Abstract
Background and Aim:
In Landspitali University Hospital, magnetic resonance imaging is used non-selectively in addition to mammogram and ultrasound in the preoperative assessment of breast cancer patients. The aim of this study was to assess invasive tumor size on imaging, compare with pathological size and evaluate the impact of magnetic resonance imaging on the type of surgery performed.
Material and Methods:
All women with invasive breast cancer, diagnosed in Iceland, between 2007 and 2009 were reviewed retrospectively. In all, 438 of 641 (68%) patients diagnosed had preoperative magnetic resonance imaging. Twelve patients treated with neoadjuvant chemotherapy were excluded and 65 patients with multifocal or contralateral disease were assessed separately.
Results:
Correlations between microscopic and radiologic tumor sizes were relatively weak. All imaging methods were inaccurate especially for large tumors, resulting in an overall underestimation of tumor size for these tumors. Magnetic resonance imaging under- and overestimated pathological tumor size by more than 10 mm in 16/348 (4.6%) and 26/348 patients (7.5%), respectively. In 19 patients (73%), overestimation of size was seen exclusively on magnetic resonance imaging. For tumors under- or overestimated by magnetic resonance imaging, the mastectomy rates were 56% and 65%, respectively, compared to an overall mastectomy rate of 43%. Of 51 patients diagnosed with multifocal disease on pathology, 19 (37%) were diagnosed by mammogram or ultrasound and 40 (78%) by magnetic resonance imaging resulting in a total detection rate of 84% (43 patients). Fourteen (3%) patients were diagnosed preoperatively with contralateral disease. Of those tumors, all were detected on magnetic resonance imaging but seven (50%) were also detected on mammogram or ultrasound or both.
Conclusion:
Our results suggest that routine use of magnetic resonance imaging may result in both under- and overestimation of tumor size and increase mastectomy rates in a small proportion of patients. Magnetic resonance imaging aids in the diagnosis of contralateral and multifocal disease.
Keywords
Introduction
Preoperative assessment of tumor size is important in breast cancer surgery, especially in breast conservation. Although many studies have assessed the value of using magnetic resonance imaging (MRI) in invasive breast cancer, its role in clinical practice is still disputed. Some studies have suggested that MRI may increase rates of mastectomies, probably by overestimating the extent of local disease (1, 2). In many of these studies, short-term clinical outcomes were the primary endpoints.
Many studies have looked at the accuracy of estimating tumor size by mammogram (MGM) and ultrasound (US) (3–6) and compared with MRI (7–9), and most of these have assessed median or mean size differences. MRI has been suggested as a complementary method in selective and high-risk patients and has become the investigation of choice in invasive lobular breast cancer (10) and dense breast tissue (9, 11).
In Iceland, over 95% of all breast cancers diagnosed in the country are treated surgically at Landspitali University Hospital and since August 2006, preoperative MRI has been used routinely in addition to MGM and US. An audit in our unit following the first year of MRI in addition to MGM and US showed that MRI changed the type of surgery performed in 14% (25/178) of cases: in 8.9% (16/178), a more extensive local excision was performed and in 5.1% (9/178), a mastectomy was performed instead of breast conservation. In 8 of these 9 patients (89%), tumor size on MRI agreed with size on pathology (unpublished data). We therefore postulated that MRI gave a better preoperative estimate of tumor size than did MGM or US. The aim of this study was to assess the size of invasive breast cancer with these three imaging modalities and compare with pathological size and evaluate the impact of MRI on the type of surgery performed.
Patients and Methods
All women diagnosed with invasive breast cancer between 2007 and 2009 found in the Icelandic cancer register were entered into the study retrospectively. Patients had both MGM and US at the breast diagnostic clinic (the Icelandic Cancer Society) and a subsequent MRI at Landspitali University Hospital, although this was frequently not performed in the very elderly (n = 79), the very obese (n = 9), patients operated in a hospital where MRI was not available (n = 35), those choosing not to have it (n = 31), and in those where MRI was contraindicated (n = 49).
The study was approved by the local scientific and ethical committee.
Study Group
From January 2007 to December 2009, 641 women were diagnosed with invasive breast cancer in Iceland. All cases of ductal carcinoma in situ alone were excluded. Preoperative MRI was done in 438 patients (68%). Twelve patients were excluded, having received neoadjuvant chemotherapy. Sixty-five patients with multifocal or contralateral disease were assessed separately. The primary patient group for analysis therefore included 361 patients with unifocal invasive breast cancer (Fig. 1). Of the 361 patients, 310 had invasive ductal cancer (IDC), 38 invasive lobular cancer (ILC), and 13 other types of invasive cancer. Patient characteristics are shown in Table 1.
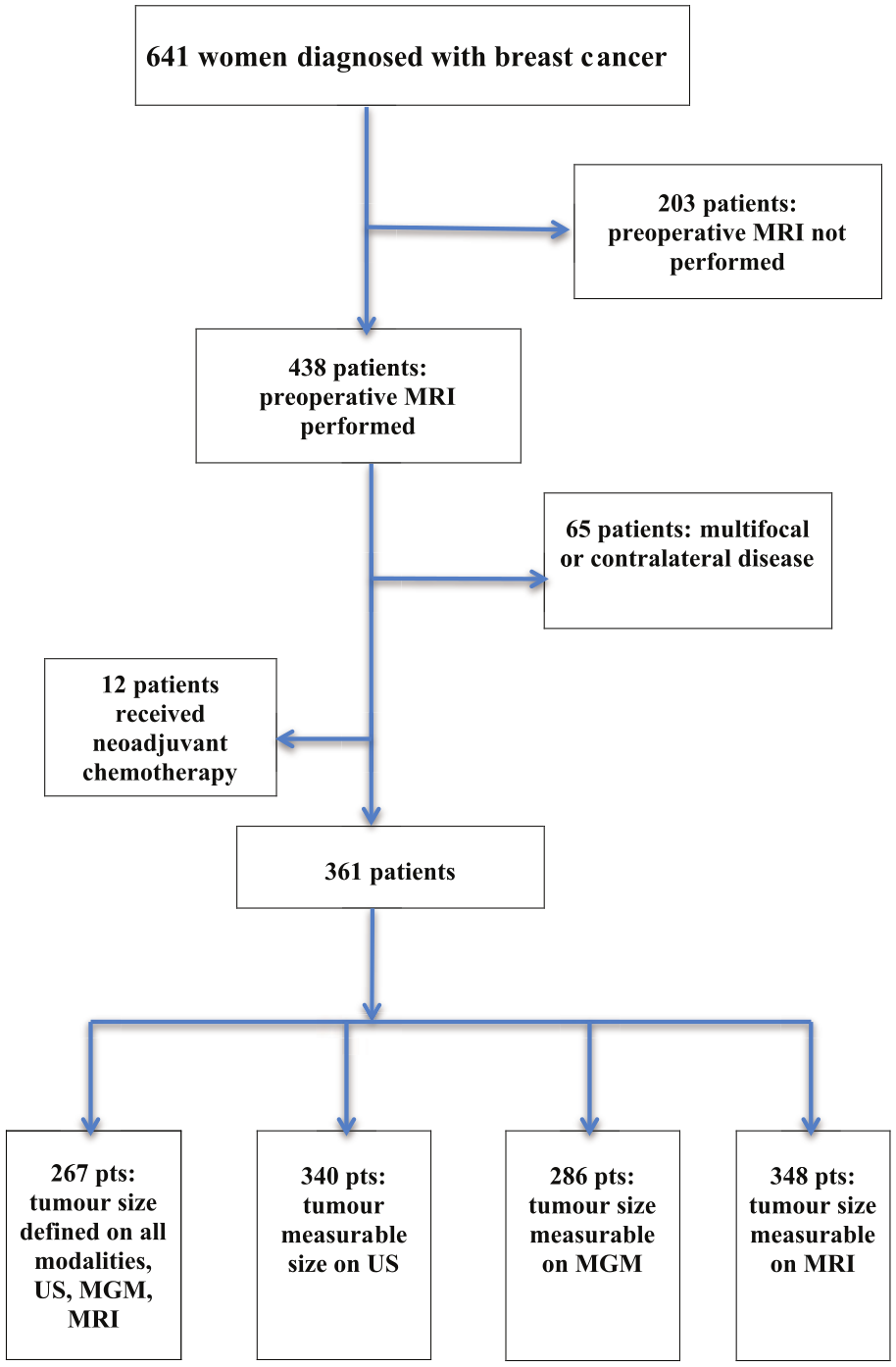
Flow chart of patients diagnosed with breast cancer in the study period.
Patient characteristics (n = 361).
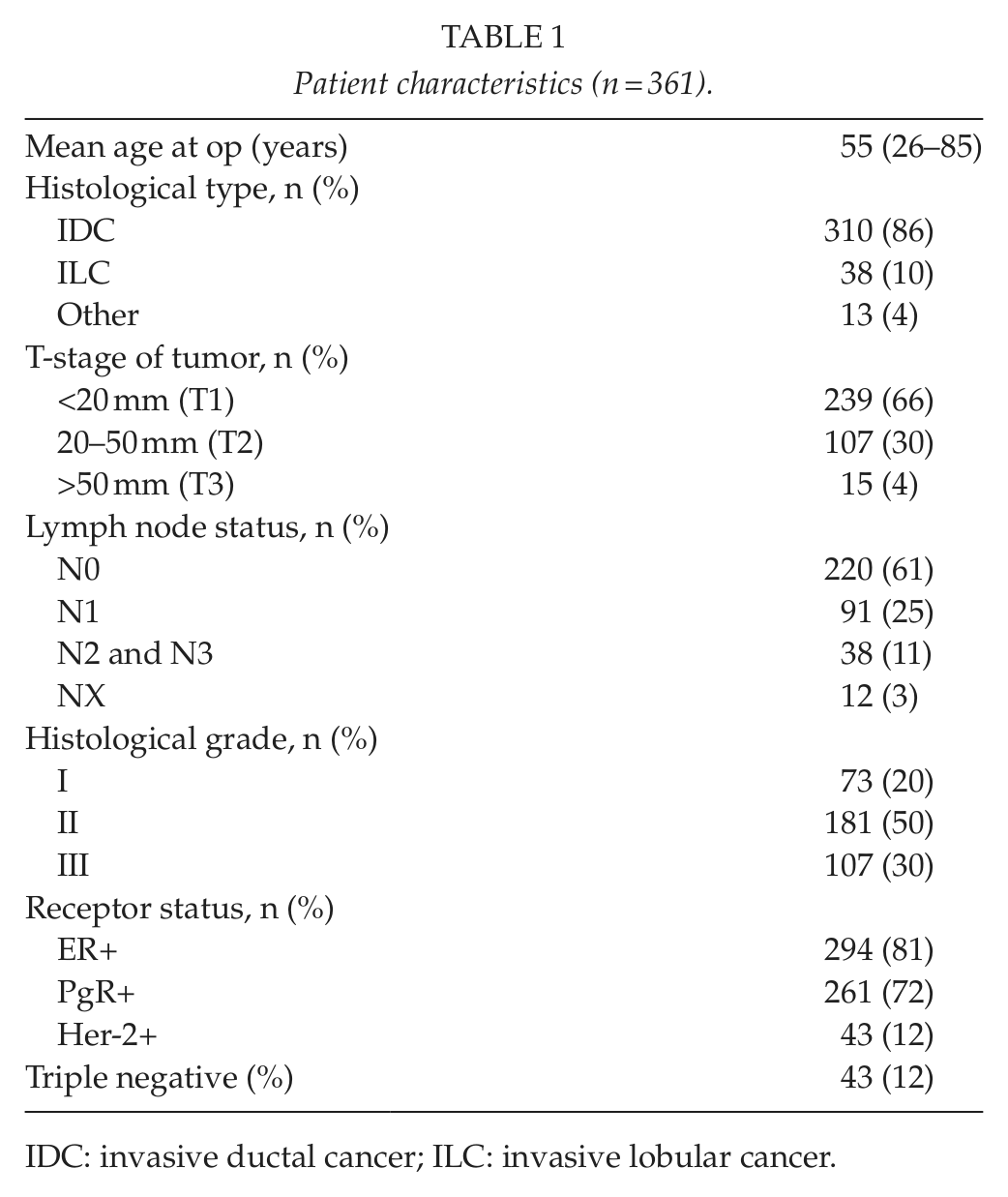
IDC: invasive ductal cancer; ILC: invasive lobular cancer.
It was possible to visualize the tumor by all three imaging modalities (MGM, US, and MRI) in 267 patients. A separate analysis was done for visible tumors by the individual imaging modalities (MGM, n = 286, US, n = 340, MRI, n = 348; Fig. 1). Thus, these evaluations of the capability of the imaging modalities to predict tumor size included only tumors visible on imaging.
In general, the modality showing the largest size has been used as the reference size for deciding the type of surgery to be performed. All patients operated with breast conservation were treated with adjuvant radiotherapy. At operation, a few millimeter margin was the goal. Indication for reoperation was a microscopic tumor margin <1 mm to invasive or in situ cancer.
Breast Radiology
In the breast diagnostic unit (The Icelandic Cancer Society), a conversion was made from analog MGM to full-field digital imaging (Siemens Mammomat Inspiration) in 2008. Irrespective of the type of MGM performed (analog or digital), three mammographic views were taken on the index breast. Tumor size was measured on all cases, and the largest diameter given as the maximal tumor size.
Ultrasonography was performed on Toshiba Aplio XG machines. Perpendicular views were taken of the tumor and measurements done in three planes and the largest diameter given as the maximal size of the tumor. A hyperechogenic rim around the tumors was not included in the size measurement. US was not done routinely on the contralateral breast.
MRI was performed within 5 days in patients with breast cancer and surgery within 3 weeks. The MRI was performed on a 1.5 T magnet (Magnetom Avanto V17, Siemens Medical Solutions, Erlangen, Germany) with a dedicated bilateral breast surface coil. The imaging was performed in a prone position using gadopentetate dimeglumine contrast media (Magnevist®, Bayer Schering Pharma AG, Berlin, Germany). The size of tumor was measured in two or three imaging planes (coronal, axial, and sagittal planes), and the largest diameter, taken from the radiological report, was used in this study.
Pathology
The specimen was palpated and the tumor located. Sections of 5–10 mm were cut from the skin surface through the whole specimen perpendicular to and down to the fascia. The specimen was fixed in 10% formaldehyde for 16–48 h. Thereafter, the tumor size and resections margins were measured. Representative areas of the tumor were cut, embedded in paraffin, and stained with hematoxylin-eosin. Evaluation of size was taken from the pathological report as well as information about receptor status and histological grading according to the Nottingham school.
Statistics
Relationships between microscopic and imaging measurements of tumor size were estimated with linear regression. Possible differences in the frequencies of over- and underestimation between MRI and US were tested with Fisher’s exact test. Calculations were made with GraphPad Prism, version 6.01.
Results
Size
The average diameter of the 267 invasive tumors, where tumor size was documented on all three modalities, was 18 mm (4–65 mm) on US, 21 mm (5–80 mm) on MGM, and 21 mm (6–80 mm) on MRI compared to 21 mm (4–75 mm) on pathology. Fig. 2 demonstrates that the correlations between microscopic and radiologic tumor sizes are quite weak and that all imaging methods underestimate tumor size for large tumors (p < 0.001 in all cases).
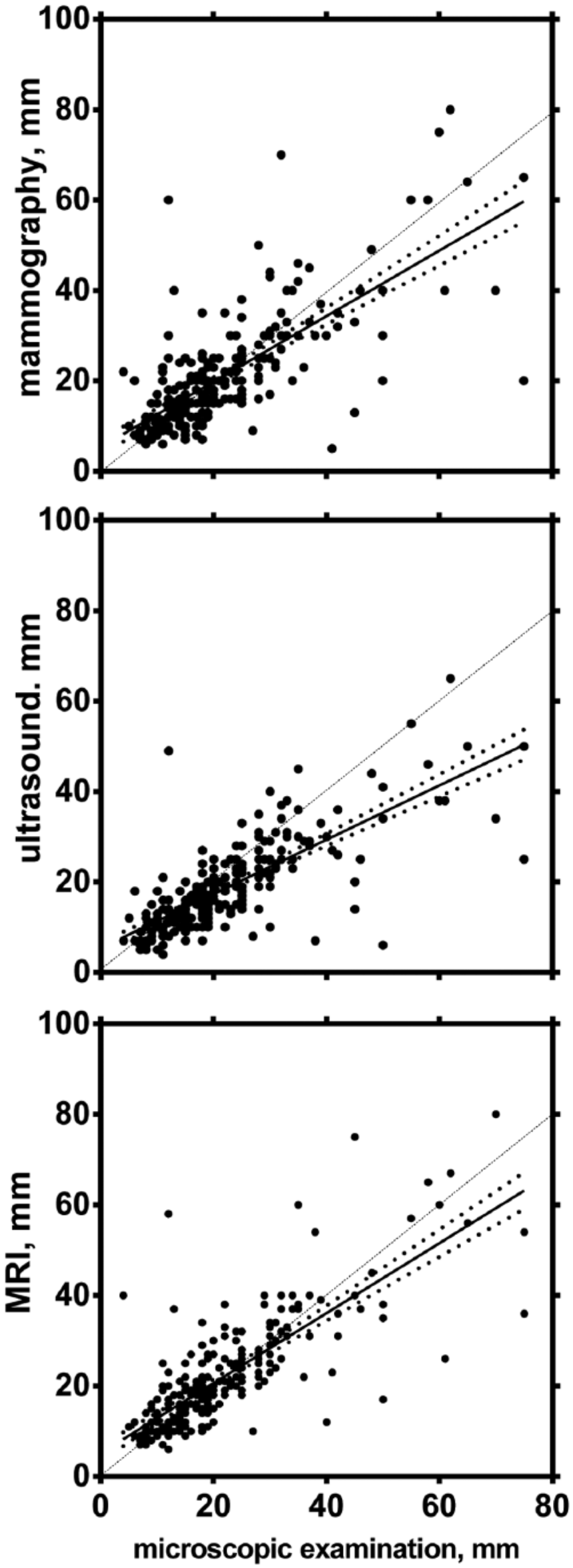
Linear regression plots between tumor size determined on microscopic examination and the imaging methods. The dashed line shows the theoretically ideal relationship between microscopic examination and imaging. The slopes of the calculated regression lines are significantly different from 1 for each imaging method (t-test in a regression model with log-transformed data, p < 0.001 in all cases). The dotted lines represent the 95% confidence intervals of the slope.
US over- and underestimated tumor size by 10 mm or more in 31 (12%) (Table 2). MRI over- and underestimated tumor size by 10 mm or more in 28 patients (10%) (Table 2). Overestimation of tumor size by 10 mm or more was more frequent with MRI than with US (Fisher’s exact test, p = 0.01). Underestimation of tumor size by 10 mm or more occurred more often with US than with MRI (Fisher’s exact test, p = 0.02).
Tumor size when measurable by all imaging modalities within the same patient (MGM, US, and MRI) (n = 267). Comparison with microscopic examination.
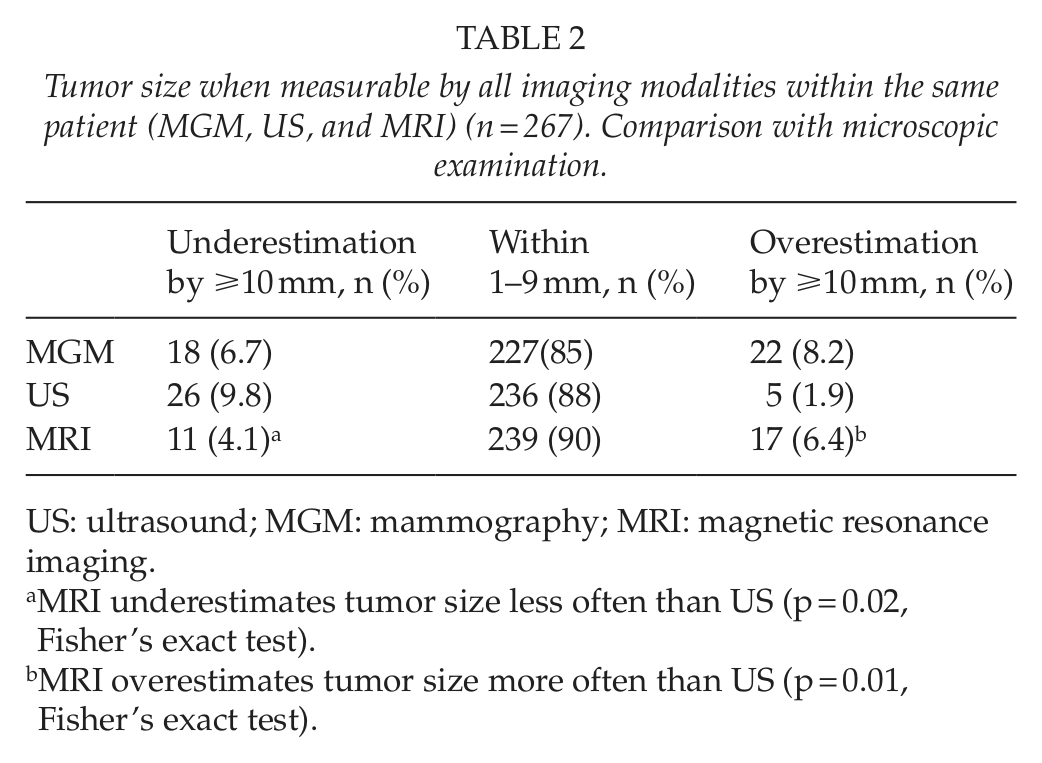
US: ultrasound; MGM: mammography; MRI: magnetic resonance imaging.
MRI underestimates tumor size less often than US (p = 0.02, Fisher’s exact test).
MRI overestimates tumor size more often than US (p = 0.01, Fisher’s exact test).
Similar results were observed when the analysis included all tumors seen on each modality (data are summarized in Table 3). In 26 patients (7.5%), MRI overestimated the size by 10 mm or more (Table 3). This size difference was documented exclusively on MRI in 19 (73%). The average size of the 26 tumors overestimated by MRI exclusively was 22 mm on pathological examination.
Tumor size when measurable by MGM (n = 286), US (n = 340), or MRI (n = 348). Comparison with microscopic examination.
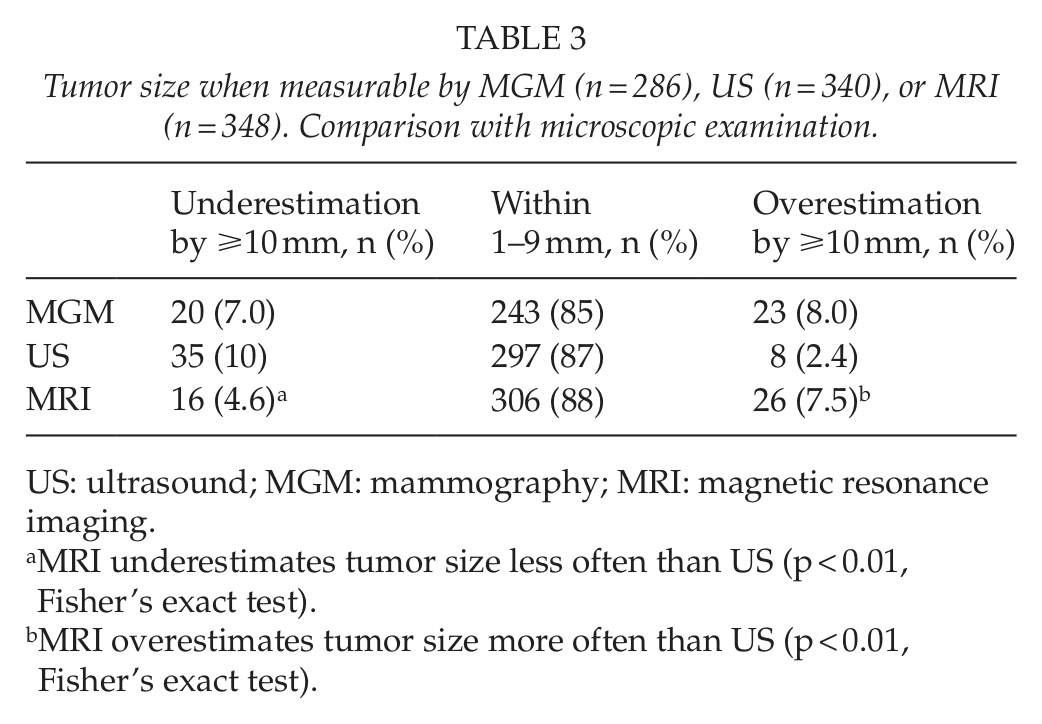
US: ultrasound; MGM: mammography; MRI: magnetic resonance imaging.
MRI underestimates tumor size less often than US (p < 0.01, Fisher’s exact test).
MRI overestimates tumor size more often than US (p < 0.01, Fisher’s exact test).
MRI was falsely negative in 13 patients (3.6%): no lesion was seen in nine cases and a benign or unspecific lesion in four. The average diameter of these missed tumors was 7.4 mm (3–19 mm). MGM was falsely negative in 58/344 (17%) patients (data were unavailable in 17 patients): no lesion was seen in 20 patients, distortion or calcification only in 38. US was falsely negative in 30/355 (8.5%) patients (data were unavailable in six patients): no lesion was seen in six patients, tumor was not measurable in 21, a benign or unspecific lesion in six.
Size Difference and Type of Surgery Performed
In all patients treated for breast cancer at Landspitali University Hospital during the study period, 57% were treated by breast conservation and 43% were treated by mastectomy with or without reconstruction. The reoperation rate (due to microscopic tumor at margins) in the breast conservation group was 6%.
When MRI overestimated size by 10 mm or more, the breast conservation rate was 35% (n = 8) and mastectomy rate 65% (n = 18). Three of eight patients having breast conservation (37.5%) had re-excisions.
When MRI underestimated size by 10 mm or more, the breast conservation rate was 44% (n = 7) and mastectomy rate 56% (n = 9). Re-excision was performed in one of the seven (14%) breast conservation patients. In three patients (0.8%), these tumors were not seen on MGM. In five patients (1.4%), this size difference was seen exclusively on MRI when compared to size on MGM and in three cases (0.8%) when compared to size on US.
ILC
Of the 361 patients, 38 (11%) had ILC. In 37 (97%), the tumor was seen and measurable on MRI, in 35 (92%) on US and in 23 (61%) on MGM. The frequency of under- and overestimation of size by 10 mm or more did not differ between MRI and US (p > 0.05; Table 4).
Invasive lobular cancer. Tumor size when measurable by MGM (n = 23), US (n = 35), or MRI (n = 37). Comparison with microscopic examination.
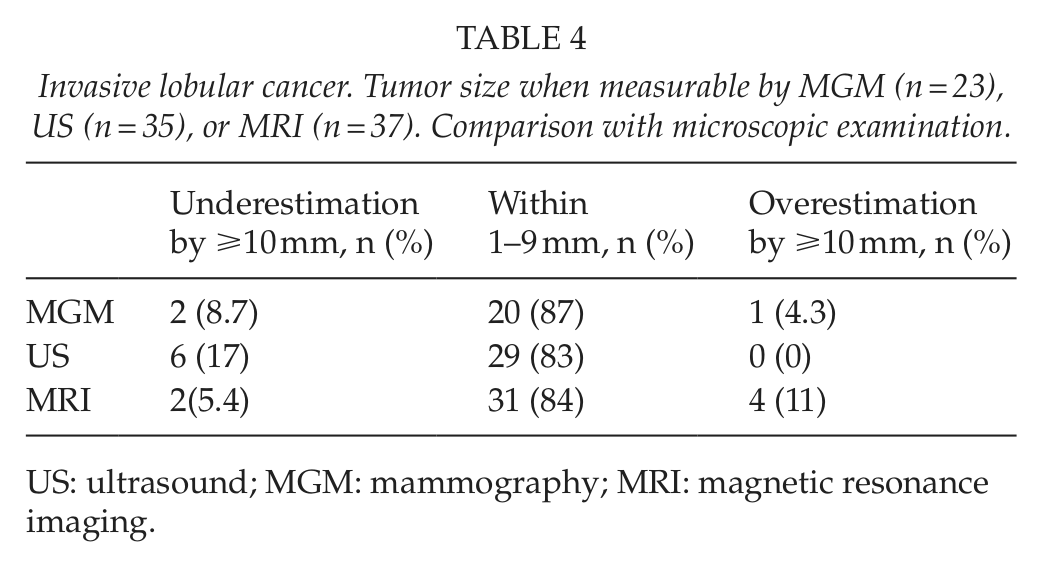
US: ultrasound; MGM: mammography; MRI: magnetic resonance imaging.
Multifocal or Contralateral Disease
In all, 51/438 patients (12%) had multifocal and 14/438 patients (3.2%) had contralateral disease on pathology. MRI demonstrated contralateral tumors in 13/14 (93%) patients, and exclusively in 7/14 (50%) patients. MGM and/or US showed multifocal disease in 19 patients (37%). MRI showed multifocality in 37/51 (73%) patients, giving 24 additional patients (47%). For patients in whom multifocal disease was correctly described by MRI (n = 37), mastectomy was performed in 22 (59%) and breast conservation in 15 (41%). Four of the latter patients (27%) had mastectomies due to inadequate margins. The end mastectomy rate was therefore 70% (26/37) in these patients.
MRI suggested multifocal disease in 28/361 patients having unifocal disease on pathology, that is, a falsely positive rate of 7.8%. The mastectomy rate in these cases was 57%. US erroneously showed multifocal disease in 15/340 patients (4.4%).
Discussion
Our results show that routine use of MRI in addition to US and MGM may be a double-edged sword in the surgical treatment of breast cancer. On one hand, MRI identifies a relatively high number of patients with multifocal disease and a small number of patients with contralateral disease. On the other hand, our results show that preoperative MRI over- or underestimates pathological tumor size in unifocal invasive disease by 10 mm or more in one-tenth of the patients. Also, MRI erroneously showed multifocal disease in 8% of patients, which led to overtreatment.
In our hospital, MRI has been used routinely together with MGM and US since 2006. Although a relatively high proportion of patients (32%) were excluded from MRI, we believe that the study group is a representative cohort of breast cancer patients.
Many breast units use MRI selectively and believe its main role is in the presence of dense breast tissue on MGM and ILC (12). Overestimation of tumor size or diagnosis of multifocality that is clinically irrelevant has been the main criticism of MRI, in addition to its cost (9, 11, 13, 14). In our study, MRI visualized ILC more often than the conventional studies.
In the preoperative planning of breast cancer, the largest size obtained by imaging is an important factor in deciding which type of surgery is performed. Our results show that US often (60%) tends to underestimate pathological size of invasive breast cancer and in 10.3% of patients by 10 mm or more. The subjectivity in estimating size on US has been well documented. Several studies have suggested that it is important to include the “halo” or rim around invasive tumors in order to decrease the risk of underestimation of size (4, 15). This was not done in the patients in this study. Our results suggest that interpretation of US together with MGM can, to some extent, lower the risk of underestimation.
Although a few millimeter size difference between imaging and pathological size is unlikely to influence the type of surgery (mastectomy versus breast conservation), we hypothesized that this may have bearing when the over- or underestimation of size by imaging is equal to or more than 10 mm. When MRI overestimated size, the mastectomy rate was 65%, which was higher than the mastectomy rate for all patients in our unit. The effect of underestimation by MRI is not possible to estimate in this study since extent of operation was based on the modality showing the largest size.
Our unit had a low, 6%, reoperation rate after breast conservation surgery, and this is significantly lower than reported in a recent UK study (16), where the average reoperation rate was 18%, and preoperative MRI was used selectively. Explanations might be differences in microscopic margin assessment and differences in acceptable margin width as well as different indications for MRI preoperatively.
The clinical relevance of pathological multifocal disease, not diagnosed by preoperative imaging, is disputed. In one study on detailed sectioning of mastectomy specimens, additional tumor foci were present in 30%–63% of patients (17). While some studies suggest that leaving behind unidentified multifocal disease is the main cause of local recurrence after breast conservation surgery, others suggest that this may be irrelevant in the presence of postoperative radiotherapy (13, 18–22). In the COMICE trial, over 1600 patients initially suitable for wide local excision after MGM/US were randomized to have or not to have preoperative MRI (1). One of the main outcome measures was reoperation rate within 6 months, the hypothesis being that diagnosing multifocal disease preoperatively would possibly result in patients having more extensive local excisions with lower re-excision rates (23). The results showed no effect on reoperation rates after use of preoperative MRI, but an increase in mastectomy rates was seen. We believe that it is important to identify multifocal disease preoperatively. Almost half (43%) of our patients with multifocal disease were diagnosed with a preoperative MRI alone and two-thirds were treated with mastectomy.
In conclusion, routine use of MRI in breast cancer has its pros and cons. The main problems are erroneous diagnosis of multifocal disease and underestimation of tumor size. The main advantages of MRI are that it can identify a substantial portion of patients with multifocal disease and that it is less prone than US to underestimate tumor size.
Footnotes
Acknowledgements
The authors thank Oskar Hagberg at the Regional Cancer Center South, Sweden for statistical advice, Maríanna Garðarsdóttir for advice on radiology, and Margrét Agnarsdóttir for advice on pathology.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
