Abstract
Background and Aims:
Severe, medically uncontrollable gastroparesis is a rare entity, which can be treated using a high-frequency gastric electric stimulator implanted surgically. Previous follow-ups have proven positive outcomes with gastric electric stimulator in patients with gastroparesis. The aim of this study was to evaluate the efficacy and safety of gastric electric stimulator in patients, in whom gastroparesis could not be controlled by conservative means in our country.
Materials and Methods:
This is a retrospective multi-center cohort comprising all patients who had been implanted gastric electric stimulator for severe, medically refractory gastroparesis during 2007–2015 in Finland.
Results:
Fourteen patients underwent implantation of gastric electrical stimulator without any postoperative complications. Laparoscopic approach was used in 13 patients (93%). Prior implantation, all patients needed frequent hospitalization for parenteral feeding, 13 had severe nausea, 11 had severe vomiting, 10 had notable weight loss, and 6 had frequent abdominal pain. After operation, none of the patients required parenteral feeding, 11 patients (79%) gained median of 5.1 kg in weight (P < 0.01), and symptoms were relieved markedly in 8 and partially in 3 patients (79%). Of partial responders, two continued to experience occasional vomiting and one mild nausea. Five patients needed medication for gastroparesis after the operation. One patient did not get any relief of symptoms, but gained 6 kg in weight. No major late complications occurred.
Conclusion:
Gastric electrical stimulator seems to improve the nutritional status and give clear relief of the symptoms of severe, medically uncontrollable gastroparesis. Given the low number of operations, gastric electrical stimulator seems to be underused in Finland.
Keywords
Introduction
Gastroparesis is a disorder of gastric emptying without underlying mechanical obstruction (1). Main symptoms include early satiety, bloating, upper abdominal pain, nausea, and vomiting leading secondarily to esophagitis and weight loss. Three etiology categories exist: diabetic, idiopathic, and postoperative. While the idiopathic form is the most common, the diabetic form leads to most severe symptoms (2, 3).
The diagnosis of gastroparesis is based on typical symptoms when mechanical obstruction is ruled out. Gastric emptying can be measured by scintigraphy, 13C-octanoic acid breath test, or dual-tracer scintigraphy (4–6). In mechanical obstruction, usually emptying of both solids and liquids is slow, but in gastroparesis emptying of liquids may be normal.
Conservative therapy of gastroparesis consists of dietary and medical treatments, which often are sufficient. Generally, small frequent meals with high protein with low fiber and fat content are recommended (7, 8). Metoclopramide, erythromycin, or domperidone can be used as prokinetic agents . Despite symptom relief, there is no clear association with enhancement of gastric emptying (9). The 5-HT4-agonist prucalopride has been proven to accelerate gastric emptying in healthy subjects and may be considered for treatment of gastroparesis (10). Furthermore, prucalopride is effective in treating obstipation, which may also relieve symptoms of gastroparesis (11–13). Pyloric botulinum toxin injections have been useful in open label studies, but not in a randomized, controlled study (14, 15). Even transpyloric stenting and total gastrectomy have been suggested as a treatment option for gastroparesis (16, 17).
Although most patients with gastroparesis have sufficient relieve by conservative means, in a small subset of patients it is uncontrollable by medical treatment. For these patients, a gastric electrical stimulator (GES) (Enterra®, Medtronic, Minneapolis, MN, USA) has become available commercially (18).
The aim of this study was to assess the efficacy and safety of implantation of GES in patients with medically uncontrollable gastroparesis. We included all patients who had undergone GES implantation in Finland between 2007 and 2015.
Materials and Methods
Patient Population and Data Selection
The hospitals, which had implanted GES in Finland, were obtained by personal communication with the manufacturer’s Finnish representative (Enterra, Medtronic). Four hospitals (Helsinki and Oulu University Hospitals; Lapland and Seinäjoki Central Hospitals) were identified and contacted, and patients who had undergone GES implantation were included in this retrospective cohort study. The patients were identified from prospective registers (Helsinki 10 patients, Lapland 2 patients, Oulu 1 patient, and Seinäjoki 1 patient).
The diagnosis of gastroparesis had been verified by dual-tracer scintigraphy in 13 patients performed directly after gastroscopy. The diagnosis required a pathologically long gastric emptying time for solids (solid intake half-time >135 min) (6), but liquid gastric emptying could be normal. No control scintigraphy studies were performed considering the x-ray burden to individual patients. Patients with no or mild symptoms were considered as marked responders, patients with moderate symptoms as partial responders, and those with severe symptoms were considered failures.
Surgical Technique
Laparoscopic operation was performed in a standard fundoplication position and setup. 5 cm incision for the Enterra device was made to the upper left quadrant of the abdomen, and a 10 mm trocar was inserted in the upper right quadrant. Two electrodes were applied to the anterior wall of the stomach 9 and 10 cm orally from the pylorus respectively. Correct position of the electrodes in the muscle layer of the stomach was confirmed by intraoperative gastroscopy. The electrodes were attached to the serosal surface of the stomach using silicon bands fixed with non-absorbable sutures and clips. The electrodes were taken out of the abdominal cavity and a subcutaneous cavity was created for the Enterra device. The impedance of the device was measured to ascertain its proper function before it was buried into the subcutaneous cavity.
Patient Follow-Up
The patients were followed at the gastroenterology outpatient clinic. The last follow-up date was defined as the last time physician either met or called the patient. The study was approved by the institutional review boards of participating centers.
Statistical Analysis
Change of weight was analyzed with the Wilcoxon matched-pairs signed rank sum test. The statistical analysis was performed by SPSS software.
Results
Fourteen patients (seven males, mean age: 40 years (range 24–54)) were included in the study (Table 1). The etiology of gastroparesis was diabetes (n = 11), idiopathic (n = 1), or postoperative (n = 2). Type 1 diabetes (n = 9) was the most common, whereas one patient had type 2 diabetes, and one patient had diabetes due to chronic pancreatitis. One patient had undergone fundoplication two times with possible vagal nerve injury and in one patient symptomatic gastroparesis was diagnosed after several abdominal operations.
Patient demographics.
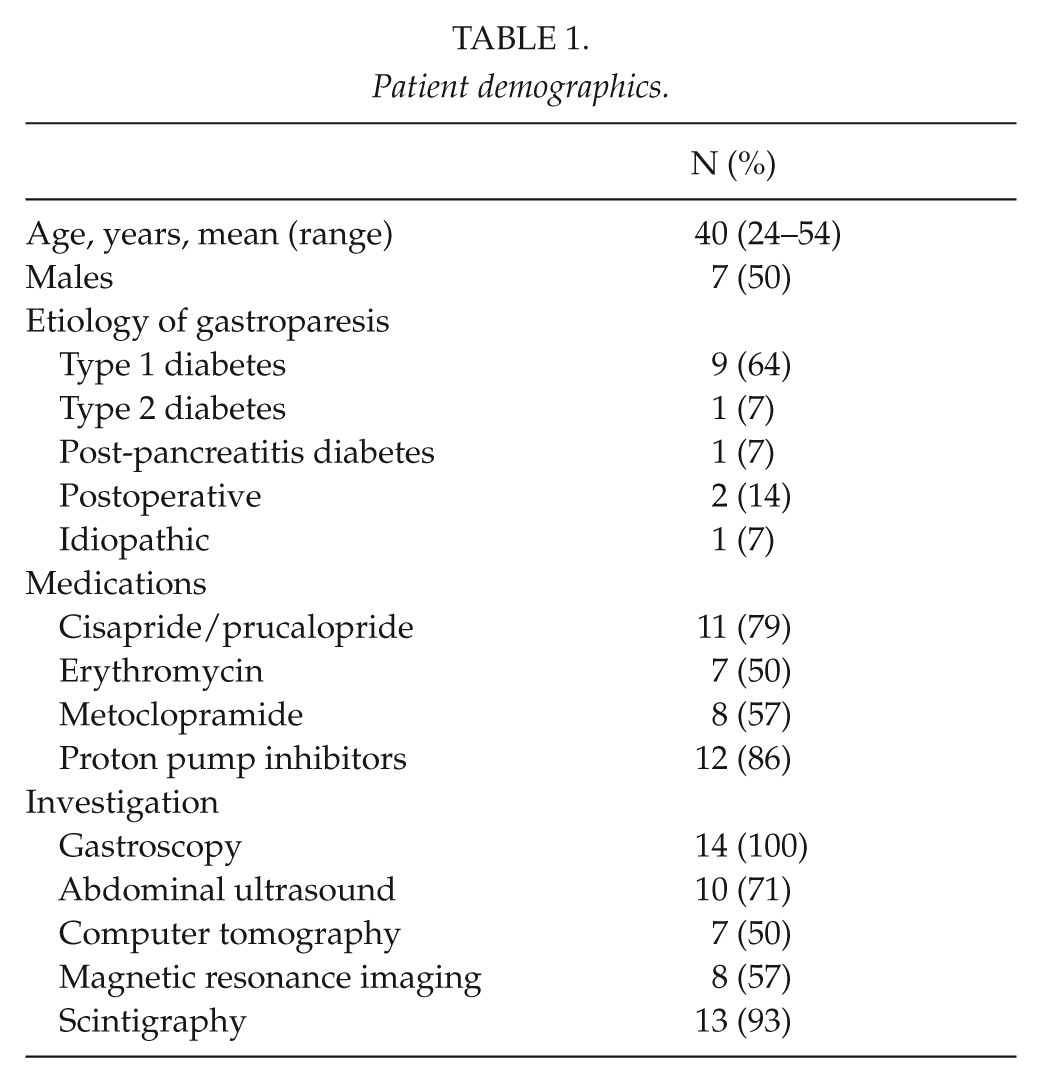
Every patient had undergone careful diagnostic work-up and medical treatment before implantation of GES. All patients had undergone gastroscopy. Scintigraphy had been performed in 13 patients (93%) and all these patients had delayed solid gastric emptying. Furthermore, all patients had undergone some form of abdominal imaging, ultrasound was performed in 10 patients (71%), computed tomography in 7 patients (50%), and magnetic resonance imaging in 8 patients (57%). Thirteen patients (93%) had undergone imaging with several modalities.
Medical therapy was tried in all patients. Twelve patients (86%) used proton pump inhibitors, eleven patients (79%) used cisapride or prucalopride, eight patients (57%) used erythromycin, and seven patients (50%) used metoclopramide. Twelve patients (86%) used several types of medications.
Nausea and vomiting were the leading symptoms, which had led to hospital admissions and periods of parenteral nutrition in 13 patients. One patient had abdominal pain as the main symptom. The number of hospital admissions varied from 2 to 13 times prior implantation of gastric stimulator. Weight of the patients varied across the preoperative period and lowest weight during conservative treatment is shown in Fig. 1. Eleven patients gained median of 10 (interquartile range (IQR) 5.3–14.3) kg in weight during intravenous nutrition treatment prior to operation, calculated after the symptoms of severe gastroparesis and the conservative treatment had started (missing data n = 3). Six (43%) patients had also abdominal pain. Median serum albumin was 38.7 (IQR 33.9–42.7) g/L preoperatively. Five patients had albumin less than 35 g/L preoperatively.
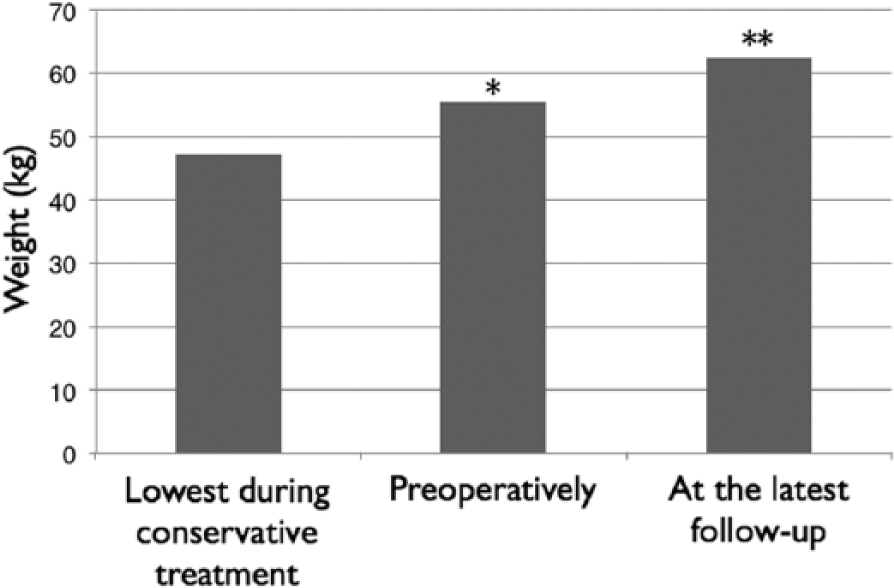
Mean weight (kg) of patients who attended all three weight measurements (n = 11).
Outcome
Thirteen GES were implanted using laparoscopic approach. One implantation was done through laparotomy due to several prior abdominal operations. No postoperative complications occurred. Discomfort at the battery pocket site was reported by one patient at 1-month follow-up. Median postoperative length of hospital stay was 4 (IQR 2–5) days, which was mainly for testing and adjusting the device. The patients were followed for median 36 (IQR 10–63) months. Two patients were lost to follow-up.
Interestingly, symptoms slowly recurred during control visits in four patients. It was found out that their GES battery was exhausted. These patients underwent battery-change reoperation. For one patient, this was done twice. Battery-change restored the functionality of the stimulator and relieved the symptoms in all four patients. No surgical complications were related to reoperations.
Symptoms of gastroparesis were relieved markedly in eight and partially in three patients (Table 2). Two partial responders continued to vomit occasionally and one had mild nausea after GES implantation. The patient who underwent open surgery continued to have abdominal pain, but vomiting decreased markedly and the patient gained 6 kg in weight after implantation. However, abdominal pain continued lead to duodenojejunostomy 6 months after GES implantation.
Symptoms and weight gain preoperatively and at the last follow-up visit.
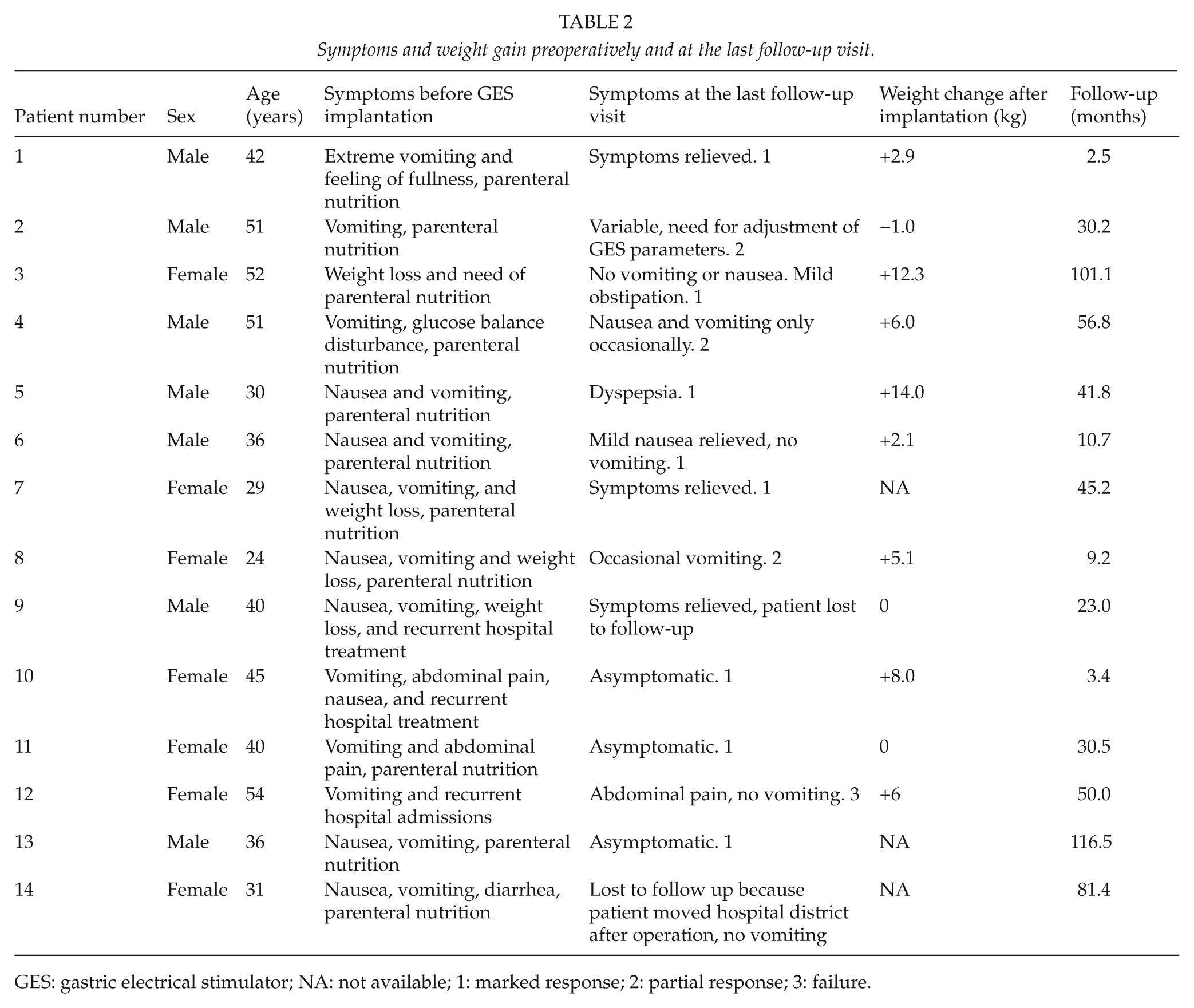
GES: gastric electrical stimulator; NA: not available; 1: marked response; 2: partial response; 3: failure.
Eleven patients (79%) gained median 5.1 (IQR 0–8.0) kg in weight (missing data n = 3), after GES implantation (Fig. 1). Five out of ten patients (50%) were without medication for gastroparesis at the latest follow-up (missing postoperative medication data n = 4).
Among patients with diabetic gastroparesis (n = 11), median HbA1c level 6 months after the implantation of GES was 67 (IQR 53–86) mmol/mol compared to the preoperative median value of 66 (54–78) mmol/mol (missing data n = 5). Thus, no change in HbA1c levels was detected postoperatively. However, hypoglycemic episodes requiring hospital treatment were very few after the GES implantation. One patient with diabetic gastroparesis continued to have hypoglycemic episodes needing hospital treatment and one other patient had one such episodes, while others did not (missing data n = 3).
Discussion
As only 14 patients have received GES in our study period, it seems that this treatment modality is underused in Finland. All patients were dependent on frequent parenteral nutrition indicating that GES is considered as the very last resort in the treatment of gastroparesis. The results of GES for treatment of medically uncontrollable gastroparesis are encouraging. None of the patients required parenteral nutrition after implantation, 80% gained weight, 70% experienced their symptoms relieved, and half of the patients could wean off medication. Furthermore, the implantation was primarily carried out laparoscopically with no complications indicating safe procedure with minimal length of hospital stay required.
Very interesting evidence for the benefit of GES on the symptoms is provided by the four patients whose GES battery was exhausted during the follow-up period. In these patients, symptoms recurred, but could again be relieved by battery-change. It thus seems that GES has long standing effect. A similar worsening of symptoms was also seen in a study of 33 patients with diabetic and idiopathic gastroparesis with GES implanted when the device was turned off (19). In two other randomized, double-blinded controlled studies, GES also significantly reduced vomiting during the 12 months open follow-up but there was no significant difference between the on and off periods during double-blind phase of the study (20, 21). In diabetic gastroparesis, GES implantation had no significant effect on HbA1c values measured after six 12 months of therapy (20).
Other centers have published similar good results of gastric electrical stimulation. McCallum and coworkers followed 182 patients for 10 years. Patients were similar compared to our study. Symptoms were significantly improved, but there was no statistical difference in gastric emptying before and after gastric electrical stimulation (22). In our study, the postoperative gastric emptying was not evaluated in all patients, but in those patients it was assessed there were no difference. The mechanisms of action of GES are not well understood. It does not seem to accelerate gastric emptying, but might act by affecting afferent vagal neural activity to control central nervous system mechanisms of nausea and vomiting (23).
The cost of the device is currently high and for this reason, the patients are highly selected. On individual basis, it is difficult to predict in which patients GES is effective. Results seem to be better in diabetic gastroparesis compared to idiopathic or postsurgical gastroparesis (24). In recent paper, Heckert and coworkers evaluated 151 patients with refractory gastroparesis who underwent GES. Improvement with symptoms was detected in 75% of patients with 43 being at least moderately improved and diabetics responded better than nondiabetic patients (24).
There are limitations in this study. This was a retrospective series with small number of patients. The follow-up time was relatively short (median 3 years). In consequence, it remains unknown how long GES will relieve the symptoms and whether the effect will wear off at some point.
In conclusion, GES seems to be effective treatment modality for severe gastroparesis in selected patients. Laparoscopy is an efficient and a safe method for GES implantation. GES implantation is scarce in Finland.
Footnotes
Acknowledgements
The authors thank Pertti Finnilä for information regarding the hospitals that have implanted Enterra® (Medtronic, Minneapolis, MN, USA) GES in Finland.
Declaration of Conflicting Interests
Jari Punkkinen has received consultant fees from Almirall, Shire, and MSD. Jari Koskenpato has received consultant fees from Abbvie, Olympus, Allergan, and MSD. Ville Sallinen has received grants from Mary and Georg Ehrnrooth’s Foundation, Vatsatautien Tutkimussäätiö Foundation, Martti I. Turunen Foundation, Helsinki University Hospital research funding, lecturing fee from Novartis, and educational travel expenses from Astellas. Other authors report no conflicts of interest. The authors alone are responsible for the content and writing of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
