Abstract
Study Design
Retrospective and prospective cohort study.
Objectives
Survival estimation is necessary in the decision-making process for treatment in patients with spinal metastasis from cancer of unknown primary (SMCUP). We aimed to develop a novel survival prediction system and compare its accuracy with that of existing survival models.
Methods
A retrospective derivation cohort of 268 patients and a prospective validation cohort of 105 patients with SMCUP were performed. Univariate and multivariable survival analysis were used to generate independently prognostic variables. A nomogram model for survival prediction was established by integrating these independent predictors based on the size of the significant variables’
Results
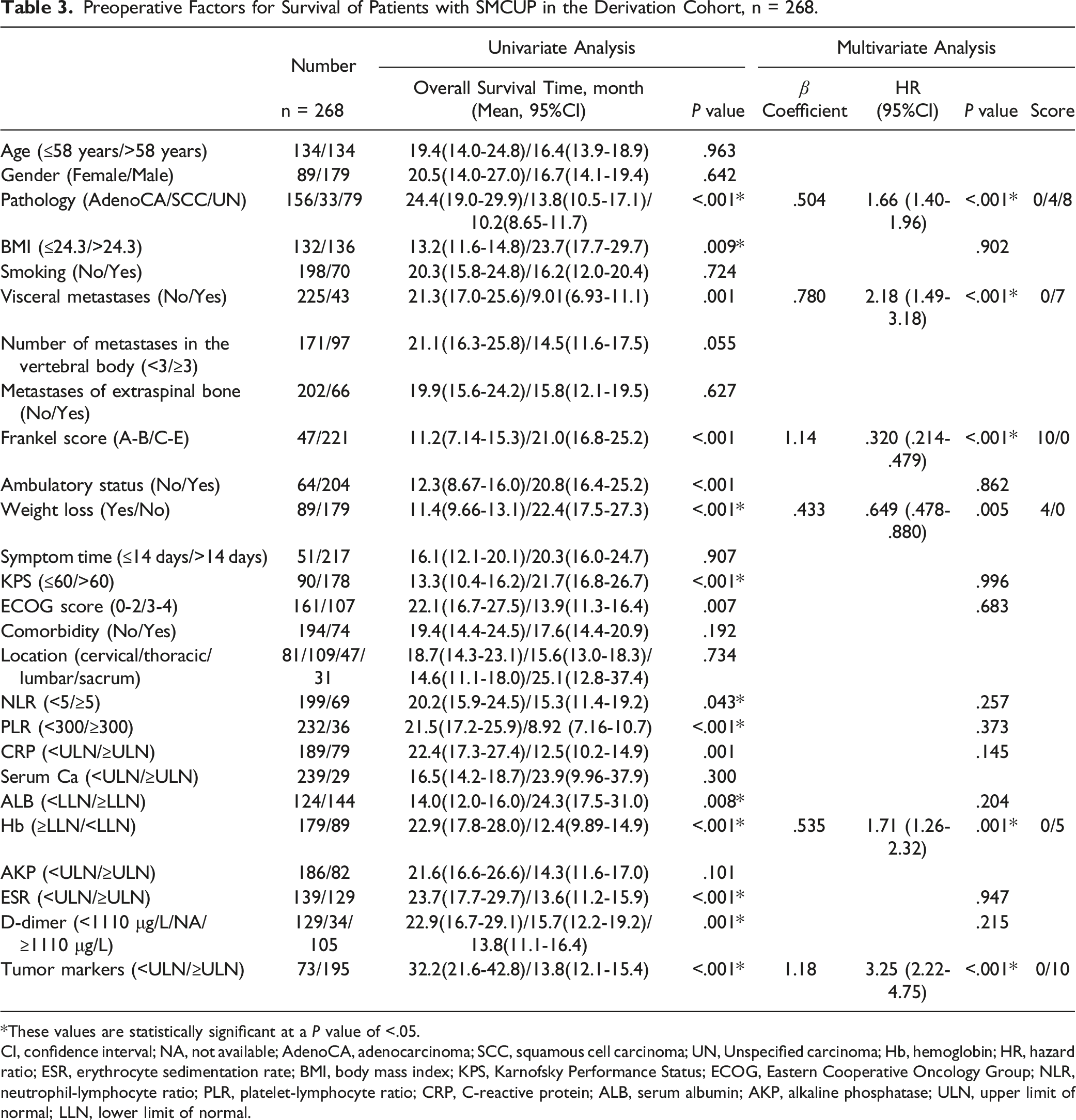
The survival prediction model included six independent prognostic factors, including pathology (
Conclusions
The novel nomogram system can be applied in survival prediction for SMCUP patients, and furtherly be used to give individualized therapeutic suggestions based on patients’ prognosis.
Introduction
Spinal metastasis from cancer of unknown primary (SMCUP) is a heterogeneous spinal metastatic malignancy with no identifiable primary cancer detected by any means present, which accounts for approximately 10% of all malignancies.1,2 SMCUP brings poor survival prognosis and debilitates life quality due to compromised neurological function including local pain, paraplegia, and corresponding complications. 3 To date, individualized, tissue-of-origin therapies did not give conclusive evidence to prolong survival prognosis.2,4 In terms of therapies on spinal metastasis, the focus emphasizes on neurological function improvement by means of surgery or radiotherapy.5,6
Until new technologies have been validated and become widely available, the fundamental principle for SMCUP estimation would be the use of focused clinical presentations. 7 Expert clinical judgment based upon survival estimation is critical in choosing the best local therapies for patients with SMCUP.1,2,8 Score models predicting survival prognosis have been derived in earlier and recent years. Mohamed et al derived an inflammation-based prognostic models using a cohort of 60 patients, and Douglas et al developed a cross-validated score model including 91 patients in both cohorts.9,10 These 2 validated models included cancer of unknown primary patients but the sample size was small. Tomita score and revised Tokuhashi score were the most used prognosis prediction systems for patients with spinal metastasis, also including SMCUP.11-13 In recent years, new predictive models for spine metastases such as SORG score were developed and demonstrated superior performance. 14 Although they included comparatively large populations, the patients were not specific and the models lacked statistically precise validation process. 15 Further, although the primary site remains occult, the pathological characteristics of the metastatic site is important as they convey histological or genetic information to guide chemotherapy or target therapies, yet until now, no score models give variables of pathology evaluation.
Hence, in this study, we asked: (1) What independent prognostic predictors are associated with survival in patients with SMCUP? (2) Can we build a comprehensive and informative score system which is useful in survival prediction for SMCUP patients? (3) Which one has the best predictive accuracy in SMCUP patients, Tomita score, revised Tokuhashi score, SORG score or the new score system?
Material and Methods
Patient Selection and Study Design
Approval was obtained from the medical ethics committee of Shanghai Changzheng Hospital (2016YFC0902100) prior to case collection. This project received an exemption from the medical ethics committee for informed consent, therefore, informed consent was not obtained. From January 2010 to December 2015, all patients diagnosed with SMCUP in Changzheng Hospital, Shanghai, were retrospectively reviewed and included in the derivation cohort. From January 2016 to October 2017, we constructed a validation cohort prospectively by consecutively including patients with SMCUP in our center. The inclusion criteria were indicated as follows1,2,9: by radiological means (CT/MRI or 18F FDG PET-CT scans) no evidence of primary tumor can be detected; the patients have no history of possible tumor or tumor mass; spinal lesions should be confirmed as metastatic malignances by biopsy or postoperative pathological studies. Overall survival (OS) refers to the interval between initiation of the diagnosis of SMCUP and death, and used as the primary end point. The study was censored on December 2020.
All 402 patients who were diagnosed with SMCUP in our institution were identified and included in the study. Five and two patients were filtered out due to incomplete laboratory or clinical data in the derivation cohort and the validation cohort, respectively. Then 17 and five patients, respectively, were excluded because of missing follow-up data. Finally, included in the study were 268 patients in the derivation cohort and 105 patients in the validation cohort, respectively (Figure 1). The flowchart for the retrospective and prospective cohorts is shown here.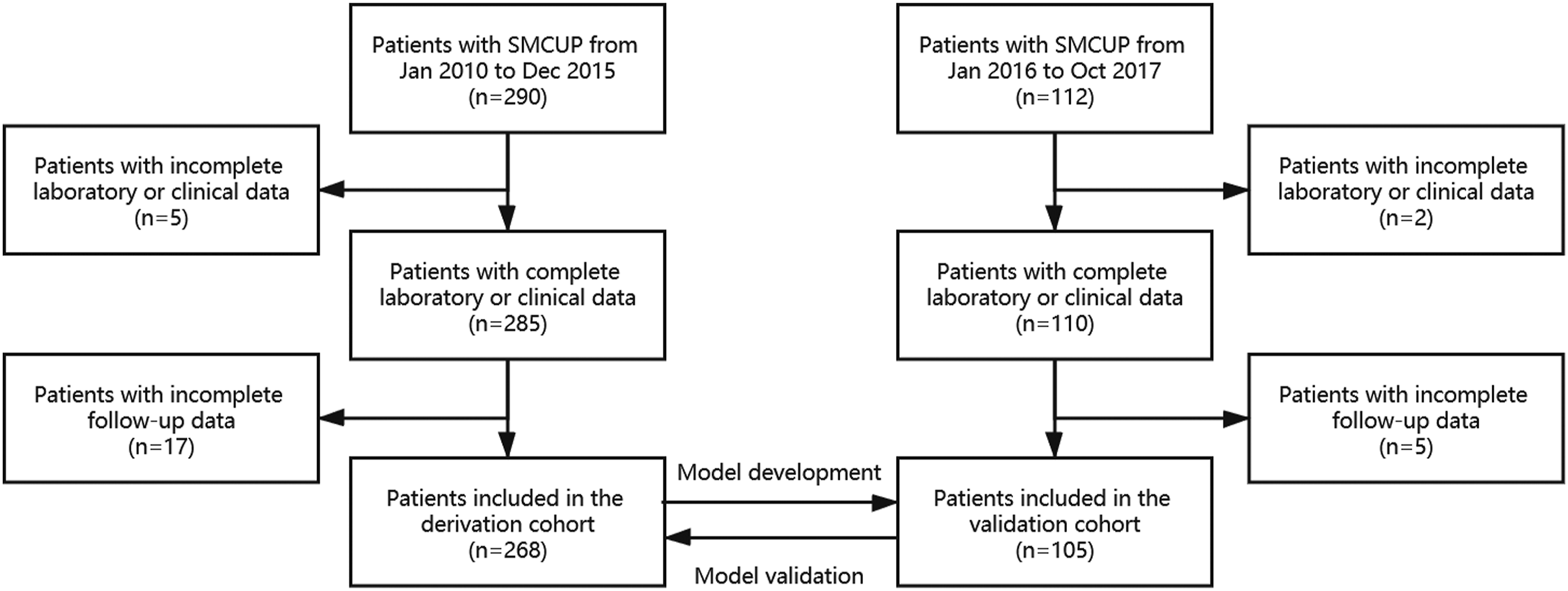
Treatment Considerations
The therapeutic strategy for SMCUP patients included surgery, radiotherapy, chemotherapy, and target therapy. 16 General conditions of patients played an important role in the therapeutic strategy decision. The patient with poor general condition was not a candidate for surgery, but for supportive care. Indications for surgery included spinal instability, progressive impairment of neurological function owing to spinal cord or nerve root compression, and intractable pain. Surgical strategies for spinal metastases were based on Tomita score and SINS score, including marginal surgery such as total en bloc spondylectomy (TES), or intralesional decompression surgery.17,18 Although all SMCUP patients had Tomita score ≥ 5, those with solitary bone lesion and no visceral metastasis were suggested to receive TES in both cohorts. Adjuvant radiation therapy was recommended following intralesional decompression surgery to achieve long-term local control. Subsequent postoperative chemotherapy and target therapy were suggested by the surgeons and physicians from multidisciplinary consultations. The final treatment choice was decided by the agreement with the patients.
Explanatory Variables
Clinical variables, laboratory tests and pathological classifications potentially affecting survival were collected according to the Delphi method, in which all the potential variables were subject to 6 rounds of oncologist consultation and consensus at national level by means of mails and visits, after which the final variables to enter the study were decided by the authors. 19
After a detailed history and a complete physical examination, blood was taken from the patients. The data of the patients were collected from themselves, their medical records, and pathology study procured by surgical or biopsy specimen. The neurological status was assessed according to the Frankel score and categorized into 2 groups (Frankel score A-B and C-E). 20 The degree of spinal cord compression was described based on the 6-point epidural spinal cord compression (ESCC) grading system. 21 The symptom duration referred to the time between the onset of neurological symptoms and the initiation of treatment, based on which spinal cord injury was categorized as acute to subacute (<14 days), and chronic (≥14 days). 14 According to the median BMI of the patients in the training cohort, patients with a BMI≤24.3 kg/m2 were categorized as a low-weight group, and otherwise as a high-weight group. Definition of weight loss was % weight loss>6%, and was calculated according to the suggestions from Martin L et al. 22 The neutrophil-lymphocyte ratio (NLR) was calculated by dividing the absolute neutrophil count by the absolute lymphocyte count, with NLR>5 considered elevated and the same calculation applied for platelet-lymphocyte ratio (PLR), with 300 as the cut-off point.23,24 The performance status was assessed according to Eastern Cooperative Oncology Group (ECOG) performance scores and Karnofsky Performance Status (KPS).16,25 Pre-treatment hemoglobin (Hb), erythrocyte sedimentation rate (ESR), C-reactive protein (CRP), alkaline phosphatase (AKP), serum albumin (ALB) and serum calcium (Ca) were evaluated and categorized based on the institutional reference values. Tumor markers including carcinoembryonic antigen (CEA), cancer antigen 125 (CA125), cancer antigen 199 (CA199), cancer antigen 724 (CA724), prostate-specific antigen (PSA, only for male) and alpha-fetoprotein (AFP) were tested before any treatment and were also categorized according to the reference values. Anyone of the above tumor markers higher than the institutional reference value was abnormal.
Pathology tissue from biopsy specimen was immediately fixed in 10% formalin, embedded in paraffin, cut in thin pieces, and stained with Hematoxylin-Eosin stain after biopsy procedure or surgical operation. 26 Histological examinations were performed by three pathologists unaware of the clinical data. Pathological types were categorized into squamous cell carcinoma, adenocarcinoma, and unspecified carcinoma.9,27
Model Derivation
Statistical survival analysis to identify independent risk factors for survival was performed by SPSS 23.0 (SPSS, Chicago, IL). Model derivation steps were performed by R project, version 3.4.3 (http://www.r-project.org/). Continuous variables are expressed as mean standard deviation (SD) and compared using an unpaired, two-tailed
Model Validation
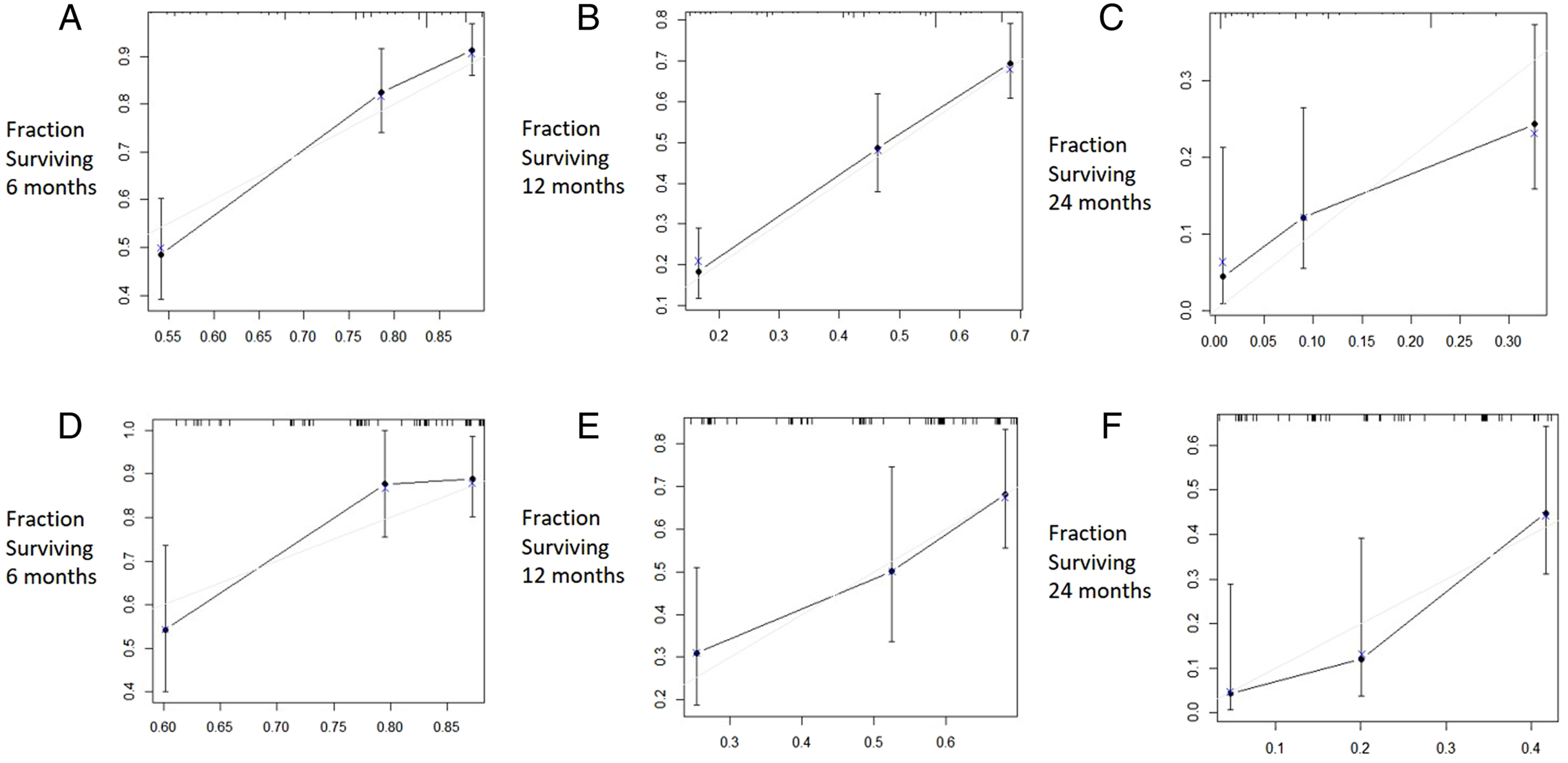
The model was subjected to 1000 bootstrap resamples for validation with the primary derivation or validation cohort by R project. 28 A concordance index (C-index) was estimated for discrimination evaluation.28,29 The value of the C-index always ranges from .5 to 1.0. Higher C-index indicates better ability to correctly discriminate the outcome with the model. Calibration refers to the agreement of predicted and actual risk. 29 We constructed a calibration plot to determine whether the predicted and observed survival probabilities were in concert at 6-, 12-, and 24-month follow-up.
Model Comparison With Existing Prognostic Systems
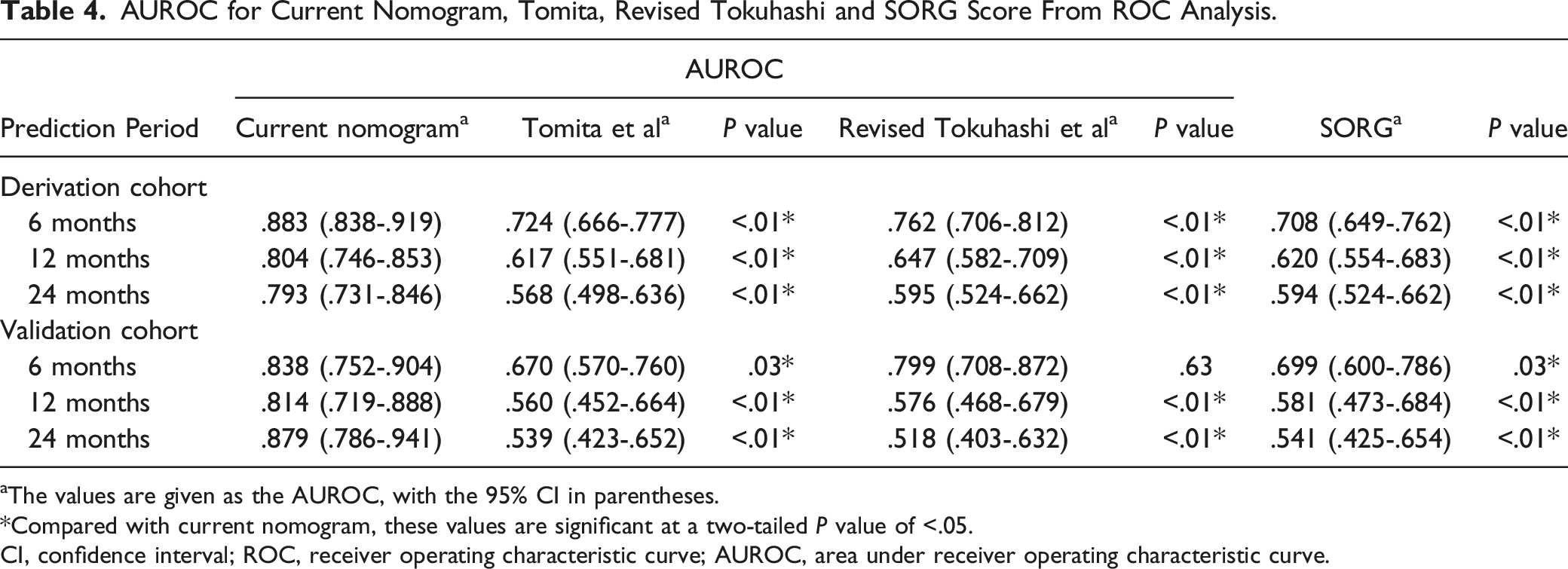
Tomita score, revised Tokuhashi score and SORG score were commonly used for survival prediction in SMCUP. Accuracy of this model was evaluated with the receiver operating characteristic curve (ROC) and area under ROC (AUROC). A cumulative risk score was calculated for each patient in both the validation and derivation cohort. ROC curves were drawn with 1-specificity and sensitivity at
Results
Demographic Characteristics
General Characteristics of 373 Patients with SMCUP.
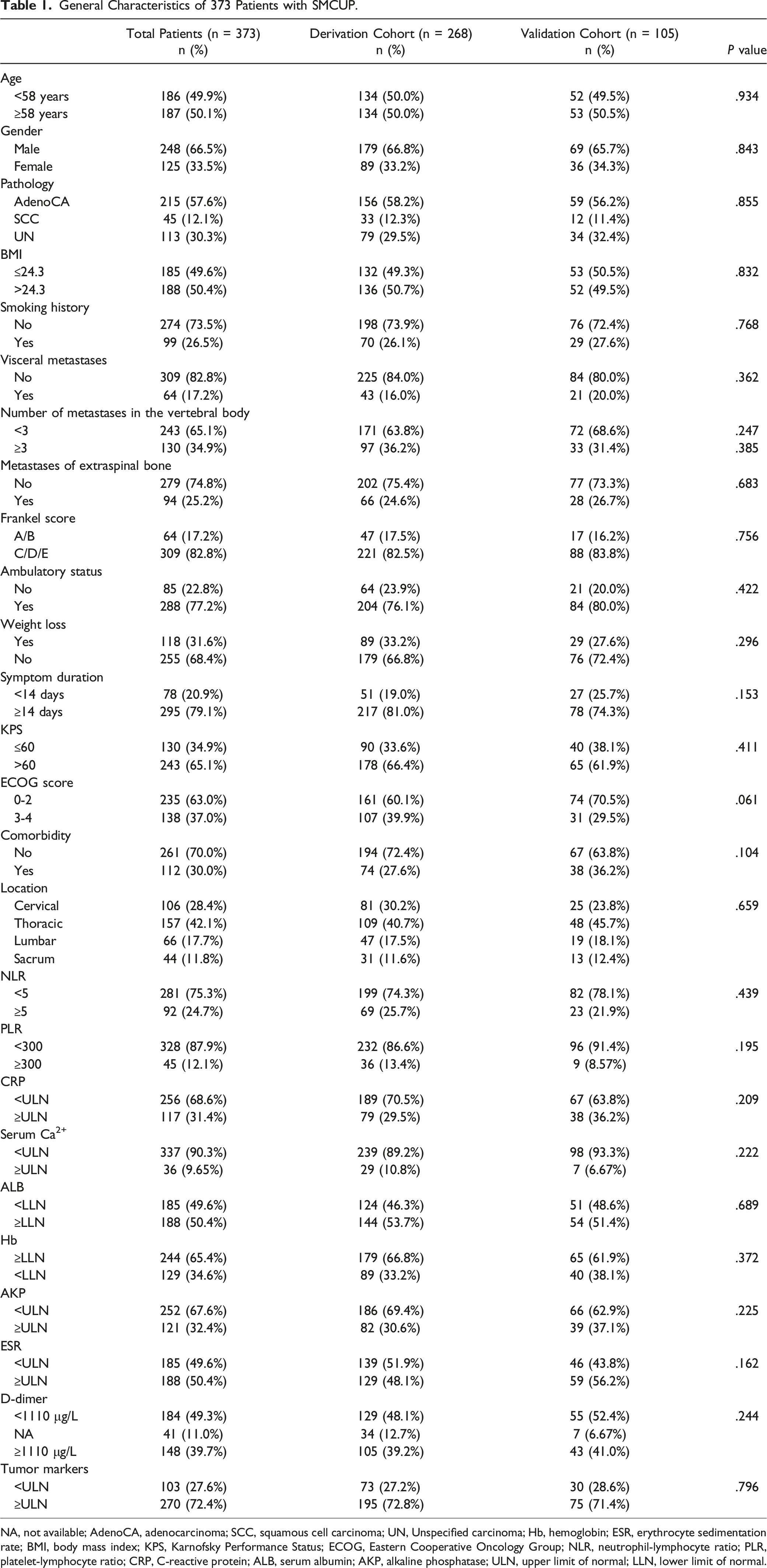
NA, not available; AdenoCA, adenocarcinoma; SCC, squamous cell carcinoma; UN, Unspecified carcinoma; Hb, hemoglobin; ESR, erythrocyte sedimentation rate; BMI, body mass index; KPS, Karnofsky Performance Status; ECOG, Eastern Cooperative Oncology Group; NLR, neutrophil-lymphocyte ratio; PLR, platelet-lymphocyte ratio; CRP, C-reactive protein; ALB, serum albumin; AKP, alkaline phosphatase; ULN, upper limit of normal; LLN, lower limit of normal.
Treatment of Patients With Spinal Metastasis From Cancer of Unknown Primary
Treatments of Patients with SMCUP.
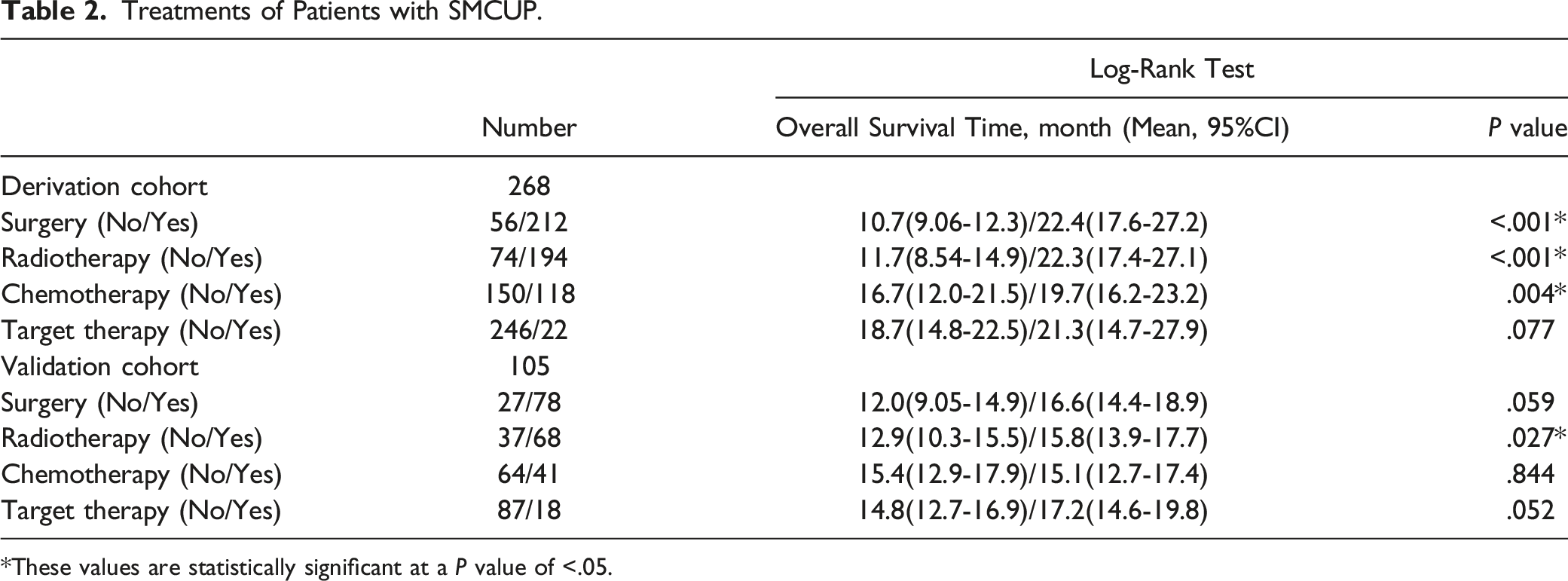
*These values are statistically significant at a
Factors Associated With Survival in Patients With Spinal Metastasis From Cancer of Unknown Primary
Univariate and multivariate analyses were based on the 268 patients in the derivation cohort. Altogether, 16 prognostic variables were statistically significant ( Kaplan-Meier survival curves are shown based on six independent factors for prognosis in the derivation cohort (A. Pathology, B. Visceral metastases, C. Frankel score, D. Weight loss, E. Serum tumor markers, F. Hemoglobin).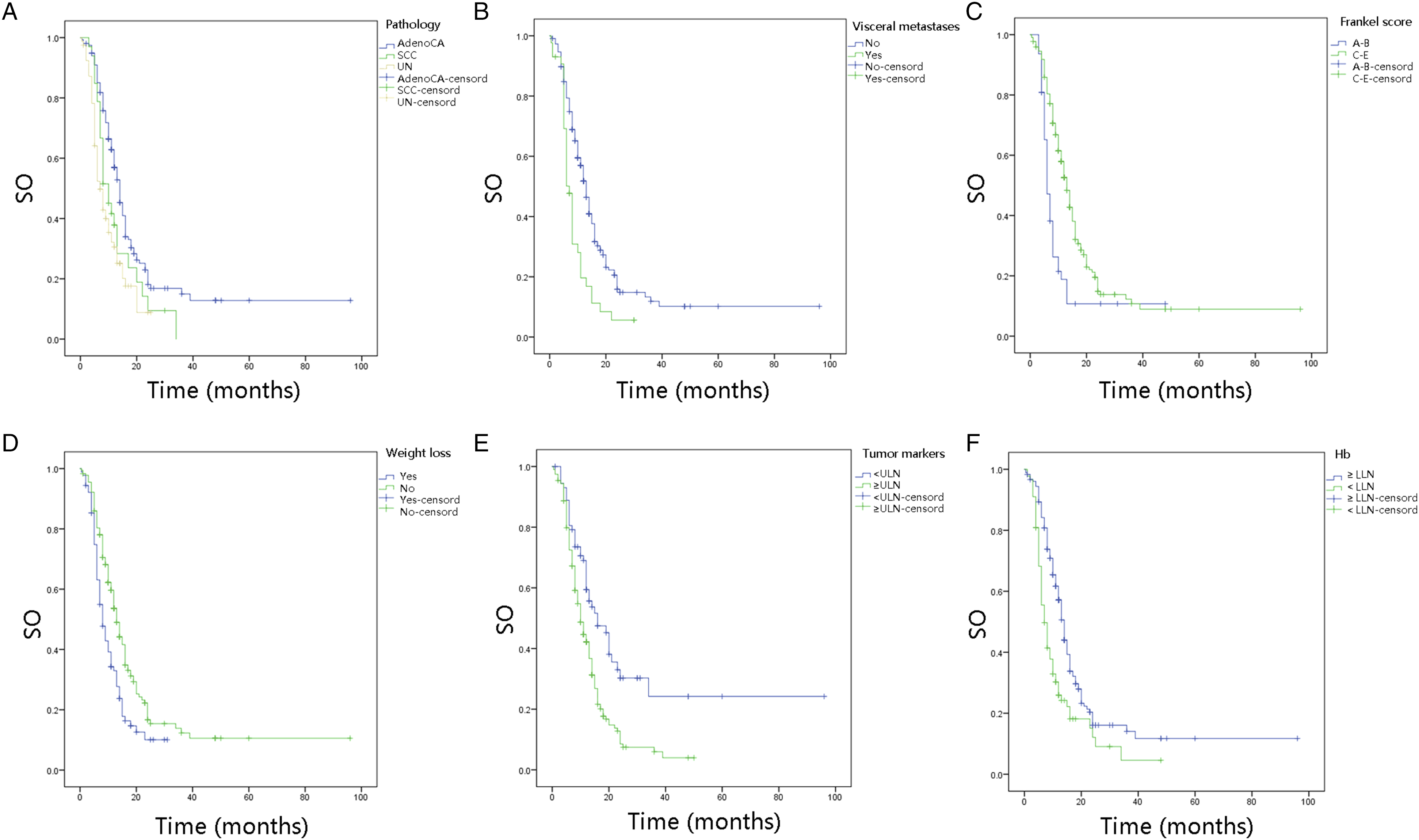
Model Derivation and Validation
The six independent variables were used to develop this nomogram model (Figure 3). The score was attributed according to the The nomogram for survival prediction of patients with SMCUP is presented here (AdenoCA, adenocarcinoma; SCC, squamous cell carcinoma; UN, Unspecified carcinoma; ULN, upper limit of normal; LLN, lower limit of normal). Preoperative Factors for Survival of Patients with SMCUP in the Derivation Cohort, n = 268. *These values are statistically significant at a CI, confidence interval; NA, not available; AdenoCA, adenocarcinoma; SCC, squamous cell carcinoma; UN, Unspecified carcinoma; Hb, hemoglobin; HR, hazard ratio; ESR, erythrocyte sedimentation rate; BMI, body mass index; KPS, Karnofsky Performance Status; ECOG, Eastern Cooperative Oncology Group; NLR, neutrophil-lymphocyte ratio; PLR, platelet-lymphocyte ratio; CRP, C-reactive protein; ALB, serum albumin; AKP, alkaline phosphatase; ULN, upper limit of normal; LLN, lower limit of normal. The calibration curves are shown for predicting patient survival at (A) 6 months in the derivation cohort, (B) 12 months in the derivation cohort, (C) 24 months in the derivation cohort, (D) 6 months in the validation cohort, (E) 12 months in the validation cohort, and (F) 24 months in the validation cohort. Nomogram-predicted probability of overall survival is plotted on the 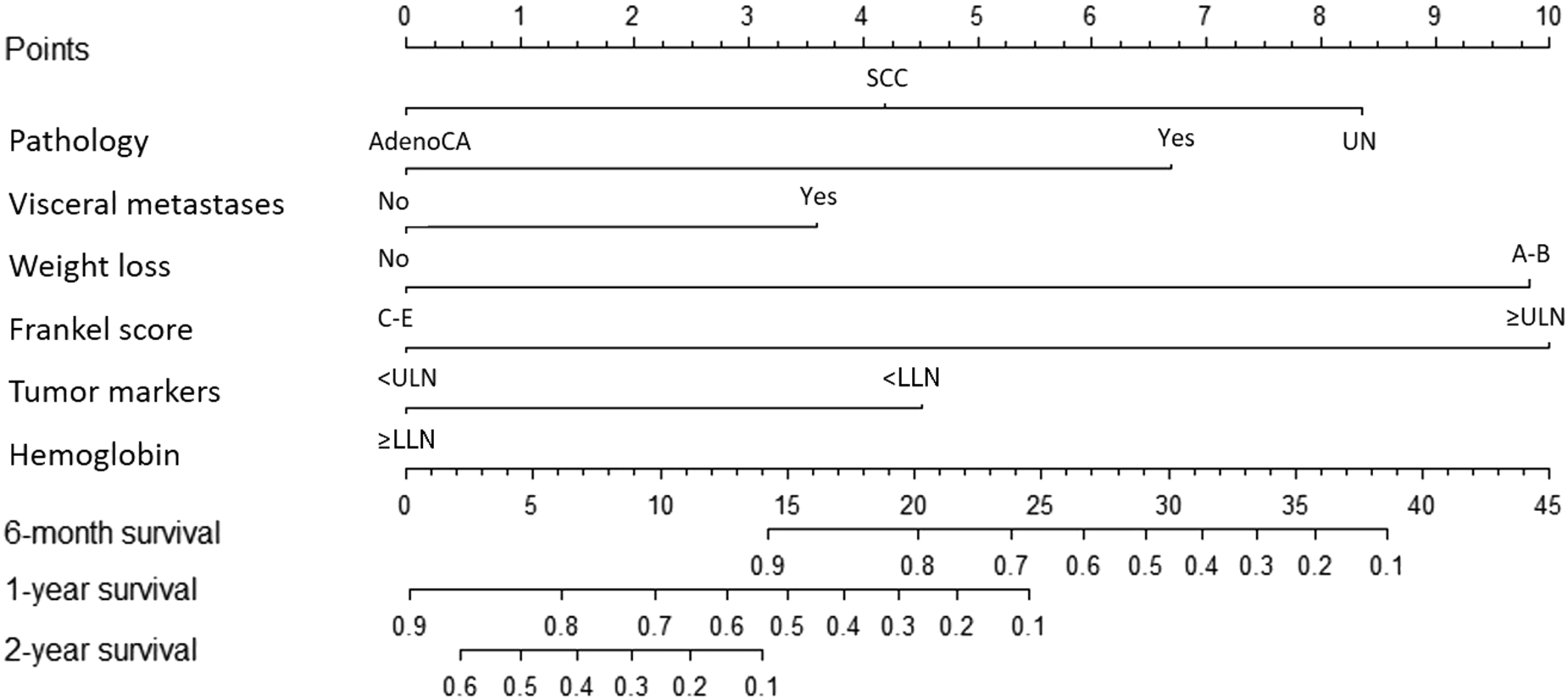
Model Comparison With Existing Score Systems
AUROC for Current Nomogram, Tomita, Revised Tokuhashi and SORG Score From ROC Analysis.
aThe values are given as the AUROC, with the 95% CI in parentheses.
*Compared with current nomogram, these values are significant at a two-tailed
CI, confidence interval; ROC, receiver operating characteristic curve; AUROC, area under receiver operating characteristic curve.
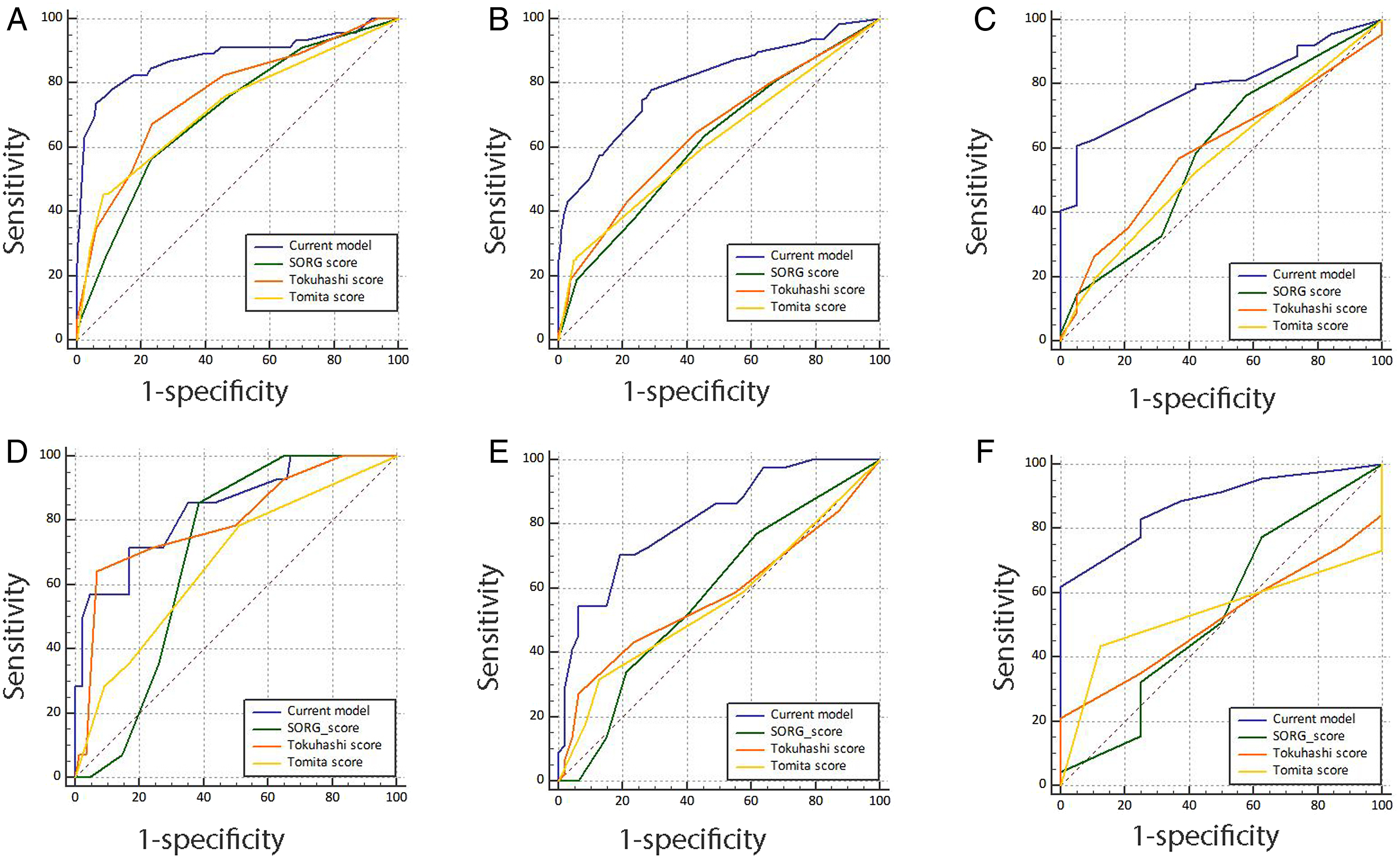
ROC curves are used for comparing current nomogram, SORG score Tomita score and revised Tokuhashi score in survival estimation at (A) 6 months in the derivation cohort, (B) 12 months in the derivation cohort, (C) 24 months in the derivation cohort, (D) 6 months in the validation cohort, (E) 12 months in the validation cohort, and (F) 24 months in the validation cohort. ROC curves are drawn with 1-specificity and sensitivity at
Discussion
Approximately 15% of cancer patients develop spinal metastasis and the main treatment modalities are surgery and radiotherapy.5,8 Spinal metastasis can cause paraplegia, severely undermined life qualities and diminished survival prognosis.16,25 Circumferential decompression followed with radiotherapy could achieve better functional outcome for patients with relatively good preoperative conditions or longer survival prognosis.5,31 To optimally stratify treatment options, it is essential to take into account overall condition with clinical variables, laboratory tests and pathological classifications.11,32 For occult primaries with spinal metastasis, or SMCUP, primary-cancer categorization or personalization method becomes impossible and these patients usually have very poor survival prognosis in comparison.27,33 Identification of a risk model to predict survival is thereby vital for individualization of treatment for patients with SMCUP. A total of 373 individual patients met the study entry criteria and were included in the analysis. According to the spinal levels which contributed to the major neurological symptoms of patients, the thoracic spine was the most common site, accounting for 42.1% of the cases, followed by the cervical spine (28.4%), lumbar spine (17.7%), and sacrum (11.8%). The 6-point ESCC scale developed and validated by Bilsky et al 21 graded the degree of epidural tumor extension and spinal cord compression based on MR imaging. Based on the 6-point ESCC scale, 15, 6, 9, 20, 248 and 75 patients belonged to grade 0, 1a, 1b, 1c, 2 and 3, respectively. This study developed a simple, readily nomogram model to estimate survival time for SMCUP. Next, a prospective cohort validation also demonstrated good performance of the model in OS prediction for SMCUP. Finally, model comparison by AUROC analysis of existing SMCUP staging systems showed that the current model had the best accuracy in survival prediction.
Clinical and laboratory variables like general conditions of patients, inflammation determinants (such as PLR and NLR) and tumor metastatic status were shown to be associated with survival of patients with SMCUP. And six independent prognostic factors were identified in the nomogram development. The nomogram illustrated Frankel score and level of serum tumor makers as sharing the largest contribution to prognosis, followed by visceral metastasis and pathological classifications. Weight loss and pre-treatment hemoglobin level showed a moderate impact on the survival. Visceral metastases, Frankel score and weight loss were commonly considered and included in previous prognosis prediction models for spinal metastasis.11,13,34 Pre-treatment hemoglobin could indicate general condition of a patient. Paulino et al 14 developed SORG model for patients with spine metastases, also demonstrated that lower hemoglobin level was independently associated with decreased survival. Serum tumor markers were generally not considered to be particularly helpful in identifying the primary tumor, especially among the adenocarcinoma. 1 However, they may be helpful in monitoring the response to treatment and prognosis prediction in various cancers.35,36 Increased level of a serum tumor maker might indicate higher tumor burden and larger tumor size. 37 In our study, serum tumor markers were tested before any treatment. Results showed that tumor markers higher than the upper limit indicated poor prognosis for patients with SMCUP. Similarly, in recent study for combined hepatocellular carcinoma and cholangiocarcinoma patients, Hou et al 38 developed a novel Tumor Marker Score (TMS) based on preoperative AFP, CEA, and CA199. Abnormally high tumor markers were associated advanced TMS as well as worse prognosis of these patients. Some variables like BMI, ambulatory status, KPS, ECOG score, NLR, PLR, CRP, level of albumin, ESR, and level of D-dimer were also proven to be candidates impacting the OS of patients with SMCUP in the univariate analysis. However, these variables failed to be included in the final model. One of the possible reasons was that different variables like BMI, KPS, ECOG, albumin, Frankel score, and ambulatory status were considered in our model, and they were all used for evaluating general condition or neurologic deficit of patients. Only Frankel score was an independent factor and included in the nomogram eventually. Another possible reason was that they might play a minor role in survival prediction.
For SMCUP, a group of heterogeneous types of cancers, indeed, although clinical variables may give strong clues to survival prognosis, it is inadequate to stratify patient survival prognosis only by clinical manifestations without proper reference to pathological aggressiveness.1,15 The current model included pathological types, which plays vital roles in clinical judgment of therapeutic regimens.2,39 For SMCUP, patients with adenocarcinoma had better prognosis than those with squamous cell carcinoma, and unspecified carcinoma indicated poor clinical outcome. Also, in another survival study for spinal metastases from non-small cell lung cancer in our institution, the patients diagnosed with adenocarcinoma seemed to have longer OS than those with squamous cell carcinoma with 18.3 months and 15.6 months, respectively. 16 Notably, previous CUP models by Tomita et al and revised Tokuhashi et al only included clinical variables, but the current model coordinated clinical presentations, results of laboratory tests as well as pathological details, and this may be probably why this novel model shows the best accuracy.11,13
The nomogram model not only has clinical implications generalizable to clinical prediction of patient survival but also serves as guidelines on multidisciplinary treatment stratification process. Based upon the predicted survival rate, it is possible to calculate the survival prognosis and thus the model may help make relevant decisions on proper management. For instance, it was suggested that patients with poor 6-month survival rate might not benefit from surgical therapy,40-43 while that for patients with higher chance of more than 3 or 6 months’ survival, it would be appropriate to include surgery or radiotherapy into decision-making process.13,34 According to the model, a patient with score >33 would have a 6-month survival rate <30% and a 12-month survival rate <5%, probably predicting poor survival after aggressive therapies considering the treatment-associated impact. However, models based on quality of life should be encouraged because they may better help clinical decision-making process and give more individualized information.
Although the study has the strength of respectable sample size and sufficient duration of follow-up time, which increased the statistical power and reliability of results, this model bears limitations. First, all the items making up this nomogram and their weightings were obtained from the cohorts in one single institution. And future multi-institutional studies to validate these results are necessary. Second, a temporal cohort was selected as the external validation set. Improved therapeutic techniques and experience from 2016 to 2017 might improve the OS of patients in validation cohort compared to that in the training cohort from 2010 to 2015. Third, the SORG score was developed from a cohort of patients who had undergone a surgical procedure for spine metastases. However, only 77.7% patients in this study underwent surgical treatments. Differences from inclusion criteria might impact an objective comparison between SORG score and our nomogram. Finally, decision-making process of SMCUP management should be based upon both post-therapeutic survival prognosis and potential elevated quality of life, and further studies are encouraged to quantifiably predict quality of life probably via validated scales or questionnaires.
Conclusions
In conclusion, the study represents a reliable and accurate nomogram model combining clinical presentations, results of laboratory tests as well as pathological details for estimation of survival rate in SMCUP patients. The model may also be a useful tool for clinicians to stratify the level of applicable management in SMCUP patients according to their survival prognosis.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Key Research and Development Project of China (2016YFC0902100), the Shanghai Science and Technology Committee (Grant No. 17411950301, 21MMC1930100 and 19411962700), the Logistics Support Department of PLA (21QNPY044) and Youth Doctor Assistance Program Funds of Shanghai Changzheng Hospital (Minglei Yang).
