Abstract
Background and Aim:
The clinicopathological demonstrations of gastric cancer vary widely between Eastern and Western countries. Turkey is situated in Europe and Middle East which acts as a bridge between east and west. We aimed to validate the two popular nomograms of east and west origin by means of patients who underwent curative surgery for gastric cancer in our country.
Material and Methods:
Of the 202 patients diagnosed with gastric cancer between the years 2006 and 2013, 145 of these patients whose data were sufficient were included in the study. For all patients, demographic, laboratory, operative, and pathologic findings were documented. For each patient, prognostic factors were incorporated into the nomograms for estimating 5-year survival probability.
Results:
For a Turkish cohort, the western nomogram showed a better discriminative capacity (AUC = 0.721, 95% confidence interval 0.637–0.806) and was better calibrated (the Hosmer–Lemeshow goodness-of-fit test p = 0.323), as compared to the eastern nomogram with AUC = 0.615, 95% confidence interval 0.522–0.708, and p = 0.077, respectively.
Conclusion:
Western nomogram was found to be more effective than eastern nomogram in prediction of estimating 5-year survival probability in patients with resectable gastric cancer in Turkish population.
Introduction
Despite the recent decline in incidence, gastric cancer (GC) is a major burden for global health. It is the fourth most common cancer and the third leading cause of cancer death worldwide (1). Radical gastric resection and lymphadenectomy is the choice of treatment for resectable GC. Despite being the only way to treat, survival for resectable GC varies widely, depending on the differences in the clinicopathologic characteristics of the patients (2). The prognosis of GC patients is mainly related to the spread of the disease. The correct prognostication is of particular importance for making informed decisions about adjuvant therapy and surveillance (3). Developments in molecular analysis have potential for prognostic ability, but it is expensive and restricted to centers with hightech laboratories and therefore not widely used. For these reasons, clinical variables are more commonly used for prediction of prognosis. The majority of institutions that treat GC patients are still dependent on accessable clinical parameters for prediction of survival (4).
In 2010, the seventh edition of the American Joint Committee on Cancer (AJCC), updated the tumor–node–metastasis (TNM) classification system to stratify GC into nine risk groups based on the depth of invasion, number of metastatic lymph nodes, and metastasis (5). In many centers, AJCC staging system is used for staging GC. However, according to the AJCC staging system, patients at the same stage can show different prognoses (6). Other factors that affect the survival of the patient, such as age, sex, and tumor location, are not part of this staging system (3).
The idea of using many clinical prognostic factors in a scoring system is of great interest. In this way, clinical decision making, planning for adjuvant therapy, and follow-up schedules can be planned (4). Prognostic nomograms are tools designed plainly for prediction. To achieve a superior method for predicting patient survival, various nomograms and scoring systems have been developed (7). So far, two of the nomograms for GC have attracted attention. Kattan et al. (7) from the United States established a nomogram derived from 1039 patients who underwent R0 GC resection at Memorial Sloan Kettering Cancer Center, New York, USA. And recently, Woo et al. created a simple but comprehensive survival nomogram using a test data set of 11,851 patients and analyzed with Cox proportional hazards model. Then, the prediction model underwent internal and external validation using four patient data sets from three different countries (South Korea, Japan, and China) (8).
The clinicopathological presentations of GC vary widely between Eastern and Western countries. These include differences in histology, location, type of surgery, stage, early detection, and other environmental and local factors (9). For this reason, further validation assessments from different centers are important to assess the applicability of these nomograms and to make them more powerful. Turkey is the only country situated in Europe and Asia and acts as a bridge between two continents. In addition, GC takes the fifth place among the most frequently seen cancer types (10). We aimed to compare the two popular nomograms of east and west origin in patients with GC who underwent curative surgery in our country and compare them with the AJCC/TNM staging system which has traditionally been used for prediction of prognosis.
Material and Methods
The study was conducted as a retrospective investigation of GC patients that had been referred to the Ankara Numune Training and Research Hospital Ankara, Turkey, between January 2006 and December 2013. A total of 202 patients who were diagnosed with GC were included. GC diagnosis was established with imaging methods and histopathological evaluation of endoscopic biopsy materials. The exclusion criteria were as follows: patients who underwent palliative surgery, patients who had distant metastasis or peritoneal dissemination that was confirmed during the operation, prior chemotherapy history, prior hematological diseases, clinical evidence of infection, or other inflammatory conditions, such as inflammatory bowel disease and rheumatoid arthritis. Of these 202 patients, 192 had local disease, and a curative resection was performed. Among these patients, 145 patients whose records were fully accessible were included in the study. A R0 resection was defined as complete histologic clear margins according to UICC/AJCC standards. For all patients, demographic (gender, age), laboratory (complete blood count, liver function tests, renal function tests, and tumor markers), operative findings (type of operations) pathologic findings were documented retrospectively. Data regarding potential prognostic factors including age, sex, preoperative laboratory measurements of neutrophil, lymphocyte, and platelet counts, postoperative tumor characteristic such as tumor location, tumor size, lymph node metastasis, TNM staging (the seventh edition AJCC TNM staging), Lauren’s classification of subtypes, and survival times were obtained from medical records. For each patient, prognostic factors incorporated in the MSKCC nomogram were extracted and entered into the nomogram calculator to generate a predicted probability of disease-specific survival at 5 years. These factors included sex, age, primary site (pylorus, antrum, body/middle third, proximal third, and gastroesophageal junction), Lauren histotype (diffuse, intestinal, mixed), tumor size, number of positive nodes resected, number of negative nodes resected, and tumor depth (M—mucosal, SM—submucosal, MP—muscularis propria, SS—subserosal, S2—serosal, S3—adjacent organ involvement). Although the nomogram generates both 5- and 9-year disease-specific survival (DSS) probabilities, validation was performed using only 5-year estimates to ensure that all patients had adequate follow-up time (7). Asian prediction model for estimating 5-year survival probability was as follows: P = 1 − P0 A, where P is the 5-year survival probability, P0 is the baseline survival probability, and A = exp[(0.024age) + (0.038men) − (0.014AMD) + (0.079APD) − (0.161MUC) + (0.009SRC) + (0.051 other histologic types) + (0.412T1b) + (0.790T2) + (1.464T3) + (1.706T4a) + (2.169T4b) + (0.050metastaticLNs) − (0.014total number of retrieved LNs) + (0.806M1) + (0.366TG)] (AMD is the adenocarcinoma moderately differentiated, APD is the adenocarcinoma poorly differentiated, MUC is the mucinous carcinoma, SRC is the signet ring cell carcinoma, LN is the lymph node, and TG is the total gastrectomy) (8). Five-year’s DSS was the primary endpoint.
Statistical Analyses
Of the 202 patients from our database, 145 records (72%) were complete regarding the information needed for the analysis. Nomogram validation comprised two activities. These are discrimination and calibration. Discrimination is the ability of the model to distinguish between true positives (mortality) and false positives (11). An important advantage of ROC analysis over traditional sensitivity-specificity analysis is that the area under the ROC curve is independent for both of the cut-point criteria chosen and the prevalence of outcome of interest. Model discrimination was measured by the area under the receiver–operator characteristic (ROC) curve. Calibration was assessed using the Hosmer–Lemeshow goodness-of-fit test χ2 statistic and the corresponding calibration curves. Number of groups (k) was 10. Chi-square statistic which is assessed using k – 2 degrees of freedom.
Results
The clinicopathologic characteristics of the patients are summarized in Table 1. The parameters including primary site of the tumor, Lauren histotype, number of resected positive and negative nodes, and depth of invasion that are used in MSKCC nomogram are also presented in the table. Eighty-two underwent a D1 lymphadenectomy (59%). The majority of patients had a tumor in the distal (48.3%) or middle stomach (29.0%). Seventy-seven (53.1%) patients were classified as Lauren diffuse-type GC. Patients with pathologic stage IIIC disease composed the largest proportion of the cohort (43.4%). The last follow-up date was 30 June 2016 and 91 patients (62.8%) had died of GC. Median overall follow-up was 26 months (1–137 months), 59 months (18–137 months) for survivors, and 16 months (1–104 months) for deceased patients.
Demographic and clinicopathological characteristics of the Ankara Numune Training and Research Hospital Cohort.
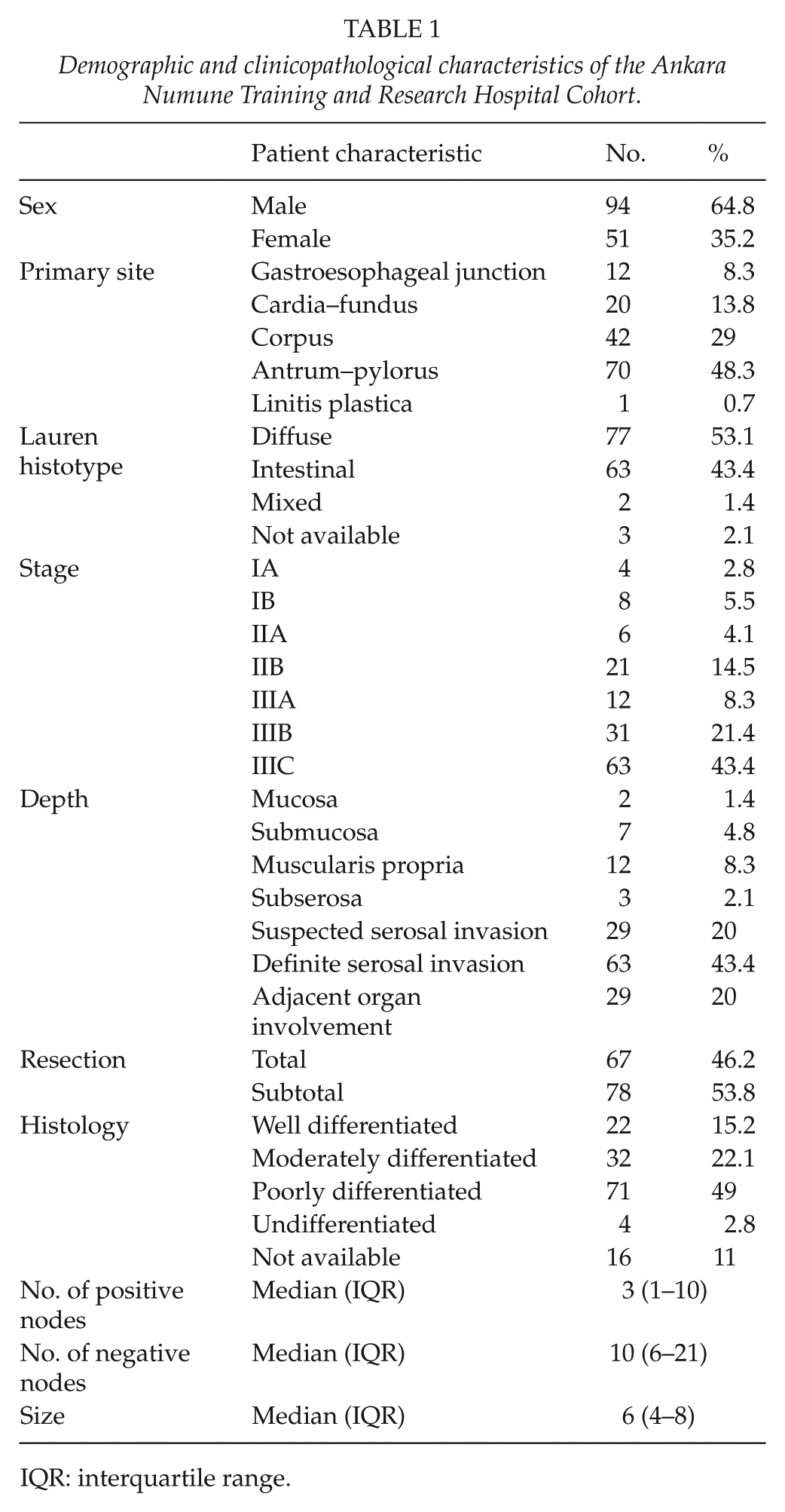
IQR: interquartile range.
Evaluation of the Nomogram
The discrimination of the MSKCC nomogram was assessed with the ROC analyses. According to ROC analyses, MSKCC showed good discriminative capacity (AUC: 0.721; 95% confidence interval (CI): 0.637–0.806) (Fig. 1). Second, calibration was assessed using the Hosmer–Lemeshow goodness-of-fit test χ2 statistic of 9.21 (p = 0.323), indicating that the nomogram can reasonably predict 5-year DSS. The discrimination of the eastern nomogram was moderate (AUC: 0.615; 95% CI: 0.522–0.708) (Fig. 1). The Hosmer–Lemeshow goodness-of-fit test χ2 statistic of 14,183 (p = 0.077), indicating that the nomogram may not well predict DSS. The nomogram can reasonably predict 5-year DSS. AUC for the AJCC/TNM staging was 0.713 (95% CI: 0.625–0.800). The Hosmer–Lemeshow goodness-of-fit test χ2 statistic of 4624 (p = 0.201), indicating that the AJCC/TNM can reasonably predict 5-year DSS. Figs 2 to 4 illustrates the Kaplan–Meier curves for patients according to nomograms. Patients were clustered into four groups according to their MSKCC and eastern nomogram survival (first quartile, 75%). As depicted, MSKCC nomogram and AJCC/TNM discriminated well, and global log-rank tests were all highly significant.
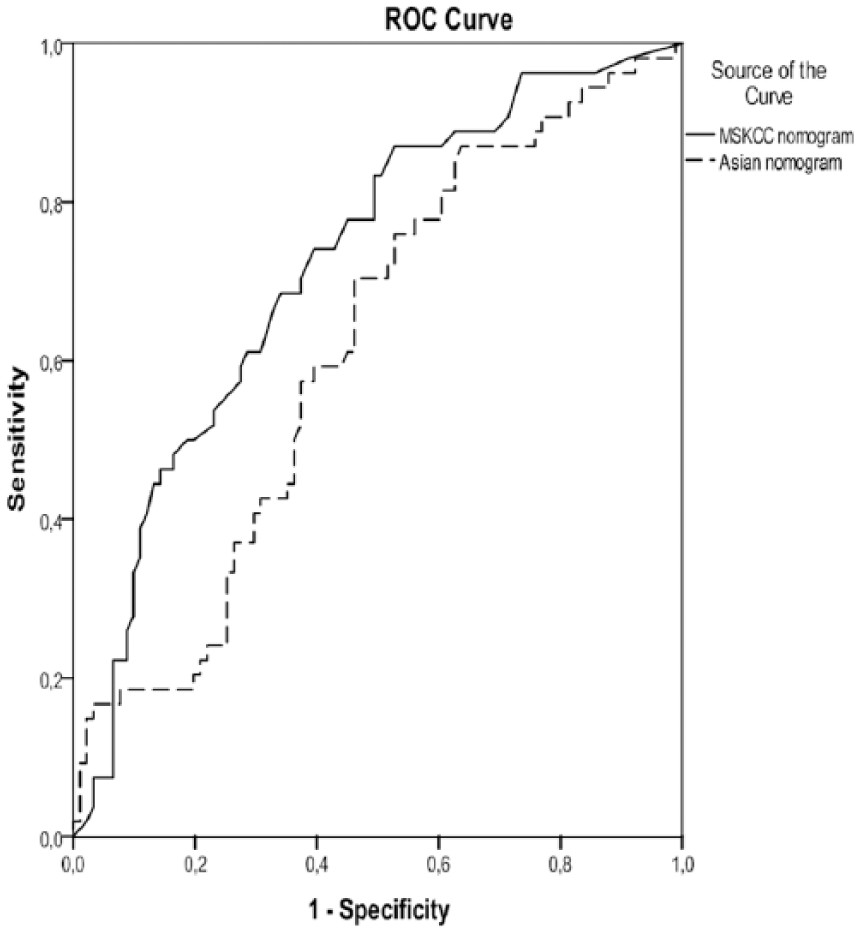
The ROC curves which represent the discriminative abilities of the MSKCC and Asian nomograms in predicting survival at 5 years.
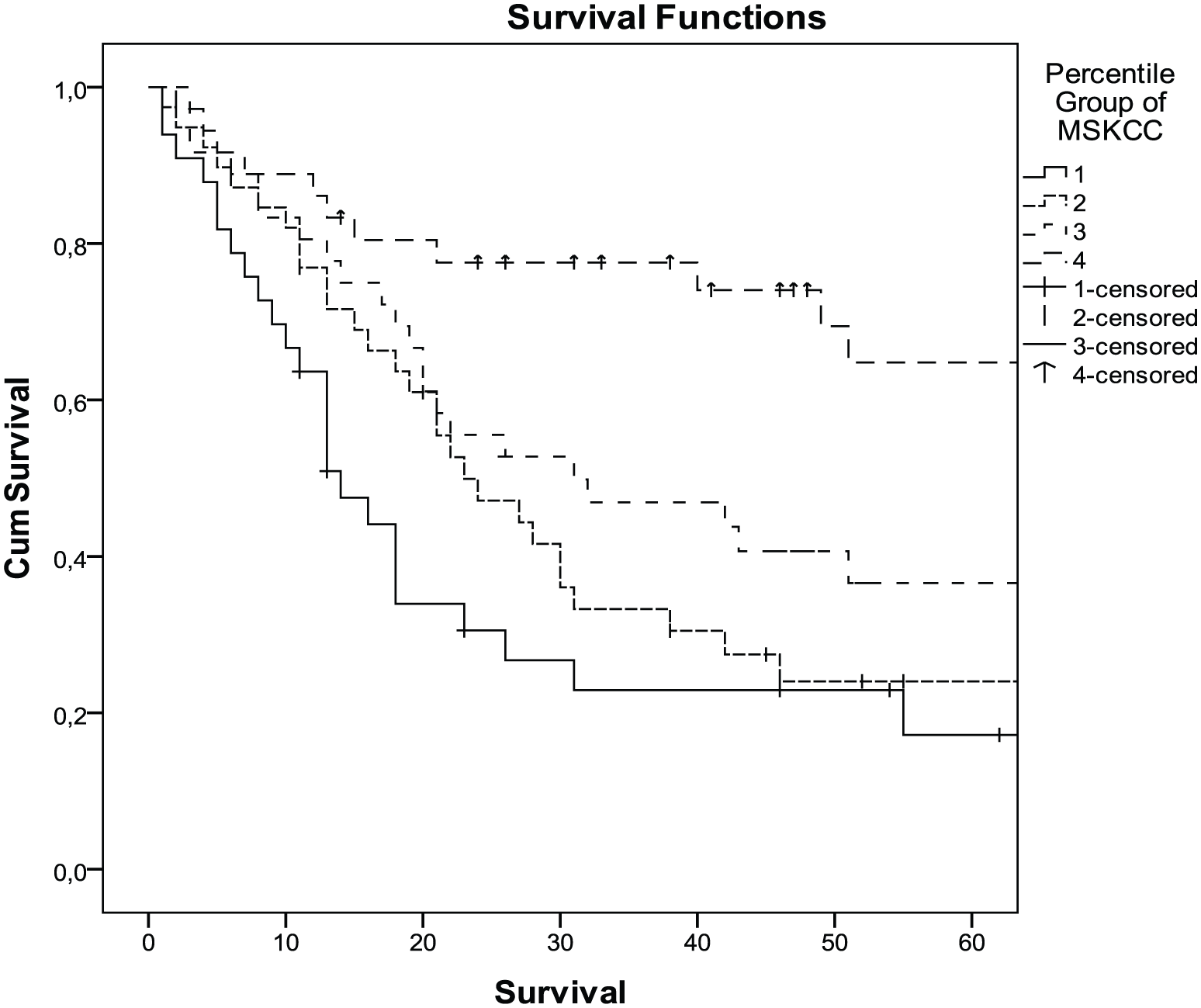
The Kaplan–Meier curves for all patients stratified by quartiles of the MSKCC nomogram predictions for 60-month overall survival. Patients in the Turkish cohort were grouped into quartiles according to the nomogram-predicted survival (first quartile, 25%; second quartile, 25%–50%; third quartile, 50%–75%; fourth quartile, 75%) (p < 0.001).
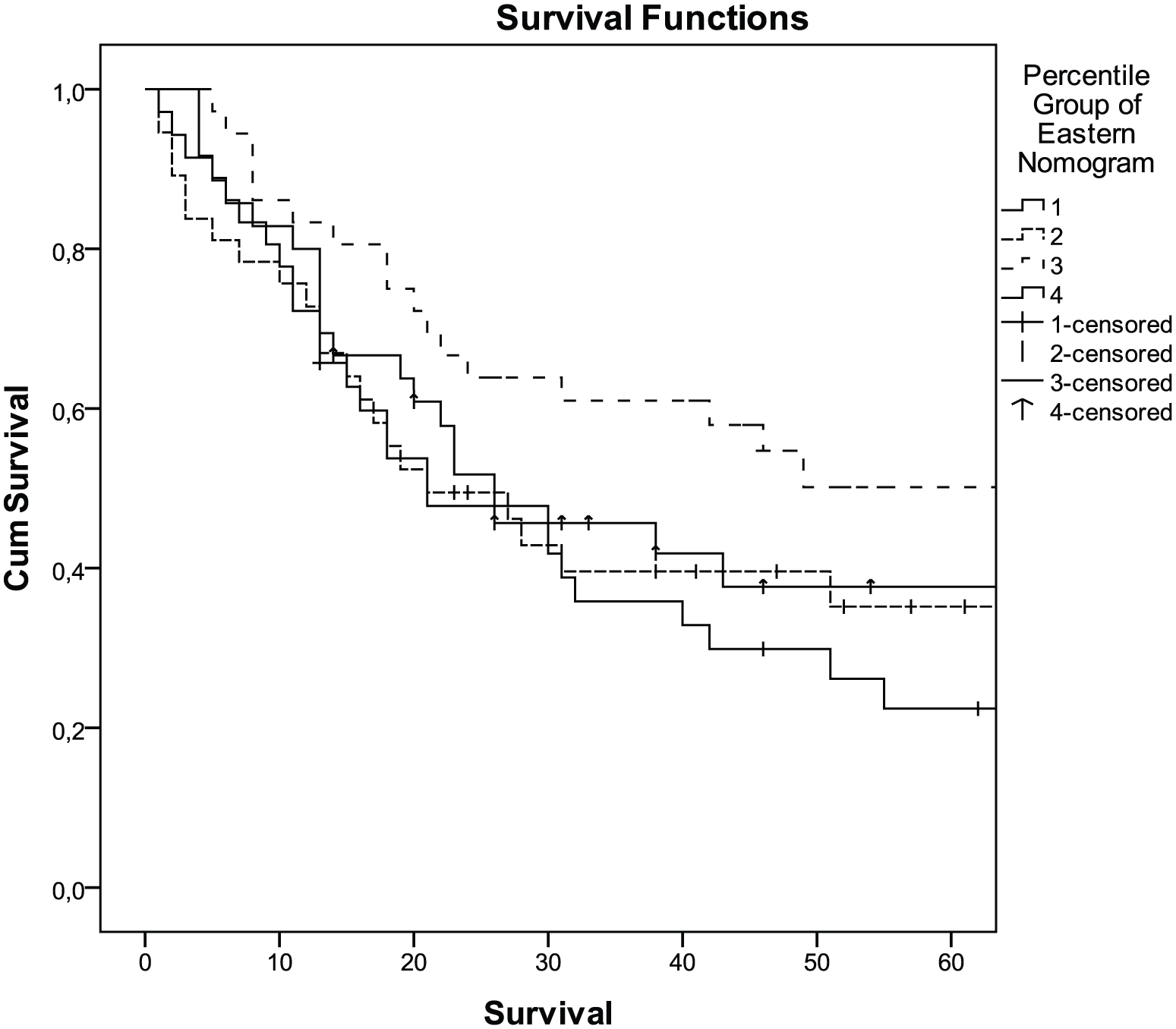
The Kaplan–Meier curves for all patients stratified by quartiles of the eastern nomogram for 60-month overall survival. Patients in the Turkish cohort were grouped into quartiles according to the nomogram-predicted survival (first quartile, 25%; second quartile, 25%–50%; third quartile, 50%–75%; fourth quartile, 75%) (p = 0.066).
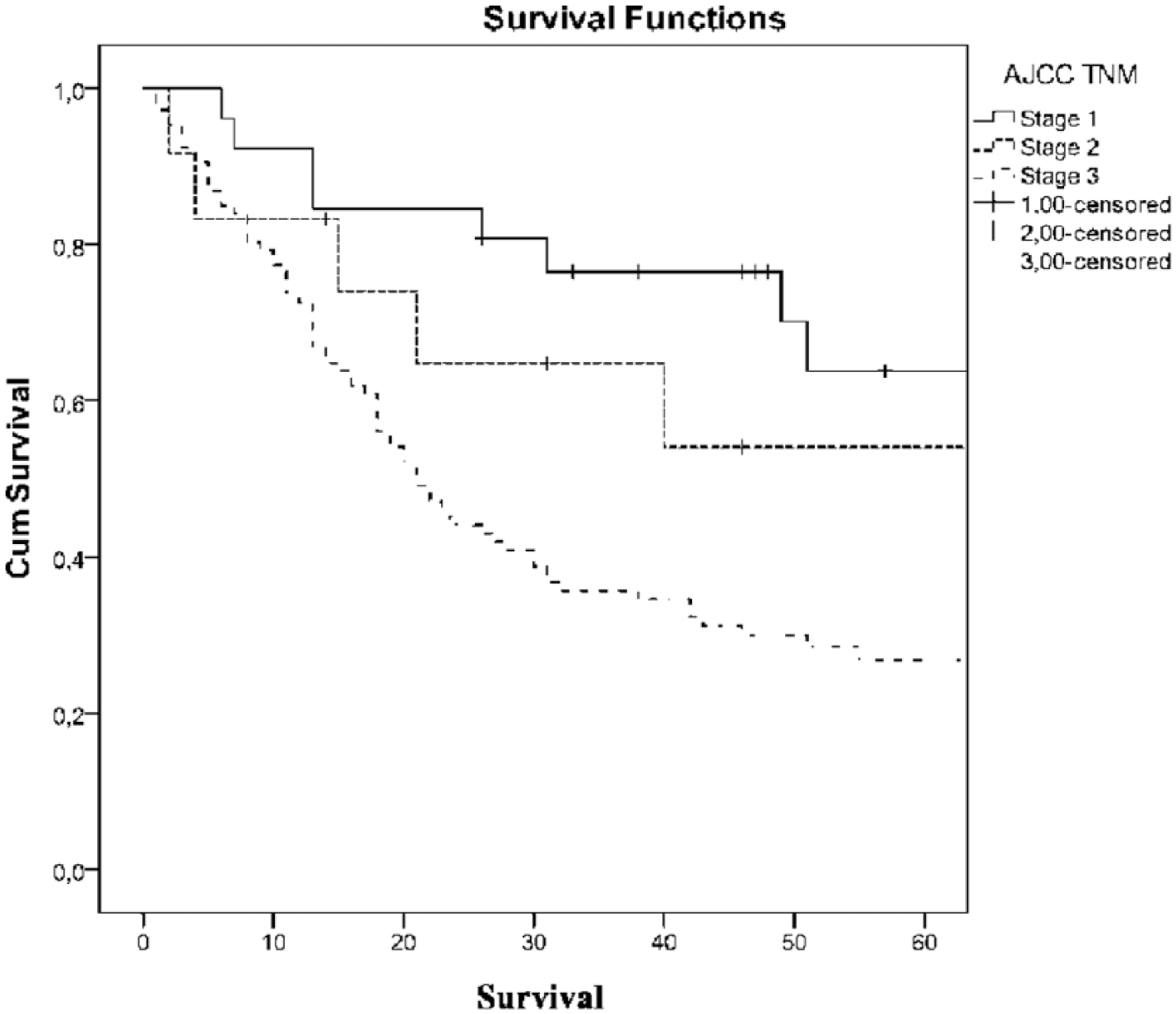
The Kaplan–Meier curves for all patients stratified by AJCC TNM predictions for 60-month overall survival (p = 0.001).
Discussion
To our knowledge, this is the first study to validate the two popular nomograms of east and west in patients who underwent curative surgery for GC in Turkey which is located as a bridge between east and west. As depicted, the MSKCC nomogram and the AJCC/TNM staging both discriminated well, with global log-rank tests highly significant. Western MSKCC nomogram proved to be much better than the eastern nomogram in predicting overall 5-year survival. At present, the most important point in GC treatment is to find out how much we can individualize therapy based on tumor characteristics and patient factors. Accurate estimation of the risk of death after GC surgery is important for both the patients and the physicians. With this information, decisions about perioperative adjuvant treatment and follow-up frequency can be made and at the same time, information about the long-term prognosis of the disease can be provided to the patient (12).
Uniform staging methods are needed to assess the outcome of treatment and to compare them with different centers. TNM staging, which is proposed by UICC/AJCC, is the most commonly used method to predict patient prognosis. However, the difference in survival of patients within the same stage according to the TNM staging system suggests that different parameters such as age, sex, tumor size, and histological type may also have an effect on survival (13–15). For example, according to the results of the study conducted by Aoyama et al. (13), 5-year survival of patients with Stage 2 or Stage 3 disease varied according to the macroscopic tumor size, as 64.9% for 70 mm, respectively.
Nomograms containing parameters not used in TNM staging system such as tumor location, extent of lymph node dissection, and presence of lymphovascular invasion can help solve this problem (16). It is a predictive tool for calculating the possible risk in the most accurate way using all proven prognostic factors with graphical demonstrations that are easy to use (17). So far, the nomogram developed by the MSKCC and the eastern nomogram, which was created afterward, have received a lot of attention. The best known GC nomogram was developed by MSKCC and the nomogram-predicted survival with a concordance index of 0.80. Chinese, Korean, German, and Dutch validations revealed good discrimination with a C-index of more than 0.70. Though, those nomograms were criticized to be based on specialized high-volume institutional data. Both the derivation and the validation samples for the nomogram were acquired from high-volume specialized centers (6, 9, 18). However, some clinicopathologic features of GCs in eastern countries are different from those in western countries, such as race difference, environmental exposure, tumor location, type of surgery. These differences may lead to incorrect estimation of the clinical outcomes of eastern patients using the same nomogram model (6, 9). Though, the MSKCC nomogram could not predict survival appropriately in Korean patients. New nomograms were developed by Korean institutions (9, 16, 19, 20). Among these nomograms, the recently published Korean nomogram of Yonsei University College of Medicine was adapted using 11,851 GC patient data. When the prediction model was applied to the external data sets, high degree of discrimination was detected. The C statistics for the external cohorts were 0.842 for Gangnam Severance Hospital, 0.868 for Gifu University Hospital, 0.839 for Soon Chun Hyang University Bucheon Hospital, and 0.798 for Nanfang Hospital (8).
These nomograms have different results when used in the eastern and western populations (9, 21). This divergence of c-indices between the east and the west may derive from the differences of clinicopathological characteristics. The clinicopathological presentations of gastric carcinoma differ extensively between eastern and western population. These differences involve histology (intestinal vs diffuse), location (distal vs proximal), environmental and dietary factors, Helicobacter pylori status, and the time of the diagnosis (9, 22). In addition, differences in overall survival can be detected between east and west because of more careful screening of the disease in the east, early detection of disease in the eastern population, and treatment of the disease in an earlier stage compared to the western population and doing more extended lymph node dissection routinely in the east (9). And also, parameters used in nomograms may vary too much . For example, unlike previously used parameters, Kim et al. (12) and Kong et al. (23) used lymph node ratio (LNR) which is the ratio of the number of metastasized lymph nodes to the number of examined lymph nodes as a parameter in their nomogram. Because of such differences between east and west, it is difficult to use nomograms in both eastern and western population, and meanwhile, testing nomogram validations in centers with high volume makes it difficult to apply these nomograms to centers with lower volume. Turkey is located at the intersection of west and east. The external validation of both nomograms in a country like Turkey and in a center with lower volume will contribute to the future studies. Besides this, the comparison of these nomograms with TNM system which is traditionally being used in Turkey is important for testing the efficiency of these nomograms. Since the type of lymph node dissection performed was not used in the nomograms for east and west comparison, variables about the presence of D1 or D2 lymph node dissection were not included in our study. There are differences between east and west regarding the necessity of D2 dissection, and there has been a debate on this issue over three decades. Our center also has not formed a consensus on this issue The type of the operation was determined according to the surgeon’s preference, and D1 dissection was preferred more frequently in our center (24). In high-volume countries like Japan or South Korea, D2 lymphadenectomy is preferred over D1. However, surgeons in Western countries generally prefer more conservative nodal dissection. This preference was influenced by the results of prospective studies of the Dutch Gastric Cancer Trial and the UK Medical Research Council (MRC), with no difference in survival between extended and limited lymphadenectomies and worse mortality with the more extensive dissection (25). However, when the Dutch trial published 15-year follow-up results, the trial showed the long-term survival benefit of D2 over D1 lymphadenectomy without morbidity or mortality in patients who had undergone the spleen preserving procedure (2). Although Turkish population differs from western and eastern populations according to clinicopathological characteristics, MSKCC nomogram was found to be more successful in prediction of overall survival in the Turkish population. Both nomograms have been generated and validated at higher volume centers; however, MSKCC nomogram was found to be more successful in our lower volume center and may show that the MSKCC nomogram is effective for lower volume centers too. Eastern nomogram validations have only been conducted in eastern populations; on the contrary, MSKCC nomogram has been implemented in Chinese, American, German, and Dutch populations. The racial diversity in the centers for validation in the MSKCC nomogram may be a factor in the success of the higher efficacy in prediction of overall survival in Turkish population (7, 8).
In conclusion, MSKCC nomogram were found to be more effective than the eastern nomogram in prediction of DSS in patients with resectable GC in Turkey. As validation continues from different centers, more powerful nomograms are created the use of these nomograms will increase worldwide.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was performed retrospectively using data collected routinely for all patients undergoing gastrectomy. The data were anonymized. Ethical approval was therefore not required. Informed consent was obtained from all individual participants included in the study.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
