Abstract
Background and Aims:
Roux-en-Y gastric bypass is widely used as treatment of morbid obesity. Weight loss, effects on obesity-related co-morbidities and quality of life are well documented post Roux-en-Y gastric bypass. Other outcome measures are less well studied. This review explores aspects of prevalence, diagnostic evaluations, etiology, and treatment of abdominal pain specific to Roux-en-Y gastric bypass.
Methods:
The review is based on PubMed searches and clinical experience with Roux-en-Y gastric bypass. Symptoms in the early postoperative phase (<30 days) were not included.
Results:
Based on limited evidence, up to about 30% of the patients may perceive recurrent abdominal pain post Roux-en-Y gastric bypass in the long term. A substantial subset of patients will need health-care evaluation for acute abdominal pain and hospital admission. The etiology of abdominal pain is heterogeneous and includes gallstone-related disease, intestinal obstruction, anastomotic ulcerations and strictures, intestinal dysmotility, dysfunctional eating, and food intolerance. Surgical treatment and guidance on diet and eating habits may allow symptom relief. The cause of pain remains undefined for a subset of patients. Impact of abdominal pain post Roux-en-Y gastric bypass on the perception of well-being, quality of life, and patient satisfaction with the procedure needs to be further evaluated and may be influenced by complex interactions between new symptoms post Roux-en-Y gastric bypass and relief of pre-existing symptoms.
Conclusion:
Abdominal pain should be part of follow-up consultations post Roux-en-Y gastric bypass. Future studies should focus on combined evaluations before and after surgery to enlighten potential casual relationships between abdominal pain and Roux-en-Y gastric bypass.
Surgery for morbid obesity is widely performed. An estimated 10,000 patients are operated annually in Nordic countries. In Norway and Sweden around 55/100,000 patients had bariatric surgery in 2016. The laparoscopic Roux-en-Y gastric bypass (RYGB) has been widely applied but is currently surpassed by sleeve gastrectomy as the most common bariatric procedure worldwide.
Weight loss, changes in obesity-related co-morbidity, and aspects of quality of life are well documented post RYGB. Side effects such as abdominal pain are less well explored. However, the RYGB may induce gastrointestinal symptoms due to anatomical and physiological alterations incorporated by the procedure. This is known also for other indications for Roux-en-Y gastrointestinal reconstruction than bariatric surgery (1, 2). Knowledge regarding the prevalence, characteristics, and consequences of abdominal pain post RYGB is limited. Some patients may not report their symptoms and may consider these an expected side effect of surgery. However, the symptoms can influence everyday life and quality of life and may be target for effective interventions (3).
This review aims to describe aspects of prevalence, etiology, and treatment of abdominal pain specific to RYGB.
Methods
This overview is based on PubMed searches and clinical experience with the patient group. The PubMed search terms included “gastric bypass and abdominal pain/gastrointestinal symptoms/chronic pain” and “Roux-en-Y and pain and gastric bypass.” Papers in English describing the prevalence of abdominal pain as reported by the patients and papers reporting abdominal pain as specific cause of hospital admission and outpatient consultations were selected. Scandinavian series were highlighted. The RYGB in these countries have typically a quite similar configuration (limb lengths and pouch size). Case series/reports and reports on symptoms in the early postoperative phase (<30 days) were not included.
Prevalence
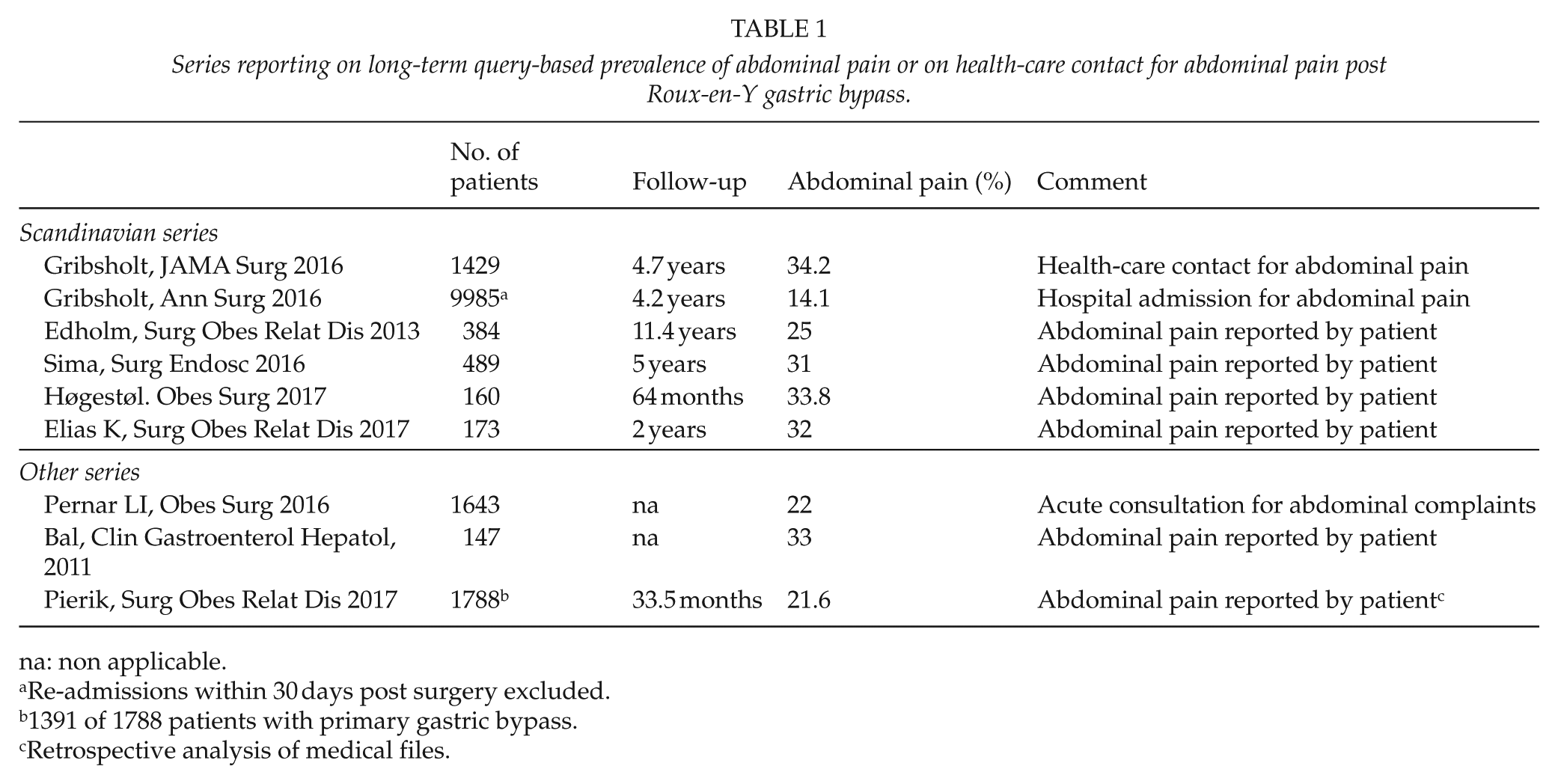
Defining the prevalence of abdominal pain after RYGB is challenging. Definitions and methods of symptom evaluation varies. Weighing of impact of findings may be difficult. Few studies are available, and results may differ between cohorts due to cultural characteristics, patient selection, duration, and rate of follow-up. Low attrition rates may bias findings. Furthermore, abdominal symptoms and pain appear to be frequent in morbid obese patients without bariatric surgery (4, 5). Symptoms of irritable bowel syndrome have been reported in up to about 30% (4). Unfortunately, combined data on perception of abdominal pain before and after surgery appears limited. Table 1 summarizes series commenting on abdominal pain post RYGB.
Series reporting on long-term query-based prevalence of abdominal pain or on health-care contact for abdominal pain post Roux-en-Y gastric bypass.
na: non applicable.
Re-admissions within 30 days post surgery excluded.
1391 of 1788 patients with primary gastric bypass.
Retrospective analysis of medical files.
In two Swedish and one Norwegian series the long-term prevalence of abdominal pain as experienced monthly or more often was 25%–33% (3, 6, 7). A recent Swedish series found that 17% of the patients reported abdominal pain prior to and 32% after RYGB (8). In the Norwegian series abdominal pain was reported by 12% of patients in a separate cohort awaiting RYGB (3). In an American series abdominal pain was reported by 33% after RYGB (9).
Although not specifically evaluated other reports indicate the presence of abdominal pain post RYGB. A Swedish register study of 28,331 patients (median follow-up 3.6 years) found an increased risk for hospital admission for gastrointestinal surgery after RYGB (relative risk 8.6). About one in four patients had additional abdominal surgery during 6 years of follow-up (10). In a Danish study, 67.6% of patients contacted health care after RYGB due to symptoms, and 29% had been hospitalized. The most common symptom leading to health-care contact was abdominal pain (Table 1; 11). Similarly, in a national Danish cohort, at least 15% was acutely admitted to hospital for abdominal pain (12). In an American series, 22% of the patients met at the emergency department with abdominal complaints that typically led to abdominal computer tomography (CT) scans (13). In a Dutch study (mostly RYGB), abdominal pain was described in medical files of 21.6% of the patients (14).
Evidence in regard to the rate of patients in need of repeated health-care contact and extended diagnostic work-up due to chronic or recurrent abdominal pain is not established.
General Symptom Characteristics
Abdominal pain post RYGB is frequently located in the upper abdomen although symptom characteristics vary. Some patients experience occasionally pain, others report recurrent symptoms over longer periods. Some symptoms may be regarded as expected post RYGB such as those caused by dumping and food intolerance. A recent study found that patients often experienced pain in the left upper quadrant (14). We found that 70% of patients with chronic abdominal pain had pain weekly or more often with symptom duration typically 1 h or less. About 50% reported pain intensity >6 on a symptom scale of 0–10. The pain affected daily activity and sleep and appeared to impact health-related quality of life (3).
General Diagnostic Considerations
Establishing an institutional algorithm for evaluation of abdominal pain probably facilitates diagnostic success. Based on symptom characteristics, biochemistry, and clinical examination, a plan for diagnostic work-up may be made. The order of the investigations depends on symptoms and suspected cause. Increased exposure to radiation post RYGB is a concern due to increased use of abdominal imaging, often without relevant findings (13, 15, 16). A comprehensive exploration of symptoms can guide initial radiologic assessment.
A plan for diagnostic evaluation is suggested in Fig. 1. First-line investigations typically include abdominal CT, ultrasonography, and gastroscopy. In acute settings and if there is a strong suspicion of internal herniation, direct laparoscopy may be justified. Early evaluation by a dietitian could reduce the need for other investigations. Colonoscopy should be considered for lower abdominal pain. For undefined epigastric pain after initial diagnostic work-up empiric treatment with proton inhibitors may be considered as could also antibiotics for symptoms of bacterial overgrowth. Follow-up should be planned after such treatments. Investigation of abdominal wall nerve entrapment should be considered during physical examination. Secretin-stimulated magnetic resonance cholangiopancreatography (MRCP) may be useful in upper right quadrant pain, particularly if associated with elevation of liver enzymes. Magnetic resonance imaging (MRI) may define conditions like intestinal strictures. Balloon or transgastric endoscopy can be considered for undefined symptoms that may be related to conditions in the gastric remnant, duodenum, or bile ducts.
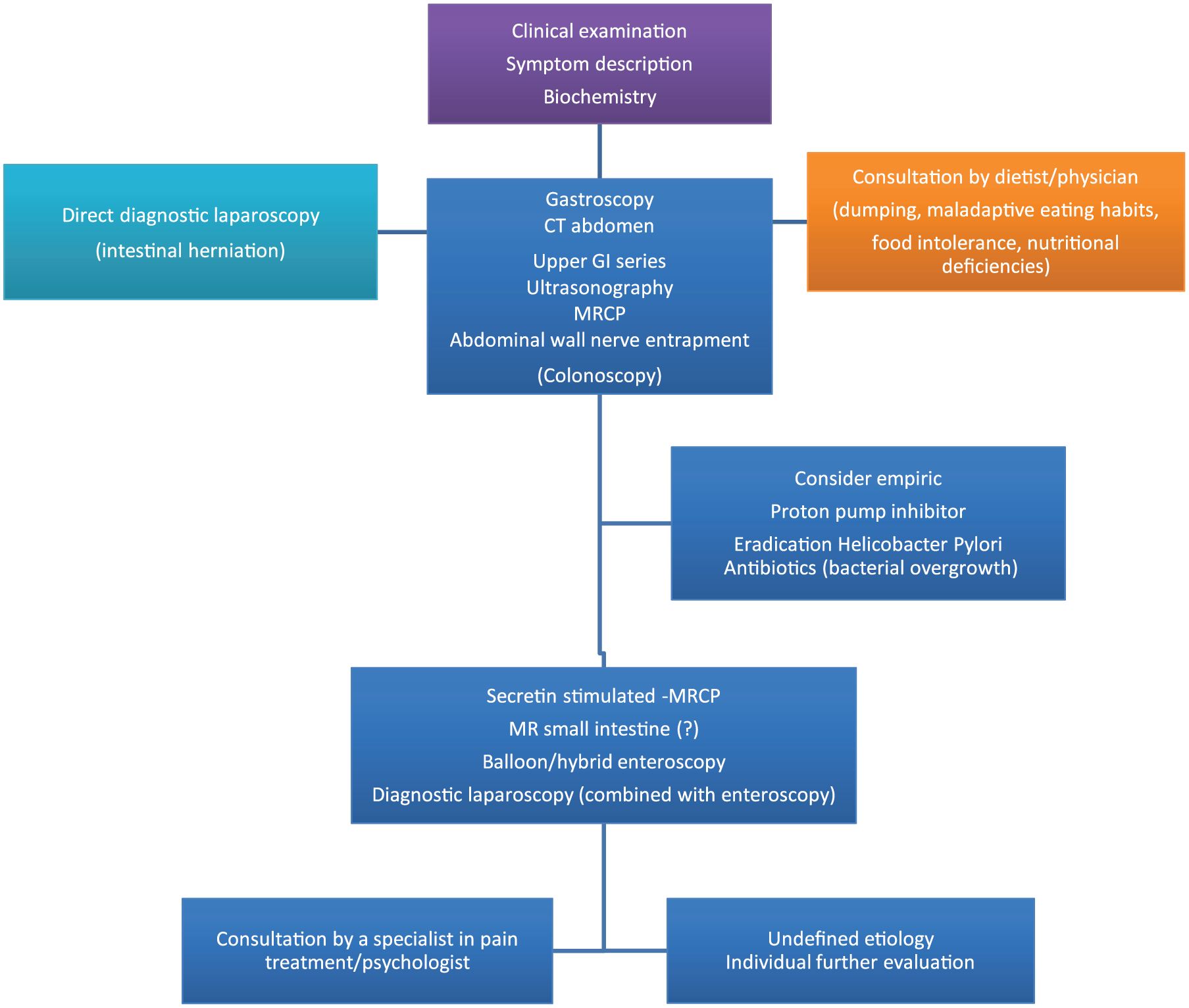
Diagnostic evaluations of abdominal pain after Roux-en-Y gastric bypass. The order of investigations depends on symptoms and findings. Individual additional targeted investigations may be relevant. MRCP: magnetic resonance cholangiopancreatography; CT: computer tomography; GI: gastrointestinal; MR: magnetic resonance imaging.
A low threshold for laparoscopy for acute abdominal pain post RYGB is recommended by many and should also be part of the diagnostic work-up for undefined recurrent abdominal pain. In an American series following normal radiological and/or upper endoscopic evaluations, a potential cause of abdominal pain was found in 57% of the patients during laparoscopy (17). A diagnostic laparoscopy should at minimum include inspection of the mesenterial defects, all parts of the Roux-en-Y configuration, the gallbladder, the abdominal wall, and the entire small bowel. Need for concomitant endoscopic inspection of the excluded stomach and duodenum during laparoscopy depend on individual evaluation.
In a subgroup of patients, the cause of abdominal pain remains unknown. In two American series, abdominal pain post RYGB was not defined in 23% and 43% of the patients (9, 17). In a Dutch study, 34.4% (7.4% of total population) remained without a diagnosis (14).
General Therapeutic Considerations
In cases of undefined cause of chronic abdominal pain, consultations by pain specialist in a multidisciplinary pain management program may be beneficial. For some, the perceived impact of pain may be associated with negative effect and previous traumatic experiences including violence and sexual abuse (18). Counseling by a psychologist may thus be of use for a subgroup of patients.
Occasionally, in situations with chronic debilitating pain without a definite cause, surgery is attempted for symptom relief. This should be done with caution and with patient understanding that outcome is uncertain and without adequate evidence. Procedures include modifying the enteroenteroanastomosis, reducing the size of blind-ended intestinal limbs, gastric pouch modifications, gastric remnant resection, and reversal of the RYGB (19–21).
Conditions Causing Abdominal Pain Post RYGB
A variety of conditions may induce abdominal pain post RYGB (Table 2). Common conditions causing hospital admission include gallstone-related disease, intestinal obstruction, particularly herniation through mesenterial defects, and probably, unspecified pain.
Conditions associated with abdominal pain related to Roux-en-Y gastric bypass.
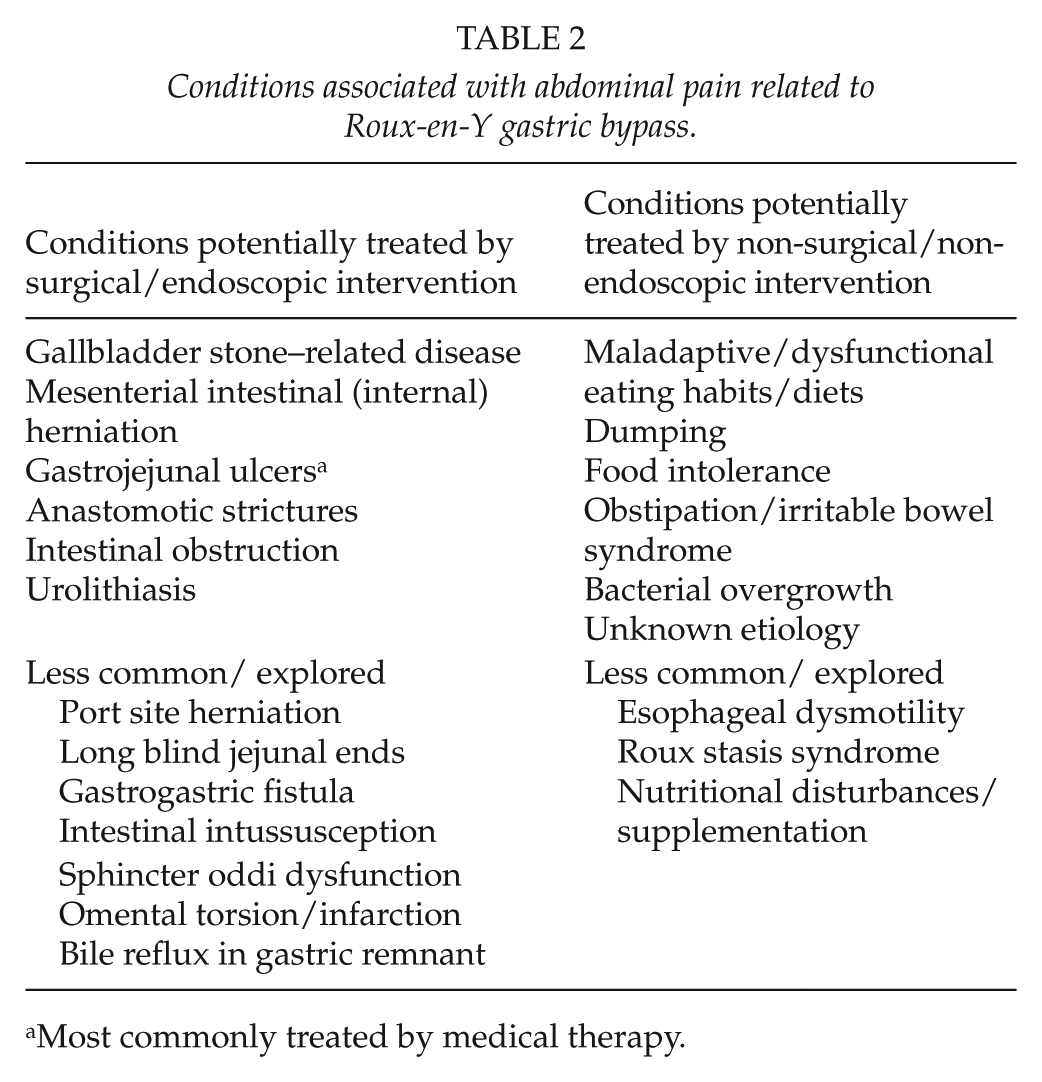
Most commonly treated by medical therapy.
Cholelithiasis
Increased risk of gallstone formation may be related to rapid weight loss and impaired gallbladder emptying. Based on Swedish registry data, the incidence ratio for cholecystectomy 6–12 months after RYGB was 3.5 times the ratio before surgery (22). In another Swedish study, the incidence ratio for undergoing cholecystectomy after RYGB was 6.1 compared to the general population (23). The symptoms of gallstone-related complications are well known and include symptoms of cholecystitis, cholangitis, and pancreatitis with typically upper central or right-side abdominal pain (24). However, post RYGB access to bile ducts for endoscopic intervention and removal of bile duct stones is challenging.
Internal Herniation
Internal herniation is reported in 5%–8% of the patients or more frequent depending on duration and rate of follow-up and routine use or not of closure of mesenterial defects (25, 26). In a recent Norwegian series, the incidence of internal herniation following non-closure of mesenterial defects were 11.7% at 60 months of follow-up (27). In a Danish study of pregnancies post RYGB, 9.7% of the patients had internal herniation (28).
Pain characteristics include intense pain that can be difficult to relieve adequately with opioids and that may radiate to the back. The pain can be provoked by eating, and may be accompanied by nausea and vomiting. In the acute ischemic setting, the pain may be continuous. These symptoms may indicate obstruction and impediment of intestinal vascular supply with risk of ischemia and perforation, and need for emergent intervention. Internal herniation may also present with subtle and intermittent symptoms.
CT may demonstrate typical findings but cannot exclude internal herniation. A low threshold for diagnostic surgery is recommended. During pregnancy diagnosing may be particularly challenging as abdominal pain is common, the threshold for radiology and laparoscopy is higher, and the need for intervention can be urgent (28).
Treatment includes reduction of the hernia and closure of mesenterial defects. Patients should be aware of the risk of re-herniation despite mesenterial defect closure. There is a risk of causing obstruction by closing the mesenterial defects at the enteroenteroanastomosis (26).
Gastrointestinal Ulceration
Ulceration at the gastrojejunostomy is reported in about 5% of the patients although the prevalence varies across reports (29). Ulcerations may cause strictures and fistulas between the gastric pouch and the gastric remnant (30). Ulcerations and strictures may cause epigastric pain, dysphagia, and even peritonitis in cases of perforation. Weight regain is associated with fistulation. Diagnosis is usually made by gastroscopy.
Gastric pouch ulcerations can be handled with proton inhibitors. Stopping smoking and eradication of Helicobacter pylori, if present, should be considered. Blocking, stenting, and reconstruction of the anastomosis may be required such as in cases of perforations (29).
Gastritis, duodenitis, and peptic and malignant ulcerations may occur in the gastric remnant and the duodenum (31). This may not be easily identified by standard gastroscopy or CT (unexpanded remnant) due to the altered anatomy (20, 31). Balloon enteroscopy or laparoscopy-assisted endoscopy through a gastrostomy may be required.
Functional (“Non-Surgical”) Causes of Abdominal Pain
Bacterial overgrowth in the gastric remnant or in the small intestine may cause abdominal pain although the mechanisms for potential associations between bacterial overgrowth and clinical symptoms are not well defined. In one series, bacterial overgrowth was stated the most common cause of chronic abdominal pain (9).
Abdominal pain post RYGB can be related to food intolerance, maladaptive eating habits such as overeating and rapid eating, and dumping syndrome. In a Dutch series, food intolerance was reported in 70.7% of the patients 2 years after RYGB and in 16.9% of the controls. A positive correlation between gastrointestinal symptoms and food intolerance was observed (32). Dysfunctional eating seems most frequent early after RYGB with possible natural adaption due to discomfort. Moderate to severe symptoms of early dumping after RYGB was reported in 9.4% of the patients in a Danish series (33).
Symptoms of functional abdominal pain are typically recurrent and ambiguous, including bloating and cramping. A dietitian may reveal dysfunctional eating habits (too fast and too much), dumping-related symptoms, and food intolerance. Food intolerance can be related to high sugar or fat content and to red meet (32). Adjusting maladaptive eating behavior can allow symptom relief as may also guidance in regard to prevention of dumping. Although without general consensus empirical administration of antibiotics for bacterial overgrowth and proton inhibitors for epigastric pain may be considered for symptom relief and diagnostic purposes (34). Bacterial overgrowth is often a diagnosis of exclusion, but hydrogen breath tests may be used for confirmation (35).
Other and Less Common/Explored Causes of Abdominal Pain
RYGB is associated with an increased risk for urolithiasis that may be linked to hyperoxaluria (36, 37). In a meta-analysis a 1.73 fold increased risk of kidney stones was observed compared to patients without RYGB (37).
Intestinal intussusception post RYGB may be related to a motility disorder. The enteroenteroanastomosis can be involved. The incidence in one series was 0.4% (38). Intussusception has been handled by simple reduction, intestinal resection, and plication to prevent recurrence (38). A long blind-ended limb at the gastrojejunostomy (“candy cane”) may cause functional problems and pain, as it may be the most direct stoma outlet (19).
Sphincter Oddi dysfunction, biliary reflux to the gastric remnant, and superior mesenteric artery syndrome may cause abdominal pain (20, 39, 40). Investigations for sphincter oddi dysfunction include secretin-stimulated MRCP and liver enzyme analyses during symptoms. Sphincterotomy has been reported successful in some patients (39, 41). Selection of patients, however, may be complex.
Esophageal or intestinal dysmotility may cause postprandial pain but are not well studied post RYGB (1, 42). Eroded sutures and staples have been associated with pain as has also omental torsion/infarction resulting from omental division (43, 44). Abdominal wall cutaneous nerve entrapment syndrome is also a suggested cause (14, 45). Nutrient disturbances may be contributors to abdominal pain (35, 46).
Measures that May Reduce the Risk for Abdominal Pain
Prescription of ursodeoxycholic acid in the postoperative period, typically for 6 months, may reduce the risk of gallstone formation and is prescribed routinely by some (47). Concomitant cholecystectomy during RYGB is occasionally performed, but this is not common practice. Closure of mesenteric defects has been shown to reduce the incidence of reoperation for intestinal obstruction (hazard ratio 0·56) during 3 years of observation (26).
A small gastric pouch may reduce the risk for gastrojejunal ulceration (48). Limiting the use of antiflogistics and smoking and eradicating Helicobacter pylori may also reduce the risk of anastomotic ulceration although the evidence for this should be substantiated. Avoiding long blind-ended intestinal segments at the anastomoses are suggested to reduce the risk of pain and bacterial overgrowth (19). Length of intestinal limb lengths and the configuration of the enteroenteroanastomosis such as the stoma size and kinking of the anastomosis may affect the risk of abdominal pain after RYGB. Knowledge, however, in this regard is limited.
Discussion
Although based on limited evidence, there are indications of a relative high proportion of patients experiencing recurrent and acute abdominal pain in the long-term post RYGB. The etiology of pain is diverse. Follow-up in institutions familiar with the RYGB procedure may facilitate diagnosis and treatment although this need to be evaluated in future studies.
Abdominal pain post RYGB is complex and the present review only presents aspects of issues related to this particular procedure. Findings in regard to prevalence should be interpreted with caution, as methodology for evaluation of symptoms varies and data from longitudinal studies are limited. Using validated procedure, specific questionnaires could facilitate our understanding of these symptoms.
Non-patient reported indicators of abdominal pain include data on gastrointestinal surgery and abdominal imaging. In an American series of mostly RYGB (mean follow-up about 3.5 years) abdominal CT was performed at least once in 36% of the patients. RYGB was identified as a risk factor for such imaging (15). In another series, 22% of the patients had abdominal CT post RYGB, most with normal scans (13). Opioid medication has been found increased from baseline 14.7% to 7 years 20.3% post bariatric surgery (49). However, surrogate indicators of abdominal pain should be interpreted cautiously.
Knowledge of the presence of symptoms prior to surgery is necessary when evaluating causal relationships. Patient-reported experience of abdominal pain both prior to and after RYGB should be the focus of future studies. An increased need for abdominal surgery during 6 years post RYGB indicates a potential effect of time that needs further exploration (10).
Some studies report reduced abdominal symptoms post RYGB (50–53). In one series, improvements were found up to 5 years post surgery for various abdominal symptoms. For abdominal pain, however, improvements were not statistically significant in the long-term (51). Another series found that abdominal pain scores as evaluated by the Gastrointestinal Symptom Rating Scale improved 1 year post surgery (52). One study found that symptoms like dysphagia and dumping decreased up to 3 years post surgery (53).
Although abdominal pain may be common, most patients appear satisfied with the RYGB. In one series, 87.4% of the patients felt better or much better although 67.6% had been in contact with health care due to symptoms, most commonly abdominal pain (11). In another study, 31% of the patients reported abdominal pain, but 88% were satisfied or very satisfied with the procedure, and 92% would recommend RYGB to an acquaintance (6). These findings should be interpreted with care, but well-being and the acceptance of symptoms may depend in part on improvement or resolution of other symptoms. Reduced rate of hospital admissions due to pulmonary and cardiovascular disease and diabetes post RYGB has been observed (12). Quality of life is typically reported improved post RYGB.
Abdominal pain should be the focus of future studies comparing RYGB with other procedures. In one series, acute consultations and hospital admissions for abdominal pain the first year following sleeve gastrectomy was 11% and 3%, respectively (54).
The Roux syndrome has been recognized as a dysmotility disorder after gastrectomies, causing abdominal pain, nausea, and vomiting (42). This syndrome was identified in 30% of the patients about 6 years post gastrectomy, and it appeared to be associated with the length of the Roux limb (1). In another series, 33% of the patients had such symptoms (2). This disorder may also be relevant for symptoms post RYGB and could be further evaluated.
Abdominal pain should be part of follow-up consultations after RYGB. Defining the etiology and ensuring symptom relief can be challenging in a subset of patients. A multidisciplinary approach may be beneficial. Research is necessary to enable more robust evidence in regard to the prevalence, burden, and consequences of abdominal pain in the context of total symptom changes post RYGB.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
