Abstract
Background and Aims:
The development of pleural infection may imply a worse state of health and prognosis. The objective of this study was to ascertain the long-term survival and causes of death after pleural infections and to compare them to those of matched controls.
Material and Methods:
Altogether 191 patients treated for pleural infections at a single University Hospital between January 2000 and December 2008 and 1910 age- and gender-matched controls were included. Survival data and the causes of death for non-survivors were obtained from national databases and compared between the groups.
Results:
The etiology of pleural infection was pulmonary infection in 70%, procedural complication in 9%, trauma in 5%, malignancy in 4%, other in 7%, and unknown in 5% of patients. The course of treatment was surgical in 82%, drainage only in 12%, and conservative in 5% of included patients. The median follow-up time was 11 years. Mortality rates were 8.4% versus 0.8% during the first 90 days, p < 0.001, and 46.6% versus 24.5% overall, p < 0.001, in patients and controls, respectively. Mortality was significantly higher in patients with pulmonary infection, procedural complication, or malignancy as the etiology of pleural infection. In multivariable analysis, advanced age, previous malignancies, institutional care, alcoholism, and malignant etiology for the infection were associated with inferior survival. Deaths caused by malignancies, respiratory diseases, and digestive diseases were significantly more common in patients than in controls.
Conclusion:
Long-term survival in patients with pleural infections is significantly inferior to that of age and gender-matched controls.
Introduction
Bacterial infection of the pleural space is a serious condition that causes significant patient discomfort, morbidity, mortality, and substantial health care costs worldwide. It is most commonly a complication of pneumonia, thoracic trauma, a malignant disease involving the lungs or the pleura, or an invasive procedure. Pleural infections comprise empyemas when presenting with frank pus in the pleura and complicated—parapneumonic when pneumonia-associated—effusions in the absence of pus but with other signs of infection in the pleural fluid analysis (1). In addition to antimicrobial therapy, hospitalization and invasive treatment are required in most cases. While pleural drainage—with or without concomitant lavage or intrapleural fibrinolysis—may suffice, the majority of patients undergo surgery. The incidence of cases requiring operative treatment has increased and is currently 11.8 per 100.000 patient years (2–5). In-hospital mortality associated with pleural infections ranges from 6% to 27% and the most common pathogens detected are streptococci and staphylococci (6–10).
The long-term prognosis of these patients is poorly described in the current literature. It is the authors’ impression that pleural infections are rare in healthy individuals and that the development of the disease implies a worse state of health. We hypothesize that even after successful treatment of the disease, the long-term prognosis in these patients is significantly inferior to that of the general population. The aim of this study was to compare the long-term survival and the causes of death between patients treated for pleural infections with age- and gender-matched controls and to identify factors associated with worse prognosis.
Material and Methods
The Study Population
The study included patients treated for pleural infections in Tampere University Hospital, Tampere, Finland, a tertiary academic referral center, between January 2000 and December 2008. The patients were identified from the institutional database by retrieving all cases associated with the International Statistical Classification of Diseases and Related Health Problems (10th revision; ICD-10), code “J86” or any subclass of “J86.” Pleural infection was confirmed or excluded by carefully reviewing each case and the relevant laboratory and imaging results. Patients without available follow-up data and those treated in the pediatric department were excluded. The demographic information and relevant medical history were recorded in each patient.
The Diagnosis And Classification of Pleural Infection
The diagnosis of pleural infection required clinical signs of infection, including fever and leukocytosis, associated with a pleural collection considered to be of significant size and consisting of either frank pus or with other findings consistent with infection in the pleural fluid laboratory analysis, that is, a positive bacterial culture or stain, pH < 7.2, lactate dehydrogenase level >1000 IU/L, or glucose <2.2 mmol/L. The etiology of pleural infection was classified according to history and presentation into the following groups: pulmonary infection, trauma, malignancy related cases, procedural complication, including cases developing after surgery, chest tube insertion, and/or percutaneous procedures involving the pleurae, other, or unknown. Malignancy-related cases comprised spontaneous pleural infections occurring in patients with intra-thoracic malignancy. If pleural infection developed as a complication of surgery or other invasive procedure performed due or in adjunct to a malignant disease, the case was classified as a procedural complication. The course of treatment in each patient was determined by the attending physician according to the clinic standards and was classified as either surgical, drainage only, or conservative. Surgery was either open thoracotomy or video-assisted thoracic surgery (VATS).
Available imaging studies obtained during hospitalization and follow-up were reviewed by a radiologist. The ventilating lung surface area of the affected lung in the chest X-ray, the maximal thickness of the pleural collection in the chest X-ray or chest computed tomography, the presence of lung abscesses, interlobar collections, and/or more than one non-communicating effusions before treatment, as well as the development of diaphragmatic elevation, disappearance of the costodiaphragmatic recess, and the increase in ventilating lung area in the chest X-rays in follow-up were recorded. Diaphragmatic elevation was diagnosed when the left hemidiaphragm was higher than the right or when the right hemidiaphragm was ≥3 cm higher than the left.
Control Material And Survival Analysis
The long-term survival of patients with pleural infections was compared to that of control subjects obtained from the Finnish Population Register Centre which contains robust data on all Finnish inhabitants and from which survival data were also obtained for the study subjects. In all, 10 controls were requested for each patient and the control subjects were matched for age, gender, and the location of residence at the time of patients’ hospital admission. The survival follow-up lasted until 18 May 2017. For non-surviving patients and controls, the diagnosis code assigned as the cause of death was obtained from the database of Statistics Finland that contained complete data on all deaths that had occurred in Finland until the end of 2015. In the statistical analysis, the causes of death were categorized according to the ICD-10 classifications main blocks, for example, “I00-I99: Diseases of the circulatory system,” and only patients surviving at least until and those whose death occurred on or before 31 December 2015 were included in the analysis. The long-term survival and the causes of death in non-survivors were compared between patients and controls and the factors associated with the prognosis were ascertained using statistical methods.
This study was performed according to the Helsinki Declaration. Institutional review board approval was obtained. Statistical analysis was performed using SPSS for Windows statistical software version 16.0 using the chi-square test and Fisher’s exact test to compare categorical data, the Mann–Whitney U test and Kruskal–Wallis H test to compare the medians between groups in nonparametric variables, and the log-rank test to compare survival between groups. Multivariable analysis for patient characteristics and factors associated with survival was performed using the Cox regression analysis. Preoperative variables that were significantly associated with survival in the univariable analysis were included in the multivariable model. Statistical significance was set at p < 0.05.
Results
Study Population
The database search from the study period yielded altogether 274 cases in which the ICD-10 code J86 was included. After critical review, a total of 81 cases were excluded, most commonly because pleural infection had been suspected but not confirmed; another diagnosis, such as malignancy with pleural involvement but no infection, was established; the patient was treated in the pediatric department; or the diagnosis code was erroneous. Of patients with established pleural infection, two cases were excluded from the analysis because of unavailable follow-up data. The final study population comprised 191 patients that were each treated for a single pleural infection episode and altogether 1910 age- and gender-matched controls. The demographic information of the study population, the patient characteristics, including the etiology of pleural infection, type of treatment given, as well as the results of the imaging studies of included patients are shown in Table 1. Surgical treatment was performed through open thoracotomy in 92% and VATS in 8% of operated patients during the study period.
The demographic information of the study subjects and the preoperative characteristics of study patients, disease presentation in imaging studies, the type of treatment given, and imaging outcomes of the study patients and the most important patient subgroups.
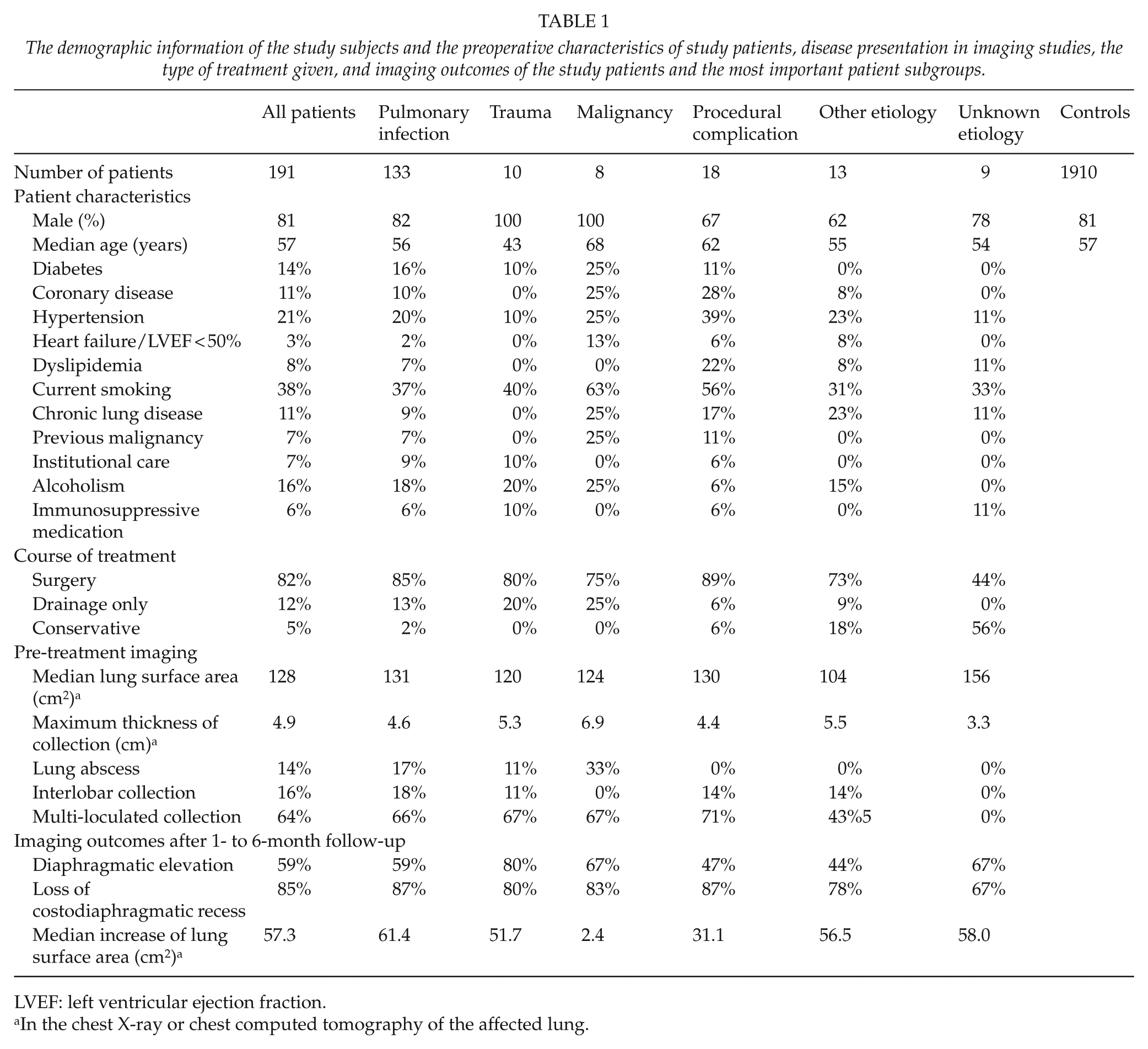
LVEF: left ventricular ejection fraction.
In the chest X-ray or chest computed tomography of the affected lung.
Survival
The median follow-up time for patients and controls was 11 (range 8–17) years and the survival of patients and controls is shown in Fig. 1 and in Table 2. The overall mortality rate during follow-up in patients and controls was 46.6% versus 24.5%, p < 0.001, respectively. The mortality rate during the first 90 days of follow-up was significantly higher in patients than in controls, 8.4% versus 0.8%, p < 0.001, respectively. However, even in patients surviving the first 90 days, the rates of death were significantly higher in patients when compared to controls during the remaining follow-up, 41.7% versus 23.8%, p < 0.001, respectively. In the subgroup analysis, survival remained statistically significantly inferior in patients with pulmonary infection, procedural complication, or malignancy as the etiology of pleural infection when compared to controls, p < 0.001 each. The median age was higher in non-survivors, both in patients, 66 versus 51 years, p < 0.001, and in controls, 73 versus 54 years, p < 0.001, respectively, when compared to surviving study subjects. Gender was not associated with the prognosis of study patients or controls.
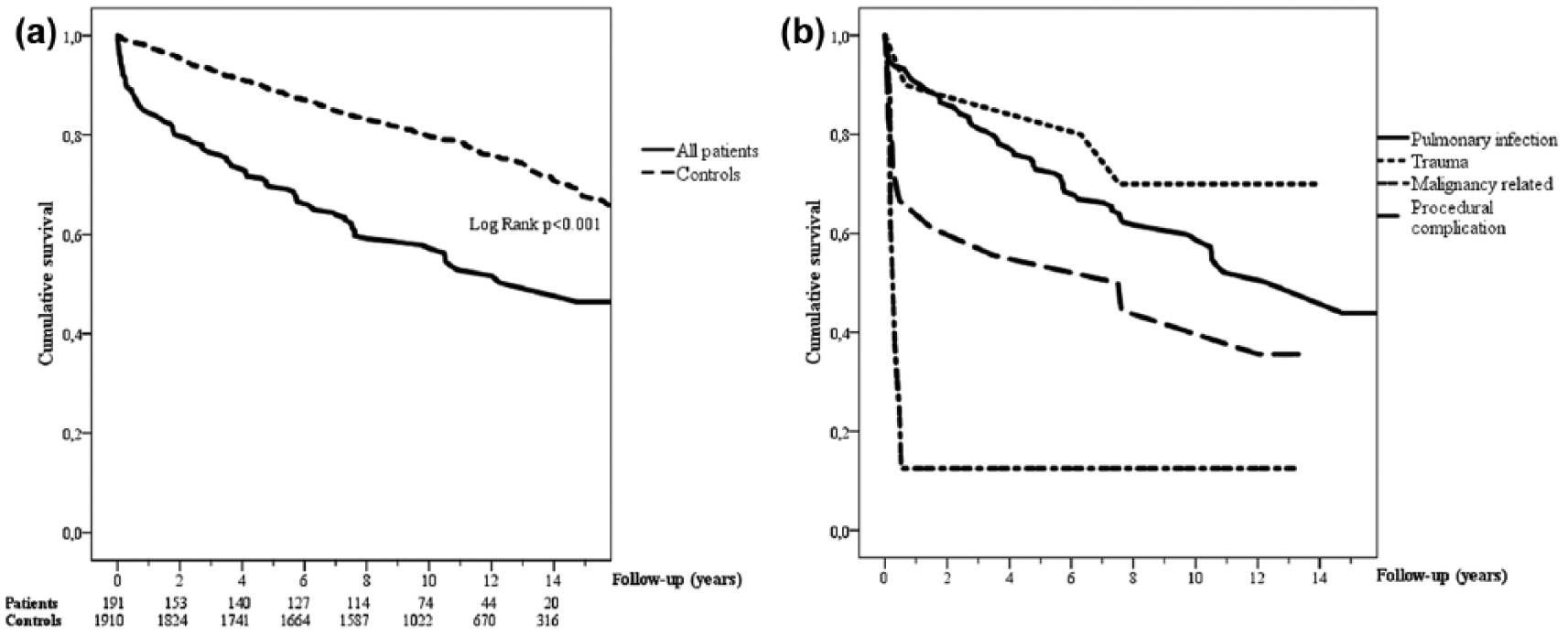
(a) Kaplan–Meier survival curve comparing patients with pleural infections and age- and gender-matched controls. The number of patients and controls at risk at each time-point are shown. (b) Survival curves in the most important patient subgroups according to the etiology of pleural infection.
Mortality rates at different time-points in patients with pleural infections and in controls.
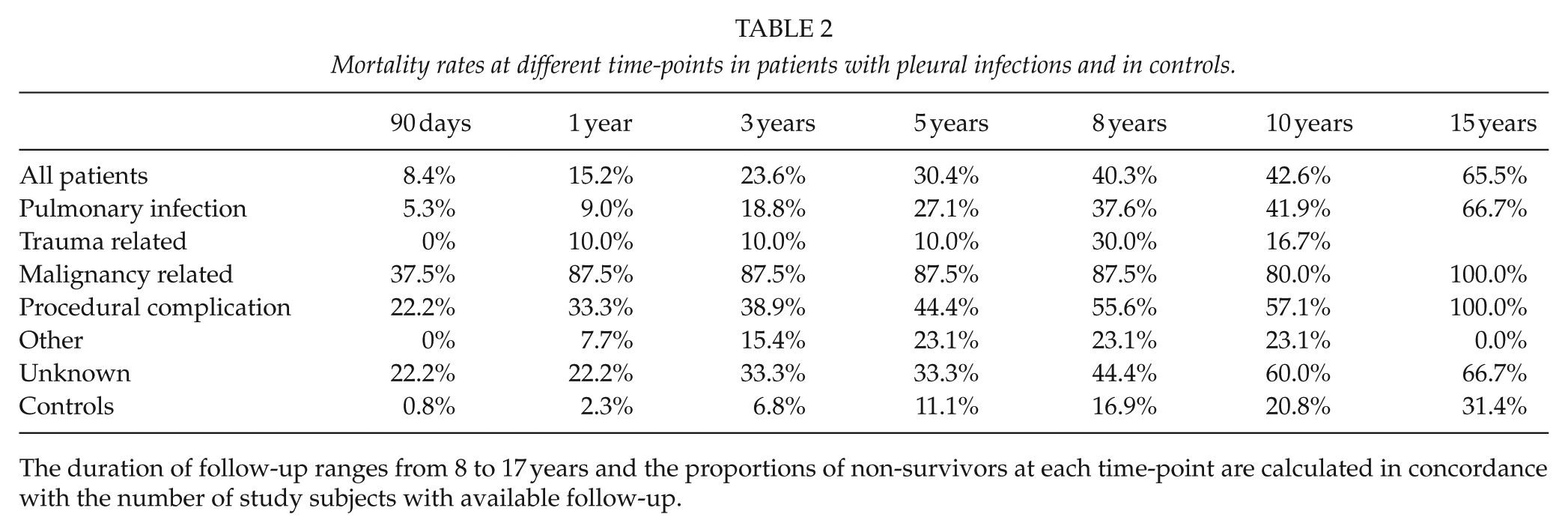
The duration of follow-up ranges from 8 to 17 years and the proportions of non-survivors at each time-point are calculated in concordance with the number of study subjects with available follow-up.
The results of the univariable survival analysis measuring the associations between patient characteristics, the etiology and radiologic presentation of pleural infection, the type of treatment given, and survival in patients with pleural infections are shown in Table 3. In multivariable survival analysis, advanced age, history of a previous malignancy, institutional care, alcoholism, and malignant etiology for the infection were independently associated with worse survival in patients with pleural infections (Table 4).
Mean survival times of patients treated for pleural infections and controls during follow-up.
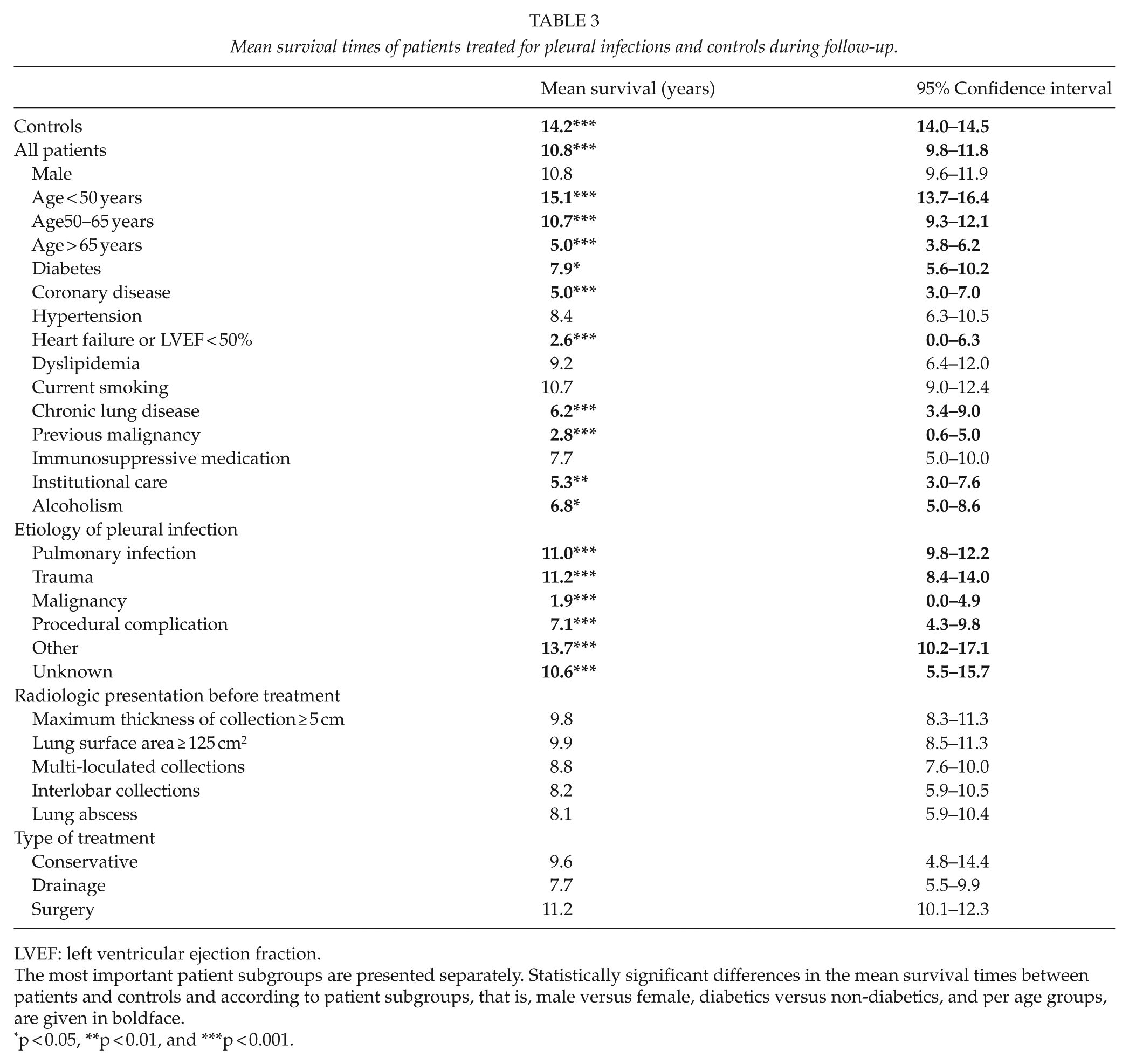
LVEF: left ventricular ejection fraction.
The most important patient subgroups are presented separately. Statistically significant differences in the mean survival times between patients and controls and according to patient subgroups, that is, male versus female, diabetics versus non-diabetics, and per age groups, are given in boldface.
p < 0.05, **p < 0.01, and ***p < 0.001.
Multivariable survival analysis with the Cox regression model in patients with pleural infections.
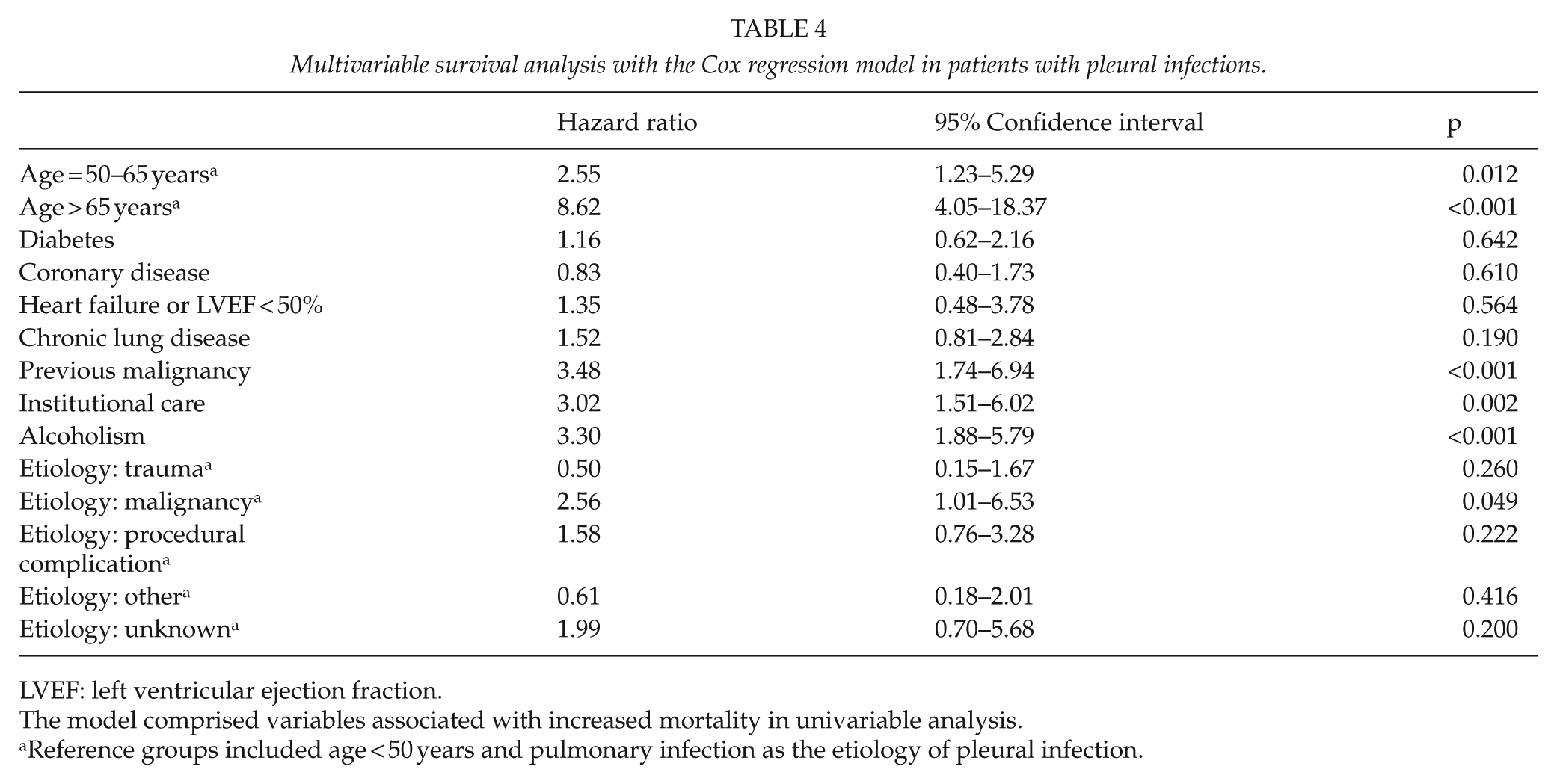
LVEF: left ventricular ejection fraction.
The model comprised variables associated with increased mortality in univariable analysis.
Reference groups included age < 50 years and pulmonary infection as the etiology of pleural infection.
The Causes of Death
The most important causes of death and their respective rates in non-surviving patients and controls are shown and compared in Fig. 2. Malignancies together with cardiovascular, respiratory, gastrointestinal, and neurologic diseases accounted for 85% of all deaths. There were no statistically significant differences in the rates of cardiovascular or neurologic causes of death between patients and controls. In contrast, deaths caused by malignancies, respiratory diseases, and digestive diseases were statistically significantly more frequent in patients, and in patients with pulmonary infection as the cause of pleural infection, than in controls.
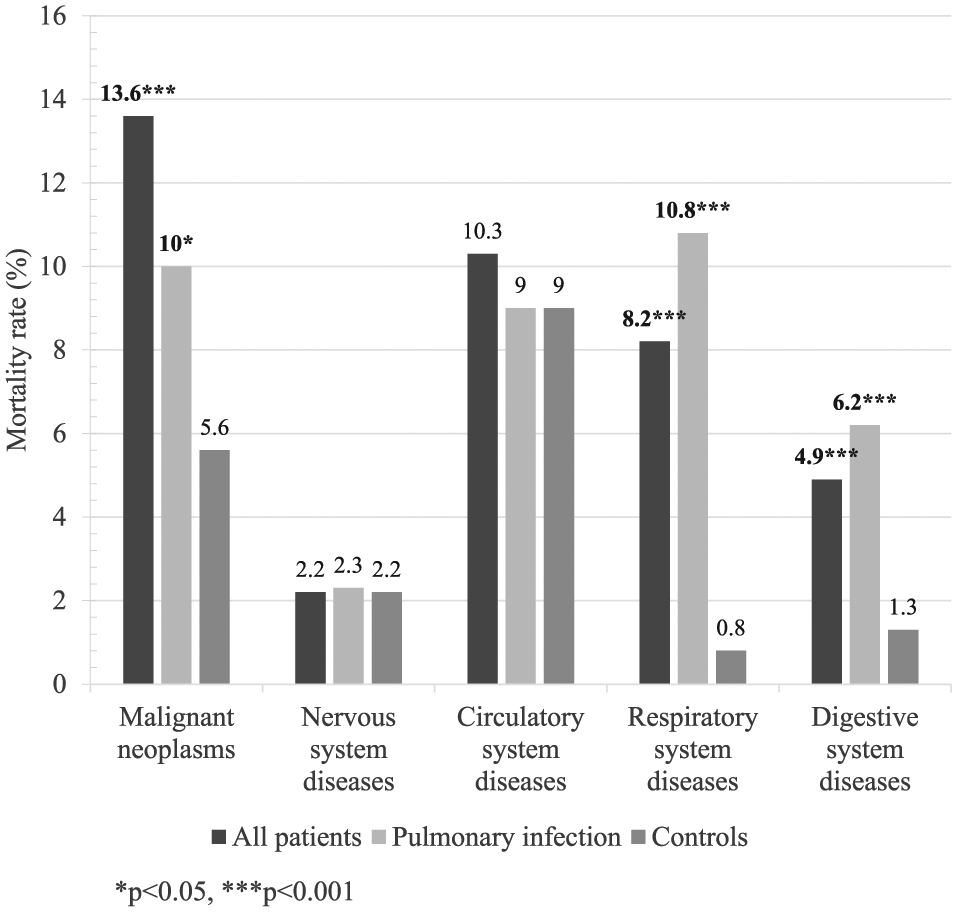
Mortality rates for the most frequent causes of death in patients with pleural infections, in patients with pulmonary infection as the cause of the infection, and in age- and gender-matched controls during follow-up. Statistically significant differences between patients and controls are represented in boldface.
Conclusion
This study sought to describe the long-term survival and the causes of death of patients treated for pleural infections and to compare these rates to those of age- and gender-matched controls. To the best of our knowledge, no similar studies with a large age- and gender-matched control material have been published. We report significantly higher early and late mortality in patients with pleural infections compared to controls and increased rates of death caused by malignancies, respiratory diseases, and gastrointestinal diseases in these patients during follow-up. Mortality rates were significantly higher even when excluding early deaths and cancer-related pleural infections.
Our results concerning the early mortality, 8.4% within the first 90 days after pleural infections, agree with the current literature. A single previous study has investigated the long-term survival after pleural infections and reported a 62% survival rate after 10-year follow-up in patients with surgically treated pleural infections caused by pulmonary infections (11), the results of which are in accordance with ours. In this study, despite a slight trend for better survival in operated patients, no statistically significant differences in the long-term survival of patients with pleural infections were observed according to the type of treatment given, that is, surgical or other. Most patients underwent surgery and the patients were not randomized between the treatment options. Our results probably indicate successful clinical decision-making in selecting patients for the most appropriate course of treatment. However, the statistical power in this regard was limited.
Compared to patients with pulmonary infections as the cause of the pleural disease, survival was clearly inferior in patients with a malignant etiology and in patients with pleural infections developing as complications after invasive procedures, many of which were probably performed due or in adjunct to malignant diseases. Malignant, pulmonary, and digestive diseases were more frequent causes of death in patients with pleural infections than in controls–even when excluding cancer-associated pleural infections. The reasons for higher death rates due to the aforementioned diseases are unclear but may be associated with shared risk factors such as smoking. In this study, 38% of included patients were actively smoking, while the corresponding rates in the general population during the study period ranged from 20% to 24% (12). Besides smoking, other surrogate markers for poor prognosis, such as alcoholism and long-term institutional care, were frequent in patients with pleural infections.
The radiologic presentation of the disease was not clearly associated with survival in patients with pleural infections. We believe that the finding both reflects our technical ability to achieve sufficient control of the infection operatively in most patients regardless of disease presentation and our limitations in the treatment of associated diseases. Several co-morbidities such as diabetes, coronary disease, and heart failure were associated with worse prognosis. However, in the multivariable analysis, only increased age, previous malignancies, alcoholism, institutional care, and malignant cause of the pleural infection remained independently associated with inferior survival.
Study Limitations
This study describes patients treated at a single University Hospital and may not accurately reflect the patient material at other institutions. Medical history of the control material was not available. The retrospective setting of the study limited the availability of some significant patient characteristics such as body mass index. Some patients might have been missed due to erroneous use of diagnosis codes. Statistical power was limited when analyzing patient subgroups.
In conclusion, the long-term survival of patients with pleural infections is clearly and significantly inferior to that of age- and gender-matched controls. The death rates for malignant, respiratory, and digestive diseases were significantly higher in patients than in controls. Advanced age, previous malignancies, alcoholism, institutional care, and malignant cause of the pleural infection were independently associated with worse survival in patients with pleural infections.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The study was supported by institutional funding and by grants received from the Tampere Tuberculosis Foundation, the Emil Aaltonen Foundation, and the Foundation of the Finnish Anti-Tuberculosis Association.
