Abstract
Introduction
Aneurysmal bone cysts (ABC) are benign yet locally aggressive bone tumours first described in 1942 by Jaffe and Lichtenstein 1 with potential for local recurrence. 2 They are relatively rare tumours, representing 1%-2% of primary bone and 15% of primary spine tumours. ABC can involve any skeletal site but most commonly manifests within the metaphysis of long bones and the spine.2,3 These tumours commonly develop during the first two decades of life,2,4 presenting as swelling, pain, and pathologic fractures. When located in the spine, they can lead to neurologic deficits by compressing the nerve roots or spinal cord.3,5 In approximately 70% of cases, ABCs may manifest as primary tumours. In comparison, in 30% of cases, they appear as secondary tumours in conjunction with conditions such as osteoblastoma, giant cell tumour of bone (GCTB), chondroblastoma, telangiectatic osteosarcoma, fibrous dysplasia, and non-ossifying fibromas.2–4
ABC are osteolytic tumours, blood-filled cavities separated by fibrous septate.2,4 The cellular component of ABC comprises osteoclast-like giant cells expressing receptor activators of nuclear factor kappa B (RANK) and neoplastic stromal cells expressing RANK ligand (RANKL). 3 On a molecular level, 65%-70% of primary ABC demonstrate translocation t (16;17) (q22; p13). This translocation results in the fusion of the promoter region of the osteoblast cadherin 11 gene (CDH11) on chromosome 16q22 with the ubiquitin protease gene (USP6) (also known as TRE2 or TRE17) on chromosome 17p13. This suggests that most primary ABCs are associated with the upregulation of USP6, triggered by the robust activity of the CDH11 promoter. 6 TRE17 induces the production of matrix metalloproteinase (MMP)-9 and MMP-10, contributing to ABC pathogenesis. 7 In addition, TRE17 inhibits osteoblastic maturation, contributing to bone loss observed in ABC. 8
Treatment options for primary ABC include both non-surgical and surgical approaches. Surgical options encompass curettage ± cavity filling and surgical resection.4, 9 Intralesional curettage carries a higher risk of local recurrence, usually within 6 to 12 months post-surgery.2,4,9 Although en bloc resection is associated with a low recurrence rate, its practicality may be impeded by the lesion’s proximity to critical neural and vascular structures.4,9 Less invasive interventions such as selective arterial embolization (SAE), sclerotherapy, intralesional injection, and radiation have shown favourable outcomes but some of these may be associated with significant morbidities.4,5
Denosumab, a human monoclonal antibody, binds to the RANKL. 10 RANKL, which activates the RANK receptor of osteoclasts, induces bone resorption. Thus, denosumab hinders RANK-RANKL interaction, suppressing osteoclast activity and replacing tumour stroma with densely new woven bone. 10 The RANKL-RANK interaction has been associated with the pathogenesis of GCTB, where its osteoclast-like giant cells prominently express RANK. 10 Considering the similarly elevated expression of RANK in the giant cells of ABCs, along with the heightened expression of RANKL in its’ neoplastic stromal cells, the potential application of denosumab therapy has been expanded to include ABCs. 9
Denosumab is FDA-approved for multiple indications, 11 including the reduction of skeletal-related events (SREs) in patients with multiple myeloma and those with bone metastases from solid tumours. It is also approved for treating adult patients and skeletally mature adolescents with unresectable giant cell tumours of bone, or in cases where surgical resection would likely result in significant morbidity. Furthermore, denosumab is indicated for the treatment of osteoporosis in postmenopausal women and for glucocorticoid-induced osteoporosis. However, denosumab is not currently FDA-approved for ABCs due to the limited available literature. 11
Thus, in this scoping review, we systematically examined the existing literature on the utilisation of denosumab in ABCs. We aim to identify the types of evidence pertaining to denosumab’s application in ABCs, focusing on its role, treatment outcomes, and adverse effects. While two previous reviews, a narrative review 12 and a systematic review, 5 have addressed the management of ABC with denosumab, our scoping review offers the most up-to-date and comprehensive analysis to date. With 29 articles (involving 67 patients), our review offers a broader perspective and captures the latest data, providing a more current understanding of ABC management with denosumab.
Materials and methods
Search strategies
This scoping review employed a rigorous search strategy aligned with the Preferred Reporting Items for Systematic Reviews and Meta-Analysis Extension for Scoping Reviews (PRISMA-ScR) checklist. 13 The search was systematically conducted across five electronic databases: Scopus, Ovid MEDLINE, Ovid Embase, PubMed, and Web of Science (WoS), from inception to 31st December 2023. To ensure comprehensive coverage of relevant literature, references from other published reviews and related published papers were meticulously reviewed to avoid missing any pertinent articles that may have been previously identified by other researchers. The Medical Subject Headings (MeSH) and keywords used were “Aneurysmal bone cyst” and “Denosumab”, in combination with Boolean operators AND/OR to refine and enhance the search.
Study selection and screening
All studies identified from the database searches were exported to a reference management software (EndNote 20), wherein duplicates were removed through the ‘Find Duplicates’ function. Two reviewers (VS, VAS) independently screened the titles and abstracts of all articles to remove review articles, conference abstracts, and studies that did not feature either ABC cases, denosumab use or both. Full-text screening of potentially pertinent studies was undertaken to evaluate adherence to the inclusion criteria, encompassing all peer-reviewed journal articles that included patients with primary ABCs treated with denosumab. Due to the scarcity of published literature on this subject, this review incorporated even case reports and case series with small sample sizes that contained the necessary parameters for analysis. Exclusion criteria involved secondary aneurysmal bone cysts, review articles, commentaries, editorials, conference abstracts, non-full text publications, non-English publications, animal and in-vitro models studies, and studies in which outcomes were not specified. Any discrepancies between the two reviewers were resolved by a third reviewer (AP). This systematic process identified the studies for final inclusion.
Data extraction and charting
Two investigators (VS, VAS) independently extracted and documented the data from each included study into a Microsoft Excel spreadsheet to minimise reporting bias. In instances of discrepancies, a third investigator (AP) intervened to resolve inconsistencies before the final findings from the specific study were recorded for further analysis.
The extracted data include the author’s name, year of publication, study type, sample size, age, gender, ABC site, presenting symptoms, interventions before denosumab (if any) and their corresponding outcome. Upon initiation of denosumab, the following data was extracted: dosage and duration of therapy, its role (neoadjuvant, primary treatment, second-line treatment or adjuvant role), clinical and radiological response, recurrence post-denosumab, adverse effect and duration of follow-up. In cases of tumour recurrences following denosumab treatment, additional data extracted included the timing of recurrence in relation to denosumab administration and the approach to managing recurrences. Similar data was gathered for patients who experienced hypercalcaemia and hypocalcaemia following denosumab treatment.
Synthesizing, analysing and results reporting
The information gathered was subsequently compiled, synthesized, and analysed. The results were then presented using tables. This review provided both quantitative and qualitative assessments of the study findings. Because of the highly heterogeneous data and diversity of research focus from included studies, which comprised case reports and case series, statistical analysis was not feasible. The Statistical Package for Social Sciences (SPSS, Chicago, IL, USA Version 20.0) was employed to create a database to analyse quantitative data.
Results
Study characteristics and demographics
The systematic searches across five databases initially revealed 390 peer-reviewed articles, as depicted in Figure 1. After removing 242 duplicates, we screened the titles and abstracts of 148 articles, subsequently excluding an additional 114 articles. Finally, a total of 34 articles underwent full-text review, of which 29 fulfilled the inclusion criteria and were ultimately included in the final review.14–42 No non-English language publications were found. PRISMA study selection flowchart showing the total of publications removed at various stages and the reasons for the exclusions.
Patients’ demography, clinical characteristics, denosumab treatment & outcome.
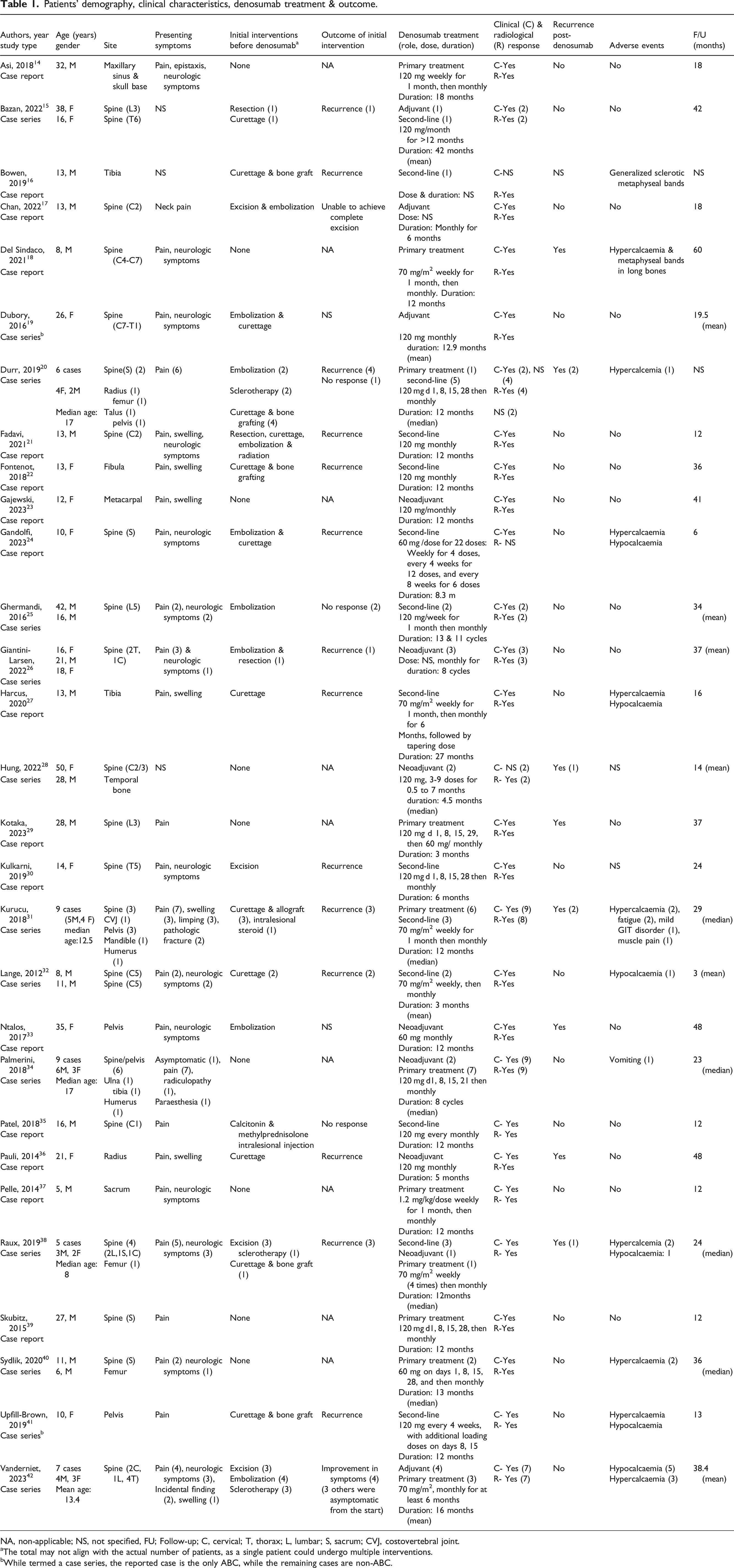
NA, non-applicable; NS, not specified, FU; Follow-up; C, cervical; T, thorax; L, lumbar; S, sacrum; CVJ, costovertebral joint.
aThe total may not align with the actual number of patients, as a single patient could undergo multiple interventions.
bWhile termed a case series, the reported case is the only ABC, while the remaining cases are non-ABC.
Denosumab therapy
Denosumab therapy: Treatment categories and applications.

In 11 cases from seven studies,23,26,28,33,34,36,38 denosumab functioned as a neoadjuvant therapy. These studies showed that neoadjuvant denosumab led to reduced tumour size, increased calcification, lesion devascularization, and better delineation of the tumours by a bony rim, enhancing the safety, feasibility, and completeness of the subsequent resection.
In 24 cases involving 14 studies,15,16,20–22,24,25,27,30–32,35,38,41 denosumab was administered as a second-line therapy either because of tumour recurrence, failed initial treatment or poor clinical/radiological response to initial interventions. These cases responded positively to subsequent denosumab therapy. Finally, in seven cases,15,17,19,42 denosumab served as an adjunct to other treatment modalities.
Amongst the other interventions used for the second-line and adjuvant groups, curettage (n = 18) was the most frequently employed, followed by arterial embolization (n = 12), tumour resection/excision (n = 10), and sclerotherapy (n = 6). These interventions were administered either alone or in different combinations. Among the combined approaches, the most frequent was the combination of surgery (curettage/excision) and selective embolization (n = 7).
Regarding denosumab administration, Table 1 reveals considerable diversity in the dosage, frequency, and duration of denosumab administration. As shown in Tables 1 and 2, all 60 patients with clinical responses reported, exhibited positive outcomes, primarily in pain and neurological symptom improvement. Likewise, 98% of the 64 patients with documented radiological outcomes demonstrated improvement after denosumab treatment.
Tumour recurrence
Recurrence post-denosumab and its management.

Six (of the 10 recurrences) occurred after denosumab discontinuation,18,20,29,31,38 at a mean duration of 11.2 ± SD 5.1 months post-cessation. The mean duration of denosumab administration for these six cases was 12.8 months ±SD 6.7 (range 3-24 months). Denosumab served as the primary treatment in five, while in one case, denosumab was administered as a second-line treatment. Of these, four recurrences occurred within 12 months, and two more within 12 to 24 months (range 3-17 months). Following the recurrences, denosumab was reintroduced in five out of the six cases, whereas in one patient, recurrence was treated with sclerotherapy.
Another three (of the 10) recurrences occurred following surgery after neoadjuvant denosumab therapy,28,33,36,with an average duration of 11 months post-surgery (range: 6-19 months). In two of these cases, recurrence was treated by resuming denosumab, while the third patient’s recurrence was managed using a combination of denosumab and curettage.
The tenth recurrence occurred in a 30-year-old female who experienced recurrence 8 months into denosumab therapy while treatment was ongoing. 20 Surgery was the primary intervention for the initial recurrence, and the commencement of denosumab treatment was postponed due to the patient’s pregnancy. Following a second recurrence 18 months post-surgery, denosumab was started; however, it was discontinued again due to the onset of her second pregnancy. A third recurrence occurred post-pregnancy, prompting denosumab resumption. In addition to the 10 recurrences outlined above, in a 10-year-old child, the tumour persisted in advancing despite undergoing 6 months of denosumab treatment. 31 Subsequently, denosumab was discontinued, and she underwent surgery. In total, less than half of the studies (21 studies comprising 31 cases) had a median/mean follow-up duration of more than 2 years.
Adverse events
Hypocalcaemia as an adverse effect of denosumab & its treatment.

Hypercalcaemia as adverse effect of denosumab & its treatment.
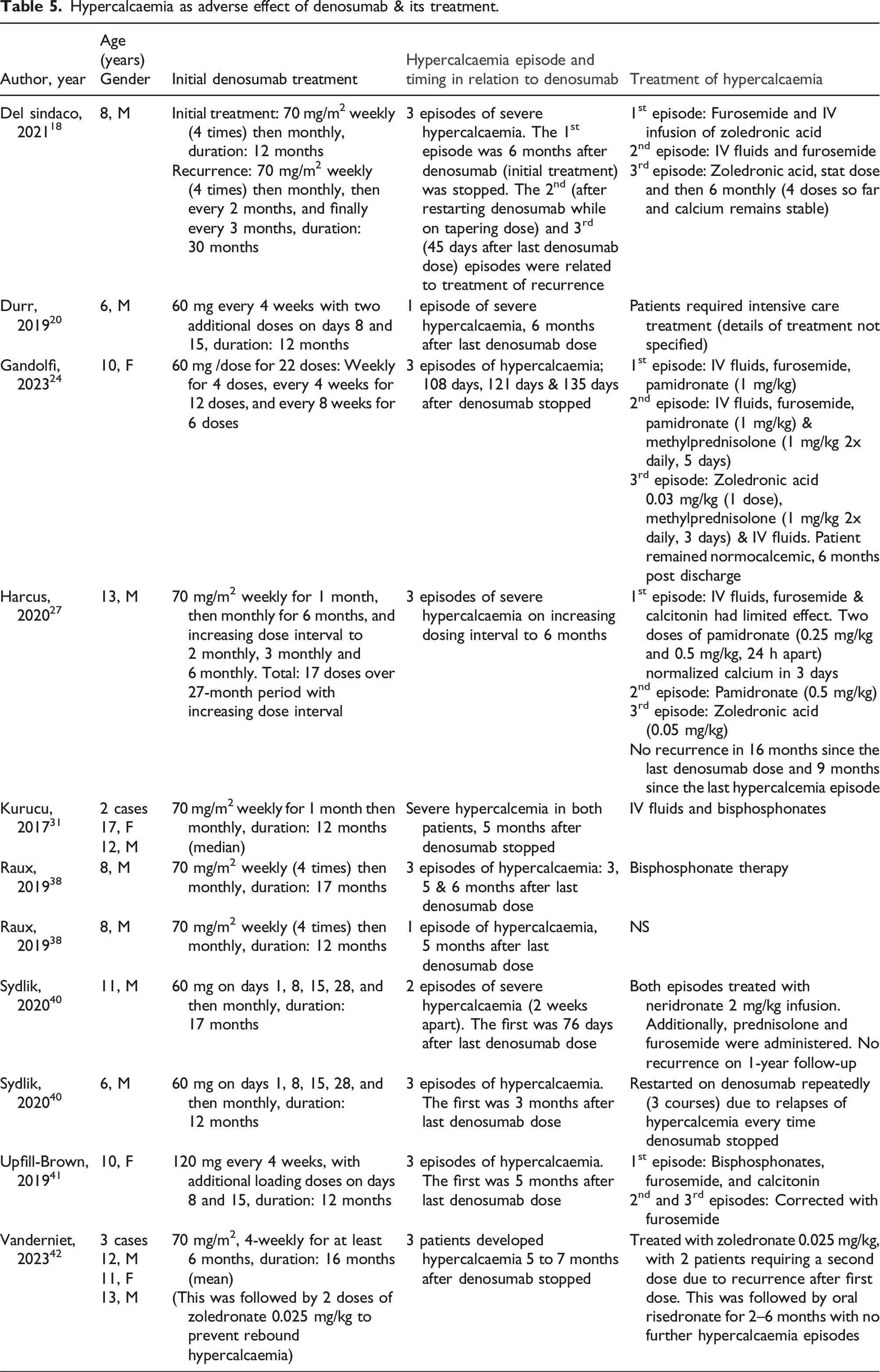
As shown in Table 5, bisphosphonates (single or repeated dose), which included zoledronate, pamidronate, neridronate, and risedronate, were the most used therapy (n = 11) in controlling hypercalcemia, even in patients experiencing multiple episodes of hypercalcemia. Other treatment approaches involved intravenous hydration and the administration of furosemide (n = 5) and the addition of calcitonin (n = 2) and/or prednisolone (n = 2) in a few cases. For one patient 40 who developed three episodes of hypercalcemia, the strategy involved repeatedly reinstating denosumab to manage hypercalcemia after each recurrence. Notably, Vanderniet et al. 42 documented three cases wherein symptomatic hypercalcemia occurred despite the prophylactic administration of two doses of zoledronate following the last denosumab dose.
Other reported adverse events (Table 1) included sclerotic metaphyseal bands in two children aged 8 and 13 years. Additional minor side effects included fatigue (n = 2), mild gastrointestinal issues (n = 1), muscle pain (n = 1), and vomiting (n = 1).
Discussion
Our scoping review presents the most current and comprehensive evaluation of denosumab use in managing ABC. The results indicate that denosumab shows promise as an effective treatment for ABC, particularly in high-risk locations like the spine and pelvis. This study highlights its diverse applications, including use as a primary treatment, neoadjuvant therapy, second-line intervention after inadequate initial treatments, and adjunct therapy; with favourable clinical and radiological outcomes observed in the patients. However, the occurrence of tumour recurrences and adverse effects such as hypocalcaemia and rebound hypercalcemia, particularly in paediatrics patients, highlights the need for careful patient monitoring and extended follow-ups. Traditionally, the standard treatment for ABC has been curettage and filling the cavity with bone graft or polymethylmethacrylate remains the standard therapeutic approach.2,43 However, it carries a 20%-30% risk of local recurrence.43,44 There is an emerging trend toward minimally invasive procedures such as selective arterial embolization, sclerotherapy, and calcitonin ±corticosteroids injections.31,43,44 Denosumab, with its expanding role, presents a relatively new alternative, particularly in surgically challenging sites such as the spine or sacrum. 43
Our scoping review found only case reports and case series, with no published clinical trials examining the use of denosumab in ABC. The majority (76.1%) of patients were aged ≤18 years, consistent with the age distribution observed in previous demographics of ABC. 2 However, there existed variations in tumour location in this review compared to the typical demography of ABC cases. While Mankin et al. 2 identified the tibia, femur and fibula as the most prevalent sites, our review revealed that the spine (62.7%) was the most common site, followed by the pelvis (10.4%). There is an obvious selection bias in our review and the observed difference likely arises from the challenges encountered in achieving curative resections of axial ABCs, where accessibility and anatomical constraints from neighbouring critical structures often hinder surgical interventions, thus favouring denosumab treatment as a preferable option.
In most cases in this review, denosumab was administered as the primary treatment (n = 25, 37.5%). In seven of the nine cases outlined by Palmerini et al., denosumab served as the primary treatment for inoperable tumours. 34 All seven cases showed clinical and radiological improvement with bone formation, ultimately obviating the need for surgical intervention. Vanderniet et al. 42 detailed three patients who received denosumab as the primary intervention due to the asymptomatic nature of their lesions, resulting in ossification and stabilization of the ABC’s size. In another case series, Kurucu et al. detailed nine patients, of which denosumab was used as primary treatment in six cases for tumours involving the vertebrae and pelvis. 31 In all cases, there was a regression of clinical symptoms within 3 months, with radiological improvement. In 35.8% (n = 24) of cases, denosumab was administered as second-line treatment after recurrence or inadequate responses following initial interventions, which included intralesional curettage, arterial embolization, excision, and sclerotherapy, either individually or in combination. In another 11 patients (16.4%), denosumab was utilized as a neoadjuvant therapy to enhance the ease and feasibility of tumour resection. In a case series involving three patients with unresectable spinal ABC, neoadjuvant denosumab was used to calcify, devascularize and decrease the size of these lesions, facilitating a safer and more thorough resection process. 26 Following denosumab administration in a 21-year-old woman with radius ABC; the tumour became delineated by a bony rim, facilitating resection and limb-preserving surgery. 36 In another case involving metacarpal ABC, denosumab reduced tumour size and induced calcification for successful resection and reconstruction. 23 Likewise, Raux et al. 38 reported a 14-year-old female with a large sacral ABC extending into the spinal canal. Neoadjuvant denosumab resulted in mineralization and cortical thickening, enabling surgical decompression and curettage.
It is important to highlight that irrespective of the denosumab regimen used, there was consistent clinical improvement, with all cases experiencing relief from pain and neurological symptoms. This will likely contribute to improvement in their functional ability and quality of life. In addition, where data was available, 98% of cases showed favourable radiological assessment after denosumab administration. Our scoping review demonstrated 10 recurrences (15%) after denosumab therapy: six occurring post-denosumab cessation, three following surgeries post-neoadjuvant denosumab, and one during ongoing denosumab treatment. Although small case numbers present challenges in drawing definitive conclusions, our data suggests that most recurrences arose within 2 years post-denosumab cessation. However, the majority of the studies included in this review indicated an average follow-up period of 2 years or less, potentially leading to an underestimation of the actual recurrence rate. Thus, longer follow-ups may provide a more accurate understanding of recurrence, guiding more informed treatment strategies. Notably, Mak et al. observed that while denosumab effectively suppressed the RANKL expression on osteoclasts, the proliferation of neoplastic stromal cells persisted unabated, suggesting their resilience in driving recurrence. 45
Treatment approaches for ABC have demonstrated recurrence rates ranging from 5% to 22%. For instance, Mankin et al. reported a recurrence rate of 22% among 121 ABC patients treated with curettage ± grafting. In comparison, patients who underwent resection exhibited a lower recurrence rate of 5%. 2 In a meta-analysis by Cruz et al., the recurrence risk after selective arterial embolization and sclerotherapies was 19% and 6%, respectively. 46
Our literature review identified 14 cases of hypercalcemia, 10 cases of hypocalcaemia, and two cases of sclerotic metaphyseal bands. There are several reported cases of hypercalcemia arising after discontinuation of denosumab treatment used for diseases such as GCTB, fibrous dysplasia, juvenile Paget’s disease, osteoporosis, osteogenesis imperfecta type VI.18,47 Of the 14 instances of hypercalcemia, 13 occurred between 2.5 and 6 months following denosumab cessation, while one occurred while increasing the dosing interval. Although adult patients accounted for 24% of the patients included in this review, none of the adults experienced hypercalcaemia. All reported cases were confined to the paediatric age range, with a notable male predominance of 71%. Likewise, a systematic review encompassing 49 instances of rebound hypercalcemia post-denosumab discontinuation revealed that 82% of cases occurred in skeletally immature individuals. 47 Additionally, the duration between denosumab cessation and the onset of hypercalcemia was notably shorter in this subset of patients. Consistent with our finding, a significantly higher number of males among the skeletally immature patients presented with hypercalcemia. 47 This trend could be linked to higher bone mass typically found in males, resulting in increased calcium storage while on denosumab during the growth phase.47,48 Hypercalcemia following denosumab withdrawal is likely due to the rapid recovery of osteoclastic activity, leading to accelerated bone resorption and calcium release into the bloodstream. 47 Skeletally immature children, with their inherently higher baseline bone turnover, are likely to be more susceptible to rebound osteoclastic activity, consequently resulting in a higher incidence of rebound hypercalcemia.47,49
While there are currently no standardized treatment guidelines, addressing denosumab-induced rebound hypercalcemia entails a multifaceted approach. In this review, a range of treatments, including intravenous hydration, diuretics, calcitonin, corticosteroids, bisphosphonates, and/or repeated dosages of denosumab, were utilized. The prophylactic administration of bisphosphonates is often employed to avert the recurrence of rebound hypercalcemia. Bisphosphonates integrate into the bone matrix, thereby modulating osteoclast activity upon discontinuing denosumab treatment. Nevertheless, rebound hypercalcemia may still occur despite this preventative measure. 42 Hypercalcemia may also be averted by gradually tapering the dosage of denosumab instead of abruptly stopping it.18,20,27,31 Currently, there are no established guidelines for timing and frequency of monitoring during denosumab therapy, 24 however, regular follow-ups are recommended upon starting and treatment withdrawal based on literature evidence. Additionally, it is important to educate patients and their families about the symptoms of hypercalcemia to ensure prompt identification and appropriate treatment. 24
This review also revealed that 10 paediatric patients (aged 8 to 14 years) experienced hypocalcaemia during the initial phases of denosumab therapy despite eight of them being administered prophylactic calcium and cholecalciferol. Treatment consisted of either escalating the dosage or commencing calcium supplements. In two instances, hypocalcaemia spontaneously resolved. Other reported adverse events included the formation of sclerotic metaphyseal bands in two children. While osteonecrosis of the jaw and atypical femoral has been documented,34,49 our review did not identify any such complications. However, mild symptoms like fatigue, minor gastrointestinal discomfort, muscle pain, and vomiting were reported.
Concerns have been raised regarding the potential of denosumab to disturb skeletal development in patients whose growth plates have yet to close. This includes disruptions in linear growth, dental development, and ossification of growth plates. 20 Despite this, our review found no evidence of impaired linear growth. However, there remains a paucity of data regarding the effects of denosumab on growth plates and linear growth, an area that needs further exploration. 31 In a histopathological study, there was persistent epiphyseal activity throughout and after denosumab therapy, coupled with the reversal of bone turnover suppression upon treatment cessation, implying denosumab did not induce substantial adverse effects on growth. 50 Considering that most cases in our review were followed up for less than 2 years, conducting longer-term follow-up to detect such adverse effects would be advisable.
This scoping review had several limitations. Firstly, the studies primarily consisted of case reports and case series. Notably, there have been no clinical trials investigating the relationship between denosumab and ABC thus far. Thus, the level of evidence-based substantiation was not high, necessitating cautious interpretation of any resulting conclusions. The data from the various studies exhibited considerable heterogeneity, including variations in the duration and total doses of denosumab, as well as the different approaches to its administration. Thus, conducting statistical analysis or meta-analysis proved unfeasible. Instead, we provided quantitative and descriptive evidence of the data.
The strengths of this review included systematic database search and meticulous data extraction, with clearly defined inclusion and exclusion criteria. Although a previous systematic review on denosumab use in ABC was published, 5 it did not categorize denosumab therapy based on treatment context—primary, neoadjuvant, second-line, or adjuvant. Our review introduces this framework, offering deeper insights into the rationale behind initiating denosumab in each category. This distinction is important for understanding how denosumab’s role varies with different treatment aims, which is essential for individualized care. For instance, denosumab is utilized in neoadjuvant therapy to shrink the lesion prior to surgery, whereas in second-line therapy, it is employed to address tumour recurrence or manage refractory cases. This categorization enhances our understanding of how denosumab can be tailored to distinct clinical scenarios, which is especially important in relatively rare conditions like ABC, where appropriate treatment plans can significantly influence outcomes. Additionally, while the previous systematic review did address side effects of denosumab such as hypercalcemia, it lacked details on the timing and management of these episodes. Our review fills this gap by offering a comprehensive analysis of hypercalcemia, detailing its frequency, timing relative to denosumab administration, and the interventions used to manage it. This provides clinicians insights into managing such complications. Furthermore, since the earlier review, 22 additional cases of ABC treated with denosumab have been reported over the last 4 years. Thus, our review represents the most comprehensive and up-to-date synthesis, incorporating the largest pool of studies available. It offers new insights into the complex relationship between denosumab and ABC, while addressing the gaps identified in previous reviews.
Conclusions
In conclusion, this scoping review highlights the effectiveness of denosumab in managing aneurysmal bone cysts, suggesting notable improvements in both clinical and radiological outcomes. Denosumab might be considered a treatment option, particularly for cases involving spinal and pelvic tumours with significant surgical risks and unresectable tumours. Other potential uses include serving as second-line therapy in cases of recurrence or failed initial intervention or as neoadjuvant therapy to enhance the feasibility of resection. A notable concern arises regarding the possibility of tumour recurrence following the cessation of denosumab, with reported recurrence occurring months after treatment discontinuation. Hence, while denosumab demonstrates promising efficacy, it necessitates extended monitoring, particularly given that the follow-up duration in most cases in this review was limited to 2 years or less. Furthermore, rebound hypercalcemia upon discontinuation or tapering of denosumab therapy remains a concern, especially in paediatric patients, suggesting the need for prophylactic measures and consistent monitoring to mitigate this risk.
Moving forward, there is a need to gain deeper insights into the effectiveness and potential long-term side effects of denosumab in the treatment of ABC. While the prevalence of ABC is relatively low, which limits the immediate pool of patients for prospective trials, there remains a critical need to explore potential use of denosumab amongst ABC patients. Given the low prevalence of ABC, conducting a prospective clinical trial may present logistical challenges; however, the feasibility could be enhanced through multi-centre collaboration to pool patients across regions. In parallel, retrospective cohort studies utilizing existing clinical databases are a valuable strategy to gather more robust preliminary data, serving as a foundational step for future trial planning.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
