Abstract
Background and Aims:
Aneurysmal bone cysts represent about 1% of primary bone tumors. The standard treatment is curettage, followed by local adjuvant treatments and bone grafting. The problem is the high recurrence rate. The purpose of this study was to evaluate retrospectively the use of bioactive glass as a filling material in the treatment of aneurysmatic bone cysts in children.
Material and Methods:
A total of 18 consecutive children (mean 11.3 years at surgery; 10 males; 11 lower, 6 upper limb, 1 pelvis; 15 with primary surgery) with histologically proven primary aneurysmal bone cysts operated with curettage and bioactive glass filling between 2008 and 2013 were evaluated after a mean follow-up of 2.0 years (range, 0.7–5.1 years).
Results:
Two (11%) patients showed evidence of aneurysmal bone cyst recurrence and both have been re-operated for recurrence. Bone remodeling was noted in all patients with remaining growth and no growth plate disturbances were recorded. Two patients needed allogeneic blood transfusion. No intraoperative or postoperative complications were recorded.
Conclusion:
We conclude that bioactive glass is a suitable filling material for children with primary aneurysmal bone cyst. Bioactive glass did not affect bone growth and no side effects were reported.
Introduction
Primary aneurysmal bone cyst (ABC) is a rare lesion with an incidence of 1.4 per 105 individuals and represents about 1% of primary bone tumors (1). The standard treatment is to remove the lesion by curettage, followed by local adjuvant treatment and bone grafting. The rate of local recurrence has varied widely (2). Recurrences are more likely in younger children, with open growth plates and within the first 2 years postoperatively (3).
A number of adjunctive therapies are used to decrease the rate of recurrence. The local recurrence can be kept to a minimum if curettage is thorough and is performed with a high-speed burr (3). Phenol is a nonselective cytotoxic agent, which destroys the remaining residual tumor and normal cells (4) and has been used in the treatment of ABCs (5).
Bioactive glasses (BG) are bone bonding, osteoconductive bone substitutes with bone-stimulative and antibacterial and angiogenesis-promoting properties (6). Bioactive glass BG-S53P4 gained European approval as a bone graft substitute in 2006. BG-S53P4 has been reported to be safe and well-tolerated and suitable for benign bone tumor surgery with good long-term results in adults (6). In one case report of a 4-year-old girl with phalangeal ABC, BG-S53P4 did not disturb the growth of bone (7). BGs dissolve slowly and the graft area remodels to bone over time. This phenomenon may be beneficial, especially in the treatment of ABCs, which are prone to recur (7).
The aim of this study was to evaluate BG as a filling material in children’s ABCs. We also aimed to describe the effect of BG on further growth and bone remodeling.
Materials and Methods
After Institutional Review Board and Ethics committee approval, the patients with aneurysmatic bone cyst were identified from three University Central Hospital databases. The medical records and radiographs of children presenting with a bone lesion treated with bioactive glass between 2008 and 2013 (40 cases) were reviewed retrospectively. Other than ABC lesions were excluded. We found 18 patients with histologically proven primary ABCs treated with curettage and bioactive glass filling.
Variables reviewed were date of birth, gender, age at presentation, pre- and postoperative imaging studies, the affected bone, cyst location versus physis, size of cyst, possible pathologic fracture, type and date of surgical procedure, the use of possible preoperative embolization or injection treatment, possible intraoperative adjuvant treatments, histology, recurrence, and other possible complications during treatment.
Preoperative and postoperative radiographs were available for all patients. In all, 17 of 18 patients had magnetic resonance imaging (MRI) of the lesion preoperatively; the remaining patient had computer tomography (CT) imaging. Three patients had both CT and MRI before treatment. Postoperatively, two patients had CT during follow-up and one patient had MRI and one both MRI and CT during follow-up to evaluate possible recurring cyst suspicion on plain radiographs. One of these patients was symptomatic (pain). The volumes of the lesions on MR and CT images were measured using Aycan Workstation OsiriXpro (version 1.04; Wurzburg, Germany) (8). CT and MR images as well as radiographs were analyzed by radiologist with special experience and interest in musculoskeletal radiology. Cysts were classified according to Capanna’s and Enneking’s classification systems (9,10).
Four patients underwent biopsy before surgical treatment. In all, 11 patients had frozen section during surgery before filling the cavity with bioactive glass. In all patients, the final histologic examination confirmed the presence of an ABC.
The operation included curettage of the lesion with high-speed burr and filling the cavity with bioactive glass, BG-S53P4 (BonAlive Biomaterials Ltd). Phenol was used as a cytotoxic agent in 12 patients.
Ethics
The study has Institutional Review Board (5 September 2012) and Ethics committee approval (19 June 2012).
Results
The mean age at the time of surgery was 11.3 years (range, 4–16 years). There were 10 male patients (mean age of 10.8 years; range, 4–16 years) and 8 female patients (mean age, 11.9; range, 7–15 years). The mean follow-up was 2.0 years (range, 0.7–5.1 years) from the date of initial surgery. Eight ABCs were type 1 (44%) in Capanna’s classification system, six were type 2 (33%), and four (22%) were type 3 cysts. Two of the cysts (11%) were inactive in Enneking’s classification system, 12 (66%) were active, and four (22%) were aggressive lesions (Tables 1 and 2).
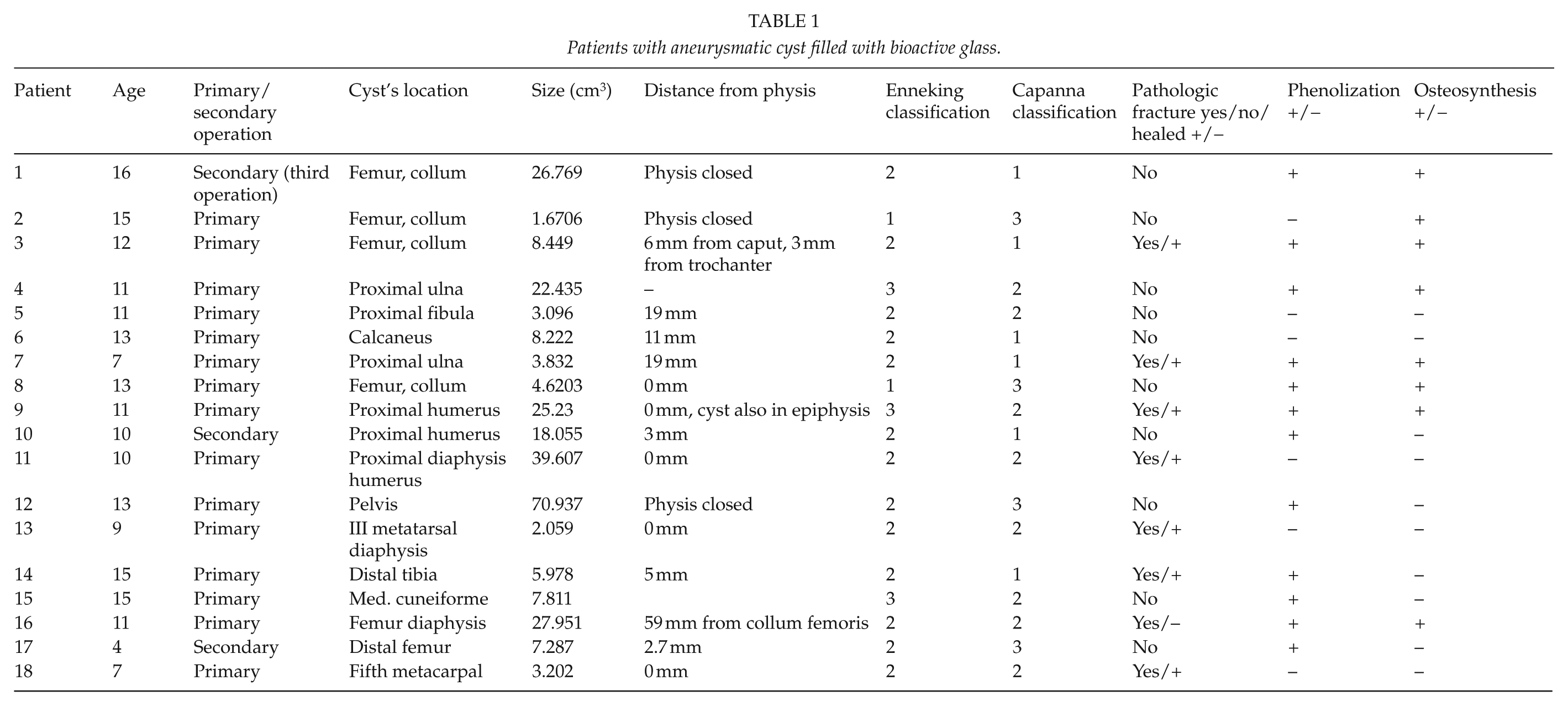
Patients with aneurysmatic cyst filled with bioactive glass.
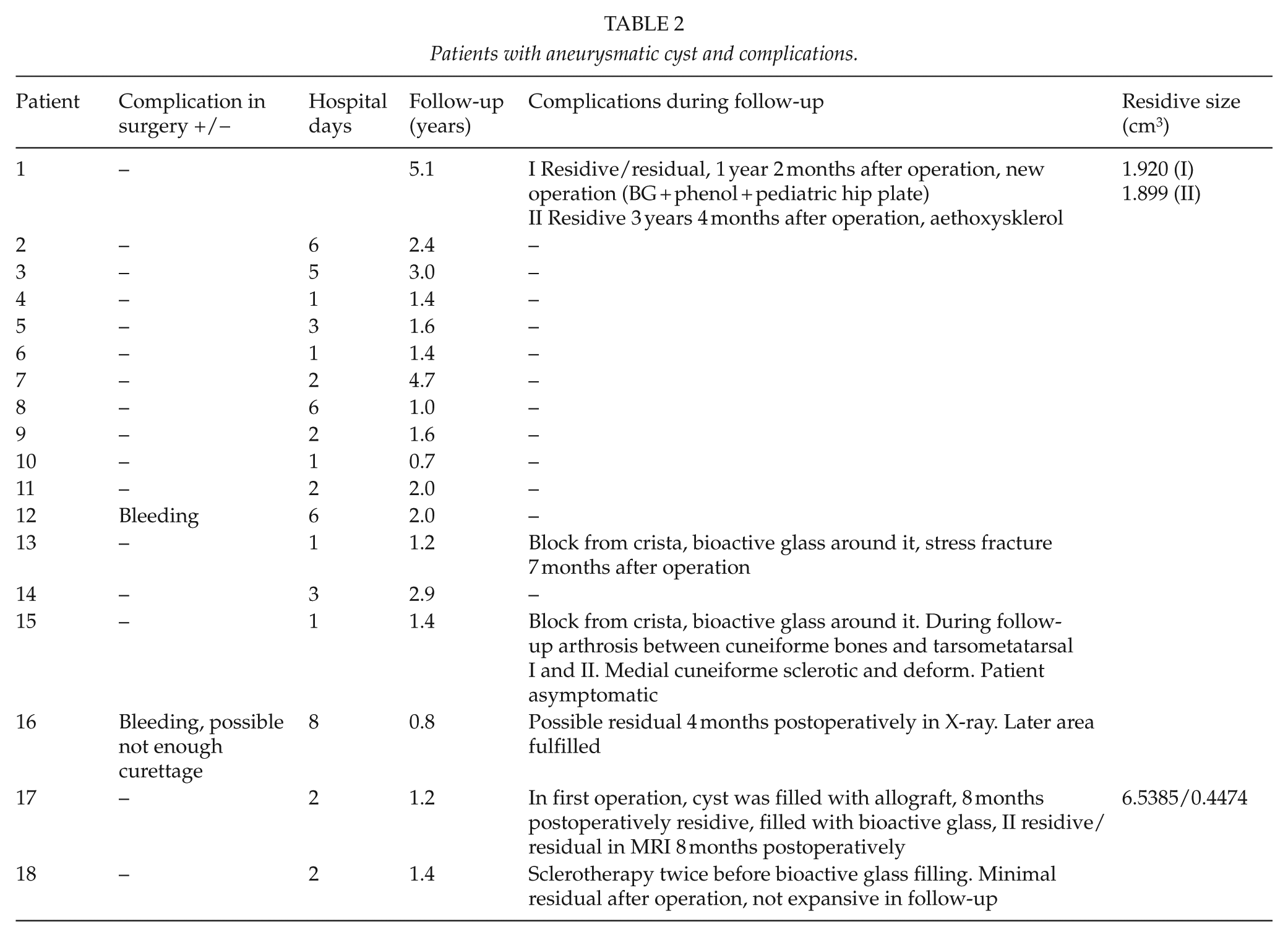
Patients with aneurysmatic cyst and complications.
The locations of the cysts are shown in Table 1. The most commonly involved bone was femur (six cases). One patient had massive aneurysmatic bone cyst of the pelvis (Fig. 1A to C). The lower limbs were involved more frequently (11 cases) than upper limbs (6 cases). Eight patients had a pathologic fracture before operation. Seven of these fractures had healed before surgery using conservative treatment. None of the patients underwent preoperative embolization. One patient had had sclerotherapy twice before operation.
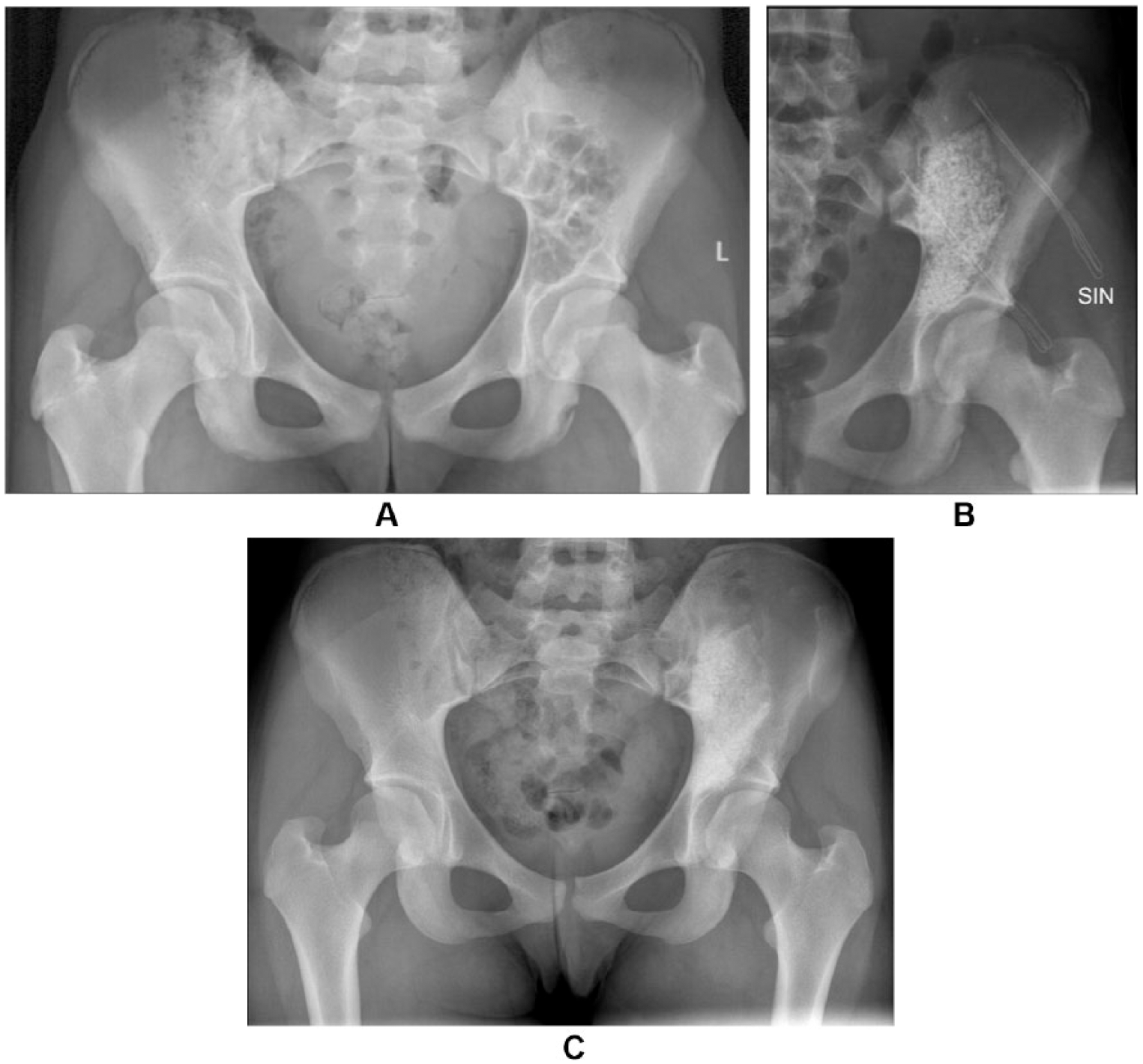
A) Preoperative radiograph from the patient who had massive aneurysmatic bone cyst of the pelvis. B) Immediate radiograph after curettage and bioactive glass filling. C) Three years from the curettage and bioactive glass filling.
Three patients had been operated previously for the cyst. In two patients, the index operation was a second and in one a third. In all three patients, allogeneic bone grafting had been used in previous operations.
Plate fixation was used in eight patients. Phenol was used in 12 patients. Bone autograft was applicated in addition to bioactive glass in one patient with cuneiforme mediale and another with third metatarsal ABC. Two patients were given allogeneic red blood cell transfusion postoperatively. No perioperative complications were recorded.
In the study period, two patients developed a recurrent lesion (Tables 1 and 2):
Patient No. 1 was 16 years at the time of the operation. The index operation was the third for the aneurysmatic bone cyst in the proximal femur. Phenol was used as adjunctive therapy. The presenting symptom was pain. A recurrence was suspected on plain radiographs and confirmed with MR and CT imaging.
Patient No. 17 was a 4-year-old boy who had revision surgery after filling with allograft. MR images confirmed the recurrence. When allograft was used, the curettage was not total because the cyst grew to the distal femur physis.
Both patients had active lesions according to Enneking’s classification system. Patient No. 1 had type 1 lesion in Capanna’s classification system and patient No. 17 had type 3 lesion.
There were no side effects to BG reported in patient’s files. All 14 patients with open growth plates continued to grow normally without any disturbances. Additionally, these patients showed bone remodeling with increased density on radiographs in the operation area. In all patients, some ill-defined glass granules were still visible among new bone at the end of follow-up.
Discussion
ABC’s origin is controversial. Over the past few years, many different genetic translocations have been described in ABC, all resulting in oncogenic activation of the USP6 gene. USP6 is an ubiquitin-specific protease that was identified as an oncogene in transfection experiments with Ewing sarcoma DNA. Recently, structurally similar USP6 fusion genes were identified in both ABC and nodular fasciitis. This suggests that these conditions are clonal neoplastic disorders that may belong to the same biologic spectrum (11).
Because of ABC’s controversial origin, no universal treatment method has been adopted. Opinion varies as to which method should be used, especially in children. Only few pediatric studies exist, the biggest ones being the study of Dormans et al. (5) and Ramirez and Stanton (12). To the best of the authors’ knowledge, there is only one case report using BG to treat a recurrent ABC in a child (7).
En-bloc resection is not a common procedure and must be considered only when there is no other alternative (2). Intralesional excision with bone grafting is usually the standard cure. However, curettage with or without bone grafting has been associated with high recurrence rates (2, 13, 14).
A number of adjunctive therapies are used to decrease the rate of recurrence (5, 15, 16). Varshney et al. (17) analyzed the literature regarding the treatment of ABCs, founding recurrence rate of approximately 29.2% when curettage and bone grafting are used. This decreased to 15.4% using high-speed burr (3, 5, 17).
Sclerotherapy is a newer treatment method. An alcoholic solution of zein (Ethibloc) has been used with good healing rates (18). Polidocanol has been used as a sclerosant with 3% recurrence rate (19). It is reported that repetitive sclerotherapy is safer method than intralesional excision and bone grafting with similar recurrence rates (17). However, there are also problems with this treatment method. Complications (pulmonary embolism, aseptic fistulae, infection, osteomyelitis, and abscess) have been reported in the literature (18, 20). Fatal brain stem and cerebellar infarction have also been described (21). Repetitive injections are usually needed and general anesthesia is required for children (17, 22). Brosjö et al. (22) found no growth disturbances in their series. When there is large ABC in weight-bearing bone, it is impossible to stabilize the bone to reduce the risk of pathologic fracture when using sclerotherapy as a sole treatment method. Therefore, the current authors would not use sclerotherapy as a sole treatment in high risk locations for pathological fractures such as proximal femoral area. In our study, internal fixation was commonly used for large cysts, especially in lower extremities, to prevent early pathologic fractures and to allow early weight bearing and motion.
Selective arterial embolization of the nutrient vessels is an alternative method in difficult locations such as spine and pelvis. Ischemia to neural and vital structures remains the main concern (15). In spine repeated embolization, when feasible, can be considered the first treatment option (23, 24), but this leads into high radiation doses.
Recurrence rates after curettage vary widely from 9% (5) to 71% (25). In a literature review, the recurrence rate of the 690 ABCs treated with curettage with or without bone graft was 31% (2). In our small series of 18 patients treated with bioactive glass, two patients developed recurrence (11%). Both were male and had active and large lesions nearby growth plates of the proximal and distal femur, respectively, and previously surgically treated using allograft bone. Additionally, phenol as an adjuvant had not been used in the first patient. Because of the retrospective nature of this study, there was no routine schema for radiographic controls. The potential recurrences were evaluated from plain radiographs. When recurrence was suspected, MR imaging was done (also CT in one patient). Only four patients had MR or CT imaging postoperatively. Patients with recurrent cysts had active cyst in Enneking’s classification and type 1 and 3 lesions in Capanna’s classification.
In our series, there were no growth arrests when evaluating the growth plate on radiographs. Other studies have reported growth problems (4%/12.5%/14.3%) (15, 17, 26). Physeal growth arrest appears to be related more to the location of the cyst and surgical technique than the filling material itself and the role of filling material in this respect is not clearly known. Juxtaphyseal ABC involvement has been reported to have growth disturbance in 55% following surgical treatment (27, 28). Appositional growth seems not to be affected by filling using bioactive glass (7). All patients in our study showed bone remodeling with dense, more sclerotic appearance in the operation area. However, limitation is that we made remodeling observations only on radiographs which may lead to an impression of a more sclerotic area than it actually is (6).
Bioactive glass proved to be a safe filling material of bone cavity after curettage of primary ABC. Its resistance to mechanical compression might be beneficial in cases of recurrent ABC. These properties may limit the expansion of the lesion as all our recurrences were small. In previous studies, the size of the recurrence has not been studied. Bioactive glass also allowed bone remodeling in all patients with remaining growth and did not disturb further growth even in small children. In conclusion, bioactive glass is a suitable filling material for children with primary ABC. Bioactive glass did not affect bone growth and no side effects were reported. The recurrence rate of primary ABC after operation appears acceptable.
Footnotes
Declaration of Conflicting Interests
I.H. has received research grants from Medtronic, Bonalive, and Baxter Healthcare Corporation and is a consultant in Medtronic. Other authors received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
The study has Institutional Review Board (5 September 2012) and Ethics committee approval (19 June 2012).
Funding
J.S. has been supported by the Turku University Central Hospital Foundation and the Finnish Paediatric Research Foundation. J.S. is a Research Fellow, O.P. is a Member, and I.H. is a leader of the Finnish Pediatric Orthopaedic Study Group, which has received funding from the Finnish Pediatric Research Foundation, Baxter, Bonalive, and Medtronic International.
