Abstract
Background:
Sarcopenia, reduced skeletal muscle mass, is associated with frailty, injuries, and mortality. The purpose of this study was to evaluate the impact of computed tomography–determined sarcopenia on surgical complications and outcomes after resection of non-small cell lung cancer.
Methods:
For a total 272 non-small cell lung cancer patients that underwent surgery between 2011 and 2016, cross-sectional area of muscle at the third lumbar vertebra (L3) was retrospectively measured using preoperative chest computed tomography images. Sarcopenia was defined as an L3 muscle index of <55 cm2/m2 for men and of <39 cm2/m2 for women. Clinical characteristics, postoperative complications, disease-free survival, and overall survival of patients with or without sarcopenia were compared.
Results:
A total of 60.3% (n = 164) were male, and mean patient age was 62.9 ± 9.6 years. The prevalence of sarcopenia was 22.4% for all study subjects, 32.9% for men, and 6.5% for women. No significant difference was observed between patients with or without sarcopenia in terms of intensive care unit or hospital stay (p = 0.502 and p = 0.378, respectively), and the presence of sarcopenia was not associated with postoperative complications. Furthermore, no significant difference was observed between the 3-year disease-free survival rate (74.3% vs 66.7%, p = 0.639) or 3-year overall survival rate (83.9% vs 87.7%, p = 0.563) of patients with or without sarcopenia.
Conclusion:
Sarcopenia as determined by preoperative computed tomography does not appear to have a negative impact on surgical outcome or overall survival for resected non-small cell lung cancer patients.
Keywords
Introduction
Lung cancer is one of the most common malignancies in developing and developed countries and a leading cause of mortality (1). Surgical resection offers the best opportunity for long-term survival and cure in patients with resectable non-small cell lung cancer (NSCLC), and its suitability in individual candidates includes preoperative staging and assessments of performance status, concurrent comorbidities, and pulmonary function.
Skeletal muscle depletion has important age-related health implications in the elderly, because sarcopenia is associated with frailty, injuries, and mortality of the population. The clinical importance of sarcopenia is also being increasingly recognized in oncologic patients as a component of cancer cachexia syndrome (2, 3), and recent studies have reported associations between preoperative sarcopenia and postoperative morbidity and mortality for different types of cancer surgeries (4–7). Sarcopenia is common in lung cancer patients, and its presence is considered to be associated with poor functional status and overall survival (OS) (8, 9). However, no study has addressed the influence of sarcopenia on postoperative complications and prognosis after curative resection of NSCLC. Accordingly, the objective of this study was to evaluate the impact of CT-determined sarcopenia on the surgical complications and long-term outcomes of patients treated surgically for NSCLC.
Materials and Methods
Patients
The radiology database and medical records system at Gachon University Gil Medical Center (Incheon, Korea) were searched, and 292 patients with newly diagnosed, pathologically proven NSCLC that underwent curative-intent surgery from January 2011 to December of 2016 were identified. After exclusion of 20 patients in whom their baseline positron emission tomography/computed tomography (PET/CT) images were unavailable for the evaluation, we included final 272 patients. Body mass index (BMI) was defined as weight divided by height squared (kg/m2), and BMI values were categorized as underweight (<18.5 kg/m2), normal (18.5–22.9 kg/m2), overweight (23.0–24.9 kg/m2), or obese (≥25 kg/m2) (10).
The institutional review board of our hospital approved this retrospective study and waived the requirement for informed patient consent.
Perioperative Assessment and Surgery
Patients were staged according to the American Joint Commission on Cancer (AJCC) Staging Manual (7th edition) (11). The routine preoperative workup included pulmonary function testing, computed tomography (CT) of the chest, whole-body 18 F-fluorodeoxyglucose (FDG) positron emission tomography (PET)/CT, bronchoscopy, and brain magnetic resonance imaging (MRI). Histopathologic evaluation of mediastinal nodes was performed by endobronchial ultrasound (EBUS)-guided transbronchial needle aspiration (TBNA). All patients were admitted the day before surgery.
Operative procedures included lobectomies, bilobectomies, sleeve resections, and pneumonectomies, and all 205 study subjects underwent video-assisted thoracic surgery (VATS) or open thoracotomy. Systematic lymph node dissection was considered mandatory. Mediastinal lymph node dissection consisted of en bloc resections of all nodes at stations 2R, 4R, 7, 8, 9, and 10R for right-sided tumors and of all nodes at stations 4L, 5, 6, 7, 8, 9 and 10L for left-sided tumors.
Early ambulation was recommended to prevent thromboembolism and low-molecular weight heparin (LMWH) was administered subcutaneously, when intensive care unit (ICU) stays exceeded 1 day. Postoperative pain was control by patient-controlled analgesia (PCA) and oral analgesics.
PET/CT Scan
All patients fasted for at least 6 h before PET/CT to ensure a normal blood glucose level. About 60 min after administering 370 MBq (10 mCi, i.v.) of FDG, imaging was performed using an integrated PET/CT device (Siemens Medical Systems, Erlangen, Germany), equipped with lutetium oxyorthosilicate crystal PET detectors, and six-slice CT detectors.
The integrated CT imaging was performed from the head to the pelvic floor without contrast administration using the following parameters: 130 kVp, 110 mAs, 2-mm pitch, 1-s tube rotation, and a slice thickness of 5 mm, which matched the slice thickness of PET images.
Image Analysis
CT images obtained from whole-body PET/CT scans at time of diagnosis were retrospectively analyzed by a radiologist. The third lumbar vertebra (L3) was selected as a landmark since the cross-sectional area of tissues in this region provide an established means of estimating total body tissue quantities in the general population (Pearson correlation coefficients range from 0.71 to 0.92 (12, 13)). Body composition analyses of two consecutive CT images extending from L3 in the inferior direction were evaluated using commercially available software (Terarecon 3.4.2.11, San Mateo, CA, USA). Tissue cross-sectional areas (cm2) of respective tissues in slices were computed automatically by summing appropriate pixels. The CT Hounsfield unit (HU) range used was −29 to 150 HU for skeletal muscle. After applying threshold using a predefined HU threshold, boundaries were corrected manually when necessary. L3 muscle index (cm2/m2) was defined as the cross-sectional area of muscle at the L3 level normalized for stature as is conventional for BMI.
Definition of Sarcopenia
Sarcopenia was defined as a L3 muscle index of <55 cm2/m2 for men and of <39 cm2/m2 for women, as proposed by international consensus for cancer cachexia (2).
Postoperative Complications
Early mortality was defined as death within 30 days of surgery or longer if death occurred during hospitalization. Complications were classified as follows: (1) respiratory, (2) cardiac, and (3) other complications. Respiratory complications were classified as major or minor complications; major respiratory complications included acute respiratory distress syndrome (ARDS), acute lung injury (ALI), pneumonia, pulmonary embolism, and pulmonary edema. ARDS and ALI were diagnosed using the Berlin definitions proposed by the European Society of Intensive Care Medicine ARDS Task Force (14). Minor respiratory complications were sputum retention and atelectasis not requiring bronchoscopy toileting. Cardiac complications were classified as arrhythmia, angina, myocardial infarction, and cardiogenic shock. Other complications included surgical/technical complications (hemothorax, early bronchopleural fistula, empyema, chylothorax, cardiac dislocation and wound infection), neurological complications, and abdominal/urinary tract complications.
Statistical Analysis
Descriptive statistics are reported as proportions or means with standard deviations. For categorical variables, comparisons between subjects with and without sarcopenia were performed using the Pearson’s χ2 test or Fisher’s exact test. Continuous variables were compared using the Student’s t-test. Univariable and multivariable logistic analyses were performed to evaluate factors associated with overall postoperative complications. Survivals were estimated using the Kaplan–Meier method and compared using the log-rank test. Disease-free survival (DFS) and OS were estimated from time of operation to recurrence, death, or last follow-up. Univariable and multivariable Cox proportional hazard models were used to identify prognostic factors of survival. Variables with p-values of <0.20 by the univariable test were included in the multivariable analysis, in which forward stepwise selection method was used. Two-sided p-values of < 0.05 were considered statistically significant. The analysis was performed using SPSS for Windows v. 19.0 (SPSS Inc., Chicago, IL, USA).
Results
Patient Characteristics and Sarcopenia
The baseline characteristics of the 272 study subjects are summarized in Table 1. There were 164 male (60.3%) and 108 female (39.7%) patients of overall mean age of 62.9 ± 9.6 years (range, 33–81 years). The overall prevalence of sarcopenia among study subjects was 22.4% (32.9% for men and 6.5% for women). The presence of sarcopenia was significantly related to male gender (p < 0.001) and smoking status (current or ex-smoker vs non-smoker, p = 0.003). Sarcopenic patients had a lower BMI (21.3 ± 2.7 kg/m2 vs 25.1 ± 3.1 kg/m2, p < 0.001) and a lower percentage forced expiratory volume in 1 second (FEV1) (87.2 ± 18.2% vs 95.8 ± 19.5%, p = 0.002). However, no significant difference was observed between sarcopenic and non-sarcopenic patients in terms of age, preoperative protein and albumin levels, and diffusion capacity of lungs for carbon monoxide (DLCO), histology, or pathologic tumor stage.
Characteristics of the study population.
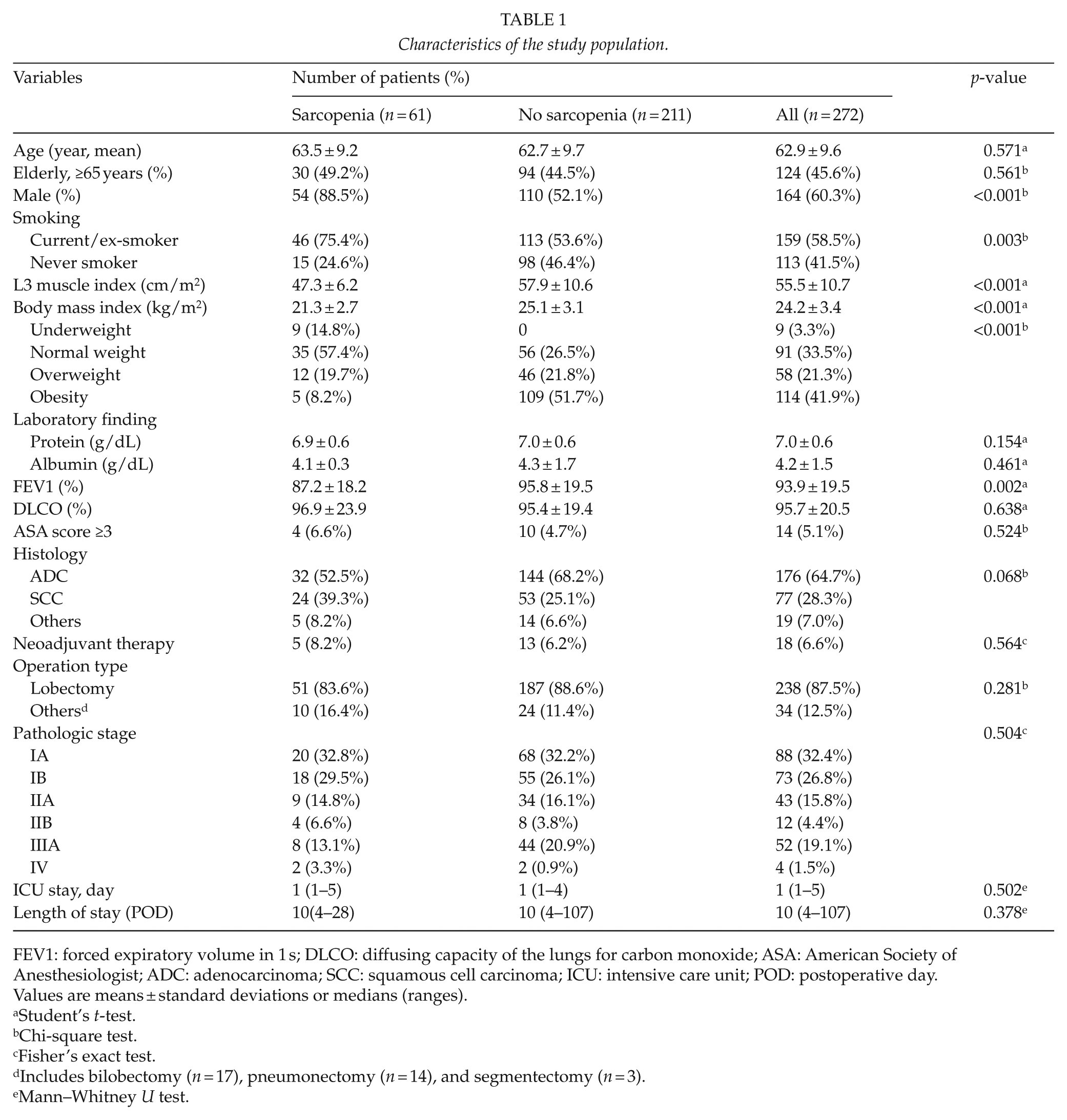
FEV1: forced expiratory volume in 1 s; DLCO: diffusing capacity of the lungs for carbon monoxide; ASA: American Society of Anesthesiologist; ADC: adenocarcinoma; SCC: squamous cell carcinoma; ICU: intensive care unit; POD: postoperative day.
Values are means ± standard deviations or medians (ranges).
Student’s t-test.
Chi-square test.
Fisher’s exact test.
Includes bilobectomy (n = 17), pneumonectomy (n = 14), and segmentectomy (n = 3).
Mann–Whitney U test.
Surgery and Perioperative Treatment
In total, 18 (6.6%) patients received neoadjuvant chemotherapy. Operative procedures performed were as follows: 238 lobectomy (87.5%), 17 bilobectomy (6.3%), 14 pneumonectomy (5.1%), and 3 segmentectomy (1.1%). In total, 151 (55.5%) received minimal invasive surgery using the VATS technique, and complete resection was achieved in all patients. No difference was found between sarcopenic and non-sarcopenic patients in terms of preoperative neoadjuvant treatments or operative procedures and techniques.
Postoperative Complications
For all study subjects, median durations of ICU and hospital stays after surgery were 1 day (range, 1–5 days) and 10 days (range, 4–107 days), respectively. No significant difference was found between sarcopenic and non-sarcopenic patients in terms of ICU or hospital stay (p = 0.502 and p = 0.378, respectively).
No early mortality occurred after surgery. The overall complication rate for all study subjects was 22.8% (n = 62), and most common complication was a cardiac complication (n = 19). Eighteen patients (6.6%) had respiratory complications; seven major (three cases of PE, three cases of pneumonia and one case of lung injury) and eleven minor complications (four cases of atelectasis and seven cases of prolonged air leakage). Other complications (n = 22) were as follows: postoperative urinary retention (n = 9), delirium (n = 7), vocal cord palsy (n = 2), chylothorax (n = 1), ischemic colitis (n = 1), azotemia (n = 1), and wound infection (n = 1). Overall postoperative complications were similar in patients with or without sarcopenia (29.5% vs 20.9%, p = 0.168); respiratory (6.6% vs 6.6%, p = 1.000), cardiac (11.5% vs 5.7%, p = 0.151), and others (9.8% vs 7.6%, p = 0.596) (Table 2).
Postoperative complications.
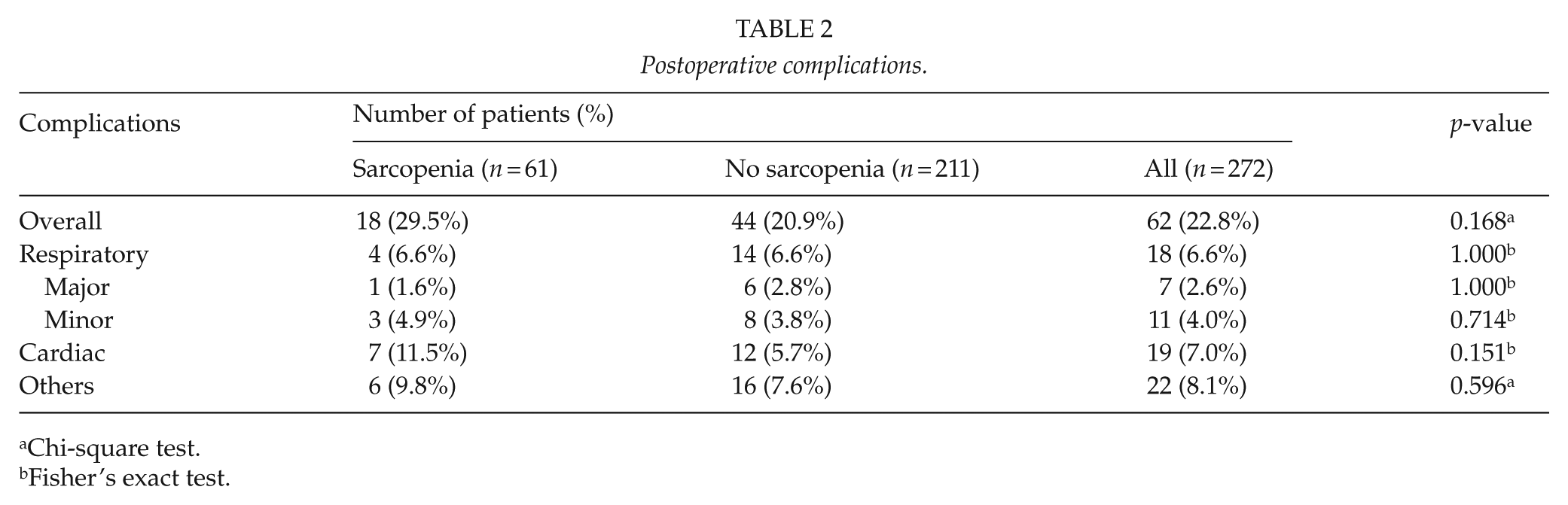
Chi-square test.
Fisher’s exact test.
There was no significant difference in the amount of skeletal muscle mass in patients who underwent postoperative complications or not (Table 3).
Preoperative CT-measured L3 skeletal muscle index (cm 2 /m2) and the development of postoperative complications.
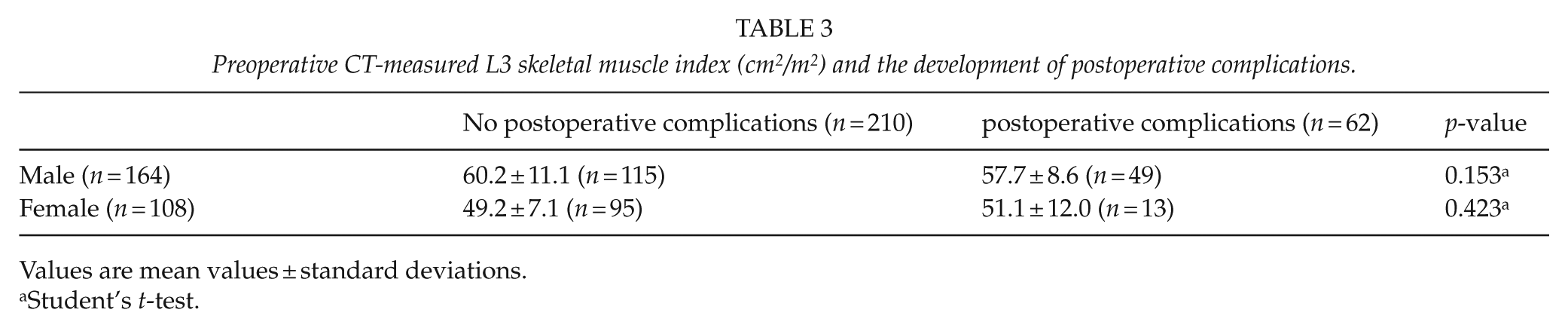
Values are mean values ± standard deviations.
Student’s t-test.
A multivariable analysis was performed to identify factors associated with overall complications; results are summarized in Table 4. Male gender (odds ratio (OR), 2.82; 95% confidence interval (CI), 1.43 to 5.56; p = 0.003) and decreased predicted postoperative FEV1 more than 50% (OR, 2.49; 95% CI, 1.08 to 5.77; p = 0.033) were significant factors associated with overall complications. The presence of preoperative sarcopenia was not an associated factor with the development of overall complications and pulmonary complication.
Factors associated with overall postoperative complications as determined by univariable and multivariable analyses.
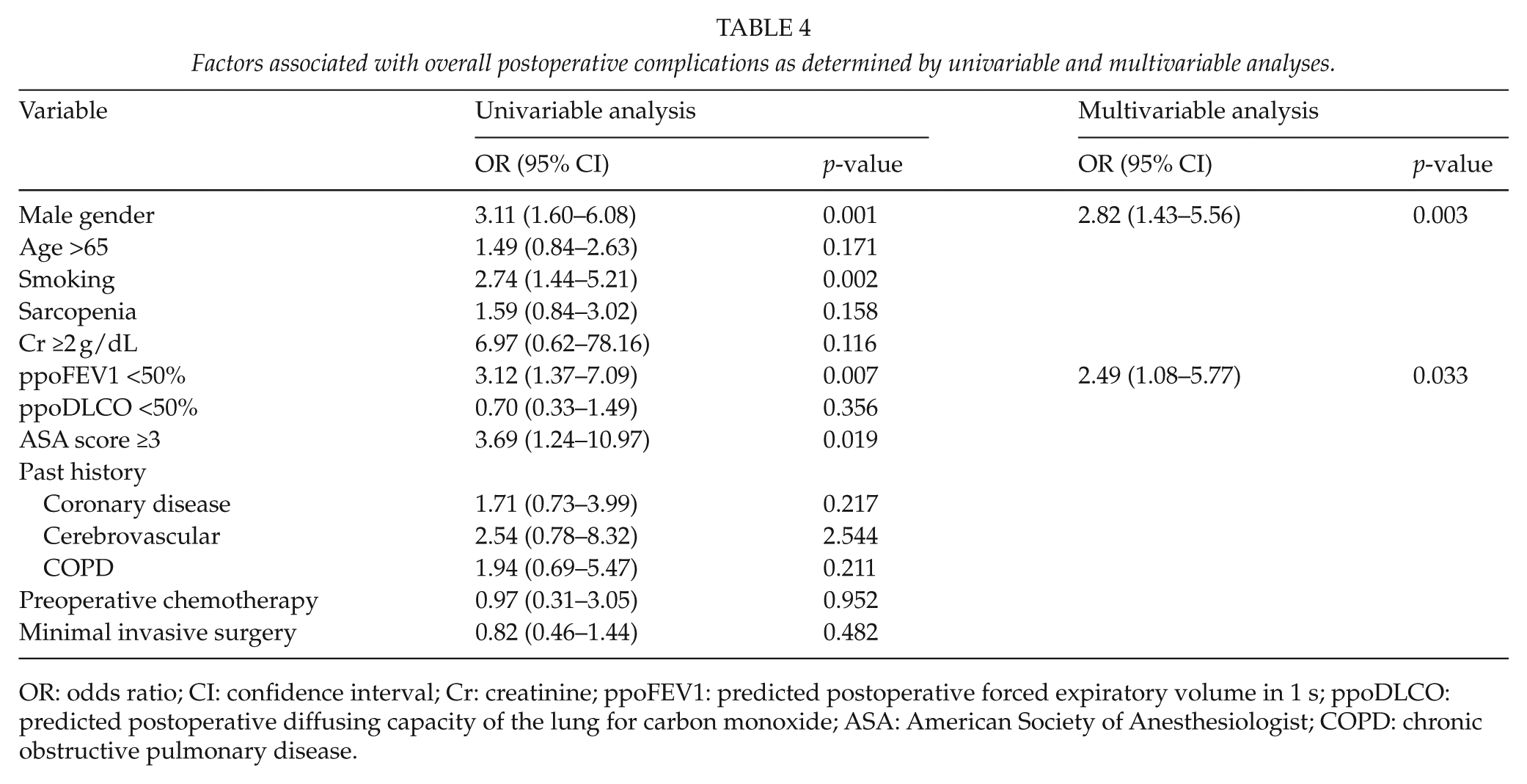
OR: odds ratio; CI: confidence interval; Cr: creatinine; ppoFEV1: predicted postoperative forced expiratory volume in 1 s; ppoDLCO: predicted postoperative diffusing capacity of the lung for carbon monoxide; ASA: American Society of Anesthesiologist; COPD: chronic obstructive pulmonary disease.
Recurrence and Survival Outcomes
During a median 26.3 months of follow-up (95% CI, 22.3 to 30.4 months), 64 patients (23.5%) recurred and 27 (9.9%) died.
Three-year DFS rate and 3-year OS rate (74.3% vs 66.7%, respectively, p = 0.639; and 83.9% vs 87.7%, p = 0.563, respectively) of sarcopenic and non-sarcopenic patients were not significantly different (Fig. 1). Multivariable analysis showed pathologic T stage (hazard ratio (HR), 2.36; 95% CI, 1.26 to 4.41; p = 0.007), pathologic N stage (HR, 2.92; 95% CI, 1.74 to 4.89; p < 0.001), and R1 resection (HR, 4.76; 95% CI, 1.44 to 15.75; p = 0.02211) significantly and independently predicted shorter DFS (Table 5), and R1 resection (HR, 11.38; 95% CI, 3.79 to 34.13; p < 0.001), pneumonectomy (HR, 3.27; 95% CI, 1.19 to 9.00; p < 0.022), and male gender (HR, 3.04; 95% CI, 1.03 to 8.96; p = 0.044) independently predicted shorter OS. The presence of preoperative sarcopenia was not found to be significantly associated with shorter OS or DFS.
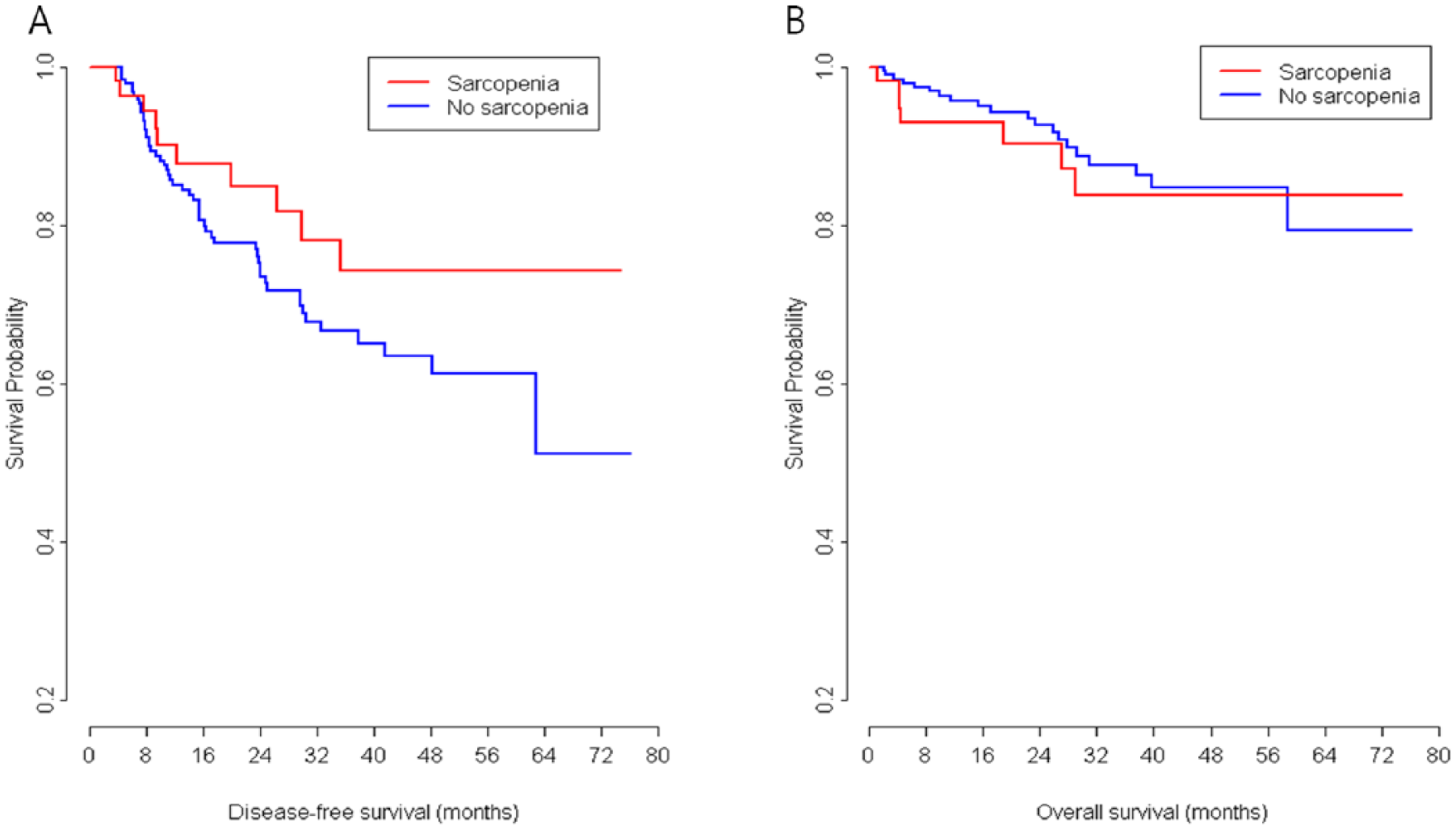
Kaplan–Meier curves of disease-free survival and overall survival for sarcopenic and non-sarcopenic patients with non-small cell lung cancer.
Factors associated with disease-free survival as determined by univariable and multivariable analyses.
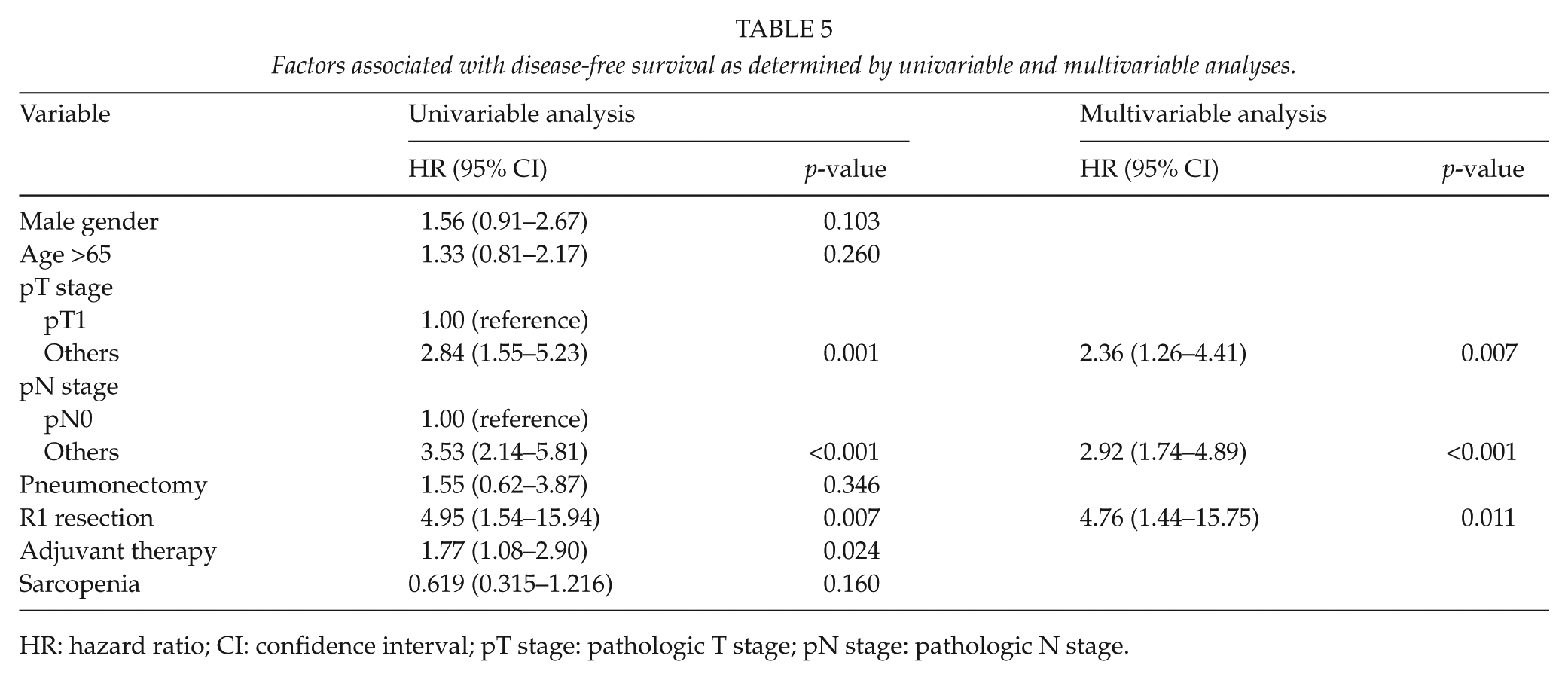
HR: hazard ratio; CI: confidence interval; pT stage: pathologic T stage; pN stage: pathologic N stage.
Discussion
The depletion of skeletal muscle mass has been shown to be a poor prognostic indicator in cancer cachexia syndrome, and thus, the identification of sarcopenia is important in oncology patients (2, 9, 15–17), since the sarcopenic status is associated with functional impairment, increased risk of chemotherapy-related toxicities, and reduced survival. The reported prevalence of sarcopenia is 16% in breast cancer survivors and 39% in preoperative colorectal cancer patients, respectively, and in NSCLC, it has a reported prevalence of 47% (18). However, in the previous study, cancer stages were locally advanced or recurrent (stages III or IV) NSCLC, and patients with early stage disease were not included. In lung cancer, the presence of sarcopenia has been reported to be associated with poor functional status, increased morbidity, and reduced survival (3, 8). However, in these studies, the poor prognostic effect of CT-determined sarcopenia was observed for all NSCLC stages combined or for advanced NSCLC, and its prognostic significance remained unknown in the resectable NSCLC population. Complete anatomic resection remains the mainstay of curative therapy for NSCLC patients with loco-regional disease, and this study is the first to report that preoperative CT-determined sarcopenia is not associated with postoperative complications, recurrence, or survival in patients that undergo surgery with curative intent for NSCLC.
Recent studies have shown associations between preoperative sarcopenia and postoperative morbidity and mortality after different types of cancer surgeries, such as resection for pancreatic cancer (4), hepatic resection for hepatocellular carcinoma (5), and for colorectal hepatic metastasis (6, 7). In resectable colorectal cancer, sarcopenia was also found to be associated with postoperative infection and delayed recovery (19). However, in endometrial cancer patients, preoperative sarcopenia was not associated with surgical complications or OS, but the sarcopenic status was associated with shorter DFS (20). The inconsistent finding was probable due to different patient physiques and types of operative procedures. In this study, no association between the presence of preoperative sarcopenia and the development of postoperative complication or long-term prognosis is probably due to two reasons. First, operable patients are in the early stages of lung cancer rather than the cachectic advanced stage, which reflects a continuum of skeletal muscle depletion during the different stages of lung cancer. In a previous study, bioelectrical impedance analysis (BIA) measured fat-free mass in preoperative NSCLC patients with stage I or II disease was similar to that observed in normal controls (21), and the overall prevalence of sarcopenia of 22.4% in our cohort is lower than its reported prevalence in advanced stage NSCLC (18, 22) or SCLC (79%) (9). In-line with the recent obesity epidemic in industrialized countries, a significant proportion of our cohort was overweight (21.3%) or obese (41.9%), and thus, sarcopenic obesity might have more clinical implications in this setting. However, we could not evaluate the significance of sarcopenic obesity due to small number of patients (n = 5) who were sarcopenic and obese. Second, as a result of technical advancements in the operative and perioperative management of lung cancer patients, complication rates after lung resection surgery have reduced.
Pulmonary complications following lung resection occur in 10%–20% of patients (23). The risk factors of postoperative pulmonary complication include advanced age, current smoker, predicted postoperative FEV1, predicted postoperative DLCO, American Society of Anesthesiologist (ASA) score, and preoperative chemotherapy (23–26). However, in this study, the incidence pulmonary complication was 6.6%, which is a slightly lower than reported previously.
Despite recent interest in the clinical implications of sarcopenia in cancer patients, data are limited on clinical outcomes by type malignancy. Furthermore, the majority of published studies have used arbitrary study-specific criteria and different imaging modalities, such as BIA, dual-energy X-ray absorptiometry (DEXA), or CT to determine the presence of sarcopenia. In this study, sarcopenia was defined using CT-determined L3 muscle indices using the standard cutoff values proposed in a recently published consensus definition of cancer cachexia (2), in which sarcopenia is defined using modality- and sex-specific cutoffs. CT examinations, as routinely conducted in oncology patients, provide a means of precisely quantifying skeletal muscle using a single cross-sectional area at the L3 as determined by secondary analysis of CT images.
Several limitations of this study should be mentioned. First, this study was performed in a retrospective manner for lung cancer patients who underwent surgery for curative intent, so early stage lung cancer patients who did not underwent surgery due to poor general condition and poor pulmonary function were not included for analysis. Second, the patients’ number was small because this study was performed at a single institution. Third, we evaluated the presence of sarcopenia at the time of diagnosis only and do not evaluate longitudinal changes of skeletal muscle loss after surgery. We suggest future studies evaluate the impact of sarcopenia and skeletal muscle loss on the short-term and long-term prognosis in operable lung cancer patients.
In summary, we investigated postoperative complications and long-term outcomes in patients that underwent curative-intent surgery for NSCLC with preoperative CT-determined sarcopenia. Our results suggest that sarcopenia does not appear to have a negative impact on surgical outcome or OS among resected NSCLC patients. Further studies, including a randomized controlled trial, will be needed to elucidate relations between sarcopenia and outcomes for these patients.
Footnotes
Acknowledgements
E.Y.K. and H.Y.L. contributed equally to this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
