infected graft, followed by in situ reconstruction. The choice to employ a single-segment graft or a composite graft, potentially extended with an autologous vein segment, depended on the need for infrapopliteal revascularization. Cases requiring extended graft lengths were managed either by consecutively anastomosing two homografts or by creating a composite graft, which involved combining Omniflow II with a vein graft.
The preparation of the Omniflow graft adhered to the instructions for use described by the manufacturing entity. 9 Concurrently, the cryopreserved homografts were methodically thawed and processed in accordance with the procedural guidelines issued by the German Society for Tissue Transplantation. 10 In this study, patients receiving homografts did not require systemic immunosuppressive therapy. Standard postoperative care, along with prophylactic antibiotic protocols, was employed to reduce the risk of infection. The homografts were cryopreserved to maintain long-term storage and availability. During the cryopreservation process, DMSO (Di-Methyl-Sulfoxide) was utilized as a cryoprotectant to prevent ice crystal formation and preserve tissue integrity during freezing. The grafts were stored in a controlled environment and were thawed using standard protocols prior to implantation to ensure optimal tissue viability and functionality.
All patients underwent excision of the infected grafts and thorough debridement of the affected soft tissues with intraoperative irrigation using antimicrobial solutions. In instances where patients presented with tissue defects of the groin, muscle flaps (sartorius muscle transposition flap or rectus femoris muscle flap) were performed. Groin incisions were deliberately left incompletely closed to facilitate the assessment of flap vitality. These partially closed incisions were managed using negative pressure wound therapy. After 5–7 days, the incisions were closed, or split skin mesh graft transplantation was performed. All cryopreserved homografts and Omniflow vascular grafts were implanted at two designated centers, and there was no crossover.
Reinfection is defined as the recurrence of infection at the same anatomical site following an initial treatment, after a period of clinical resolution. Postoperative infections, including cases where sepsis persisted postoperatively or where the new graft became infected immediately, were not considered reinfections. Instead, these were classified as continuous infections. This definition includes instances where the patient experienced a return of clinical symptoms or positive microbiological cultures indicating the same or a new pathogen within a specified period after the initial infection was considered resolved. No cases of early postoperative infections were observed in our study.
Antibiotic therapy
When a diagnosis was suspected, consecutive blood culture samples were collected upon admission. Following the collection of specimens from the infected graft and adjacent tissue during surgery, empirical intravenous antibiotic treatment was initiated. Subsequent antibiotic adjustments were made based on the microbiological results, resistance profiles, and recommendations from a microbiology expert. Intravenous antibiotic treatment was changed to oral therapy after normalization of the inflammation parameters. Antibiotic treatment was administered for a maximum of 6 weeks.
Postoperative data
Prior to hospital discharge, each patient underwent a clinical assessment and duplex ultrasound. Antiplatelet therapy consisted of 100 mg acetylsalicylic acid. For cases involving infrapopliteal bypass, lifelong oral anticoagulation therapy was initiated using phenprocoumon, targeting an International Normalized Ratio therapeutic range of 2–3. Follow-up appointments were scheduled every 3–6 months in an outpatient setting, where patients underwent clinical, laboratory, and duplex ultrasound assessments to check for signs of reinfection and evaluate graft patency. The complete follow-up protocol can be viewed under Table 1.
Follow-up protocol.

CRP: C-reactive protein; PSV: peak systolic velocity.
Study endpoints
The primary endpoint of the study was freedom from reinfection. Primary patency and amputation-free survival were secondary endpoints. Primary patency and amputation-free survival were defined according to the recommended standards for reports dealing with lower extremity ischemia. 11 The 30-day results included 30-day mortality, 30-day amputation, 30-day graft failure, and morbidity data. Thirty-day mortality was defined as death within 30 days of the operation. Thirty-day amputation was defined as any above-ankle amputation of the index limb within 30 days of the index operation. Thirty-day graft occlusion was defined as occlusion of the bypass graft within 30 days of the index procedure. The recorded morbidity data consisted of surgical site and medical complications. The last available data regarding their status were used in the study for the patients who became unavailable during the follow-up period.
Statistical methods
In this retrospective study, a priori sample size calculation was performed. We included as many patients as possible to maximize the power for first valid and reliable estimates for predefined clinically relevant endpoints when using biosynthetic collagen prostheses for infrainguinal graft infections.
All categorical variables are presented as absolute and relative frequencies, while age, white blood cell (WBC) count, C-reactive protein (CRP), and length of intensive care unit (ICU)/hospital stay are shown as median and interquartile range (IQR) and were compared using Mann–Whitney U test. Comparisons between graft materials were performed using an analysis of variance and Pearson’s chi-square test of independence for categorical variables. The median follow-up time was assessed using the reverse Kaplan–Meier method of overall survival (OS). Freedom from reinfection, primary patency, and amputation-free survival were defined from the date of bypass surgery to the date of the respective event. In the case of no event, the patient was censored at the last date, known as event-free. The impact of graft material on all endpoints was analyzed using the Kaplan–Meier method, and groups were compared using the log-rank test. Due to the small sample size and the low number of events, no multivariable Cox Regression models could be used. R software version 4.2.1 (The R Foundation for Statistical Computing) was used for statistical analyses.
Results
Patient characteristics
Between 2009 and 2019, 63 patients (38 males; average age 67.7 years) were treated for vascular graft infections with bypass reconstruction. Of these, 34 (20 males, average age 67.4 years) received an Omniflow II graft, referred to as group I, while 29 (18 males, average age 67.9 years) were treated with cryopreserved arterial homografts, referred to as group II. Clinical presentations included fever in 24 patients (38.1%), wound infection in 13 (20.6%), and active bleeding in 11 (17.5%). The median CRP level was 49.60 mg/dL, and the median WBC count was 9.30 g/L. Notably, group 1 had higher rates of hyperlipidemia (82.4% vs 27.6%, p = .001), smoking (61.8% vs 27.6%, p = .01), and chronic obstructive pulmonary disease (32.4% vs 3.4%, p = .01). Patient characteristics are summarized in Table 2.
Patient characteristics.
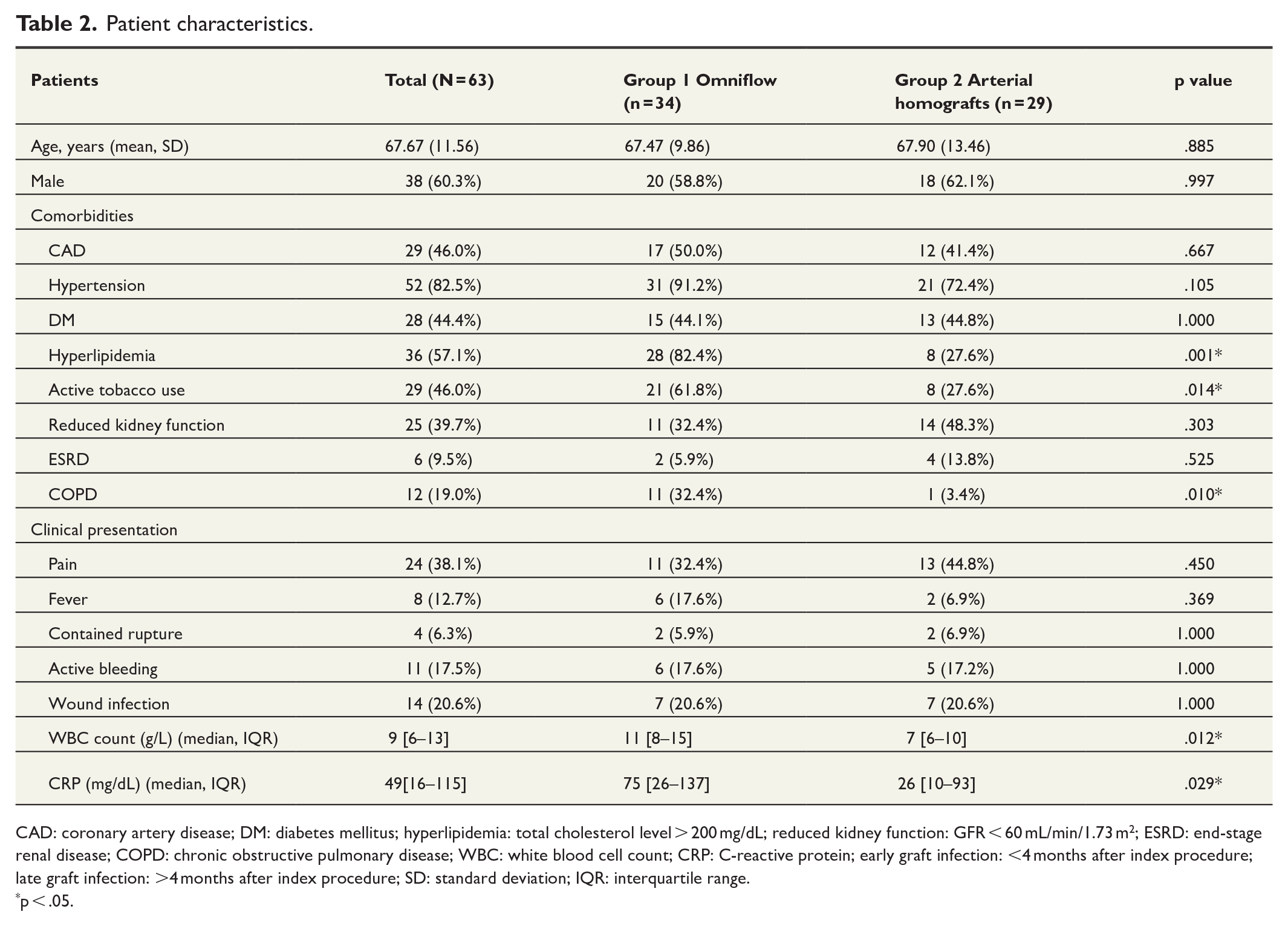
CAD: coronary artery disease; DM: diabetes mellitus; hyperlipidemia: total cholesterol level > 200 mg/dL; reduced kidney function: GFR < 60 mL/min/1.73 m2; ESRD: end-stage renal disease; COPD: chronic obstructive pulmonary disease; WBC: white blood cell count; CRP: C-reactive protein; early graft infection: <4 months after index procedure; late graft infection: >4 months after index procedure; SD: standard deviation; IQR: interquartile range.
p < .05.
Operative details
In group 1, 34 patients received 18 single-segment grafts with Omniflow II (52.9%), 2 combined grafts with Omniflow II (5.9%), and 14 composite grafts with autologous vein and Omniflow II (41.2%). In group 2, 29 patients received 12 single homografts (41.4%), 14 combined grafts with homografts (48.3%), and 3 composite grafts with homografts and autologous veins (10.3%). There was no statistically significant difference between graft localization across the groups. Muscle flaps were employed in 31 patients (91.2%) in group 1 and in 5 (17.2%) in group 2, with a statistically significant difference (p = .001). Cultures from intraoperative samples were positive in 28 patients (82.4%) in group 1 and 10 (34.5%) in group 2, with gram-positive bacteria identified in 27 patients (79.4%) in group 1% and 9 (31%) in group 2. Operative details and microbiologic findings are summarized in Table 3.
Operative details and microbiologic findings.
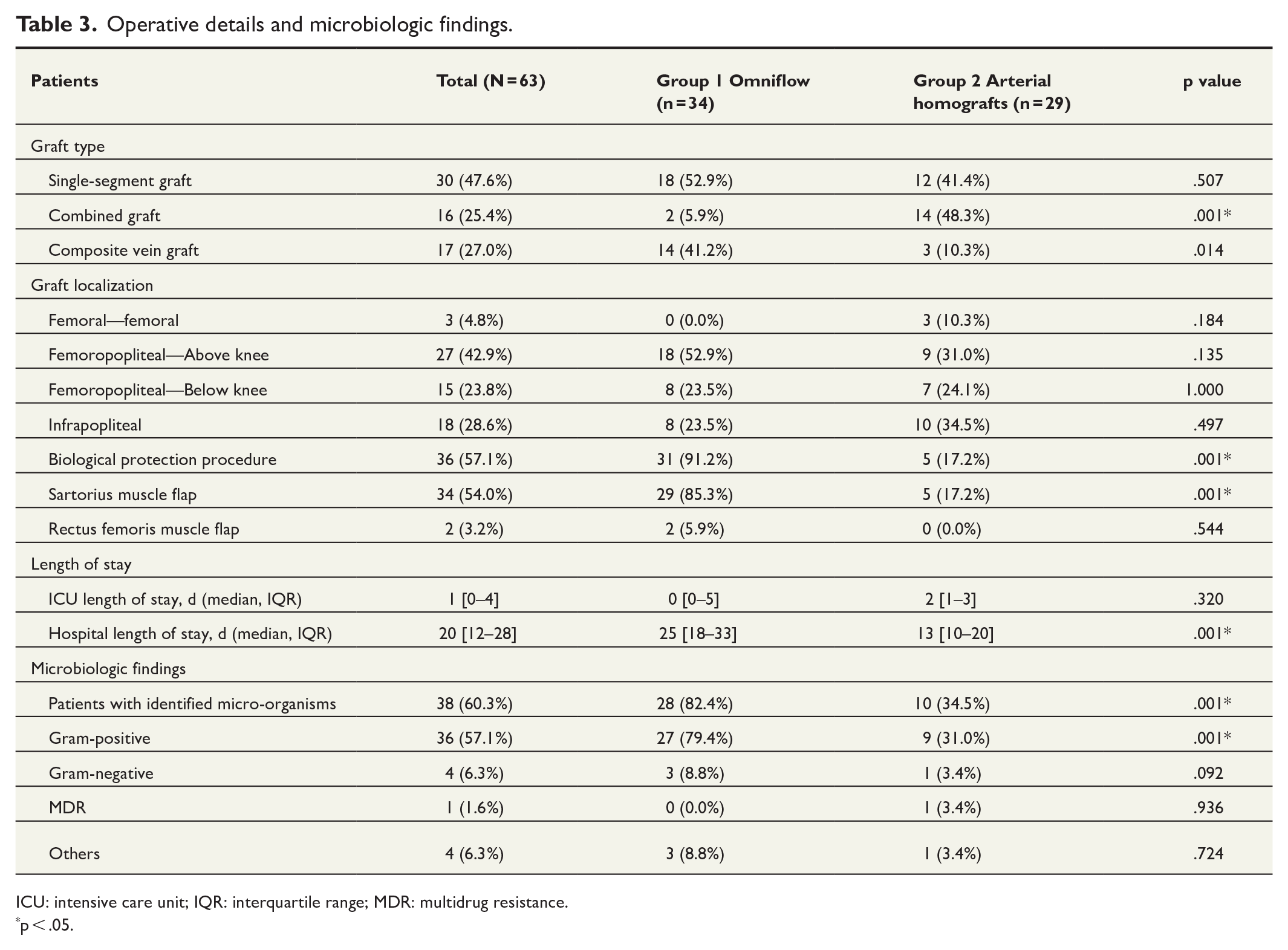
ICU: intensive care unit; IQR: interquartile range; MDR: multidrug resistance.
p < .05.
Thirty-day results
Overall, one patient died; thus, the 30-day mortality rate was 1.6%. A graft occlusion occurred in eight patients (12.7%), in six (17.6%) in group 1 and two (6.9%) in group 2. Major limb amputations due to graft occlusion occurred in two patients per group, corresponding to 5.9% in group 1 and 6.9% in group 2.
Postoperative complications included groin hematomas at the access site in three patients (8.8%) in group 1, which did not require further surgical correction. In group 2, four patients (13.8%) had superficial wound infections, with one patient requiring surgical intervention beyond antibiotic treatment. In this case, a superficial infection was surgically excised. The graft itself remained covered and unaffected.
Cardiac complications were also noted, with heart failure occurring in three patients (10.3%) in group 2. Respiratory complications were observed in both groups, with one patient (2.9%) in group 1 and two (6.9%) in group 2 developing pneumonia. Acute renal failure was reported in one patient in group 2 (3.4%), while another required transient dialysis. Overall three patients developed hospital acquired pneumonia, one in group 1 (2.9%) and two in group 2 (6.9%). Thirty-day mortality and morbidity data are summarized in Table 4.
Thirty-day mortality and morbidity data.
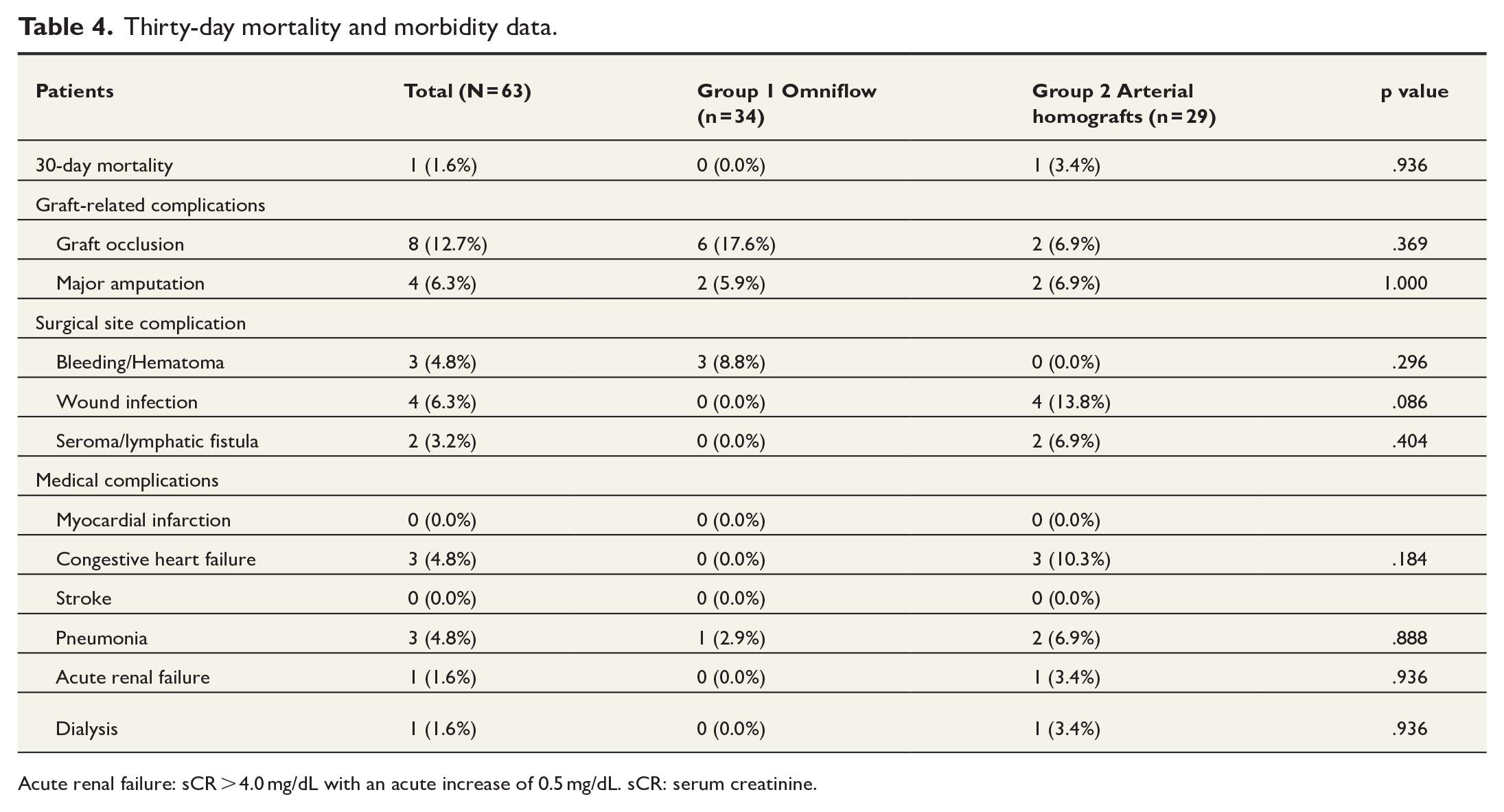
Acute renal failure: sCR > 4.0 mg/dL with an acute increase of 0.5 mg/dL. sCR: serum creatinine.
Long-term results
No patient was lost to follow-up. Freedom from reinfection is shown in Figure 1. Freedom from reinfection was 97.1% at 1, 3, and 5 years in group 1. Group 2 showed similar results, with 96.6%, 93.1%, and 93.1%, respectively (p = .4).
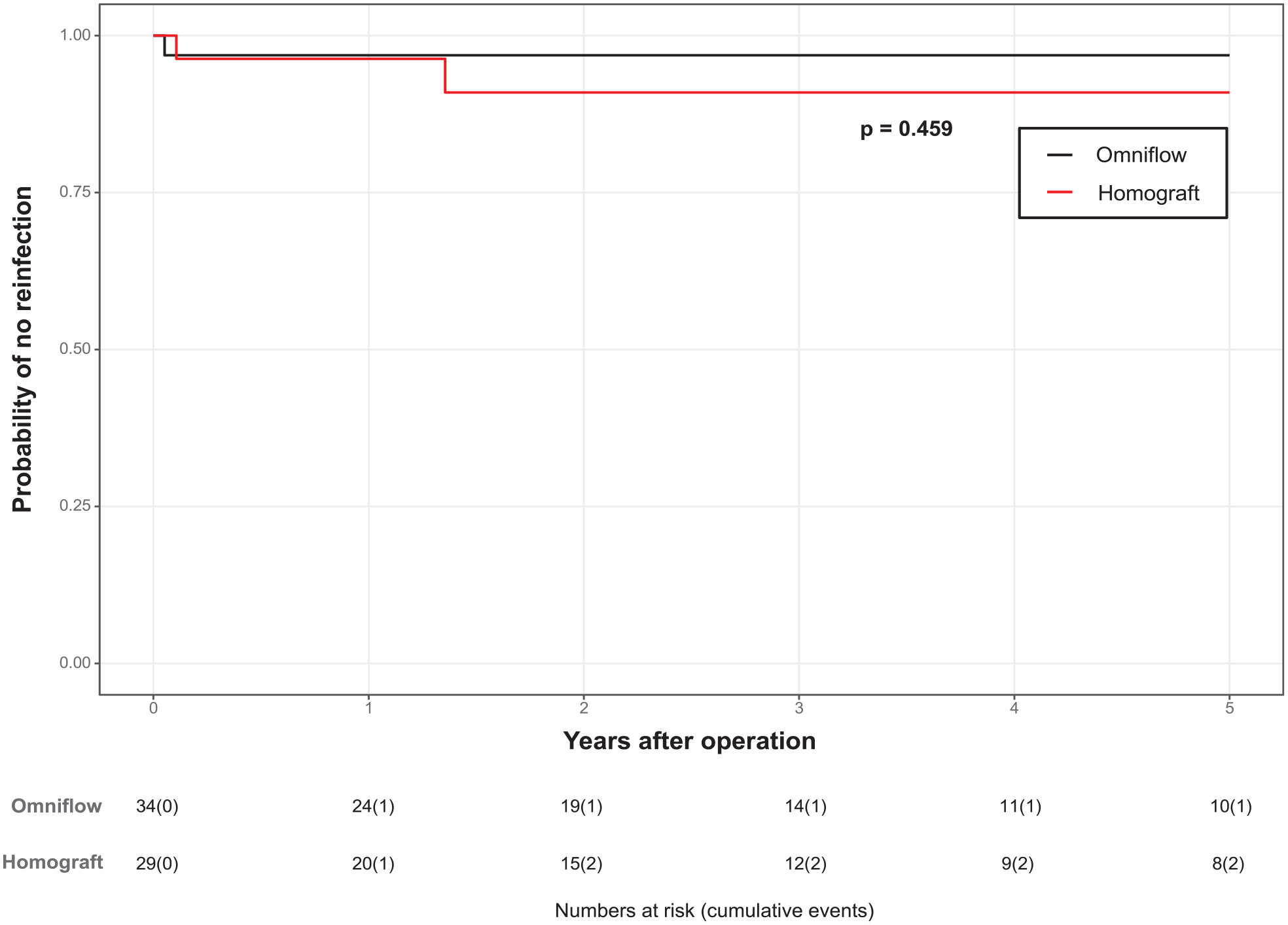
Freedom from reinfection.
The primary patency rates are shown in Figure 2. The primary patency rates in group 1 were 61.8%, 55.9%, and 50% at 1, 3, and 5 years, respectively. In group 2, the corresponding intervals were 72.4%, 58.6%, and 55.2%.
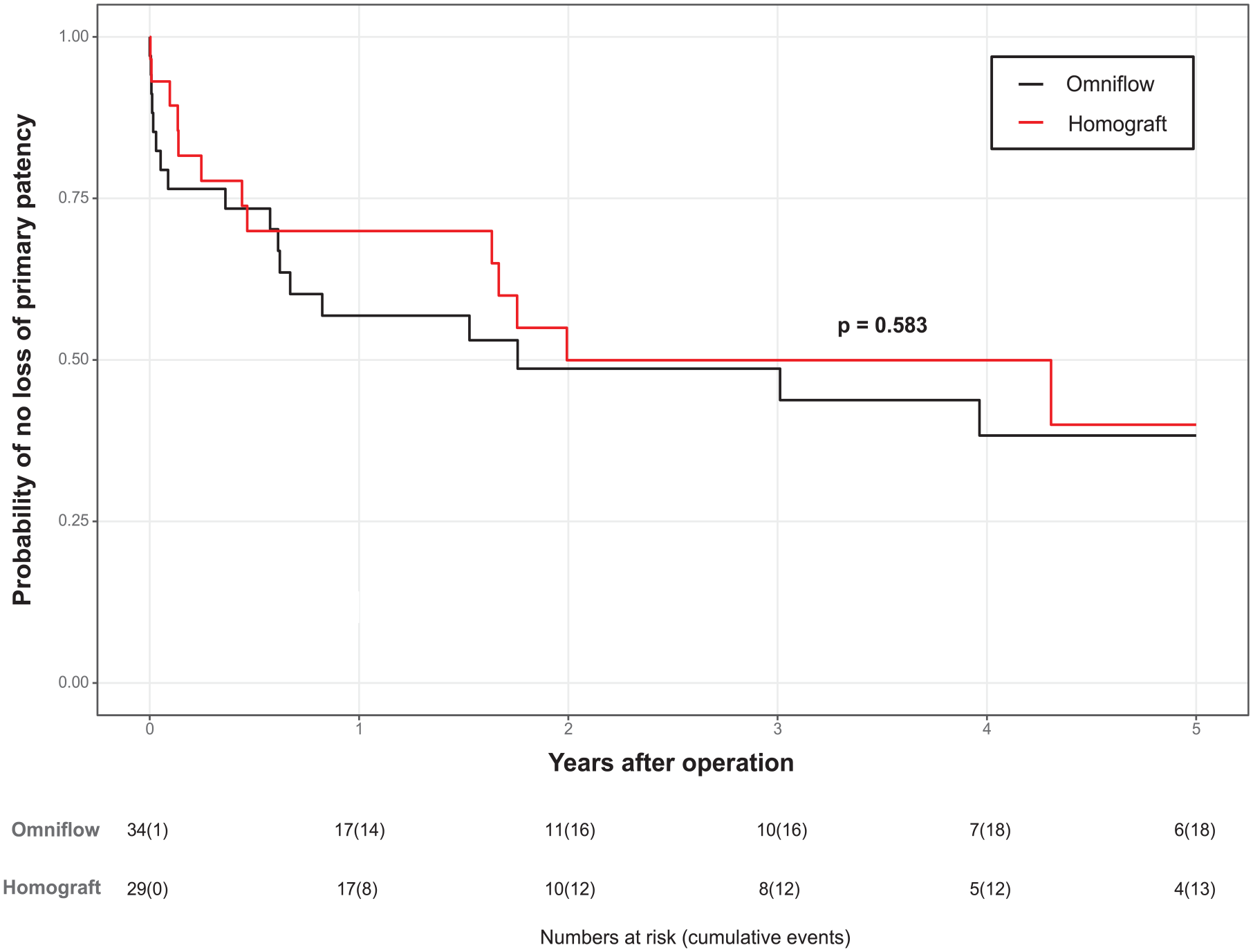
Primary patency rate.
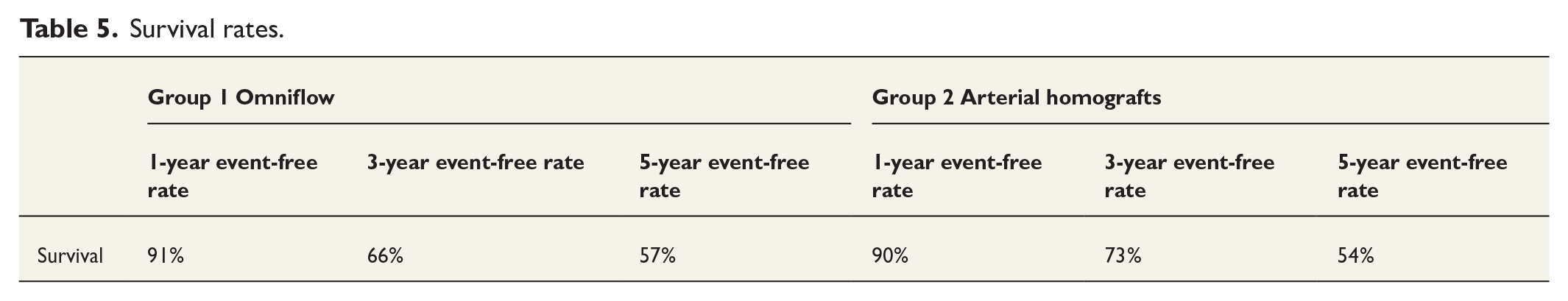
The amputation-free survival rates are shown in Figure 3. The amputation-free survival rates were 82.2%, 58.8%, and 52.9% in group 1 and 75.9%, 69.0%, and 55.2% in group 2. The overall survival rates are presented in Table 5. Survival rates at 1 year, 3 years, and 5 years were 91%, 66%, and 57% in group 1, and 90%, 73%, and 54% in group 2, respectively. The median follow-up period was 34 months. None of the differences between the groups were statistically significant.
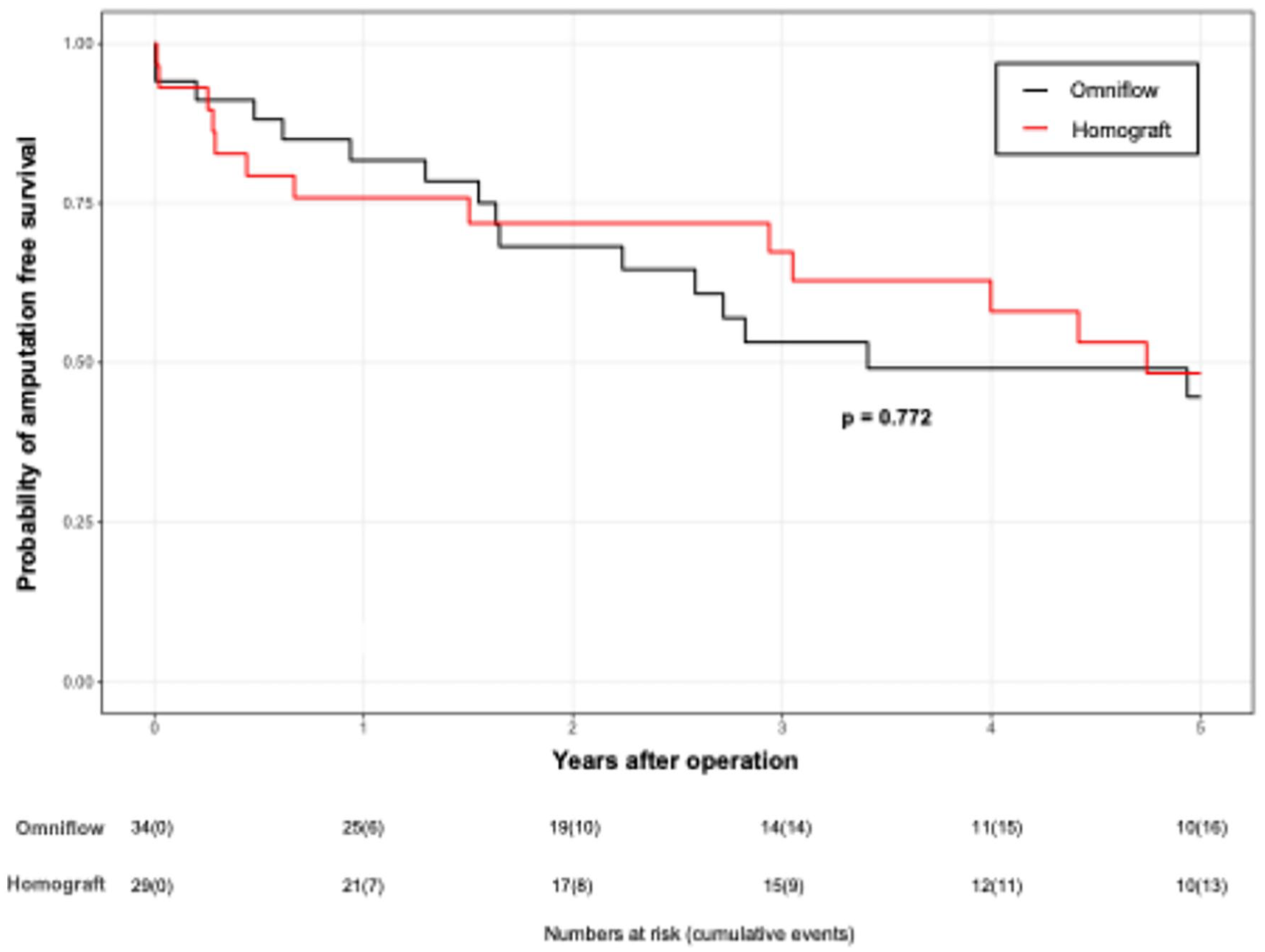
Amputation-free survival.
Survival rates.
Discussion
This study assessed the immediate and extended outcomes associated with two different graft materials in patients with infrainguinal VGI. Both materials demonstrated similarly low rates of reinfection with no long-term graft-related mortality. Given the absence of significant differences in 5-year outcomes and considering their availability, cost, and user-friendliness, Omniflow II grafts may be a suitable alternative to cryopreserved arterial homografts.
However, the gold standard for managing infrainguinal VGI is using autologous material, such as the greater saphenous or femoral vein, in suitable patients.1,12 Although superior survival rates and higher resistance to reinfection have been published with these graft material, harvesting the superficial femoral vein requires a longer operative time, can lead to venous hypertension in the affected limb in up to 15% of patients, and cannot be used for arterial reconstructions below the knee owing to size mismatch.13–15 In addition, 10%–40% of patients have no suitable autologous vessels left for reconstruction due to previous operations or trauma. 16 Cryopreserved arterial homografts have been an alternative for reconstructive surgeries addressing graft infections, provided they are arranged in advance. Nevertheless, the use of these graft materials in emergency reconstruction is limited by their scarcity, highlighting the need for an off-the-shelf alternative that can be deployed immediately.
Moreover, the economic burden associated with these grafts is typically greater than that associated with standard off-the-shelf products. In addition, there is a notable incidence of graft-related reinterventions, with literature documenting rates as high as 20% during extended follow-up periods. This is attributed to complications, including aneurysmal degeneration, graft dilation, and the risk of graft rupture. 17
Graft degeneration has also been reported in Omniflow II grafts, with studies indicating an aneurysmal degeneration rate ranging from 1% to 12.6% during extensive follow-up periods. 18 In contrast, no aneurysmal degeneration or graft dilatation was observed during follow-up in our study in neither of groups.
Efforts are ongoing to identify durable graft materials for the treatment of infrainguinal VGI in cases where the use of autologous veins is not an option or not feasible. Omniflow I and II grafts, first developed in 1992, are currently recognized as viable options for patients without adequate autologous veins.18,19 While an animal study by Bozoglan et al. 20 suggested increased susceptibility to methicillin-resistant Staphylococcus aureus infections, we do not have sufficient data on isolated bacteria to draw similar conclusions in our study. A recent in vitro study by Tello-Diaz highlighted that Omniflow II grafts possessed notable resistance to biofilm formation. Unfortunately, this study did not investigate the resistance properties of cryopreserved arterial homografts. 21
In addition, the rate of freedom from reinfection within 5 years was similar between the group treated with Omniflow II grafts (97.1%) and that treated with cryopreserved arterial homografts (93.1%). These results are comparable with those reported by Siracuse et al., Chang et al., and Jensen et al.1,13,14 This resistance to infection could be attributed to neovascularization and swift integration with the tissue with the use of Omniflow II grafts.22,23 However, in addition to tissue integration, aggressive surgical intervention, muscle flap coverage, and appropriate antibiotic therapy certainly play an important role in the treatment of VGI. There is a substantial difference between the groups regarding the use of muscle flaps, with significantly more patients in the Omniflow II group requiring this intervention. This discrepancy may reflect differences in clinical decision-making and the observed prevalence of positive bacterial cultures, particularly Gram-positive bacteria, in the Omniflow II group. Muscle flap coverage likely served as an additional measure to mitigate infection risks in this group, addressing the specific clinical challenges encountered during treatment. To minimize the overall risk of reinfection, it is essential to optimize comorbidity management, ensure meticulous wound care, and promote strict adherence to antimicrobial protocols.
Studies focusing solely on infrainguinal vascular reconstruction using the Omniflow II vascular graft prosthesis or cryopreserved arterial homografts are limited by small patient numbers and short follow-up periods. A key challenge in the existing literature is the inclusion of mixed cohorts combining aortic and peripheral reconstructions, making direct comparisons difficult. Töpel et al. reported encouraging results in 29 patients with infected vascular grafts, with primary patency rates of 67.6% at 1 year and 61.5% at 2 years. 7 These outcomes are comparable to those reported by Caradu et al. 24 Furthermore, Castier et al. presented the largest series of patients undergoing cryopreserved arterial allograft implantation for peripheral vascular graft infections during a 7-year study period. In this cohort of 17 patients, the 18-month primary and secondary allograft patency rates were 68% and 86%, respectively. 25
In our patient group, the amputation rate was relatively low at 6.3%, and there was no significant difference in outcomes between the two types of graft materials used. Although VGI is generally recognized as a negative indicator of limb preservation, with Mertens et al. reporting an amputation risk of nearly 40%, our findings did not reflect this effect. 26
Our study has several limitations. The retrospective design introduces information bias. In addition, the small sample size hindered our ability to adjust for potential confounding factors, complicating direct comparisons across groups. This limitation must be considered when interpreting the results. Furthermore, the absence of a standardized protocol for conduit selection and reliance on individual surgeon preferences could introduce selection bias, particularly the tendency to use Omniflow II grafts in higher-risk patients.
In conclusion, this study provides valuable insights into the management of vascular graft infections, supported by long-term follow-up data. However, larger studies with greater numbers of patients are needed to confirm these findings and to provide more robust evidence for refining treatment protocols and improving long-term graft performance.
Despite these constraints, our study represents the most extensive analysis of biosynthetic grafts for lower-limb VGI to date and is pioneering the field in comparing these particular graft types.
Customized treatment is essential for the diverse populations affected by VGI. Graft selection must be judiciously personalized, considering the specifics of the anastomosis site, the diameter of the target vessel, and local clinical practice. Biosynthetic grafts are viable, cost-effective, and user-friendly alternatives to cryopreserved arterial homografts, supporting their widespread application in clinical settings. Such personalized approaches are vital for managing VGI.
Footnotes
Acknowledgements
There are no acknowledgements to declare for this study.
Author contributions
M.Z.: Conceptualization; Data curation; Formal analysis; Investigation; Project administration; Methodology; Writing—original draft.
K.P: Writing—original draft; Writing—review & editing.
W.S: Writing—review & editing.
F.Z: Writing—review & editing, statistical analysis
I.T: Writing—review & editing.
M.S: Writing—review & editing.
T.B: Writing—review & editing. Conceptualization; Methodology; Supervision; Writing—review & editing.
Data availability
Due to the sensitive nature of the data regarding patient confidentiality, some restrictions apply to the availability of these data, which are not publicly available. Data are, however, available from the authors upon reasonable request and with permission of the University medical center Regensburg.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical considerations
This study was approved by the local ethics committee before data collection by the University Medical Center Regensburg (Approval Number 14-101-0017). Written informed consent was obtained from all participants included in this study.
Consent to participate
Applicable.
Consent for publication
Applicable.
