Abstract
Background:
The surgeon’s intraoperative assessment of the curative potential of tumor resection following gastrectomy adds new information that could help clinicians and patients by predicting survival.
Methods:
All patients in Sweden undergoing gastric cancer resection between 2006 and 2018 were grouped according to a prospectively registered variable; the surgeon’s intraoperative assessment of the curative potential of surgery: curative, borderline curative, or palliative. Factors affecting group allocation were analyzed with multivariable logistic regression, while survival was analyzed using multivariable Cox regression and the Kaplan–Meier method. Positive predictive value (PPV) and negative predictive value (NPV) were calculated.
Results:
Of 2341 patients undergoing gastric cancer resection, 1547 (71%) were deemed curative, 340 (15%) borderline curative, and 314 (14%) palliative (140 missing assessments). Advanced stage increased the risk of borderline curative resection (Stage III, odds ratio (OR) = 6.04, 95% confidence interval (CI) = 3.92–9.31), as did emergency surgery OR = 3.31 (1.74–6.31) and blood loss >500 mL; OR = 1.63 (1.06–2.49). Neoadjuvant chemotherapy and multidisciplinary team (MDT) discussion both decreased the risk of borderline curative resection, OR = 0.58 (0.39–0.87) and 0.57 (0.40–0.80), respectively. In multivariable Cox regression, the surgeon’s assessment independently predicted worse survival for borderline curative (hazard ratio (HR) = 1.54, 95% CI = 1.29–1.83) and palliative resections (HR = 1.76, 95% CI = 1.45–2.19), compared to curative resections. The sensitivity of the surgeon’s assessment of long-term survival was 96.7%. The PPV was 50.7% and the NPV was 92.1%.
Conclusion:
The surgeon’s intraoperative assessment of the curative potential of gastric cancer surgery may independently aid survival prediction and is analogous to prognostication by pathologic Staging. Advanced disease, emergency surgery, and a high intraoperative blood loss, increases the risk of a borderline curative or palliative resection. Conversely, neoadjuvant treatment and MDT discussion reduce the risk of borderline curative or palliative resection.
Context and Relevance
Investigation the prognostic value of the surgeon’s own intraoperative assessment of the curative potential of a gastric cancer resection has not previously been performed. Such prognostication may be important to tailor patient information and care in the period after surgery but before pathological analysis of the specimen has been concluded. This study shows that predictions of survival based upon the surgeon’s intraoperative assessment is sensitive and comparable to predictions based on pathologic tumor stage. In addition, there is the added benefit of being immediately available in the post-operative period. Survival was markedly reduced in patients where the operating surgeon deemed the resection borderline curative or palliative—in contrast to when the operating surgeon deemed the resection curative. This study finds important risk factors associated with an increased risk of non-curative resection and the study highlights the ability of the surgeon to predict long-term survival.
Introduction
Gastric cancer is currently the fifth most common form of cancer diagnosed globally with a dismal prognosis representing the third leading cause of cancer death. 1 A radical surgical resection, often combined with neoadjuvant or peri-operative chemotherapy as part of multimodal treatment, is paramount for the curative treatment of patients with localized disease.2–4 Survival following gastric cancer resection is influenced by many well-researched factors, such as age, comorbidities and tumor stage. Previous studies have also outlined the importance of tumor-free resection margins and clearance of any lymph nodes containing malignant disease. Different methods to ascertain radical resection with negative margins have been explored. Intraoperative frozen sections, alongside a standardized surgical approach, including appropriate lymphadenectomy, 5 reduces the risks of leaving residual tumor 6 and involved regional lymph nodes.
While some gastric cancer resections are straightforward and not technically demanding, others require complex intraoperative decision-making to enable a complete resection. Furthermore, despite meticulous preoperative work-up, the operating surgeon will often learn new information about the disease at the time of surgery. During the procedure, the operating surgeon will ascertain details about the extent of disease, including nodal involvement, allowing an expert opinion to be formed as to whether the operation will be curative or palliative in nature. This opinion on the curative potential of the operation is prospectively reported to the Swedish National Registry for Esophageal and Gastric Cancer (NREV) in all patients undergoing surgery.
In common practice, pathological assessment of the resected specimen is the most utilized factor to predict survival in patients having undergone radical surgery. Previous studies show an association between the American Joint Committee on Cancer (AJCC) and Union for International Cancer Control (UICC) tumor/node/metastasis (TNM) classification system for tumor stage 7 and resection margin involvement, 8 and survival.
The present study aims to assess whether the surgeon’s own intraoperative assessment of the curative potential of the resection can, independently, predict survival. In addition, we aimed to identify any risk factors for the operation being deemed borderline curative or palliative.
Methods
Study sample
This was a retrospective, multi-center, nationwide cohort study. The source population included all patients with gastric cancer (International Classification of Disease, 10th version (ICD-10) C16.0C-C16.9), diagnosed in Sweden between 1 January 2006 and 26 February 2018. Patients were identified in the Swedish NREV. The individual patient data were cross-linked between NREV, the Swedish Prescribed Drug Register, Cause-Of-Death Register, Cancer Register, Patient Register, and the Longitudinal Integrated Database for Health Insurance and Labor Market Studies (LISA database). All the above registries are well described, researched, and validated.9–16 Patients who had undergone gastric cancer resection with or without oncologic treatment were included. Patients who underwent endoscopic resections and patients with clinical tumor Stage 0 were excluded.
Exposures
Since the start of NREV in 2006, the register has included a mandatory variable reported by the operating surgeon (usually immediately following the resection) that could assume the three separate values of either “curative resection,” “borderline curative resection,” or “palliative resection.” The variable constitutes a professional opinion on the curative potential of the resection by the operating surgeon. The patients in the present study were grouped and analyzed according to this variable.
Pre- and postoperative information on tumor and patient characteristics, alongside details regarding treatment, were retrieved from NREV. Patients had undergone standard preoperative clinical staging with endoscopy, computed tomography (CT), and in some cases, staging laparoscopy, with no preoperative clinical suspicion of widespread disease.
Data on comorbidities at the time of diagnosis were compiled from NREV, the National Patient Register and the Prescribed Drug Register. For each patient, a Charlson comorbidity index (CCI) was calculated. 17 Data regarding operative volumes of individual surgeons were not available for analysis.
Statistical analysis
Group comparisons were made between patients who underwent either curative, borderline curative, or palliative resection, as assessed by the operating surgeon. In a directed acyclic graph (DAG) model, the following covariates and confounders were identified to be associated with the surgeon’s assessment of the curative potential of the tumor resection: age (below 50 and thereafter in 10-year intervals up to 90 and above), sex, comorbidities (CCI; 0, 1–2, ⩾3), clinical stage according to the AJCC/UICC TNM classification system eighth edition (Stages I–IVb), neoadjuvant treatment (yes or no), timing of surgery (elective or emergency), intraoperative blood loss (less or more than 500 mL), tumor location (proximal or distal), treatment recommendation in a multidisciplinary team (MDT; yes or no) discussion, and staging laparoscopy (yes or no). Risk factors for the cancer resection being assessed as borderline curative or palliative were analyzed with uni- and multivariable logistic regression presented as odds ratios (OR) with 95% confidence intervals (CI).
Survival was calculated from the date of diagnosis and patients were followed until death, emigration or the end of follow-up (17 April 2018). Unadjusted overall survival for patients was displayed using Kaplan–Meier plots. The impact of the surgeon’s assessment of the curative potential of the resection on survival was analyzed using the Cox proportional hazard model calculating hazard ratios (HRs) with 95% CI, including adjustment for age, sex, comorbidities, clinical or pathologic TNM, neoadjuvant treatment, timing of surgery, intraoperative blood loss, staging laparoscopy, MDT discussion, resectional margin involvement, and reported extent of lymphadenectomy.
The impact of the surgeon’s assessment on survival for sub-groups of clinical or pathologic stage as well as with or without MDT discussion was calculated and presented with separate supplementary Kaplan–Meier plots.
Sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV) accuracy of the surgeon’s intraoperative assessment (curative/palliative), as well as for clinical and for pathologic staging were calculated. The chosen outcome for positive prediction was survival more than 5 years from diagnosis (yes/no).
The study was approved by the regional ethical review board in Stockholm (2013/596-31/3 and 2016/1486-32).
Results
Patients
Between 2006 and 2018, 6713 patients were diagnosed with gastric cancer in Sweden. Of these, the following patients were excluded: 3814 patients (57%) did not undergo surgery, 487 patients (7.3%) underwent surgical exploration without resection due to locally advanced or metastatic disease, 37 patients (0.6%) had an endoscopic removal alone of superficial gastric cancer, and an additional 34 patients (0.5%) had clinical Stage 0 disease. The remaining 2341 patients (35%) all had a gastric cancer resection (51% subtotal, 43% total, and 6% were atypical resections), and comprised the final study population for further analysis (Fig. 1).
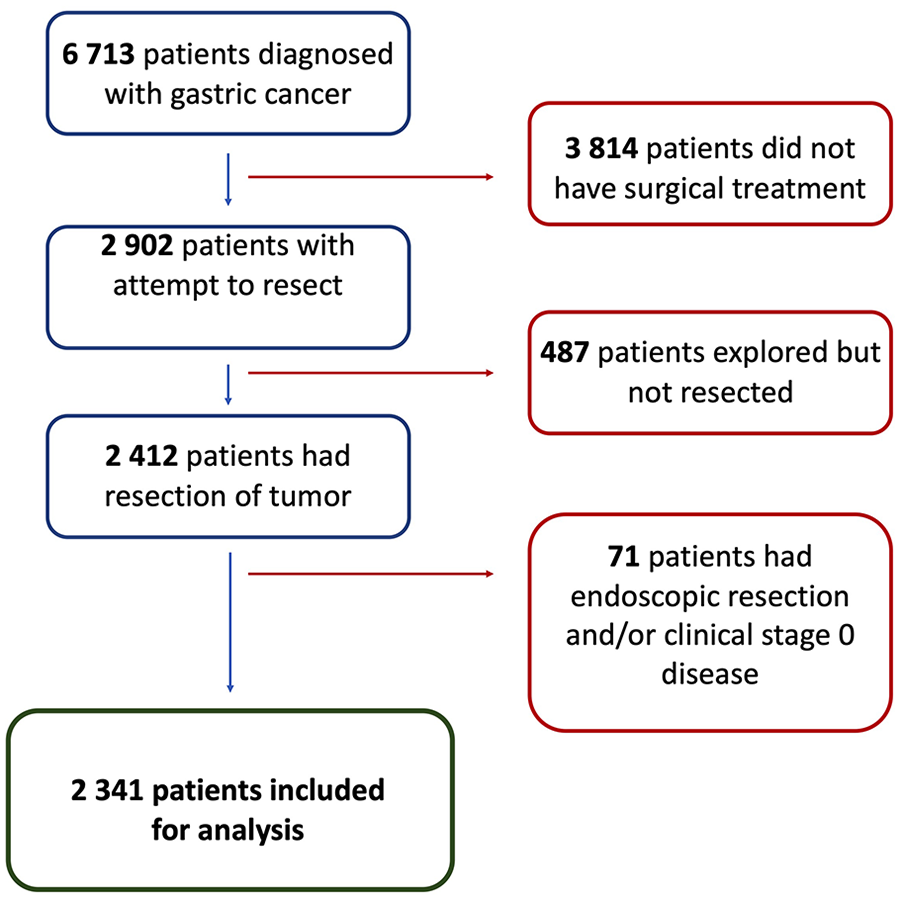
Flow chart displaying patient selection for main analysis.
In 2201 (94%) of the resected patients, there was a recording of the operating surgeon’s assessment on the curative potential of the procedure, while in 140 patients (6%) that information was missing. The operating surgeon deemed the resection curative in 1547 patients (71%), borderline curative in 340 (15%), and palliative in 314 patients (14%).
Baseline characteristics are presented in Table 1.
Baseline characteristics of 2341 patients resected for gastric cancer between 2006 and 2018 in Sweden, grouped according to the operating surgeon’s assessment of the curative potential.
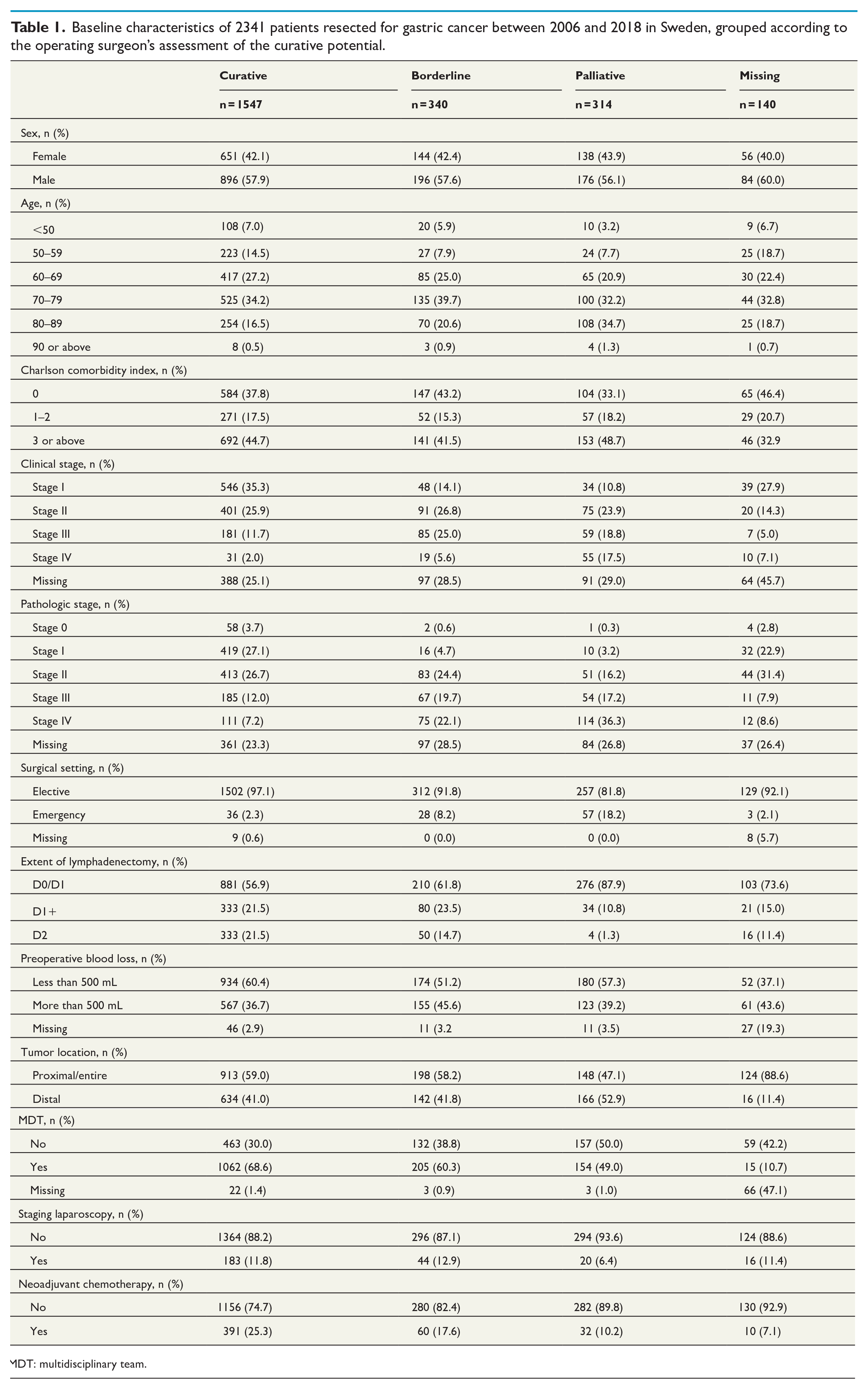
MDT: multidisciplinary team.
Risk factors for borderline curative resection
Following analysis by multivariable logistic regression, factors associated with an increased risk of the operation being assessed as borderline curative included higher clinical stage (Stage II: OR = 2.75, 95% CI = 1.85–4.09; Stage III: OR = 6.04, 95% CI = 3.92–9.31; Stage IV: OR = 6.94, 95% CI = 3.50–13.8), emergency resection (OR = 3.31, 95% CI = 1.74–6.31), and increased intraoperative blood loss (>500 mL: OR = 1.63, 95% CI = 1.06–2.49). Patients who had their case presented at an MDT (OR = 0.57, 95% CI = 0.40–0.80), or received neoadjuvant chemotherapy (OR = 0.58, 95% CI = 0.39–0.87) had a reduced risk of an operation being deemed borderline curative. Age, sex, comorbidities, tumor location, and previous staging laparoscopy were not associated with a borderline curative outcome (Table 2).
Multivariable logistic regression model for the risk of the outcome borderline curative gastric cancer resection (a) or palliative gastric cancer resection (b), as assessed by the operating surgeon.
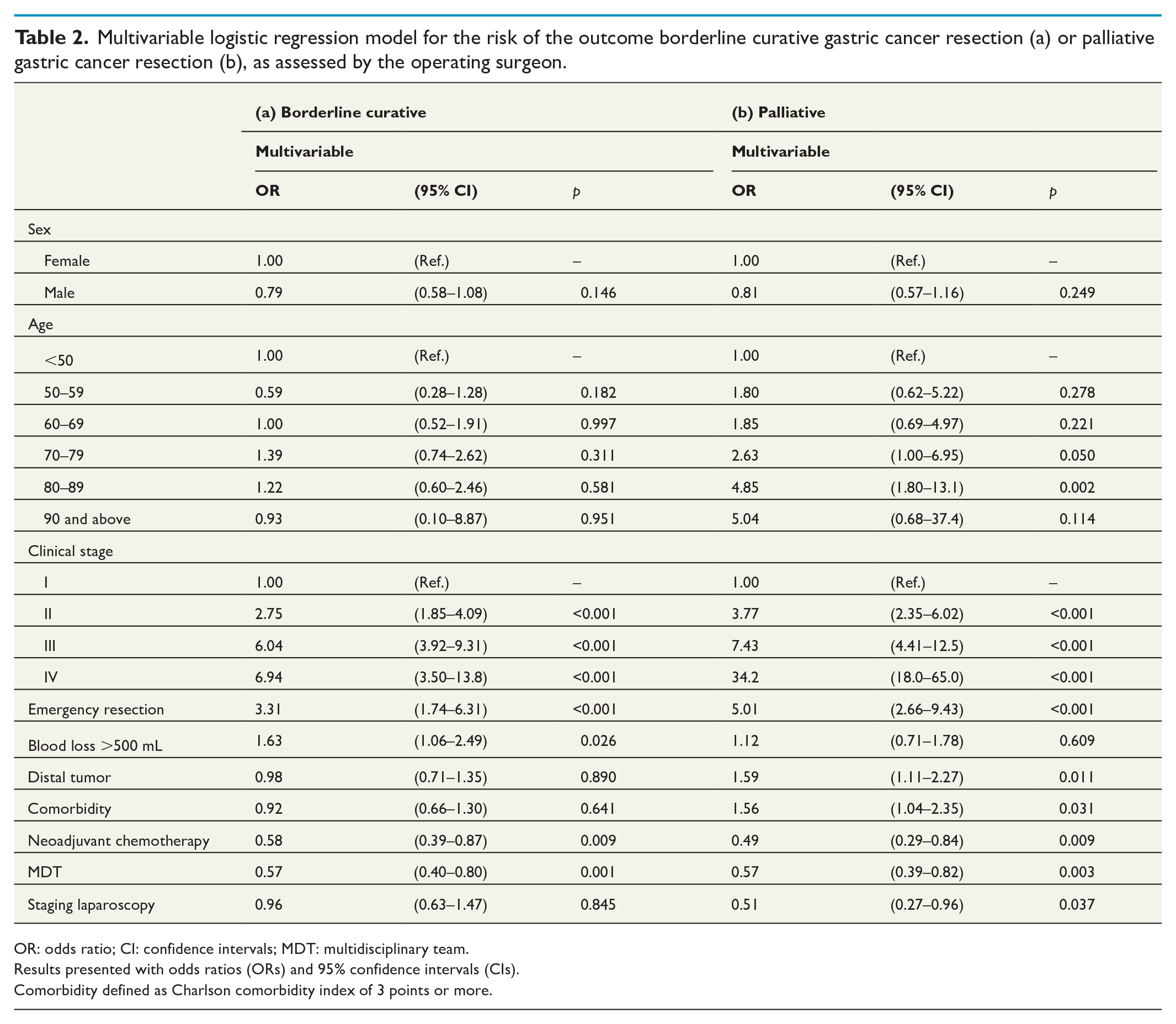
OR: odds ratio; CI: confidence intervals; MDT: multidisciplinary team.
Results presented with odds ratios (ORs) and 95% confidence intervals (CIs).
Comorbidity defined as Charlson comorbidity index of 3 points or more.
Risk factors for palliative resection
Patients who were instead deemed to have had a palliative resection more often had a distal tumor or an emergency operation (Table 1). Risk factors and point estimates for having a palliative resection were very similar to those of patients who were deemed to have undergone a borderline curative resection (Table 2). Notable differences to borderline curative patients were that excessive blood loss was not associated with a palliative outcome and a preoperative staging laparoscopy seemed to reduce the risk of palliative resection (OR = 0.51, 95% CI = 0.27–0.96). Distal tumor location (OR = 1.59, 95% CI = 1.11–2.27) and advanced comorbidity were significantly associated with the resection being deemed palliative (CCI 3 or above: OR = 1.56, 95% CI = 1.04–2.35).
The surgeon’s assessment of the curative potential of the resection and survival
The overall 5-year survival for the study population (n = 2341) was 38% with a median survival of 36 months. Among patients assessed as having a curative resection, the 5-year survival was 49.8% (95% CI = 47.1–52.5), for borderline curative, 15.7% (95% CI = 11.8–20.1), and palliative 6.7% (95% CI = 4.1–10.0), with corresponding median survival of 59.4, 17.8, and 10.5 months, respectively (Fig. 2A). Separate survival analysis indicated that survival was dependent upon both clinical and pathological tumor stages as shown in Fig. 2B and C.
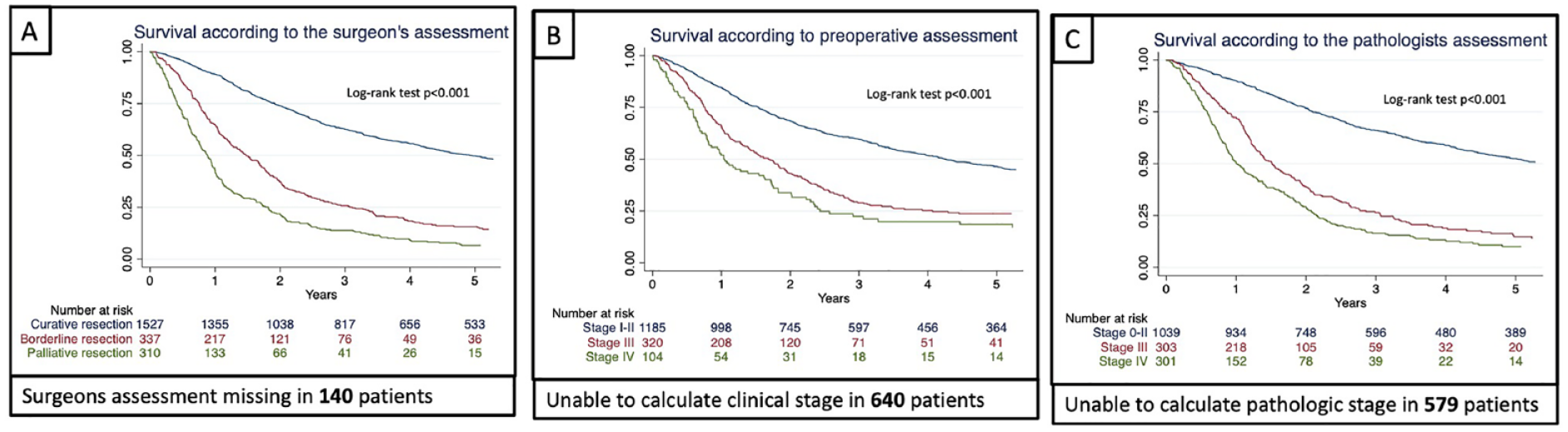
(A) Kaplan–Meier curves in 2341 patients who underwent gastric cancer resection displaying survival according to the operating surgeon’s assessment of the curative potential. Missing information as indicated below each graph. (B) Kaplan–Meier curves in 2341 patients who underwent gastric cancer resection displaying survival according to clinical staging. Missing information as indicated below each graph. (C) Kaplan–Meier curves in 2341 patients who underwent gastric cancer resection displaying survival according to pathologic staging. Missing information as indicated below each graph.
Clinical- and pathological-stage-specific analysis of survival according to the surgeon’s assessment as well as survival analysis stratified on whether the patients had been presented at an MDT all displayed a prognostic quality of the surgeon’s intraoperative assessment of the curative potential of the operation (Supplemental Figs 1 to 3).
The multivariable Cox regression model with adjustments for covariates known at the time of surgery, including clinical TNM stage, showed that the operating surgeon’s assessment of the curative potential of the resection was independently associated with survival; borderline curative had a two-fold risk of death ( = 2.05, 95% CI = 1.71–2.45), and palliative resections a three-fold risk of death (HR = 2.98, 95% CI = 2.46–3.60) as compared to curative resection. Emergency surgery and increased intraoperative blood loss were also independently associated with decreased survival (Table 3). Two variables independently associated with improved survival were MDT discussion (HR = 0.79, 95% CI = 0.68–0.91) and neoadjuvant chemotherapy (HR = 0.80, 95% CI = 0.68–0.96).
Hazard ratios (HRs) with 95% confidence intervals (CIs) from Cox proportional hazards models adjusted for covariates available at (a) the time of surgery and (b) at the time of pathological review of the specimen.
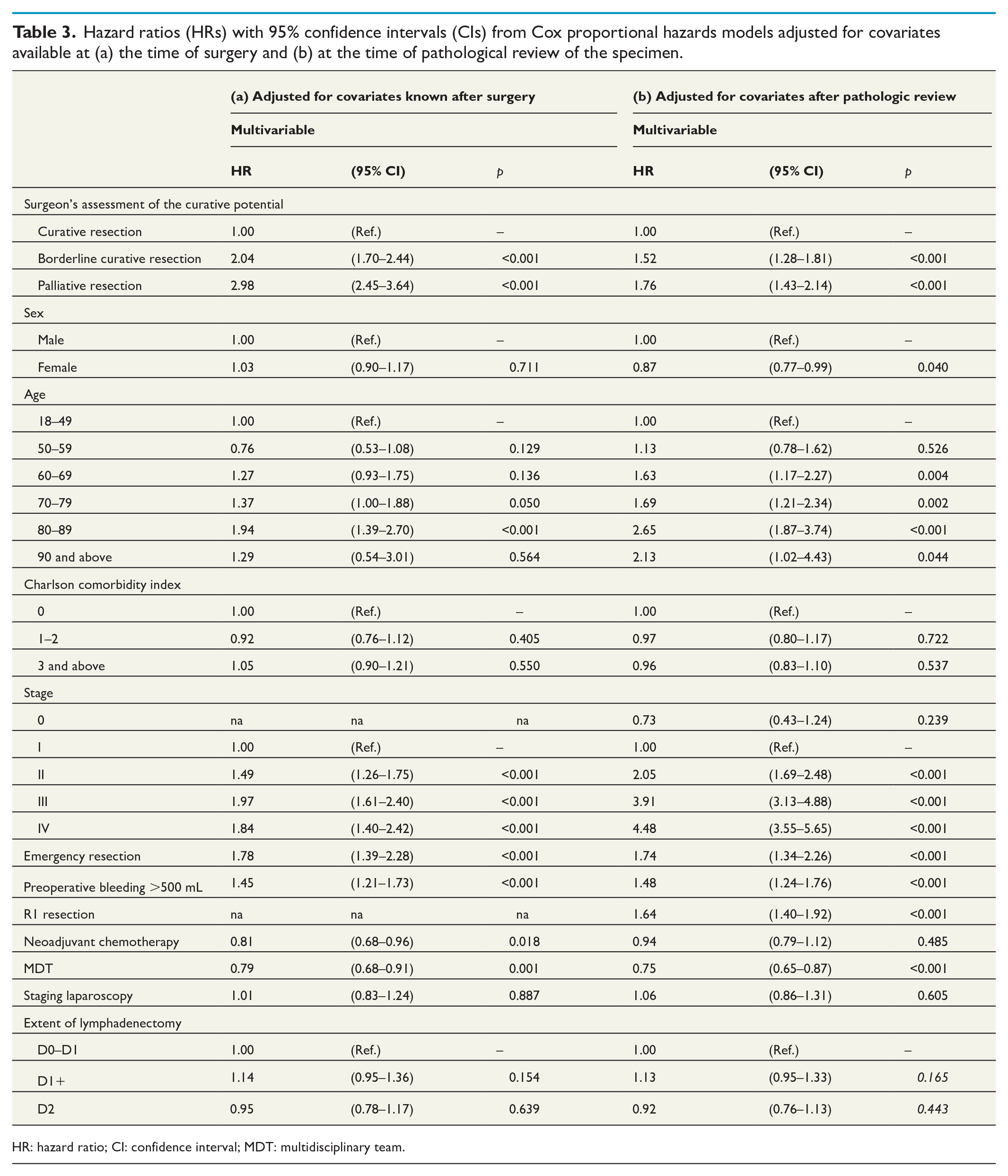
HR: hazard ratio; CI: confidence interval; MDT: multidisciplinary team.
The second Cox regression model adjusting for pathologic TNM, instead of clinical TNM, and microscopic examination of surgical margins as well as for the extent of lymphadenectomy produced similar results although microscopic non-radical resection was associated with decreased survival (HR = 1.65, 95% CI = 1.41–1.94) and adjustments for pathologic stage somewhat weakened the operating surgeon’s assessment of the curative potential as an independent predictor for survival (Table 3). The extent of lymphadenectomy was not associated with survival.
Accuracy and predictive value of the surgeon’s assessment
Analyzing cases where the curative potential of the operation was assessed either curative or palliative, the sensitivity of the surgeon’s assessment for predicting long-term survival (5 years) was 96.7% (95.3–97.8 (95% CI)) compared to 93.2% (91.1–95.0) for pathologic staging (Stages 0–II = curative or Stage IV = palliative). The specificity was low for the surgeon’s prediction 29.1% (26.3–32.0) in similarity with pathologic staging at 35.7% (32.2–39.3). The PPVs were 50.7% (48.8–53.2) and 53.2% (50.3–56.2), whereas the NPVs were 92.1% (88.6–94.9) and 87.0% (82.7–90.5), respectively. Overall accuracy of the surgeon’s intraoperative assessment was calculated at 58.2% (55.9–60.4) and for pathologic staging at 61.0% (58.4–63.5) in long-term survival prediction. In all aspects, the sensitivity, specificity, PPV, NPV, and accuracy were lower when prognosticated by clinical stage (Table 4).
Sensitivity and specificity analysis of three different prognostic modalities for the outcome long-term survival (>5 years) in 2341 patients undergoing gastric resection.
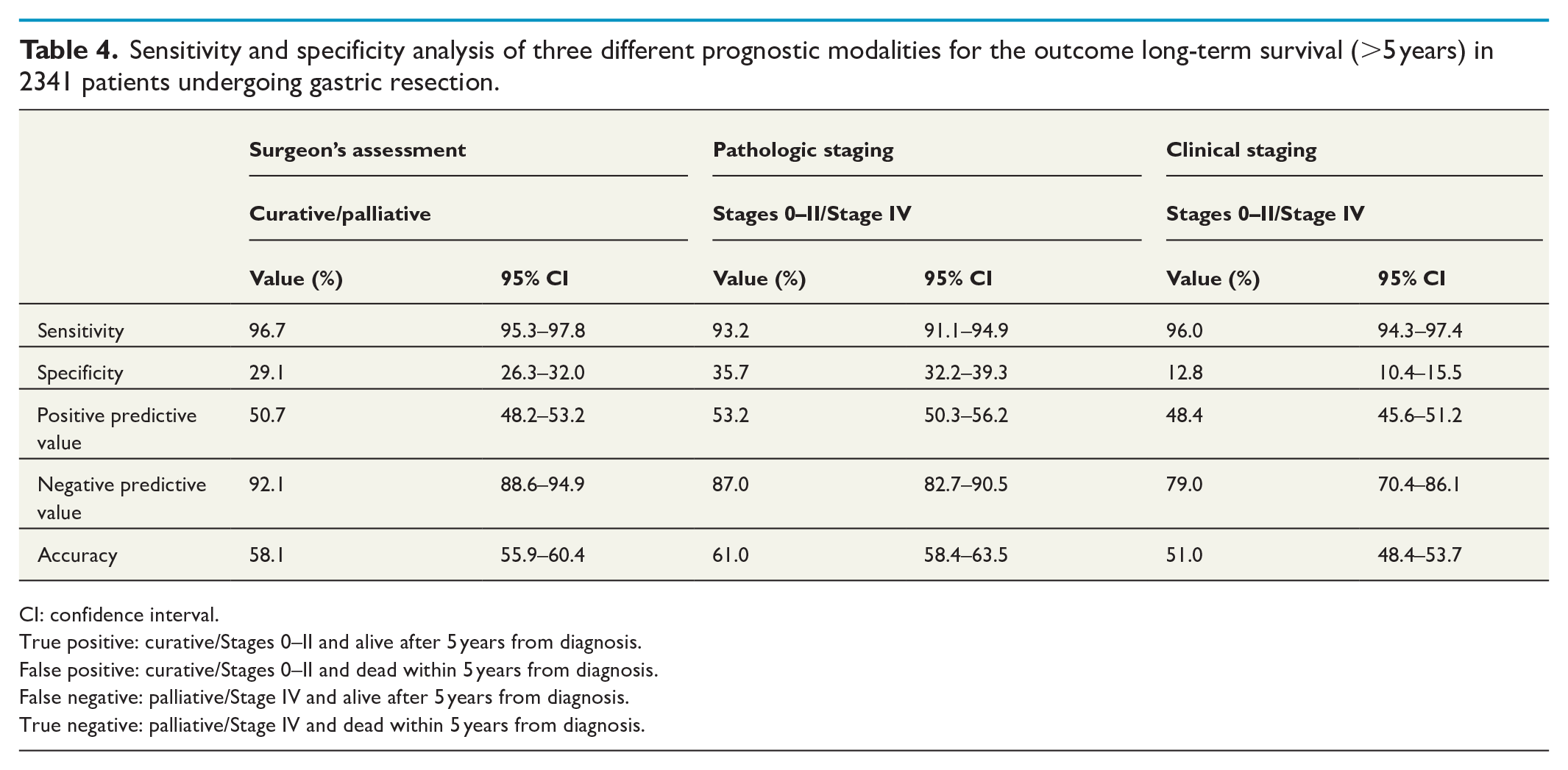
CI: confidence interval.
True positive: curative/Stages 0–II and alive after 5 years from diagnosis.
False positive: curative/Stages 0–II and dead within 5 years from diagnosis.
False negative: palliative/Stage IV and alive after 5 years from diagnosis.
True negative: palliative/Stage IV and dead within 5 years from diagnosis.
Discussion
This study has shown, for the first time, that the surgeon’s intraoperative assessment of the curative potential of surgery is predictive of long-term survival. Surgeons’ typically base the assessment of curative intent on a weighted combination of several key factors, such as age, comorbidity, performance status, tumor stage/localization, preoperative MDT discussion, response to neoadjuvant chemotherapy, degree of radicality, intraoperative complications, technical difficulties, and bleeding. Most of these factors have independently been shown to affect survival, but the literature on a composite evaluation such as the present study is sparse. The surgeon’s composite evaluation, or intraoperative assessment, may also influence the extent of lymphadenectomy as well as the radicality on resection margins. The mechanisms behind this are proposed to relate to the chances of cure, carefully balanced with steering the patient through their postoperative journey. In a patient where it is evident intraoperatively that the risk of harm (i.e. complication) outweighs the likelihood of cure (e.g. in the context of lymph node spread or an advanced tumor), the surgeon may prioritize a palliative approach to resection. This may be one of the key aspects of the surgeon reporting his or her assessment to the registry immediately after the operation. The surgeon knows what ambition has gone into the operation and why, combining all intraoperative findings, and ultimately makes a prognostic assessment on the curative potential of the operation. Information of this sort is very rarely available to the pathologist that might explain why the operating surgeon’s assessment is as accurate as the present study finds. In gastric cancer surgery, the Japanese originally standardized D1, D1+, and D2 lymphadenectomy, 5 which are now internationally recognized standards with aim to counteract the surgeon-to-surgeon variability in gastric cancer resections, and hence improve overall survival. Nevertheless, intraoperative decision-making by the individual surgeon will still have impact on the probability to perform a curative resection.
Protic et al. 18 assessed patients undergoing surgery for colorectal liver metastasis and intraoperative assessment of resection margins by an experienced hepatobiliary surgeon improved the prognostic impact on recurrence rate and disease-free survival compared to standard postoperative histopathological assessment by a pathologist. In the present study, patients with pathological Stages 0–2 had better overall survival than patients where the operating surgeon had deemed the operation curative. Conversely, in patients where the operating surgeon assessed the resection as palliative, overall survival was even lower than in patients with pathological Stage 4. This could indicate a better discrimination by the surgeon in determining a poor prognosis, strengthened by the high NPV for prognostication of long-term survival. For borderline curative potential and pathological Stage 3, the predictive value was similar as indicated by the overlapping Kaplan–Meier survival curves and corresponding estimates for 5-year survival (Fig. 2).
The predictive value of the operating surgeon’s assessment could be useful in the clinical setting as it seems to be the best estimate of early postoperative prognosis and as such could have practical consequences on the subsequent care of the patient. That the predictive value of the operating surgeon is on par with pathologic staging (and better than clinical staging) suggests that it should be held in the highest regard until confirmed by the pathology report of the resected specimen. The survival analysis stratified on disease stage found interesting variations in survival when predicted by the surgeon’s assessment of curative potential. For example, analyzing patients with clinical or pathological stages 1–2 (Supplemental Figs 2 and 3), the 5-year survival was more than double in patients where the surgeon deemed the operation curative compared to borderline curative. This highlights the importance of intraoperative findings. It should be noted that the surgeon’s assessment of curative potential was reported by many different Swedish surgeons over the study period and there is no record of separate surgeon volume or experience. What is deemed borderline curative for one surgeon may not be reported as such by another surgeon indicating that inter-individual or inter-center variability could exist. This needs further evaluation.
In the present study, 4.7% of resected patients were clinically staged with limited metastatic, or oligometastatic disease. Even meticulous clinical staging will sometimes under- or over-stage patients, illustrating the difficulties with diagnostic accuracy in the clinical staging of gastric cancer.19–21 Improving clinical staging by performing laparoscopy with peritoneal washings for cytology in advanced tumors, is a useful adjunct. That said, some patients are still under-staged with this modality and the true extent of disease only becomes evident at the time of resection. In this study, patients with advanced clinical tumor stage underwent borderline curative or palliative resection more frequently. This is likely due to the fact that this patient group have the highest risks of metastatic disease and tumor progression in the interval from clinical staging to the operation. 22 Staging laparoscopy was associated with a reduced likelihood of the operation being deemed palliative. This could be interpreted in favor of staging laparoscopy as an important tool to rule out peritoneal disease. However, there were no associations between staging laparoscopy and survival indicating that staging laparoscopy is mainly a tool to refine patient selection.
Emergency surgery was associated with an increased risk of borderline curative or palliative resection. In patients undergoing gastric resection as an emergency procedure, many will have presented with a perforated or bleeding gastric cancer or suffer from gastric outlet obstruction syndrome (GOOS). 23 Even though only a small number of gastric cancers perforate, 24 the severity of such a presentation will greatly reduce the chances of survival due to peritoneal spread 25 and ultimately influence the surgeon’s opinion of the curative potential. In patients hospitalized due to severe GOOS, with limited but non-curable disease, a palliative resection may be favorable to a stent or gastrojejunostomy. 26 Distal gastrectomy is a recognized method of symptom control and has even translated to short term survival benefits 26 when performed to treat severe GOOS in young, fit patients with limited metastatic disease, where metastatic spread is an intraoperative finding. 27 Previous evidence from the REGATTA trial showed that gastrectomy and chemotherapy cannot be justified for patients with known metastatic disease, over palliative chemotherapy alone. 28
Several previous studies29–31 have attributed reduced overall survival to major intraoperative hemorrhage in upper gastrointestinal cancer surgery. The present study identified intraoperative bleeding as both a factor increasing the likelihood of borderline curative or palliative resection and also as an independent negative predictor of survival in keeping with previous studies. Impaired visualization and decreased distinction of dissection planes due to excessive bleeding may play an important role in reducing the likelihood of a curative resection.
This study identified two important protective factors—neoadjuvant chemotherapy and preoperative discussion at an MDT. Patients receiving neoadjuvant therapy had a reduced risk of undergoing a borderline curative or palliative resection. The downstaging effect of chemotherapy may explain this finding and it may also be due to patient selection. Patient treatment recommendation is often decided in an MDT, the benefits of which have previously been reported. 32 This includes improved survival for patients who are planned for palliative treatment. 33
Limitations of the study are the retrospective nature making firm causal conclusions difficult to draw. Factors that appear to influence the surgeon’s assessment of the curative potential are not necessarily risk factors but may be proxy associations. In addition, the proportion of missing data with respect to the clinical and pathologic stage may affect the accuracy of stage allocation for a proportion of patients. Another potential weakness is there are no data on individual surgeon volume, making it difficult to know the experience of the lead operator. This may influence the surgeon’s assessment of the curative potential of the operation.
The nationwide coverage of gastric cancer surgery in Sweden, when compared to patients in the Swedish Cancer Registry, for the studied years (96%), and the prospective manner in which data are reported to the NREV are strengths of the present study. The NREV has also previously undergone a validation study, published in 2016, and these data have been found to be of high quality. 12 The utilization of National population registers secured complete follow-up.
In conclusion, the current population-based study investigating all patients undergoing gastric cancer surgery in Sweden between 2006 and 2018 has assessed the prognostic value of the surgeon’s intraoperative assessment of curative potential. Predicted long-term survival was independent of, and more precise than, predictions based upon clinical tumor stage. This was also comparable to predicted survival according to pathological stage. The sensitivity of the surgeon’s intraoperative assessment to prognosticate long-term survival was near perfect but with low specificity, mainly indicating a high NPV. Survival was reduced in patients where the operating surgeon deemed the resection borderline curative or palliative. Key factors, such as advanced stage, emergency surgery, and large-volume intraoperative blood loss were identified to increase this risk, while neoadjuvant treatment and MDT decreased the risk of borderline curative or palliative resection.
Supplemental Material
sj-docx-1-sjs-10.1177_14574969231216594 – Supplemental material for Intraoperative assessment of the curative potential to predict survival after gastric cancer resection: A national cohort study
Supplemental material, sj-docx-1-sjs-10.1177_14574969231216594 for Intraoperative assessment of the curative potential to predict survival after gastric cancer resection: A national cohort study by Gustav Linder, Richard J. McGregor and Mats Lindblad in Scandinavian Journal of Surgery
Supplemental Material
sj-jpg-2-sjs-10.1177_14574969231216594 – Supplemental material for Intraoperative assessment of the curative potential to predict survival after gastric cancer resection: A national cohort study
Supplemental material, sj-jpg-2-sjs-10.1177_14574969231216594 for Intraoperative assessment of the curative potential to predict survival after gastric cancer resection: A national cohort study by Gustav Linder, Richard J. McGregor and Mats Lindblad in Scandinavian Journal of Surgery
Supplemental Material
sj-jpg-3-sjs-10.1177_14574969231216594 – Supplemental material for Intraoperative assessment of the curative potential to predict survival after gastric cancer resection: A national cohort study
Supplemental material, sj-jpg-3-sjs-10.1177_14574969231216594 for Intraoperative assessment of the curative potential to predict survival after gastric cancer resection: A national cohort study by Gustav Linder, Richard J. McGregor and Mats Lindblad in Scandinavian Journal of Surgery
Supplemental Material
sj-jpg-4-sjs-10.1177_14574969231216594 – Supplemental material for Intraoperative assessment of the curative potential to predict survival after gastric cancer resection: A national cohort study
Supplemental material, sj-jpg-4-sjs-10.1177_14574969231216594 for Intraoperative assessment of the curative potential to predict survival after gastric cancer resection: A national cohort study by Gustav Linder, Richard J. McGregor and Mats Lindblad in Scandinavian Journal of Surgery
Footnotes
Acknowledgements
This research was not preregistered. The authors declare that any republication of the data (e.g. in secondary analysis) will not constitute redundant publication, will not breach copyright, and will reference the original publication.
Author contributions
All authors made substantial contributions to the conception and design, the acquisition, analysis, and interpretation of data for the work. The work was drafted and critically revised by all authors. All authors approve of the final version to be published. All authors agree to be accountable for all aspects of this work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Author GL received a general research grant from the Ihre Foundation. Funding bodies had no role in study design, data collection, data analysis, manuscript preparation, and publication decisions. All authors had complete access to the study data that support the publication.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
