Abstract
Background and Aims:
Hernia formation is associated with alterations of collagen metabolism. Collagen synthesis and degradation cause a systemic release of products, which are measurable in serum. Recently, we reported changes in type V and IV collagen metabolisms in patients with inguinal and incisional hernia. The aim of this study was to determine if the altered collagen metabolism was persistent after hernia repair.
Material and Methods:
Patients who had undergone repairs for inguinal hernia (n = 11) or for incisional hernia (n = 17) were included in this study. Patients who had undergone elective cholecystectomy served as controls (n = 10). Whole venous blood was collected 35–55 months after operation. Biomarkers for type V collagen synthesis (Pro-C5) and degradation (C5M) and those for type IV collagen synthesis (P4NP) and degradation (C4M2) were measured by a solid-phase competitive assay.
Results:
The turnover of type V collagen (Pro-C5/C5M) was slightly higher postoperatively when compared to preoperatively in the inguinal hernia group (P = 0.034). In addition, the results revealed a postoperatively lower type V collagen turnover level in the inguinal hernia group compared to controls (P = 0.012). In the incisional hernia group, the type V collagen turnover was higher after hernia repair (P = 0.004) and the postoperative turnover level was not different from the control group (P = 0.973).
Conclusion:
Patients with an inguinal hernia demonstrated a systemic and persistent type V collagen turnover alteration. This imbalance of the collagen metabolism may be involved in the development of inguinal hernias.
Keywords
Introduction
Hernia formation is accompanied by deficiencies of the connective tissue (1 –3). Collagens are the main proteins of the extracellular matrix and are the primary components to ensure the tensile strength of connective tissue such as ligaments, tendons, skin, and fascia (4). The maintenance of the collagen structures, and hence the quality of the extracellular matrix, relies upon an ultrafine balance between the synthesis and degradation of the collagen proteins (5). Matrix metalloproteinases (MMPs) are a family of 23 different human endopeptidases that remodel the extracellular matrix components and collagens (6). As a result of the synthesis and degradation processes of collagen, small fragments called neo-epitopes are released into the extracellular space and taken up systemically. In the last decade, specific assays have been developed to detect many of these cleavage products of collagen (5, 7).
Type V collagen is a fibril-forming collagen and is important for collagen fibrillogenesis (8, 9). Type V collagen is involved in the pathogenesis of the classic type of Ehlers–Danlos syndrome. The inherited collagen disorder caused by mutation in type V collagen is characterized by skin hyperextensibility, widened atrophic scars, and joint hypermobility. Importantly, patients with Ehlers–Danlos syndrome have a higher frequency and an earlier onset of abdominal wall hernias (10). Type IV collagen is the main collagen type of the basement membrane (8, 11).
Recently, it has been reported that serum biomarkers of collagen type V and IV metabolism were altered in patients presenting with inguinal or incisional hernia (12). These novel findings indicated major systemic alterations of type V and IV collagen in hernia patients. It remains unknown whether these observations in collagen turnover represent a systemic predisposition to hernia disease or a local phenomenon related to the hernia.
Our hypothesis was that patients with hernias are predisposed to develop hernias due to persistent systemic alteration of collagen turnover. To address this, we wanted to confirm that the altered collagen turnover existing prior to hernia repair was also present years after hernia repair. In this study, type V and IV collagen turnover was measured in the serum of patients followed up for 35–55 months after hernia repair.
Material and Methods
Patients
This study included patients who were previously operated on for abdominal wall hernia. Prior to the operation, venous blood samples were collected (12). A total of 60 patients were included from 2009 to 2011. The patients were enrolled in three predefined groups subjected to elective (1) repair of a primary inguinal hernia, (2) repair of an incisional hernia, and (3) laparoscopic cholecystectomy without the presence or history of hernias (control group). Male patients older than 40 years and postmenopausal females not receiving any hormone replacement therapy were included.
In this study, 56 patients of the original 60-patient cohort were invited by letter to participate (Fig. 1). Venous blood was collected and the patients were clinically examined for hernia in the outpatient clinic. Data on smoking status, alcohol consumption, body mass index (BMI), American Society of Anesthesiologists (ASA) score, the presence of other diseases, and medicine intake were recorded.
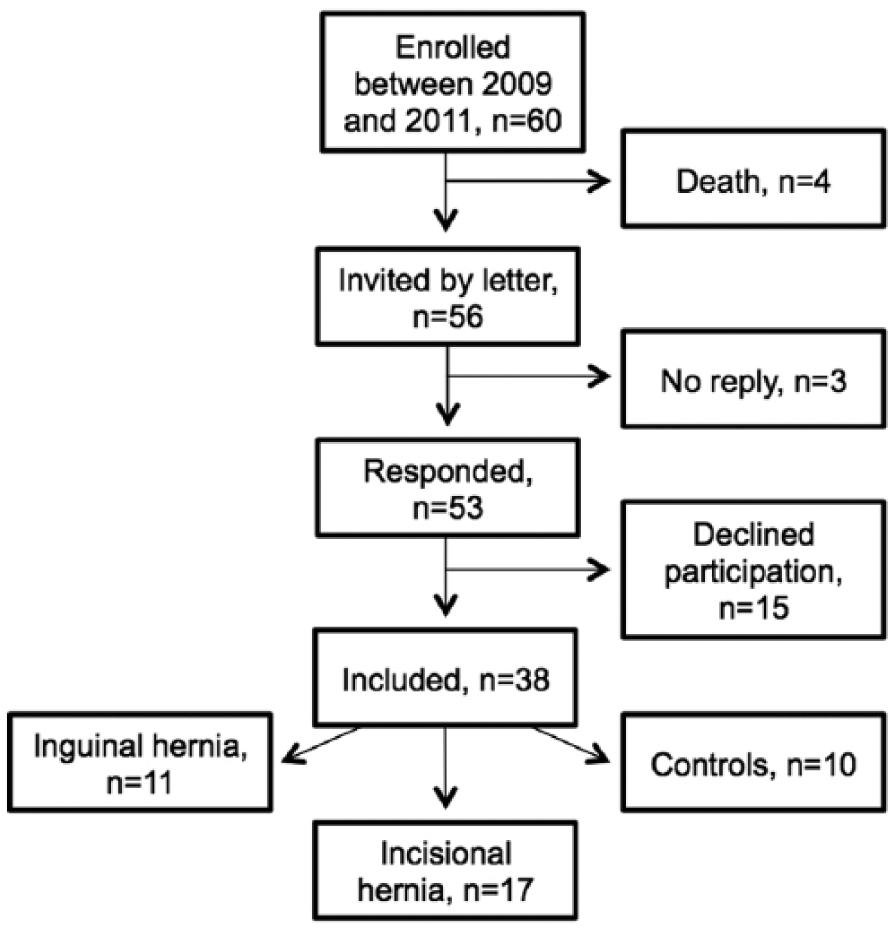
Flowchart of included patients.
The project was approved by the regional Research Ethics Committee (Project-ID H-3-2014-020) and registered by The Danish Data Protection Agency (BBH-2014-016). Written and verbal informed consent was obtained from all participants included in the study.
Biomarker Assays
Blood samples were coagulated for 30 min before centrifugation at 1800 × g at 4 °C for 10 min. The serum fraction was stored at −80 °C until analysis. The preoperative and postoperative serum samples were analyzed by a solid-phase, competitive, enzyme-linked immunosorbent assay (ELISA) to detect neo-epitope fragments from synthesis and degradation of the collagens. The methods were developed by Nordic Bioscience A/S, Herlev, Denmark. A neo-epitope is a specific amino acid sequence generated by enzymatic cleavage. All assays used mouse monoclonal antibodies that react with enzymatically processed collagen. Type V collagen synthesis was measured as the C-terminal pro-peptide of type V procollagen (Pro-C5) and type V collagen degradation as MMP-2- and MMP-9-mediated fragmentation of type V collagen (C5M) (13, 14). Type IV collagen synthesis was measured as the N-terminal peptide of type IV procollagen (P4NP) and type IV collagen degradation as a pepsin/MMP-9-generated fragment of mature type IV collagen (C4M2) (7). The type V collagen turnover was expressed by the Pro-C5/C5M ratio and the type IV collagen turnover by the P4NP/C4M2 ratio.
Technically, 96-well plates were precoated with streptavidin (Roche Diagnostics, Mannheim, Germany) and incubated with the respective biotinylated antigens for 30 min at room temperature. The samples were diluted in an incubation buffer containing 10 mg/mL of bovine serum albumin (A-7906; Sigma-Aldrich, St. Louis, MO, USA). In total, 20 µL of the standard kit controls and serum samples were incubated together with 100 µL of horseradish peroxidase–conjugated monoclonal antibodies for 1 h at 20 °C (Pro-C5, P4NP, and C4M2) or 3 h at 4 °C (C5M). The wells were washed with buffer containing 25 mmol/L Tris-HCl (pH 7.2), 50 mmol/L NaCl, 1.5 mmol/L Bronidox L5, and 0.915 mmol/L Tween-20, using a standardized microplate washer (BioTek Instruments, Winooski, VT, USA). Tetramethylbenzidine was added (100 µL/well), the plate was incubated for 15 min at room temperature, and then 102 mmol/L H2SO4 was added (100 µL/well). All plates were agitated at 300 rpm during incubations. Optical densities were read at 450 and 650 nm (reference) using an ELISA reader (VersaMAX; Molecular Devices, Wokingham, UK). The standard curves were fitted by 4-parametric mathematical models and used for calculations of the concentrations of the unknowns.
Statistical Analyses
Pre- and postoperative biomarker levels of each group were compared using mixed analysis of variance (ANOVA) test. If the ANOVA test was significant for the respective biomarker, the dependent variables were compared by paired t test. One-way ANOVA test was performed to compare the postoperative biomarker levels between the groups. If the ANOVA test was significant, the control group and the specific hernia groups were compared by an unpaired t test. Data were not normally distributed and were log-transformed before the analyses. A P value < 0.05 was defined as statistically significant. SPSS version 22.0 (IBM, Chicago, IL, USA) was used for data analysis.
Results
A total of 38 patients were included (Table 1). These patients had undergone elective operation for inguinal hernia (n = 11), incisional hernia (n = 17), or gallbladder stones (controls, n = 10). The median time from the primary surgery until study inclusion was 42 months (range: 35–55 months).
Patient characteristics (postoperatively).
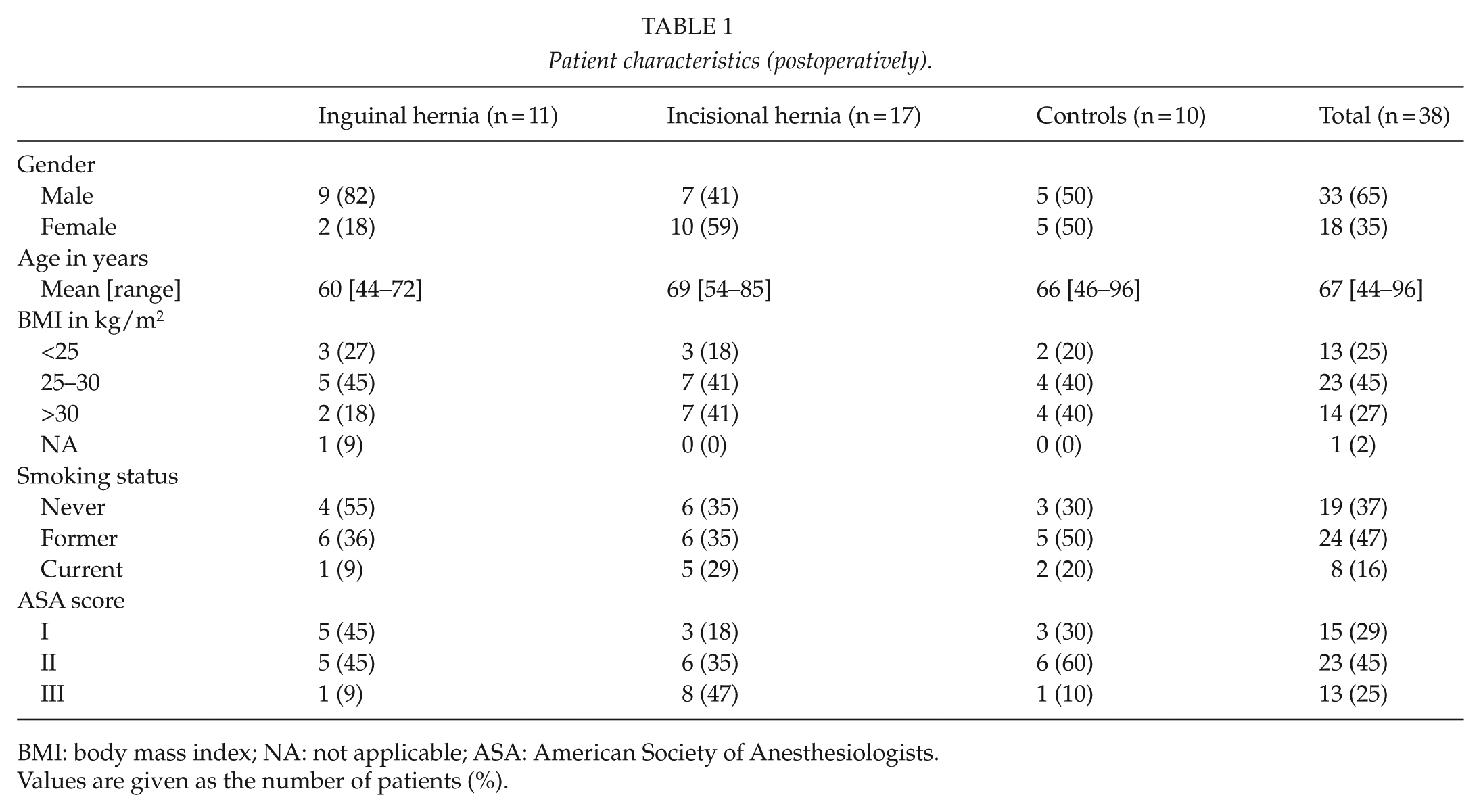
BMI: body mass index; NA: not applicable; ASA: American Society of Anesthesiologists.
Values are given as the number of patients (%).
The turnover of type V collagen (Pro-C5/C5M) was slightly higher postoperatively as compared to preoperatively in the inguinal hernia group (P = 0.034; Fig. 2). Postoperatively, Pro-C5/C5M was significantly lower in the inguinal hernia group compared to the controls (P = 0.012). In the incisional hernia group, the type V collagen turnover was significantly higher after hernia repair as compared to the preoperative level (P = 0.004). Postoperatively, no difference was found between the type V collagen turnover in the incisional hernia group compared to controls (P = 0.973).
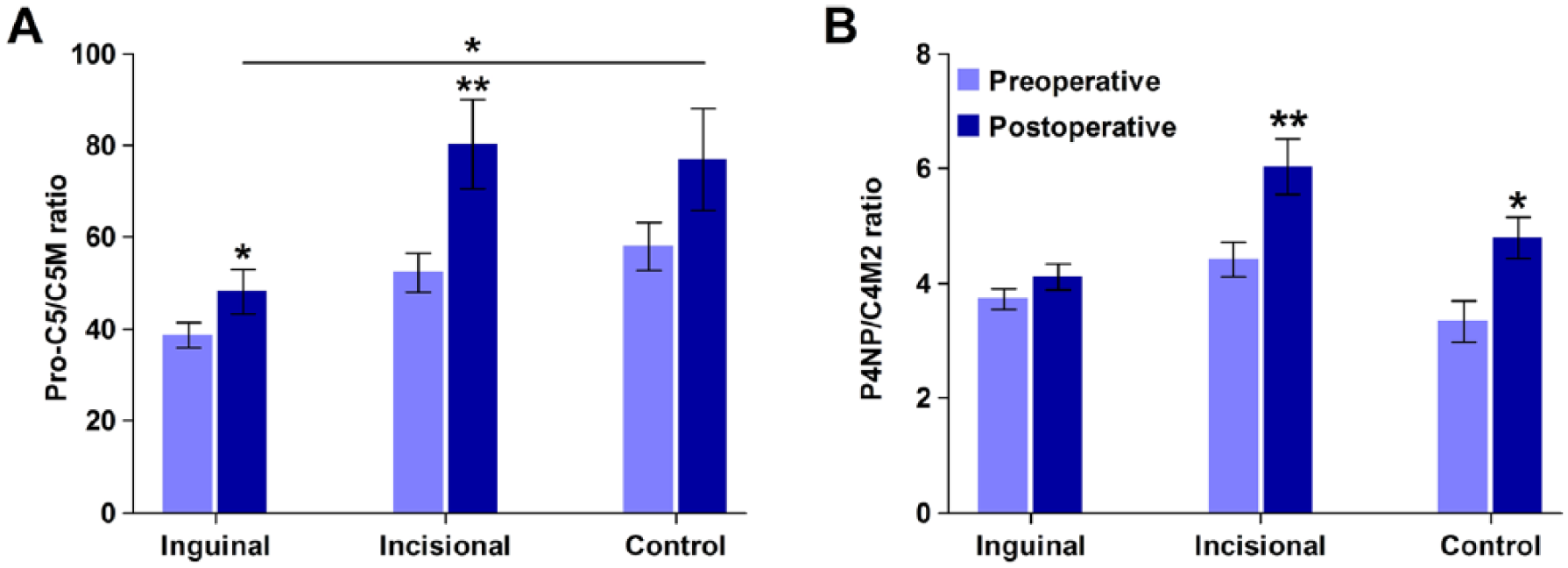
Preoperative (light blue bars) and postoperative (dark blue bars) serum biomarker levels of (A) type V collagen turnover (Pro-C5/C5M) and (B) type IV collagen turnover (P4NP/C4M2) in inguinal hernia patients (n = 11), incisional hernia patients (n = 17), and control patients (n = 10). Mean ± SEM. Postoperative median (all groups included): type V collagen turnover = 59.4 (range: 25.8–160.6); type IV collagen turnover = 4.7 (range: 2.8–9.8). *P < 0.05, **P < 0.005.
In the inguinal hernia group, the type IV collagen turnover (P4NP/C4M2) did not change significantly after hernia repair as compared to the preoperative levels. Conversely, the type IV collagen turnover was higher after hernia repair in the incisional hernia group (P < 0.0005). No difference was found in type IV collagen turnover between the inguinal hernia group and the control group postoperatively nor between the incisional hernia group and the controls (P = 0.153 and P = 0.092, respectively). In the control group, the type IV collagen turnover was higher postoperatively compared to preoperatively (P = 0.036).
Overall, the type V and IV collagen turnover was significantly higher postoperatively compared to preoperatively (P < 0.0005 and P < 0.0005, respectively).
There were no significant differences in biomarker levels regarding gender, smoking, BMI, and ASA score.
Discussion
In our previous study (12), preoperative samples from patients with inguinal and incisional hernias showed lower type V collagen turnover but higher type IV collagen turnover as compared with the controls. In this study, the same patient population was followed up 35–55 months after their primary surgery to examine if the alterations observed preoperatively were persistent after hernia repair. The results here demonstrate a persistently low type V collagen turnover in patients who had undergone inguinal hernia repair compared to controls. Conversely, the level of type V collagen turnover was higher after incisional hernia repair and thereby reached the level of the control group.
Type V collagen plays an essential role in the regulation and initiation of collagen fibril nucleation and thereby fibril formation (15). A recent study demonstrated that patients with Ehlers–Danlos syndrome and mutations in type V collagen genes had altered structure of tendons with large and irregular collagen fibrils (16). These clinical findings are consistent with those in type V collagen–deficient mice that display irregular collagen contour and decreased number of collagen fibrils (9). Even though type V collagen quantitatively is a minor component in most collagen fibrils, a decrease in type V turnover may impair matrix quality. Because the low type V collagen turnover level persisted in the patients previously operated for inguinal hernia, it is reasonable to assume that their abnormality of type V collagen turnover was present prior to hernia formation. The type V collagen alteration may predispose to the development of inguinal hernia.
The type V collagen turnover of the incisional hernia group was higher after hernia repair, thereby approaching the level of the control group. This change reveals that the preoperative type V collagen turnover alteration may be a consequence of a present incisional hernia rather than a general predisposition to hernia disease. In addition, the results indicate that the incisional hernia formation may be caused by factors other than the alteration of the type V collagen metabolism. These patients have previously been operated on facing a risk of incisional hernia formation that is influenced by external factors such as smoking, male gender, high BMI, surgical site infection, and surgical technique (17, 18). In this study, the incisional hernia group had a higher proportion of patients with BMI > 30 kg/m2 than the inguinal hernia group. Adipose tissue of obese people shows increased expression of type V collagen compared to persons with normal BMI, whereas no significant difference was found in type IV collagen expression (19). In the study by Sand et al. (20), no association between BMI and type IV collagen biomarkers was found in patients with chronic obstructive pulmonary disease. The increased risk of incisional hernia in patients with high BMI may be due to a combination of high abdominal pressure and alteration of the extracellular matrix. Further research is required to clarify this.
The significant change of type IV collagen turnover over time in the control group complicates the interpretation of the biomarker levels in the remaining groups. This difference between the pre- and postoperative levels in the control group could be explained by the fact that these patients also had undergone a surgical procedure. The overall turnover level (all groups included) of type V and IV collagen increased significantly over time, indicating a potential influence of surgery on the turnover level. It is also possible that the overall higher turnover level is due to aging. Karsdal et al. (21) found that both collagen type V collagen and type IV collagen turnover levels in rats change during their lifetime, suggesting a change in quality and quantity of collagens over time. In this study, there was no significant correlation between age and turnover level. This can be due to high inter-individual variability and a relatively small sample size.
The number of patients included in this study was a limiting factor. Ranges of both type V and type IV collagen turnover levels were wide. Since this study was conducted as a late follow-up of patients included in a previous study (12), no power calculation was performed prior to data collection, and it was not possible to enroll more patients to increase the power. Another limitation was that the systemic levels of biomarkers may not reflect tissue-specific conditions related to hernia disease. It would be relevant to follow individuals initially without a hernia in a longitudinal study to see if changes in collagen biomarkers relate to the development of hernia. However, an estimated lifetime risk of inguinal hernia repair is 27% for men and 3% for women (22), meaning that such a study will require a large number of patients and a long follow-up period.
Based on the present findings, the type V collagen turnover was persistently diminished in patients operated on for inguinal hernia and may reflect poor matrix quality due to change in formation, size, and structure of the collagen fibrils. The development of an inguinal hernia may be a consequence of an impaired type V collagen metabolism. Conversely, the type V collagen turnover in the incisional hernia group reached the level of the control group after hernia repair, suggesting that the preoperatively high turnover was a consequence of a present hernia. The results reveal that inguinal hernia and incisional hernia are different pathologies with divergent predisposing factors.
In conclusion, abnormality of type V collagen may be an important contributor in the development of inguinal hernia disease, and collagen turnover may serve as a biomarker for poor extracellular matrix quality and possible future inguinal hernia formation. Future studies with a larger number of patients are needed to validate the results derived from this study. Additional longitudinal studies should investigate if the alterations are present before hernia formation.
Footnotes
Acknowledgements
The authors thank the project nurse Tina Lee Brøndum for assistance with patient contact and collection of blood samples.
Declaration of Conflicting Interests
L.L., N.A.H., M.S.Å., and L.N.J. declare no conflicts of interest. P.J., J.H.M., and M.A.K. are employed at Nordic Bioscience A/S.
Ethical Approval
Informed consent was obtained from all participants included in the study. The project was approved by the regional Research Ethics Committee (Project ID: H-3-2014-020) and registered by The Danish Data Protection Agency (BBH-2014-016).
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
