Abstract
Study Design:
A retrospective cohort study of consecutively operated neuromuscular scoliosis patients.
Background and Aim:
Surgical correction of neuromuscular scoliosis can be complicated by early gastrointestinal complications, but data on the extent and severity of them is scarce. The aim of the study was to determine the incidence, course, and risk factors of gastrointestinal complications after neuromuscular scoliosis correction.
Material and Methods:
Ninety-one patients (<21 years of age) were consecutively operated on for neuropathic neuromuscular scoliosis during 2000–2011. Patients who developed marked postoperative gastrointestinal complications were identified and clinical, operative, and radiographic records, death certificates, and post-mortem reports were examined.
Results:
The average age at surgery was 14.5 (SD 2.9) and follow-up time was 4.9 (SD 2.3) years. Gastrointestinal complications occurred in 12 (13%) patients and included prolonged paralytic ileus (7%, 6/91), dysphagia (7%, 6/91), and gastroparesis (1%, 1/91). Hospital stay was 22 (SD 11) days in patients with gastrointestinal complications and 16 (SD 20) days in non-complicated patients (p = 0.005). Dysphagia required permanent feeding gastrostomy in one patient whereas other complications were transient and none caused death. The risk factors for postoperative gastrointestinal complications were preoperative main curve correction <30% in traction/bending radiographs (Relative Risk (RR) = 28 (95% Confidence Interval (CI) 4.4–180); p < 0.001), preoperative main curve >90° (RR = 5.5 (95% CI 1.3–23); p = 0.020), disturbance in intraoperative spinal cord monitoring (RR = 6.0 (95% CI 1.1–34); p = 0.043), and intravenous opioid medication over 5 days postoperatively (RR = 7.9 (95% CI 1.8–35), p = 0.006).
Conclusion:
Gastrointestinal complications occurred in 13% of patients after neuromuscular scoliosis correction. Marked gastrointestinal complications extended postoperative hospitalization period, but they were transient in majority (92%) of cases and none caused death. Rigid scoliosis was the most significant risk factor for gastrointestinal complications. Gastrointestinal complications appear to be less frequent after posterior only spinal fusion with total pedicle screw instrumentation and Ponte osteotomies.
Keywords
Introduction
Gastrointestinal disorders (e.g. dysphagia, gastroesophageal reflux (GER) and impaired gastric emptying and motility) are common in patients with neuromuscular disease (1). Neuromuscular scoliosis has been linked to pulmonary morbidity and decreasing functional ability but the connection between scoliosis and gastrointestinal morbidity is poorly understood. It is known that surgical correction of neuromuscular scoliosis can cause a variety of early gastrointestinal complications (e.g. superior mesenteric artery syndrome (SMAS), paralytic ileus, pancreatitis), but the incidence, severity, and risk factors of these complications are unclear (2–8).
We performed a retrospective cohort study with an objective to determine the incidence, severity, course, and risk factors of gastrointestinal complications after surgical correction of neuromuscular scoliosis. Based on the earlier literature and our clinical estimation, our hypothesis was that one-fifth of patients have transient postoperative gastrointestinal morbidity, and that the main risk factors for these complications were spastic neuropathic neuromuscular disease, combined anteroposterior operative approach, and severe spinal deformity (6, 9, 10).
Material and Methods
Patients
In total, 118 consecutive patients under the age of 21 underwent instrumented spinal fusion for neuromuscular scoliosis, kyphoscoliosis, or kyphosis at Helsinki University Central Hospital during 2000–2011. We included all patients (n = 91) with neuropathic neuromuscular disease and at least a 2-year follow-up (in patients who were alive at the time of the study). Three patients (one male, two females) with less than 2-year follow-up, one female patient with previous spine surgery for other indication, and 23 patients with myopathic neuromuscular disease were excluded. We excluded patients with myopathic neuromuscular scoliosis from the study population because their neuromuscular disease, severity and type of scoliosis, operative treatment, and perioperative examinations were significantly different than in patients with neuropathic neuromuscular scoliosis.
In the end, the study population consisted of 91 patients of which 55 were females. The age at surgery averaged 14.5 (standard deviation (SD) 2.9) and follow-up time 4.9 (SD 2.3) years. Fifty-three (58%) patients had spastic and 38 (42%) had flaccid neuropathic disease. Neuropathic diseases included cerebral palsy (CP; n = 42), syndromic disorders (n = 24), spinal cord injury (n = 3), myelomeningocele (n = 7), spinal muscular atrophy (n = 3), polyneuropathy (n = 4), and unknown disorders (n = 8). Sixty-five (71%) patients were non-ambulatory. Ten (9%) patients died during the follow-up period. No perioperative mortality was observed.
Study Design
This was a retrospective cohort study with consecutively operated patients. Clinical and radiographic evaluation of patients was conducted according to normal pre- and postoperative follow-up protocol. Two independent observers collected and analyzed the data. Patients with gastrointestinal complications were identified after clinical, operative, and radiographic data collection was complete. We identified risk factors for these complications by performing a group-comparison between patients with gastrointestinal complications and the rest of the study population and by binary logistic regression.
Gastrointestinal Complications
Patient was evaluated to have a postoperative gastrointestinal complication if the symptoms of the complication began immediately after surgery during the postoperative hospitalization period. The marked gastrointestinal complications that were encountered in the study population were prolonged paralytic ileus, gastroparesis, and significant dysphagia. A prolonged paralytic ileus was defined as an ileus that was verified on radiography, had no signs of mechanical bowel obstruction, and required nasogastric decompression or parenteral nutrition over 1.5 weeks. The cutoff point was selected to be 1.5 weeks, which was based on our clinical experience that it represents abnormally prolonged ileus after neuromuscular scoliosis surgery. Gastroparesis was defined as delayed gastric emptying (without mechanical outlet obstruction), that needed parenteral nutrition or nasojejunal feeding tube. Significant dysphagia was defined as a swallowing difficulty requiring postoperative readmission to the hospital for parenteral nutrition or implantation of a feeding gastrostomy during the following 2 years after surgery.
Operative Methods
The indication for scoliosis surgery was a progressive scoliosis >50° with poor spinal balance. Operation was either posterior only (n = 57) or combined anteroposterior (one- or two-stage; n = 34) instrumented spinal fusion. The anterior procedure was used in large, rigid curves (>60° on traction radiographs) that could not be corrected with the earlier instrumentation (hybrid, Luque) by posterior spinal fusion alone (11). The anterior approach was thoracotomy for the lower thoracic spine or thoracoabdominal for the T12-L2 area and included anterior release of the anterior longitudinal ligament, multi-level discectomies, and spinal fusion using autologous rib grafting. The posterior instrumentation was a total pedicle screw (TPS; n = 43), hybrid (claws, hooks, and pedicle screws; n = 39), or Luque (sublaminar wires; n = 9) construct with extension to pelvis (iliac screws or Luque–Galveston rods; n = 44) when pelvic obliquity exceeded 10°. Ponte type osteotomies were performed in 33 (36%) patients to correct kyphosis and increase spinal mobility during correction (12). Halo-femoral traction was used perioperatively to decrease spinal rigidity in 12 (11%) patients (13). Vertebral column resections were performed in three (3%) patients.
Prophylactic antibiotics and proton pump inhibitors (
The pre- and postoperative pain, nausea, and vomiting management protocol did not change during the study period. Preoperatively patients were given acetaminophen. Postoperative pain treatment consisted of acetaminophen, tramadol, or oxycodone given intravenously, orally or by patient-controlled analgesia (PCA) intravenous infusion pump. None of the patients received epidural analgesia. Postoperative nausea was treated with metoclopramide or ondansetron. The type and amount of medication that was given was determined by the severity of symptoms (self-evaluation of pain on visual analog scale, secondary clinical signs of pain and nausea), patient weight, and patient’s mental ability. In certain patients (# 3, 14, 19) with marked paralytic ileus or gastroparesis, gastroprokinetic medications (cisapride and tegaserod) were given based on the evaluation of the pediatric surgeon (Table 1).
Gastrointestinal complications after neuromuscular scoliosis correction.
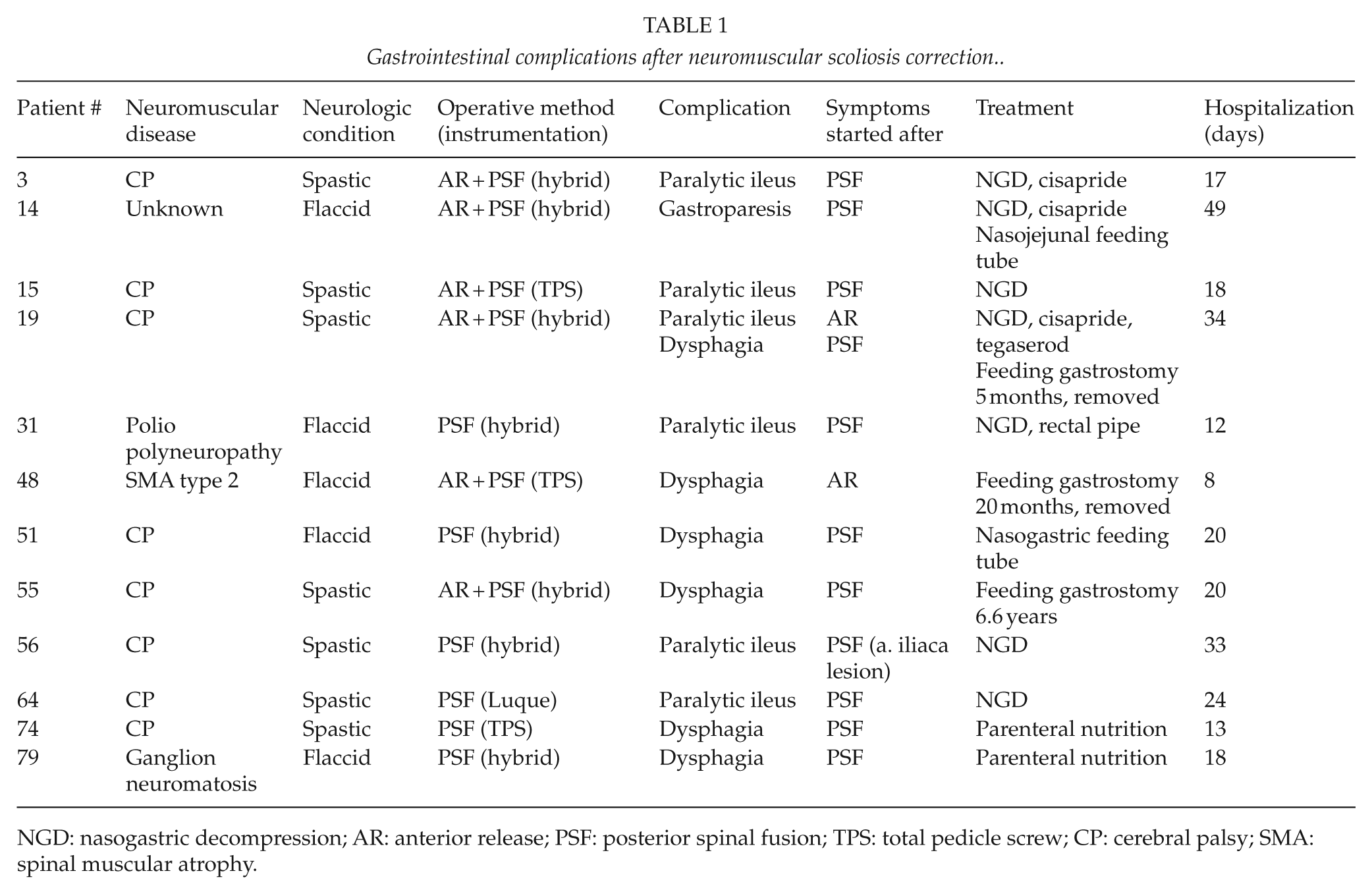
NGD: nasogastric decompression; AR: anterior release; PSF: posterior spinal fusion; TPS: total pedicle screw; CP: cerebral palsy; SMA: spinal muscular atrophy.
Clinical Evaluation
Standard preoperative clinical examination included complete neurological evaluation by a pediatric neurologist. Depending on the preference of the orthopedic spinal surgeon, patients were referred to a pediatric gastroenterologists and nutritionist to evaluate nutritional status and make appropriate adjustments. Body mass index (BMI) was measured before surgery and at final follow-up.
Radiographic Evaluation
Standard supine, sitting, or standing posteroanterior (PA) and lateral radiographs were taken of the entire spine pre- and postoperatively, and at final follow-up. The thoracic (apex T1-T11), thoracolumbar (T12-L1) and lumbar (L2-L5) scoliosis curves, coronal balance, and pelvic obliquity were measured from the PA-radiographs. Thoracic kyphosis (T2-T12), lumbar lordosis (T12-S1), and sagittal vertical axis were measured from the lateral radiographs (15, 16). Main curve rigidity was assessed with preoperative traction or side bending radiographs. Rigid scoliosis curves had small correction in traction and side bending radiographs (16).
Statistical Analysis
Nonparametric quantitative data were evaluated using the Mann–Whitney U test. The Pearson and Spearman rho test was used in bivariate correlations. Chi2 test was used in comparisons of categorical data. Binary logistic regression was used to assess relative risk (RR) and 95% confidence interval (CI) of independent variables for the development of gastrointestinal complications after neuromuscular scoliosis surgery. When several statistically significant risk factors for gastrointestinal complications were identified in binary logistic regression, we used multivariate logistic regression to evaluate the RR and CI of these risk factors in a multivariate model that was adjusted for age, gender, and neuromuscular condition. P-values <0.05 were considered statistically significant.
Results
Gastrointestinal Complications
Significant gastrointestinal complications occurred in 12 (13%) patients. None of the patients with gastrointestinal complications died during follow-up (Table 1).
Prolonged paralytic ileus was observed in six patients (five spastics, one flaccid). One of these patients underwent emergency laparotomy due to an intraoperative iliac artery lesion. The arterial injury was caused by a misplaced sacral pedicle screw that was inserted during posterior spinal fusion. All patients with paralytic ileus were treated successfully by nasogastric decompression, intravenous fluid supplementation, and enemas.
One female patient (# 14) developed gastroparesis after surgery. Her radiographs and endoscopy revealed gastric retention without signs of mechanic obstruction. She was treated with jejunal feeding tube for 6 weeks and recovered fully.
Significant swallowing difficulty (requiring re-hospitalization for parenteral fluid intake within 1 month of the operation or implantation of feeding gastrostomy) was seen in six patients (three spastics, three flaccids). One of these patients also had a prolonged paralytic ileus immediately postoperatively. The treatment of these patients consisted of intravenous fluid supplementation with or without nasogastric tube feeding and antiemetic medication. Symptoms resolved fully in three patients after 3–6 weeks but three patients required feeding gastrostomy. Two patients resumed sufficient oral intake and gastrostomy was removed after 6 and 20 months, whereas one patient remains dependent on feeding gastrostomy 6.6 years after surgery. According to pediatric neurologists, there were no findings to suggest that postoperative dysphagia was caused by progression of the primary neurological disease.
Operative Results
The operative time averaged 7.1 (SD 2.3) h and the mean blood loss (in relation to blood volume) was 120% (SD 110%). Patients with gastrointestinal complications had a similar mean operative time (7.7 (SD 2.3) vs 7.0 (SD 2.2) h; p = 0.199) and blood loss (140% (SD 95%) vs 110% (SD 120%); p = 0.149) when compared to other patients. However, the average length of postoperative intravenous opioid medication (5.3 (SD 2.1) vs 4.3 (SD 2.9) days; p = 0.028) and hospitalization time (22 (SD 11) vs 16 (SD 20) days; p = 0.005) were significantly longer in patients with gastrointestinal complications.
Intraoperative SSEP or MEP disturbance was observed more often in patients who developed gastrointestinal complications (4/11) when compared to other patients (8/76) (p = 0.020). Ponte osteotomies were used less often in patients with gastrointestinal complications (1/12 vs 32/79; p = 0.031). In patients who underwent posterior only spinal fusion, TPS instrumentation was used less often in patients who developed postoperative gastrointestinal complications (1/6) when compared to patients who did not (34/51) (p = 0.017). The anterior release (6/12 vs 28/79; p = 0.331), pelvic fixation (3/12 vs 41/79; p = 0.082), halo-femoral traction (0/12 vs. 12/79; p = 0.147), or vertebral column resections (1/12 vs 2/79; p = 0.351) were not used significantly more often in patients with gastrointestinal complications.
Radiographic Outcomes
The average preoperative main curve was 85° (SD 21°) and postoperative main curve correction 69% (SD 17%). Patients with gastrointestinal complications had significantly smaller main curve correction in traction/bending radiographs before surgery than other patients (26% (SD 12%) vs 41% (SD 11%); p < 0.001). The radiographic parameters can be seen in more detail in Table 2.
Radiographic results.
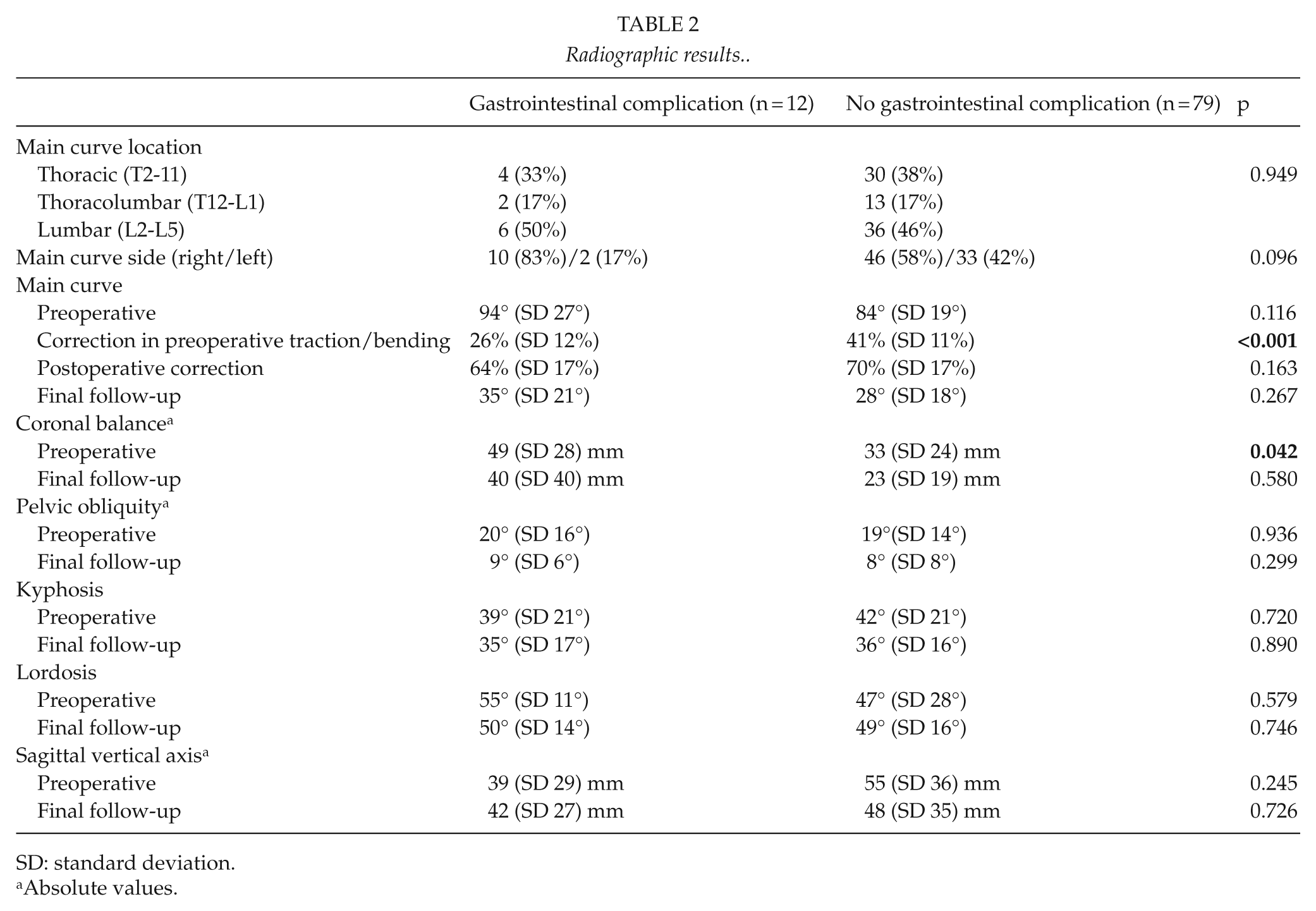
SD: standard deviation.
Absolute values.
Non-Gastrointestinal Complications
The complication rate was 44% (40/91). Pulmonary complications were seen in 10%, instrumentation breakage in 13%, wound infections in 8%, urinary retention in 8%, and neurologic complications (transient or permanent lower extremity paresis) in 4% of patients. Postoperative urinary retention was more common in patients with gastrointestinal complications (3/12 vs 4/79; p = 0.016) when compared to other patients. Other non-gastrointestinal complications were seen in equal amounts in patients with gastrointestinal complications when compared to other patients (4/12 vs 28/79; p = 0.887).
Clinical Status
Patients with gastrointestinal complications did not have significant difference in age at surgery (14.4 (SD 3.0) vs 14.6 (SD 2.9); p = 0.694), gender (2/12 vs 34/79 males; p = 0.082), mental retardation (7/12 vs 47/79; p = 0.939), or ambulatory status (10/12 vs 55/79 non-ambulatory; p = 0.327) when compared to other patients. The preoperative BMI averaged 14.0 (SD 3.4) in patients with postoperative gastrointestinal complications and 16.7 (SD 3.6) in other patients (p = 0.074). At final follow-up, BMI averaged 18.2 (SD 3.1) and 18.1 (SD 4.7) (p = 0.562), respectively. One of the six patients who underwent preoperative high caloric diet due to poor nutritional status developed a gastrointestinal complication. Gastrointestinal complications were not observed in patients who had undergone preoperative Nissen fundoplication (n = 6) or who had a feeding gastrostomy (n = 10) at the time of surgery. One out of 10 patients, who had GER diagnosis before surgery, developed a marked gastrointestinal complication postoperatively.
Gastrointestinal Complications Over Time
All marked gastrointestinal complications in this study occurred during 2000–2007. In total, 65 patients were operated on during 2000–2007 and 26 patients during 2008–2011. The first operation with TPS instrumentation was performed at our institution in 2005. In our institution, the use of anterior release and hybrid instrumentation was significantly more common in 2000–2007 than in 2008–2011 (anterior release 33/65 vs 1/26, respectively; hybrids 37/65 vs 2/26, respectively; p < 0.001 for both comparisons), while the use of TPS instrumentation (19/65 vs 24/26) and Ponte osteotomies (15/65 vs 18/26) was more common after 2007 (p < 0.001 for both comparisons).
The duration of postoperative intravenous opioid medication decreased over time (R2 = –0.084; p = 0.007), while the degree of average preoperative main curve (R2 = –0.003; p = 0.661) and scoliosis rigidity in traction/bending (R2 = 0.009; p = 0.428) remained unchanged. The duration of postoperative opioid medication was significantly longer after the combined anteroposterior spinal fusion (5.7 (SD 3.2) days) than after posterior spinal fusion alone (3.6 (SD 2.6) days) (p < 0.001).
Risk Factors for Gastrointestinal Complications
After adjusting for age (>14 years at surgery), gender (male), and neuromuscular condition (spastic), the most significant risk factors for postoperative gastrointestinal complications were main curve correction <30% in preoperative traction/bending radiographs (RR = 28 (95% CI 4.4–180); p < 0.001), the duration of postoperative intravenous opioid medication over 5 days (RR = 7.9 (95% CI 1.8–35), p = 0.006), disturbance in intraoperative neurophysiological spinal cord monitoring (SSEP or MEP) (RR = 6.0 (95% CI 1.1–34); p = 0.043), and preoperative main curve >90° (RR = 5.5 (95% CI 1.3–23); p = 0.020). After performing multivariate logistic regression analysis which included all the above risk factors (adjusted for age, gender, and neuromuscular condition), the only statistically significant risk factor to remain was main curve correction <30% in preoperative traction/bending radiographs (RR = 14 (95% CI 1.4–134); p = 0.023) (Table 3).
Risk factors for gastrointestinal complications after neuromuscular scoliosis correction.
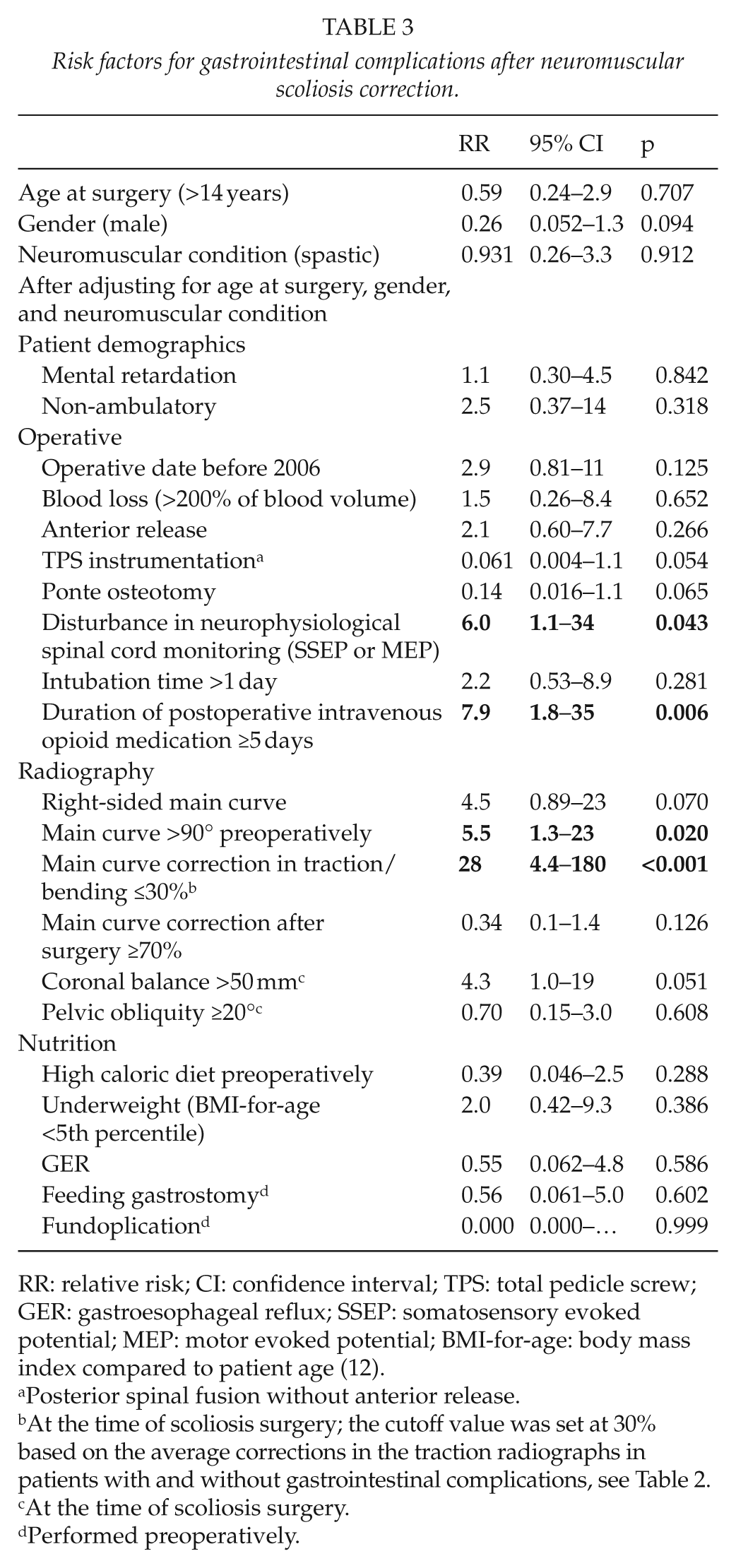
RR: relative risk; CI: confidence interval; TPS: total pedicle screw; GER: gastroesophageal reflux; SSEP: somatosensory evoked potential; MEP: motor evoked potential; BMI-for-age: body mass index compared to patient age (12).
Posterior spinal fusion without anterior release.
At the time of scoliosis surgery; the cutoff value was set at 30% based on the average corrections in the traction radiographs in patients with and without gastrointestinal complications, see Table 2.
At the time of scoliosis surgery.
Performed preoperatively.
Discussion
To the best of the authors’ knowledge, this is the first cohort study evaluating the incidence, severity, course, and risk factors of gastrointestinal complications after neuromuscular scoliosis surgery in a general level. We found out that majority of patients did not experience significant gastrointestinal complications after neuromuscular scoliosis correction. The incidence of gastrointestinal complications was 13 % in our study. These complications were predominantly reversible and associated with gut motility (paralytic ileus, dysphagia). None of them caused death. We observed that preoperative scoliosis rigidity was significantly associated with the development of these complications.
The study design was retrospective with a medium-term follow-up time. Two independent observers collected the data from extensive clinical, operative, and radiographic records and analyzed the results. Our study has some limitations in addition to the retrospective nature of the study. Patients were operated on during a period exceeding 10 years and therefore, significant variety was observed in operative methods over time. We had a sufficiently large patient population (n = 91) but significant heterogeneity was observed in patients’ neurological disorders and other demographics. The magnitude of preoperative main scoliosis curve and postoperative correction was similar to previously reported results in the literature (17). The objective evaluation of nutritional status (e.g. BMI) and gastrointestinal symptoms in non-ambulatory patients with neurodevelopmental disorders is challenging and may give inaccurate results. In addition to GER and dysphagia, other less evident preoperative gastrointestinal diseases could not be identified.
In the previous literature, gastrointestinal complications have been variably reported (incidence 0%–37%) after neuromuscular scoliosis surgery (6, 9, 10). Several studies of postoperative pancreatitis and SMAS exist while there is only one prospective study by Jalanko et al. with small study sample investigating gastrointestinal problems in a more general level after scoliosis surgery (4–7, 9, 18–20). In our study, the rate of gastrointestinal complications (13%) was similar to previously published results by other authors (7, 9, 18, 21).
Gastrointestinal complications in our study consisted of paralytic ileuses, swallowing difficulties, and gastroparesis. Majority (92%) of these were reversible and treated successfully with conservative methods, such as nasogastric decompression, intravenous fluid supplementation, antiemetic medicine, and laxatives. These complications extended postoperative hospitalization period significantly and in some cases, the symptoms persisted for several years. Feeding gastrostomy was postoperatively implanted on three patients with dysphagia. One patient has been dependent on the gastrostomy to this day. Postoperative pancreatitis was not observed in our study. We did not measure serum amylase levels systematically and therefore, mild postoperative pancreatitis may have gone overlooked. Our data suggest, however, that majority of the most significant gastrointestinal problems after neuromuscular scoliosis surgery are caused by gut dysmotility (4, 5, 7).
There were several risk factors for gastrointestinal complications in our study: rigid scoliosis, large preoperative main curve, disturbances in intraoperative spinal cord monitoring, and postoperative opioid medication. According to multivariate analysis, preoperatively rigid scoliosis was the most significant risk factor of all. A similar kind of finding has been made in the study by Braun et al. They observed that thoracic spinal rigidity was associated with postoperative SMAS after scoliosis surgery (4).
The magnitude and rigidity of scoliosis are interrelated. Other risk factors, such as the degree of opioid pain medication and disturbances in the intraoperative spinal cord monitoring, can be regarded to be secondary to the operative correction of severe and rigid scoliosis. Patients who undergo surgical correction of rigid scoliosis require more often anterior releases and more forceful maneuvers to correct the deformity and hence are more painful after surgery. It is likely that the higher amount of opioid pain medication that is postoperatively needed contributes to the formation and prolongation of paralytic ileus after surgery.
The specific mechanism how rigid scoliosis leads to gastrointestinal complication after scoliosis correction can be speculated. The greater rigidity and tension in the corrected spine can lead to neuropathy (neurapraxia or axonotmesis) in the visceral nerves and spinal cord. This assumption is supported by the finding of more intraoperative disturbances in intraoperative spinal cord monitoring in patients who developed gastrointestinal complications. Also, neuropathic neuromuscular disease may make patients more susceptible to the detrimental effects of neuropathy caused by instrumented scoliosis correction.
The hypothesis of neuropathic damage to gut function has been presented previously. Vande Velde et al. described five orally fed patients with CP, who underwent posterolateral spinal fusion and postoperatively developed severe gastric dysmotility requiring jejunostomy feeding that persisted for several years in some. Interestingly, in two cases, scoliosis was not corrected but only stabilized (8). Zein et al. (22) concluded that recurrent nausea and emesis following scoliosis surgery is caused by delayed gastric emptying due to postprandial antral hypomotility that is likely to be caused by neuropathy resulting from continuous traction of the spine.
Contrary to our hypothesis, the degree of spinal deformity correction, neurological primary disease, or combined anteroposterior surgical approach were not significantly associated with the development of these gastrointestinal complications. It appears that as long the deformity is flexible, one can correct it as much without adverse effects on gastrointestinal function. Preoperative nutritional status or gastrointestinal disorders did not associate with the development postoperative gastrointestinal complications in our study. Also, patients with gastrointestinal complications did not appear to have significantly larger amount of other complications. Interestingly, however, urinary retention was more common in patients with postoperative gastrointestinal complications than other patients.
ERAS protocols are popular in children with adolescent idiopathic scoliosis but those kinds of methods were not implemented in the neuromuscular scoliosis patients in this study (23). In children with neuromuscular scoliosis, early mobilization is probably the single most effective method to expedite recovery after surgery. Neuromuscular scoliosis patients present with a heterogeneous group of conditions. Therefore, it is difficult to set up a specific protocol for this whole group of patients.
While spinal rigidity had a good specificity (97%) for gastrointestinal complications, its sensitivity was low (45%) and therefore cannot be used to predict which patients ultimately develop gastrointestinal complications. The causes behind these complications are undoubtedly multifactorial. One of our patients, for example, developed paralytic ileus after undergoing a laparotomy to correct an intraoperative iliac artery lesion. The selection of operative method plays a significant role. The incidence of gastrointestinal complications has declined over time as the use of hybrid instrumentations and anterior releases has decreased and the use of TPS instrumentation and Ponte osteotomies has increased in our institution. Ponte osteotomies increase spinal mobility during scoliosis correction with TPS instrumentation and may also allow spinal deformity correction by shortening, thus possibly reducing the tension in the visceral nerves and spinal cord. The significance of halo-femoral traction in the development of gastrointestinal complications remains unclear. None of our 10 patients with halo-femoral traction developed marked gastrointestinal complications.
Conclusion
Marked gastrointestinal complications were common and occurred in 13% of patients after surgical correction of neuromuscular scoliosis. While majority of these complications were reversible, they extended postoperative hospitalization and some of them caused long-term gastrointestinal morbidity. Gastrointestinal complications were not associated with death. Rigid scoliosis was the most significant risk factor for these complications. Significant gastrointestinal complications appear to be less frequent after posterior only spinal fusion with TPS instrumentation and Ponte osteotomies.
Footnotes
Acknowledgements
The authors would like to thank orthopedic surgeon Mikko Mattila and research nurse Katariina Mattila for their help and cooperation and Rinnekoti Research Centre for their support.
Declaration of Conflicting Interests
I.H. has received scientific funding from Medtronic International.
Funding
Grants were received from the Rinnekoti Research Center. Scientific support was received from Medtronic International to hire a research nurse who helped us to collect the perioperative data.
Ethical Aspects
We obtained permission to perform this study from the Ethics Committee of the hospital where the study was conducted (ref. # 162/13/03/03/2008).
