Abstract
Background and Aims:
Preoperative magnetic resonance imaging has become an important complementary imaging technique in patients with breast cancer, providing additional information for preoperative local staging. Magnetic resonance imaging is recommended selectively in lobular breast cancer and in patients with dense breast tissue in the case when mammography and ultrasound fail to fully evaluate the lesion, but the routine use of magnetic resonance imaging in all patients with invasive ductal carcinoma is controversial. The purpose of this randomized study was to investigate the diagnostic value of preoperative magnetic resonance imaging and its impact on short-term surgical outcome in newly diagnosed unifocal stage I invasive ductal carcinoma.
Material and Methods:
A total of 100 patients were randomized to either receive preoperative breast magnetic resonance imaging or to be scheduled directly to operation without magnetic resonance imaging on a 1:1 basis. There were 50 patients in both study arms.
Results:
In 14 patients (28%), breast magnetic resonance imaging detected an additional finding and seven of them were found to be malignant. Six additional cancer foci were found in the ipsilateral breast and one in the contralateral breast. Magnetic resonance imaging findings caused a change in planned surgical management in 10 patients (20%). Mastectomy was performed in six patients (12%) in the magnetic resonance imaging group and in two patients (4%) in the control group (p = 0.140). The breast reoperation rate was 14% in the magnetic resonance imaging group and 24% in the control group (p = 0.202). The mean interval between referral and first surgical procedure was 34 days in the magnetic resonance imaging group and 21 days in the control group (p < 0.001).
Conclusion:
Preoperative magnetic resonance imaging may be beneficial for some patients with early-stage invasive ductal carcinoma, but its routine use is not recommended without specific indications.
Introduction
Preoperative breast magnetic resonance imaging (MRI) is increasingly used as a complementary imaging technique in patients newly diagnosed with breast cancer. MRI is expected to improve diagnostic accuracy in terms of local staging, providing additional information facilitating the planning of surgery and reducing reoperations and local recurrences of breast cancer.
The diagnostic accuracy of breast MRI has been well documented in several cohort studies and meta-analyses (1,2). MRI is more sensitive than mammography or ultrasonography (US) in detecting malignant or suspicious lesions of the breast (3). MRI is able to detect otherwise occult additional tumor foci in both breasts, leading to altered surgical treatment in 18% of women with breast cancer (4).
The role of MRI has however been a subject of debate, and current evidence does not support the routine use of preoperative breast MRI in all patients with primary breast cancer (5). MRI is recommended to be used selectively in patients with invasive lobular cancer, occult primary breast cancer, and dense breast tissue in the case when interpretative difficulties or controversial findings in conventional imaging are present (6).
The most common type of invasive breast carcinoma is invasive breast carcinoma of no special type (NST), commonly known as invasive ductal carcinoma, comprising 60%–80% of all breast cancer tumors (7). In most patients with clinically local, early-stage invasive ductal breast cancer, the prognosis is favorable after breast conserving surgery, adjuvant chemotherapy and radiotherapy. However, in these patients, there might be demand for improved preoperative local staging due to the heterogeneity and the multifocal nature of the disease.
The purpose of this study was to evaluate the short-term clinical outcome and the added value of breast MRI in patients with early-stage ductal breast cancer in comparison with control patients undergoing standard preoperative triple assessment (clinical examination, mammography/US, and histopathological examination of core needle biopsy) alone without properative MRI.
Materials and Methods
In the Department of Plastic and General Surgery, Turku University Hospital, Turku, Finland, on average, 450 patients with primary breast cancer are undergoing surgeries annually. The catchment population is 470,000 residents within the hospital district. The patients are referred to surgery from breast screening units or general practitioners. Turku University Hospital is the only hospital performing breast cancer operations within the area. Mammography, US, and core needle biopsy are needed in all patients. Two standard mammography views per breast are needed. US axillary staging is also needed and documented. The findings of mammography and US are reviewed by a breast radiologist, and if the evaluation is sufficient, no additional radiological examinations are needed. The treatment of choice in stage I breast cancer is breast conserving surgery (lumpectomy with histopathologically clear resection margins), sentinel node biopsy (SNB), and postoperative breast radiotherapy. Oncoplastic surgical techniques or mastectomy are considered in stage I breast cancer, if the lesion is multifocal/-centric or locally extensive. Preoperative breast MRI is not used routinely in this patient group. Adjuvant chemotherapy, targeted therapy, or hormonal therapy is considered individually in each case.
Patient Selection and Randomization
In this prospective randomized trial, consecutive women aged 35 years or older with newly diagnosed unilateral and clinically unifocal stage I (unifocal tumor ⩽ 20 mm in greatest dimension based on palpation, mammography, or US before surgery) invasive ductal carcinoma were enrolled in the study at the Department of Plastic and General Surgery, Turku University Hospital, Turku, Finland. A total of 100 patients were randomized to either receive preoperative breast MRI (MRI arm) or to be scheduled directly to operation without MRI (control arm) on a 1:1 basis. The first patient was enrolled in February 2011, and the last patient in February 2013. Exclusion criteria were previous breast cancer, any previous breast surgery, bilateral breast cancer, pregnancy, lactation, or any known contraindication to MRI (severe kidney insufficiency with glomerular filtration rate < 30 mL/min/1.73 m2, ferromagnetic or otherwise MRI-incompatible implants or devices, and claustrophobia). Also, patients with breast parenchymal pattern DY were excluded (according to the classification of Wolfe’s parenchymal pattern) because MRI is performed frequently in patients with very dense breasts in the case when mammography and ultrasound fail to fully evaluate the lesion. Eligible patients with confirmed breast cancer diagnosis were invited to participate in the study by a consultant breast surgeon during their first preoperative outpatient visit. The randomization process was implemented by one breast surgeon (author N.B.). A total of 43 patients refused to participate. After discussion and decision to participate, all included patients provided written informed consent. The allocation process was manual. After the decision of participation, a folded paper note was blindly picked up and the study arm was determined according to “MRI” or “CONTROL.” The trial was approved by the Research Ethics Committee of the Hospital District of Southwest Finland.
Radiology
A standardized MRI protocol was performed on a Siemens Avanto Magnetom 1.5 T MRI scanner (Siemens Medical Imaging, Erlangen, Germany) with bilateral four-channel breast array coil and patient in prone position. Coil configuration allowed the use of Generalized Autocalibrating Partially Parallel Acquisition (GRAPPA) technique to accelerate acquisition. Technique was applied in three-dimensional (3D) sequences using acceleration factor of 2. All patients were imaged using axial T2-weighted fat-suppressed Short Tau Inversion Recovery (STIR) sequence with repetition time (TR) of 13,240 ms, inversion time (TI) of 130 ms, and echo time (TE) of 77 ms. The field of view (FOV) was 360 × 360 mm2, the voxel size 0.9 × 0.7 × 3.0 mm3, the number of slices 39, and the acquisition time 6:52. 3D T1-weighted gradient echo sequence with fat suppression was performed before contrast agent administration using TR of 28 ms, TE of 4.7 ms, flip angle of 20°, FOV of 360 × 360 mm2, the number of slices 160, and nearly isotropic voxel size of 1.1 × 0.8 × 1.0 mm3. Acquisition time was 5:42 and this same sequence was repeated after dynamic contrast enhancement in static phase. The dynamic sequence was 3D T1-weighted gradient echo sequence without fat suppression with TR of 8.1 ms, TE of 4.7 ms, flip angle of 25°, and voxel size of 1.1 × 0.8 × 1.0 mm3. The dynamic series of images covered whole breasts (FOV 340 × 340 mm2, the number of slices 144), the time resolution of one image set was 87 s, and the post-contrast series was repeated five times. Contrast enhancement was performed with gadotheric acid (Dotarem, Guerbet, Roissy CdG Cedex, France) with an automatic power injector at a flow rate of 3 mL/s. Post-contrast image acquisition began immediately after administration of 0.2 mL/kg of contrast agent and saline bolus. Subtracted 3D images as well as axial and coronal maximum intensity projection (MIP) images were reconstructed from each post-contrast phase. Except breast, the imaging sequences covered both axillary areas so that the axillary lymph nodes could be seen on images. The MRI images were interpreted by a radiologist (author R.P.), who has 5-year experience in breast imaging including breast MRI, and by another radiologist (not author) who has 20-year experience in mammography and breast US and 10-year experience in breast MRI. The interpretation of MRI images was done separately and it was always performed side by side with all previous mammography and US images. The MRI images of patients were assessed using ACR BI-RADS® lexicon (Breast Imaging Reporting and Data System by American College of Radiology) (8). The images and findings of all MRI examinations, mammography, and US were always scrutinized and discussed at a multidisciplinary breast cancer meeting including surgeons, radiologists, medical oncologists, and pathologists. If a lesion was seen only on MRI, a second-look US was performed and US-guided core needle biopsy taken if possible. Second-look US was usually performed by a different breast radiologist than MRI. During the course of this study, no MRI-guided biopsies were required.
Preoperative MRI images were also evaluated with intention to determine axillary lymph node status. Axillary lymph node size, long axis to short axis ratio, cortical thickness to anteroposterior diameter ratio, the presence of a fatty hilum, and contrast enhancing patterns (homogeneous or heterogeneous) on post-contrast series were noted as possible signs of suspicious metastasis. In patients with suspicious finding, a targeted US was discussed at the breast meeting.
The additional findings of MRI were recorded according to their locations in the ipsilateral breast, in the contralateral breast, or in the axilla. The size of the tumor was recorded, compared with the corresponding size in the mammography and US, and with the histopathological tumor size.
Surgical Procedure
In all included patients, a first plan for breast conserving surgery and SNB was discussed by the multidisciplinary breast cancer team. All patients were also discussed at the postoperative meeting. The patients with nonpalpable tumors underwent preoperative US-guided localization marking. The marking method was either radioguided occult lesion localization (ROLL) or sentinel node and occult lesion localization (SNOLL) technique. The goal of tumor resection was to achieve tumor-free margins confirmed by histopathological examination. A reoperation was indicated if the cells of invasive cancer were found to extend out to the edge of the resected tissue (“cancer cells in the ink”) or if the histopathologically measured margin between ductal carcinoma in situ (DCIS) and the edge was less than 2 mm. A change from suggested breast conservation to mastectomy was discussed if breast conserving surgery was found to be inadequate in removing the tumor because of high tumor-to-breast volume ratio, multicentric cancer, extensive DCIS, or extensive intraductal component (EIC). Axillary lymph node clearance was performed if isolated tumor cells, micrometastases (<2 mm), or macrometastases (⩾2 mm) were detected in SNB or preoperative US-guided biopsy. All mastectomies and reoperations were discussed and planned by the multidisciplinary breast cancer team.
Histopathology
The resection of the serially sectioned tumor specimen was confirmed by using specimen imaging. The tumor specimen was sliced to the macroblocks and orientated by different inks according to the radiological images. The evaluation of the tumor size, the extent of DCIS, multifocality, resection margins, and other prognostic parameters like lymphovascular invasion was done on the large sections. Tumor size was taken to be the diameter of the largest focus in surgical specimens.
In this study, 4-µm thick serial paraffin section were cut from tumor tissue and stained with hematoxylin and eosin (HE). The breast cancer histology was assessed according to the World Health Organization classification (7), and tumor grading was based on the recommendations made by Elston and Ellis (9). “Extensive DCIS” was defined as DCIS occupying several ducts and quadrants throughout the breast. “Extensive intraductal component” was defined as when the component of intraductal carcinoma constitutes more than 25% of the primary tumor with intraductal foci. Tumor grades are classified as follows: Grade 1 is well-differentiated, Grade 2 is moderately differentiated, and Grade 3 is poorly differentiated. Immunohistochemical staining of needle core biopsies for estrogen and progesterone receptors (ER, PR), proliferation marker Ki-67, and human epidermal growth factor receptor-2 (HER2) were performed from subsequent sections. Four different ready-to-use rabbit monoclonal antibodies were used from Ventana Medical Systems/Roche Diagnostics: ER (SPI, rabbit), PR (IE2, rabbit), HER2 (4B5, rabbit), and Ki-67 (30-9, rabbit) with BenchMark XT immunostainer and ultraVIEW Universal DAB Detection Kit (Ventana/Roche, Tucson, AZ, USA). The percentage of nuclei with immunoreactivity to ER, PR, and Ki-67 was classified as continuous data from 0% to 100%. ER-positive and PR-positive cases showed staining in at least 1% of the tumor cell nuclei. HER2 expression was evaluated as membrane staining of invasive tumor cells and scored from 0 to 3. Carcinomas revealing 2+ or 3+ immunostaining were retested for HER2 gene amplification with chromogenic in situ hybridization (CISH) to determine HER2 positivity. Tumor size was taken to be the diameter of the largest focus in surgical specimens. Axillary lymph node status was achieved through sentinel lymph node biopsy or axillary lymph node dissections.
Adjuvant Therapies and Follow-Up
The data of adjuvant therapies and most recent follow-up were obtained from the electronic patient records.
Statistics
In statistical analyses, categorical variables were analyzed by Pearson’s χ2 test and continuous data by the nonparametric Mann–Whitney U test. A two-sided p-value of less than 0.05 was considered statistically significant. Statistical analyses were performed with IBM SPSS Statistics for Windows, Version 21.0 (IBM Corp., Armonk, NY, USA).
Results
A total of 100 women were enrolled into the study. In all, 50 patients were randomized to the breast MRI arm and 50 to the control arm. The baseline characteristics of the study patients are presented in Table 1. The flow chart of the study is presented in Fig. 1.
Baseline characteristics of patients and tumors.
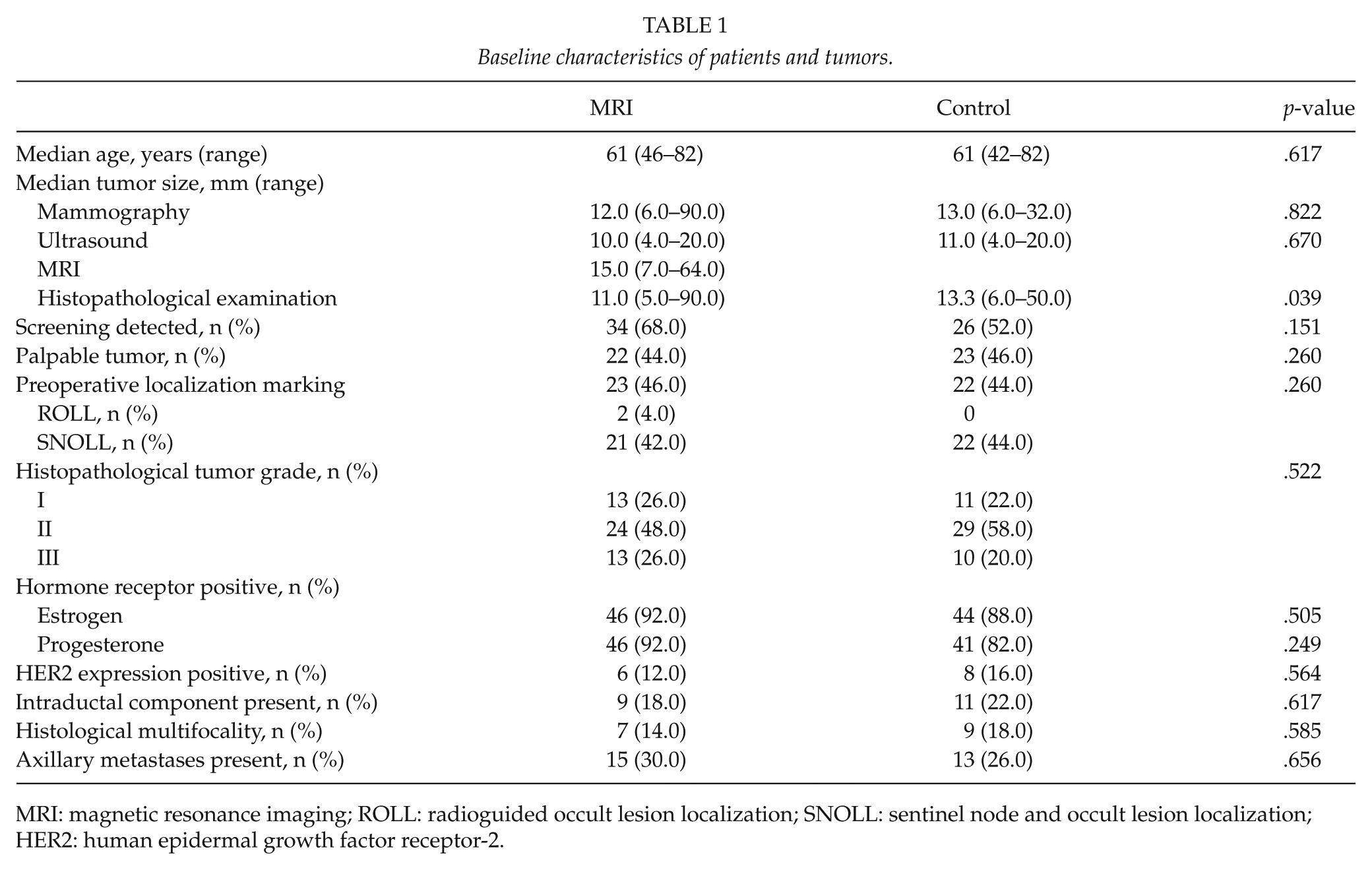
MRI: magnetic resonance imaging; ROLL: radioguided occult lesion localization; SNOLL: sentinel node and occult lesion localization; HER2: human epidermal growth factor receptor-2.
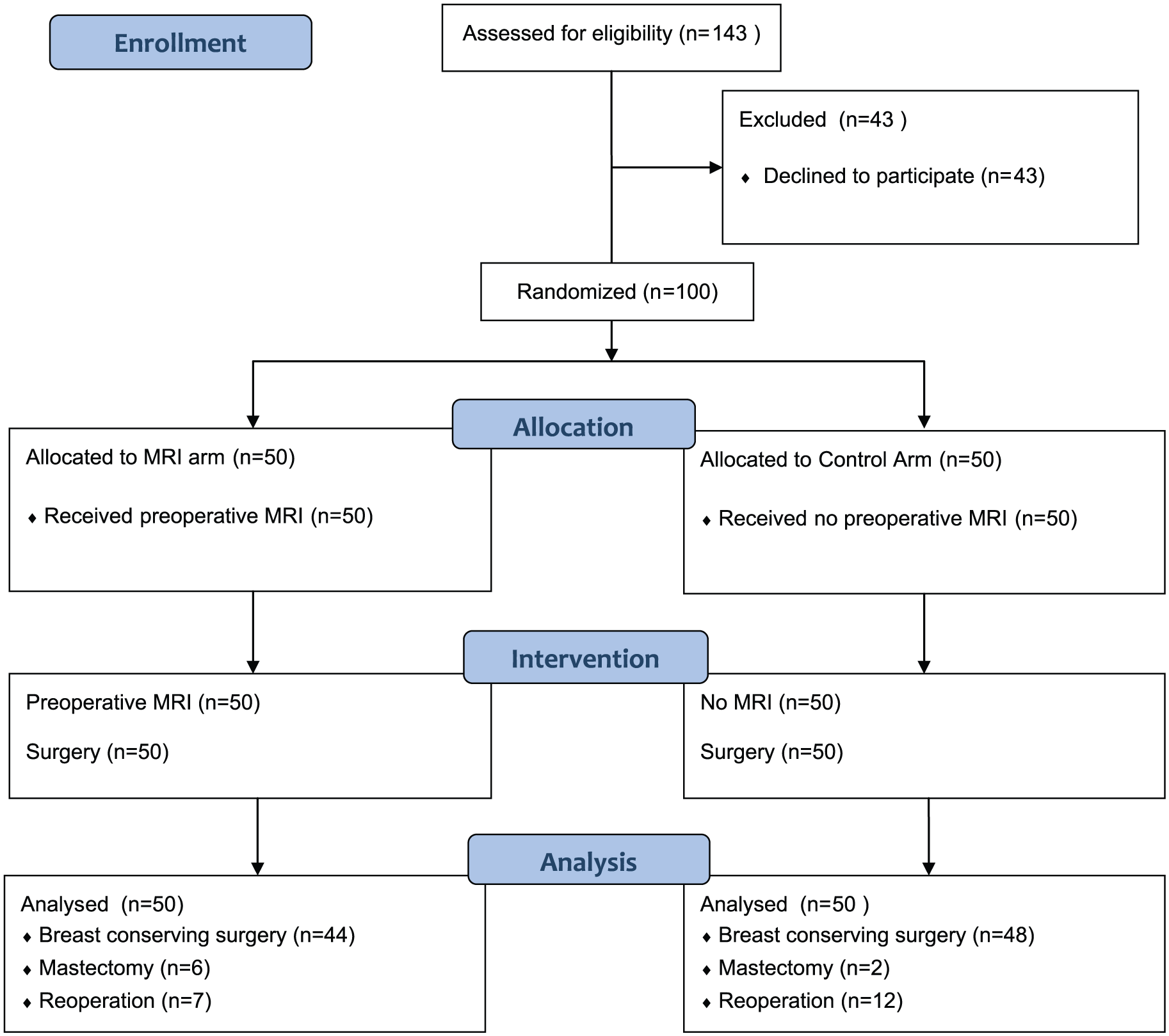
Flow chart of the study.
Mri Results and Additional Findings
All of the index cancer lesions were detected in the MRI. Preoperative MRI detected 19 additional findings in 14 patients (28%), and 10 of these lesions (53%) in seven patients (14% of patients undergoing MRI) were confirmed to be malignant. All of the additional breast findings were mass lesions. The median size of these lesions was 8.0 mm (mean, 8.2 mm; range, 5–14 mm). Six additional cancers were histopathologically verified in the ipsilateral breast and one in the contralateral breast in seven patients. Five patients with additional BI-RADS 3 lesions underwent a control MRI after a period of 6–12 months and no additional cancers were found during their follow-up. The data of additional findings are summarized in Table 2.
Additional findings on breast MRI.
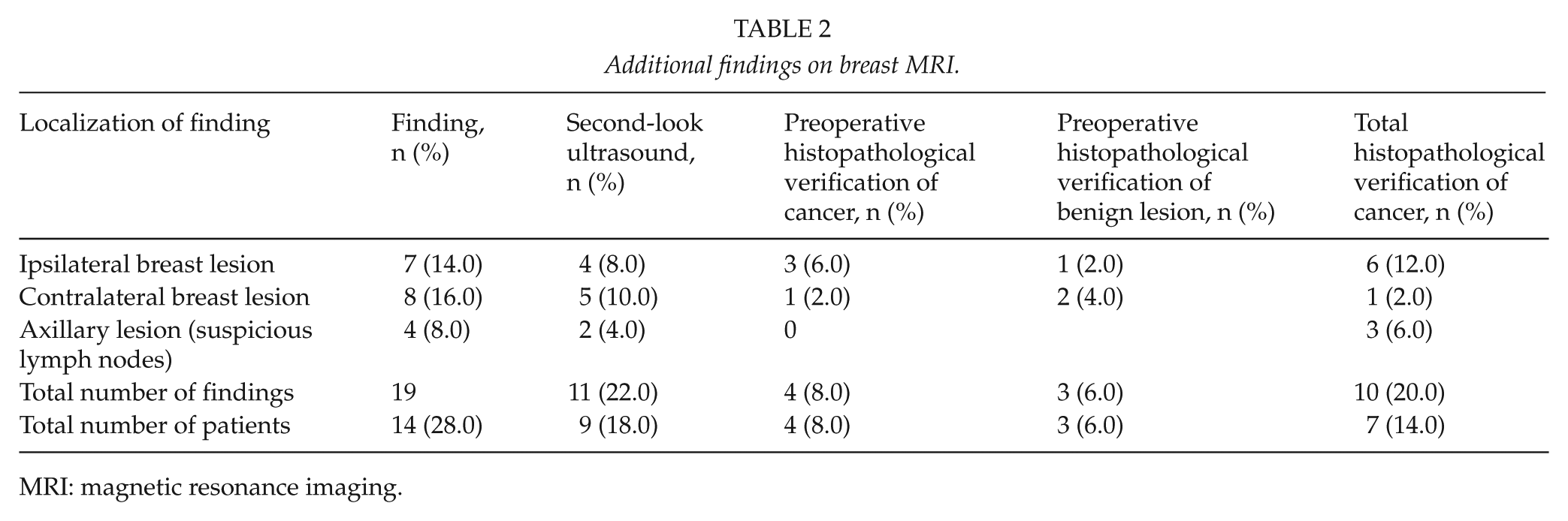
MRI: magnetic resonance imaging.
Second-Look Us
Nine patients underwent second-look US because of additional MRI finding in the ipsilateral or contralateral breast, or in the axilla. Four of these lesions were cancer foci, which were verified with US-guided core needle biopsy. In three patients, the lesion was biopsied and found to be benign (adenoma, fibrosis, and fibrocystic disease). In two patients, additional breast lesions were invisible in US, and these patients underwent MRI after a follow-up of 6–12 months because the lesions were of BI-RADS 3.
Tumor Size
In the MRI patient group, the median size of the index tumor was 15.0 mm (range, 7–64 mm) in MRI, 12.0 mm (range, 6.0–90.0 mm) in mammography, 10.0 mm (range, 4.0–20.0) in US, and 11.0 mm (range, 5.0–90.0 mm) in histopathological examination, respectively. In five patients (10%), the diameter in MRI was at least 10.0 mm higher than the maximum diameter in mammography or US. In five patients (10%), MRI also overestimated the tumor size more than 10.0 mm compared with the histopathology. This overestimation was considered to be associated with a false positive mastectomy in one patient with a discrepancy of 25 mm regarding the estimation of tumor size. In six patients, MRI underestimated the tumor size more than 10.0 mm compared to histopathology. In four of these six cases, the histopathological extent of the disease was associated with extensive DCIS or EIC. Three of these patients underwent reoperation, which was breast conserving resection in two patients, and mastectomy in one patient.
Initial Breast Surgery
Preoperative MRI resulted in a change in surgical management in 10 patients (20%). The MRI findings and altered surgical treatment of these 10 patients are presented in Table 3. The initial operation was mastectomy in five patients in the MRI group (10%), but there were no initial mastectomies in the control group (p = 0.022). The indication for initial mastectomy was multifocal tumor in three patients and extensive tumor size in two patients. Extensive DCIS or EIC was also a contributing factor in three patients. MRI prolonged the waiting time to the operation: the mean interval between referral and first surgical procedure was 34 days in the MRI group (range, 16–68 days) and 21 days in the control group (range, 8–53 days). There was a statistical difference between the groups (p < 0.001).
Altered surgical treatment in 10 patients after preoperative MRI.
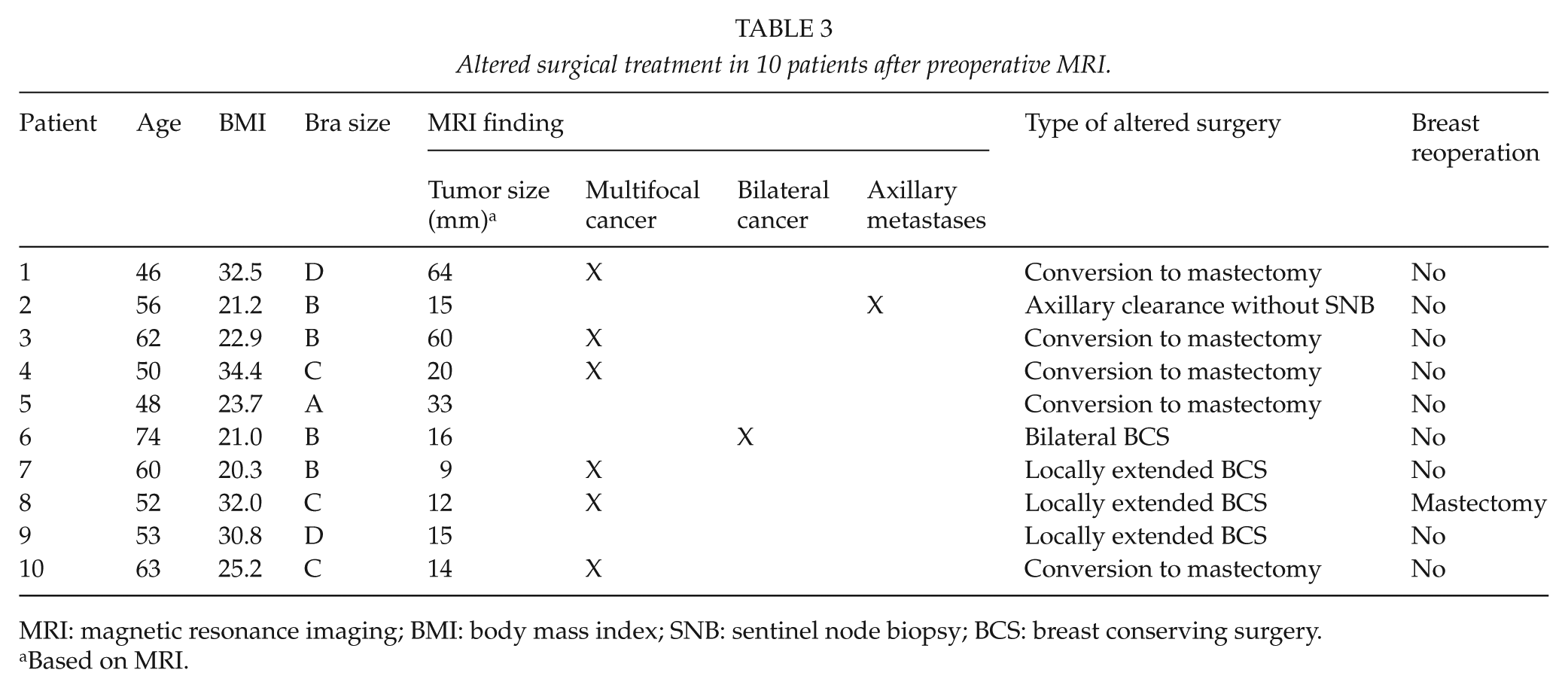
MRI: magnetic resonance imaging; BMI: body mass index; SNB: sentinel node biopsy; BCS: breast conserving surgery.
Based on MRI.
Breast Reoperations and Definitive Mastectomy Rate
Seven patients (14%) underwent breast reoperation in the MRI group and 12 patients (24%) in the control group. The difference was not statistically significant (p = 0.202). The indication for breast reoperation was tumor-positive resection margins in each case. In the MRI group, the reoperation was mastectomy in one patient and breast conserving resection in six patients. In the control group, the reoperation was mastectomy in two patients and breast conserving resection in 10 patients. The definitive mastectomy rate was 12% (6 patients) in the MRI group and 4% (2 patients) in the control group. The difference was not statistically significant (p = 0.140).
Axillary Surgery
A total of 15 patients (30%) had metastatic axillary lymph nodes and underwent lymph node dissection in the MRI group and 13 patients (26%) in the control group, respectively. The difference was not statistically significant. Four clinically node-negative patients had suspicious MRI finding in the axilla. Two of them underwent targeted US and US-guided fine needle aspiration cytology with negative result. These two patients underwent SNB, and metastatic sentinel nodes were found in the frozen section analysis resulting in axillary lymph node dissection. The latter two patients underwent surgery without US. One of them underwent axillary lymph node dissection based on MRI finding and metastatic lymph nodes were confirmed in the histopathological analysis. The last patient underwent SNB and the sentinel nodes were found to be benign.
Adjuvant Therapies and Follow-Up
In the MRI group, 24 patients received adjuvant chemotherapy, 45 patients had radiotherapy, and 37 patients had hormonal therapy. In the control group, 27 patients received chemotherapy, 49 patients had radiotherapy, and 38 patients had hormonal therapy. The differences between study arms were not statistically significant. No patient received neoadjuvant therapy. After a median follow-up time of 49 months, there were no local recurrences. In the control group, recurrent distant disease was detected in three patients. One patient died in the control group due to cancer-unrelated cause.
Post Hoc Power Analysis
We conducted a post hoc power analysis and found that this study had a 24.5% power to detect a significant difference in breast reoperation rate between the study groups and a 51.6% power for mastectomy at the two-sided significant level of 0.05. A total of 412 patients would be needed for a power of 80% for mastectomy and 480 patients for a breast operation.
Discussion
In this prospective randomized study, we found that in the absence of a specific clinical need, the routine use of preoperative MRI is not indicated. Exposing patients with early-stage ductal breast cancer to additional, more accurate preoperative local staging does not always result in improved outcome. This conclusion is in accordance with the Finnish Health Technology Assessment (HTA) report, which was published recently on this topic by the National Institute for Health and Welfare (10). This HTA report does not support widening the indications of preoperative MRI to include routinely stage I ductal tumors.
The overall diagnostic accuracy of breast MRI is excellent, which has been documented in several cohort studies, systematic reviews, and meta-analyses (1–3,11). The sensitivity of MRI is superior to mammography or US, and unlike with conventional imaging techniques, the sensitivity is not dependent on tumor size, the type of tumor, or the density of breast parenchyma (12). However, the superior diagnostic accuracy of MRI does not always translate into clinical outcome. The tumor-to-breast volume ratio is not a standardized measure and is subjective. This is further complicated by the low specificity of MRI and by the fact that MRI is done in the prone position while both surgery and physical examination are done in totally different positions. Therefore, adapted changes in the surgical management approaches might be the key to improve outcomes. Also, the preoperative supine breast MRI might help in the future to improve surgical planning.
MRI may alter the surgical management plan resulting in locally more extensive resection, mastectomy, or bilateral approach (Fig. 2). In our study, this change occurred in 20% of patients. This result is in concordance with the recent Swedish prospective randomized study (Preoperative MRI of the Breast (POMB)) by Gonzalez et al. (4). In the POMB trial with a study population of 440 patients, this change was also observed in 18% of the patients. Logically, more accurate diagnostics and more targeted surgery should result in decreased reoperation rate. In our study, 14% of MRI patients underwent breast reoperation and 24% of the control patients, but the difference was not statistically significant. In the POMB trial, the correspondent rates were 5% versus 15% respectively, achieving statistical significance (p < .001). This result is contradictory to the randomized British Comparative Effectiveness of MRI in Breast Cancer (COMICE) trial with an extensive study population of 1623 patients, where reoperation rates were not different between the MRI group and controls (13). In this study, breast conserving therapy (BCT) failure was mainly due to extensive DCIS or EIC. Furthermore, it can be speculated that we have failed to correctly identify the true extent of DCIS in some cases, which might have resulted in presumable positive margins.
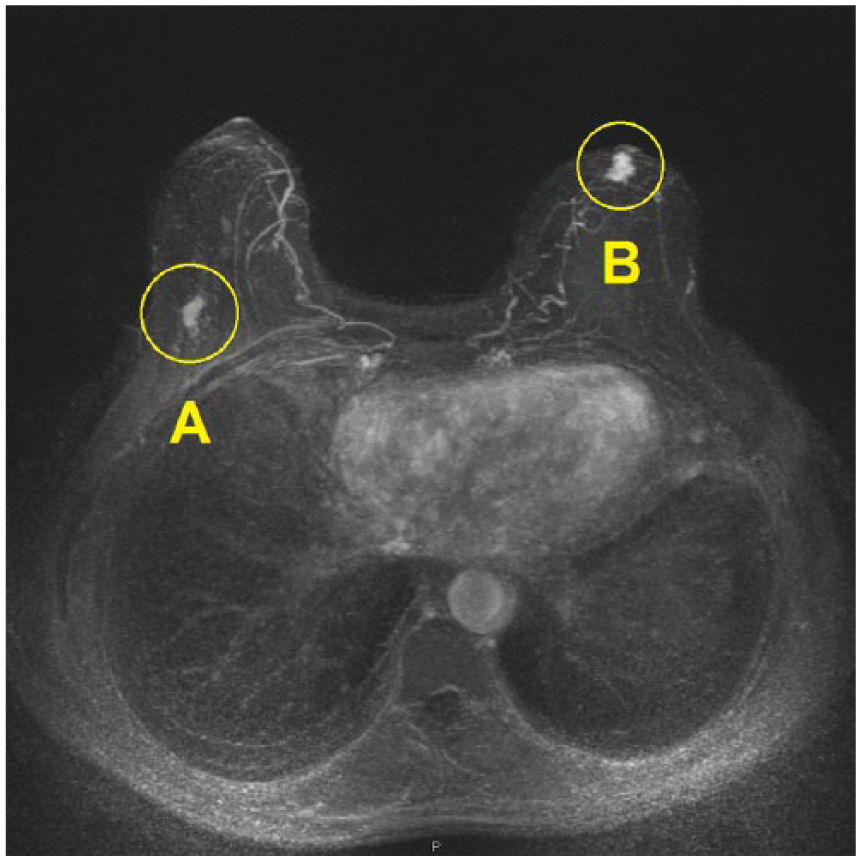
Preoperative MRI revealing (A) ductal cancer in the right breast and (B) an additional 1-cm lesion in the left breast, which was not detected on mammography. Targeted US-guided core needle biopsy confirmed the left breast lesion to be invasive lobular cancer. The patient underwent bilateral breast conserving surgery.
There have been suspicions whether the increasing use of breast MRI causes increased mastectomy rates or even unneccessary mastectomies. Half of the additional MRI-detected lesions are benign, and MRI also overestimates the size of cancer in 33% of patients, and this overestimation may result in unnecessary extensive surgery (14). DCIS seems to be an additional risk factor for both over- and underestimation (15). In this study, MRI either overestimated or underestimated the size of tumor more than 10 mm in 22% of patients in comparison with histopathological tumor size, and one mastectomy was considered to be overtreatment. In the POMB trial, the mastectomy rates between study arms were quite equal: 43% in the MRI group and 41% in the control group (4). In the COMICE trial, the rates were 7% and 1%, respectively (12). In the current study, the definitive mastectomy rate was 12% in the MRI arm and 4% in the control arm. Thus, preoperative MRI was associated with a trend towards increased mastectomy rate, although the difference was not statistically significant.
An additional MRI finding alone should not be an indication for more extensive surgery without histopathological verification. These lesions should be evaluated with targeted second-look US and US-guided biopsy (16). If the lesion cannot be detected with US, MRI-guided biopsy should be used in patients with BI-RADS 4–5 lesions. In patients with BI-RADS 3 lesion, a control MRI can be performed after a follow-up interval of 6, 12, or 24 months, if the lesion remains stable. In this study, this approach was used in patients with BI-RADS 3 lesions. The disadvantage of false positive and borderline MRI findings is that they cause additional investigations, costs, and anxiety for patients.
If breast cancer is locally extensive, some proportion of mastectomies can be avoided using oncoplastic resection techniques. In this study, three patients underwent locally more extensive but conventional resection based on MRI finding. Five patients underwent mastectomy because oncoplastic option was not considered to apply to them. Also, surgeon’s opinion at the multidisciplinary meeting is of major importance and plays an important role. At our preoperative meetings, the chairman is always a plastic surgeon having no interest favoring mastectomy.
If a new technique improves diagnostical quality and alters the surgical treatment, it does not necessarily provide therapeutic benefit in terms of prolonged disease-free survival or disease-specific overall survival. The therapeutic benefit associated with breast MRI in primary breast cancer has limited evidence. There is no evidence that MRI would decrease local breast recurrences or improve overall survival (17). In general, local recurrence rate is very low after breast conserving surgery, adjuvant systemic therapy, and radiotherapy. The landmark study by Veronesi et al. (18) was conducted before MRI era and it reported a local recurrence rate of only 8.8% after a median follow-up time of 20 years. Because the incidence of local recurrence is relatively low with standard triple assessment, a significant additional value of MRI would be unlikely.
Furthermore, MRI causes extra delays to the breast cancer care pathway. We found out that the delay between referral and initial surgery was on average 34 days in our MRI patients. MRI was performed rapidly within 1 week after randomization, but the delay was caused by consequent arrangements such as second-look US, biopsies, and multidisciplinary breast meetings. A fear of delays is common among patients with newly diagnosed breast cancer. Although such delay probably has no effect on clinical outcome, it may cause considerable anxiety and unnecessary distress for the patient. An additional delay of 19–41 days associated with MRI has also been reported in other studies (19).
To date, this is the fourth prospective randomized controlled trial (RCT) investigating the preoperative use of MRI in breast cancer (4,13,20). This study is the only RCT focusing on a selected population of patients with early-stage invasive ductal carcinoma. We are aware of the main limitation of this study: the number of our study patients is low, which makes the statistical analyses underpowered to demonstrate significant differences between the two study arms. There were severe factors limiting the sample size, such as financial limitations and an estimation of slow accrual, which is a common problem with small single-center trials. Our study groups were well balanced in terms of clinical and histopathological characteristics, but there was a significant difference in the histopathological tumor size, favoring the MRI group with slightly smaller tumors. This may influence the reoperation rates. The number of clinical endpoint events was also low, which can be associated with selection bias and misinterpretations.
In conclusion, the results of this prospective randomized study indicate that, in a setting of a small volume institution, the routine use of preoperative MRI does not give clear advantage to improve the outcome in patients with stage I invasive ductal breast cancer. In clinical practice, MRI should not be used routinely in this patient population and the individual indications for MRI should be discussed by the multidisciplinary breast cancer team.
Footnotes
Acknowledgements
Presented at the Annual Meeting of the Finnish Societies of Surgery and Anaesthesiology, Helsinki, Finland, 18 November 2016.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by a grant from the University Hospital of Turku (EVO).
