Abstract
Background:
Cardiac surgery–associated acute kidney injury is a frequent and serious postoperative complication of cardiac surgery and is associated with an increased risk of morbidity, mortality, and length stay. In this study, we hypothesized that persistent elevation in inflammation in the first 48 h might be a powerful predictor of clinical outcome. Our aim was to elucidate the usefulness of interleukin-6 and procalcitonin postoperative levels in predicting mortality and renal complications in cardiac surgery patients.
Methods:
A total of 122 cardiac surgery patients were enrolled. Procalcitonin and interleukin-6 concentrations were measured on the second postoperative day, and their levels were evaluated versus a number of conditions and endpoints.
Results:
Procalcitonin has a good predictive value for adverse renal outcome (p < 0.05). Interleukin-6 has a good predictive value for 30 days and overall mortality in cardiac surgery population (p < 0.05). We did not observe a significant difference in procalcitonin and interleukin-6 levels among patients with different types of surgery and different extracorporeal circulation time, but the levels of both the molecules increase significantly depending on number of transfusions received by patients (p < 0.01).
Conclusion:
We speculated that procalcitonin and interleukin-6 could be two effective biomarkers. There is a possibility of having a combined inflammatory multi-biomarker panel, with procalcitonin for predicting renal outcome and interleukin-6 for predicting mortality.
Introduction
Cardiac surgery–associated acute kidney injury (CSA-AKI) is a frequent and serious postoperative complication of cardiac surgery (CS) (1). Common contributors to CSA-AKI are coronary artery bypass surgery and valve replacement surgery with cardiopulmonary bypass (CPB), with consequently extended intensive care unit (ICU) stay, hospital stay, increased need for short- and long-term renal replacement therapy (RRT), progression of chronic kidney disease (CKD), and increased short- and long-term mortality (1). Several proofs exist to sustain that CSA-AKI is caused by a variety of factors, including exogenous and endogenous toxins, ischemia and reperfusion injury, inflammation, and oxidative stress (2). Hence, CSA-AKI is characterized by an abrupt deterioration in kidney function following CS as evidenced by a reduction in the glomerular filtration rate. Unfortunately, this deterioration may not be detected in the first 24–48 h using conventional monitoring method. Diagnosis criteria for AKI depend on serum creatinine (sCr) levels that are unable to differentiate prerenal from intrinsic AKI (3). Due to the difference in baseline characteristics and in surgery type, the incidence of CSA-AKI ranges from 8.9% to 39% based on RIFLE (Risk, Injury, Failure, Loss of kidney function, and End-stage kidney disease) or AKIN (Acute Kidney Injury Network) criteria (4, 5). Applying AKIN and RIFLE criteria, the mortality rate (hospital discharge or 30-day mortality) ranges from 3.8% to 54.4% in patients developing CSA-AKI and increases progressively with the degree of renal impairment (3, 6).
Of note, a number of studies examining the inflammatory response after CS demonstrated a strong increase in inflammatory markers in the first 24 to 48 h after CS. Furthermore, several studies demonstrated the rises in the pro-inflammatory cytokines, such as tumor necrosis factor (TNF)-α, interleukin-6 (IL-6) and interleukin-8 (IL-8) (7), and the correlation between the magnitude of cytokine response and the severity of organ injury (8).
In this study, we hypothesized that persistent elevation in inflammation in the first 48 h may be a powerful predictor of clinical outcome. Specifically, the scope of this study was to elucidate the usefulness of postoperative levels of IL-6 and procalcitonin (PCT) in predicting mortality and renal complications in CS patients.
Materials and Methods
Subjects
This study was conducted over a 5-month period in the CS Department, at St. Bortolo Hospital in Vicenza, Italy. All consecutive patients (>18 years) undergoing on-pump cardiac surgical procedure were eligible for this study. Patients with exposure to radiocontrast media in the 72 h preceding AKI were excluded. We excluded patients with the second postoperative day on Saturday and Sunday. A total of 122 CS patients were enrolled and their medical histories were reviewed. Clinical characteristics, comorbidities, Logistic European System for Cardiac Operative Risk Evaluation (EuroSCORE) (9), laboratory data, intervention characteristics, and parameters were recorded for all patients.
Subsequently, we identified patients who developed AKI due to CS during the course of hospitalization and were classified as CSA-AKI. CSA-AKI was defined according to the current classification system and to clinical findings (10). AKI was presumed to be related to cardiac dysfunction after exclusion of other possible causes of renal damage based on the review of the clinical course of the patients. AKI was defined by AKIN criteria (11). Applying AKIN criteria, the strict definition was used for increments of sCr within 48-h interval from in-hospital admission. We considered sCr level at in-hospital admission as the baseline value (preoperative value).
The procedures were in accordance with the ethical standards laid down by the Helsinki Declaration. All patients were informed about the experimental protocol and the objectives of the study before providing informed consent and biological samples. The protocol and consent form were approved by the Ethics Committee of San Bortolo Hospital.
Sample Collection
Peripheral venous blood samples were collected from all 122 CS patients into ethylenediaminetetraacetic acid (EDTA)-containing tubes at the second postoperative day. We choose this time point for sample collection to avoid spurious inflammatory response and the effects of the hemodilution related to the CPB pump prime on the biomarkers concentration (12, 13). Samples were subsequently centrifuged for 10 min at 1600 g. Following centrifugation plasma was carefully and immediately separated from blood cells and stored at −80 °C until use. All samples were processed within 1 h after collection.
Pct and Il-6 Evaluations
Quantitative analysis of PCT was performed using BRAHMS PCT sensitive KRYPTOR, (ThermoFisher Scientific, Hennigsdorf, Germany). The functional assay sensitivity is 0.06 µg/L and the direct measuring range is 0.02–50 µg/L.
The quantitative determination of IL-6 in the plasma was performed by the human instant enzyme-linked immunosorbent assay (ELISA) kit (eBioscience, San Diego, CA). Cytokine determination was performed according to the manufacturer’s protocol and instructions. Optical density was read by VICTOR™ X4 Multilabel Plate Reader (PerkinElmer Life Sciences) at 450 nm. The assay sensitivity is 0.92 pg/mL. The amount of IL-6 was calculated from the standard curve (ranging from 3.1 to 200 pg/mL) according to the manufacturer’s protocol. All tests were performed in duplicate.
Laboratory Parameters
sCr was measured by enzymatic method with an automatic analyzer (Dimension Vista; Siemens Healthcare, Tarrytown, NY, USA). White blood cells (WBC), hemoglobin (Hb), and hematocrit (Hct) were measured by the automated hematology analyzers XN 9000 (SYSMEX; KOBE, Japan).
Condition and Endpoints
PCT and IL-6 concentrations were measured on the second day after the CS and their levels were evaluated versus a number of conditions and endpoints: extracorporeal circulation (ECC) duration (four groups: no ECC, ECC less than 70 min, ECC 71–120 min, and ECC more than 120 min); type of surgery (coronary, valvular, coronary and valvular, and others); death (in hospital, 30-day death, and overall (= total death at 6 months after CS); length of stay in ICU; bleeding, number of transfusion (defined as the summary of the number of red blood cell package, platelet apheresis, and fresh frozen plasma units), and composite renal outcome (defined as AKI, need for RRT and/or worsening of CKD stage any time from admission to discharge).
Statistical Analysis
Statistical analysis was performed using the MedCalc® software package, version 13.2.2.0. Categorical variables were expressed as percentages; continuous variables were expressed as means ± standard deviation (normally distributed variables) or median and interquartile range (IQR) (non-normally distributed variables). The Kruskal–Wallis test or analysis of variance (ANOVA) was applied to compare groups, as appropriate. The area under the receiver operating characteristic (ROC) curves (AUC) was compared to predict the prognostic power of each variable. A p-value of <0.05 was considered statistically significant.
Results
The median age of all 122 CS patients was 67.0 years (IQR: 59.0–75.5) and 83 (68.0%) were male. The median Logistic EuroSCORE was 4.27 (IQR: 2.2–8.39). The baseline demographic, surgical, and clinical characteristics of the patients are shown in Table 1, and pre- and postoperative laboratory data are shown in Table 2.
Baseline demographic, surgical, and clinical characteristics.
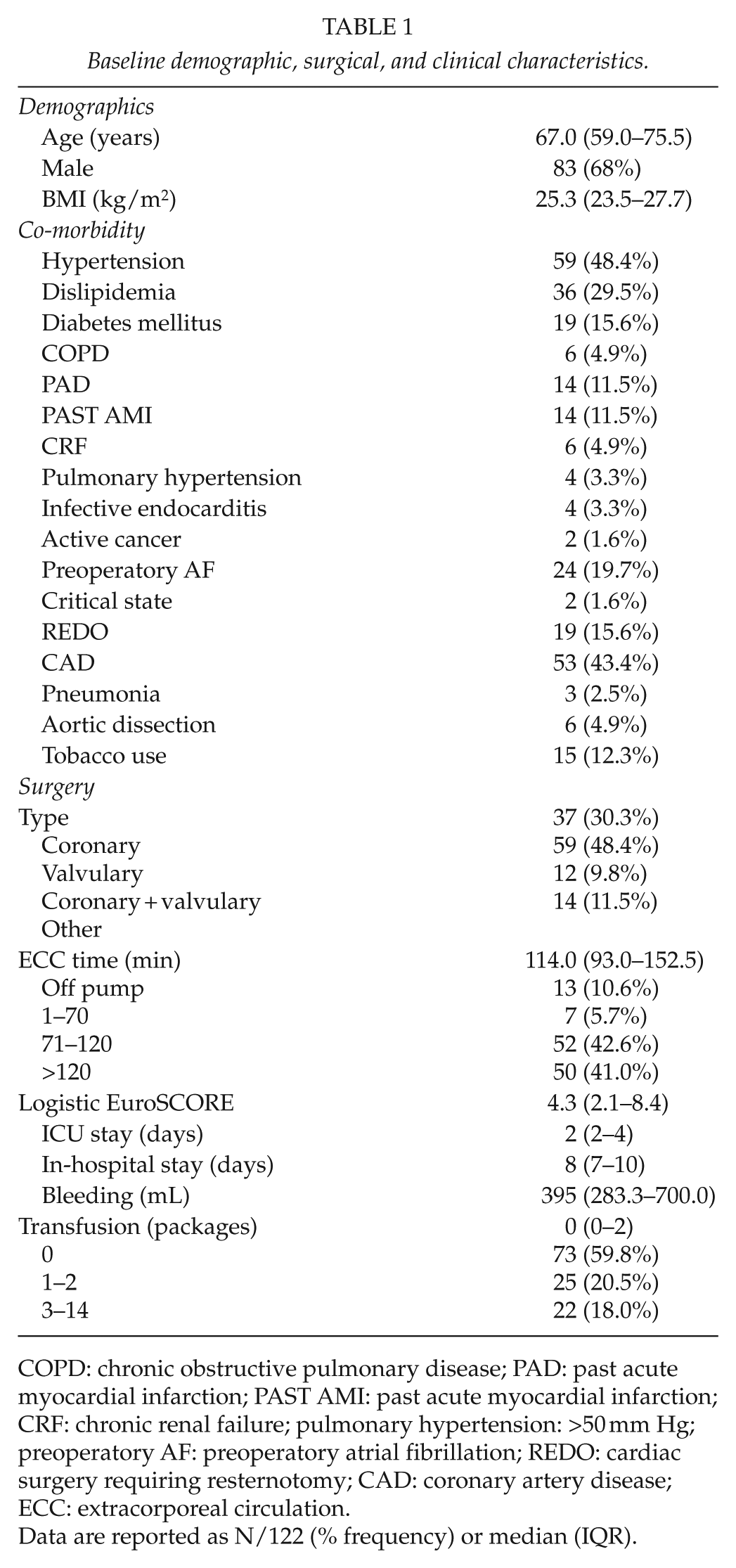
COPD: chronic obstructive pulmonary disease; PAD: past acute myocardial infarction; PAST AMI: past acute myocardial infarction; CRF: chronic renal failure; pulmonary hypertension: >50 mm Hg; preoperatory AF: preoperatory atrial fibrillation; REDO: cardiac surgery requiring resternotomy; CAD: coronary artery disease; ECC: extracorporeal circulation.
Data are reported as N/122 (% frequency) or median (IQR).
Pre- and postoperative values for complete blood count and sCr.
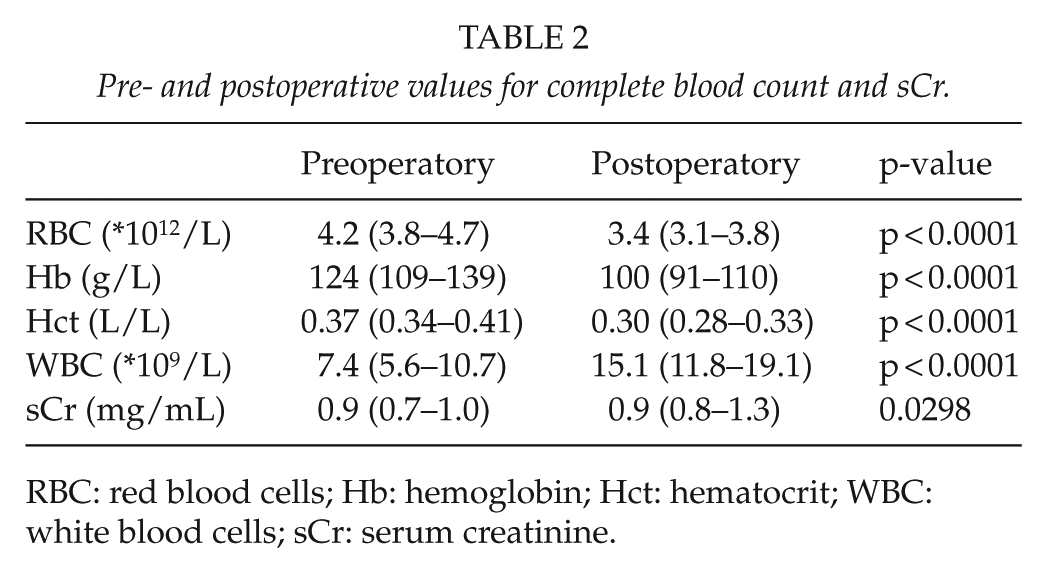
RBC: red blood cells; Hb: hemoglobin; Hct: hematocrit; WBC: white blood cells; sCr: serum creatinine.
We classified patients in groups according to different parameters. Types of surgery were classified into four mutually excluding groups: 37 (30.3%) patients underwent coronary surgery, 59 (48.4%) to valvulary procedures, 12 (9.8%) to coronary + valvular procedures, and 14 (11.5%) were subjected to other surgeries (Table 1). The median bleeding in the first 24 h after CS was 395 mL (IQR: 283.3–692.5). Patients who needed transfusion were 47/122 (38.5%). Of them, 25 (20.5%) needed 1–2 packages and 22 (18.0%) needed 3–14 packages. Median number of transfusions was 0 (IQR: 0–2) during in-hospital stay. Furthermore, we divided patients into four groups based on ECC duration: 13 (10.6%) patients had an off-pump surgery, 7 (5.7%) with an ECC time between 1 and 70 min, 52 (42.6%) with an ECC time between 71 and 120 min, and 50 (41.0%) patients with an ECC time longer than 120 min.
Median ICU stay was 2 days (IQR: 2–4), and median in-hospital stay was 8 days (IQR: 7–10). In-hospital mortality and 30-day mortality was 5.7% (seven patients) and overall mortality was 7.4% (nine patients).
We measured PCT and IL-6 levels at the second postoperative day: median PCT was 0.9 ng/L (IQR: 0.3–3.7) and median IL-6 was 15.6 pg/L (IQR: 8.4–31.5). In Table 3, we reported the values of PCT and IL-6 according to different parameters. The difference in PCT and IL-6 levels among the types of surgery and ECC duration is not statistically significant, but the levels of both the molecules increase significantly depending on number of transfusion received by patients (p < 0.001) (Table 3).
PCT and IL-6 levels classified for different ECC time, type of surgery, and transfusion needed.
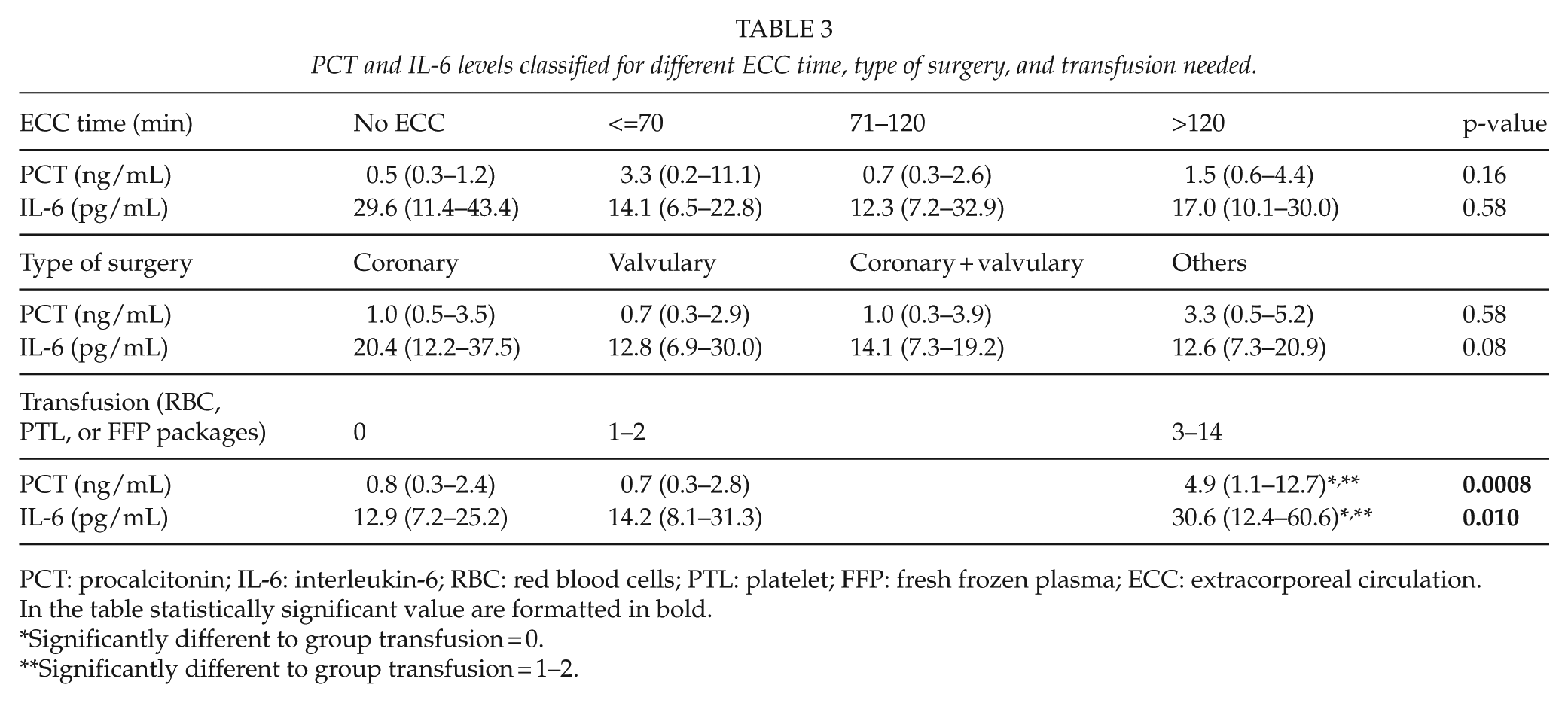
PCT: procalcitonin; IL-6: interleukin-6; RBC: red blood cells; PTL: platelet; FFP: fresh frozen plasma; ECC: extracorporeal circulation.
In the table statistically significant value are formatted in bold.
Significantly different to group transfusion = 0.
Significantly different to group transfusion = 1–2.
The relationship between biomarker concentrations and clinical outcomes was tested by ROC curve evaluation. Fig. 1 shows the AUC (95% confidence interval (CI)) for PCT and IL-6 on mayor clinical outcomes. AUC for IL-6 is significantly higher than the 1 for PCT at 30-day death and overall death (p < 0.05) (Fig. 1). The predictive value for an ICU stay longer than 5 days was 0.656 (0.564–0.740) for PCT and 0.611 (0.518–0.699) for IL-6, while AUC for ICU stay 10 days or longer is 0.806 (0.724–0.873) for PCT and 0.601 (0.507–0.689) for IL-6, without having a statistically significant difference among these two biomarkers. Neither PCT nor IL-6 concentrations seem to be good predictors of bleeding, estimated as the number of transfusions needed (AUC 0.605 and 0.598, respectively).
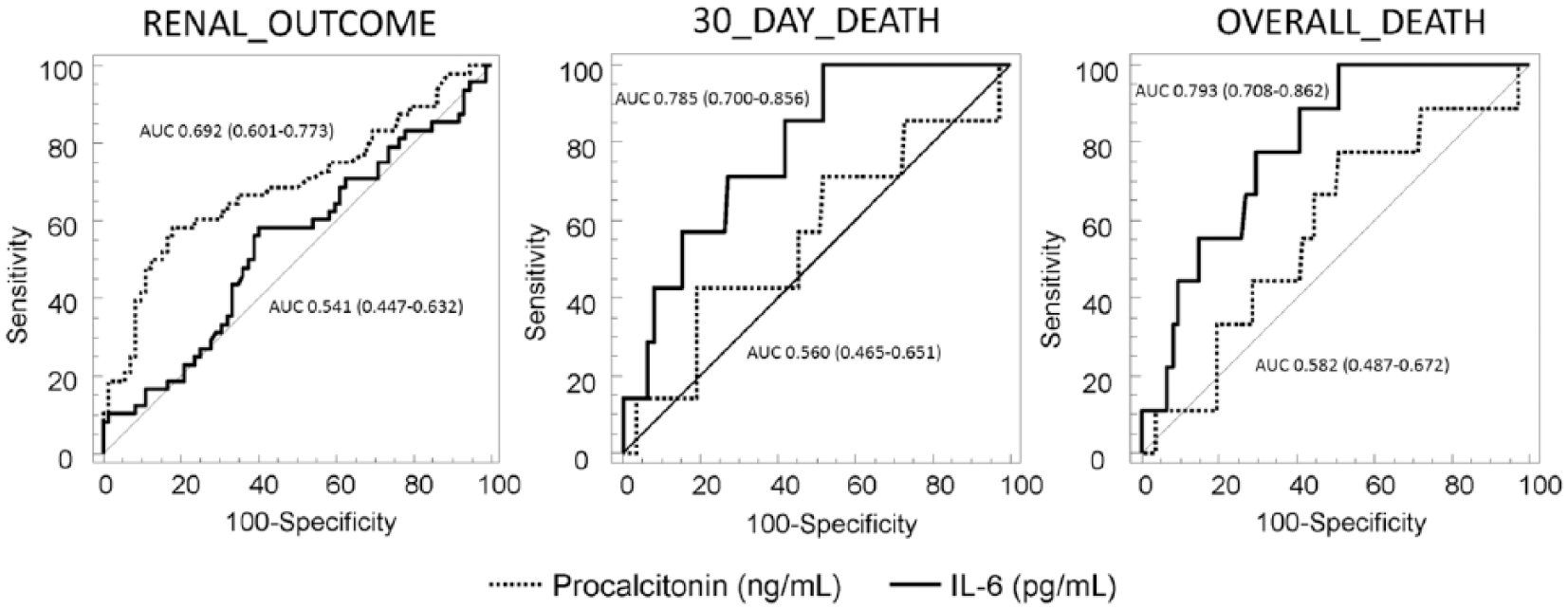
ROC curves examining PCT and IL-6 in predicting renal outcome, 30-day and overall death after CS.
CSA-AKI occurred in 28.7% (35 out of 122) of patients of whom 14.3% (5 out of 35) were treated with postoperative RRT before hospital discharge. Four out of five patients died in hospital and one out of five died in the postoperative 6 months. The progression of CKD stage occurs in 7 out of 122 patients (5.7%). Five patients moved from CKD stage 1 to 2 and two patients from stage 2 to 3. The adverse composite renal outcome, as previously defined, occurs in 42 out of 122 (34.4%) patients. The values of PCT and IL-6 in patients with and without CSA-AKI are reported in Table 4. PCT showed to be statistically higher in patients with AKI respect patients without AKI. Instead, IL-6 was statistically higher in patients who underwent RRT. In patients with composite renal outcome, PCT concentration was 3.3 (0.8–7.5) ng/mL and plasma IL-6 was 18.6 (8.8–35.8) pg/mL. Furthermore, in patients with progression of CKD stages, PCT was 0.8 (0.3–2.1) ng/mL and IL-6 was 20.3 (10.7–46.1) pg/mL. As show in Fig. 1, PCT has a stronger predictive value for adverse renal outcome (PCT: AUC of 0.692; 0.601–0.773, versus IL-6: AUC 0.541; 0.447–0.632, p < 0.05).
PCT and IL-6 values in patients with and without CSA-AKI.

PCT: procalcitonin; IL-6: interleukin-6; AKI: acute kidney injury; RRT: renal replacement therapy.
In the table statistically significant value are formatted in bold.
Discussion
In this study, we evaluated the usefulness of postoperative IL-6 and PCT in predicting mortality and renal complications in CS patients. Presently, we showed that PCT could be a novel biomarker in predicting renal outcome in patients after CS and IL-6 could be useful in predicting mortality in this population.
AKI is an important and common complication after CS and is an independent risk factor for a series of adverse outcomes. We described a 28.7% of AKI in our study population. In order to detect CSA-AKI at the early stage and to be able to promptly provide interventions, the advent of novel several biomarkers of AKI have opened a new era of early detection and prognosis prediction for AKI.
Largely, numerous studies in animals and humans have suggested that there is a significant inflammatory response that occurs after CS; consequently, many investigators have proposed that interventions to diminish the inflammatory response might improve outcomes after CS (14). Furthermore, acute systemic inflammatory response is considered as one of the key factors contributing to increased postoperative morbidity and mortality (15).
PCT has been reported to be a biomarkers associated with mortality in CS patients (16). In light of above, PCT levels have been shown to be correlating with several severity-of-illness scoring systems used in clinical practice, including APACHE II (Acute Physiology and Chronic Health Evaluation II), SAPS (Simplified Acute Physiology Score) II, and SOFA (Sepsis-related Organ Failure Assessment) scores (17). Additionally, PCT levels are also related to the development of postoperative complications, including postoperative myocardial infarction, Systemic Inflammatory Response Syndrome (SIRS), respiratory failure, need for positive inotropic support, renal failure, and hepatic failure (18). It is well established that CSA-AKI is associated with intrarenal and systemic inflammation. Accordingly to these data, in our study, PCT levels were statistically increased in patients who developed CSA-AKI after CS, independently from the type of surgery.
With regard to cytokines and inflammation, the results of this study suggest that IL-6 has a good predictive value for 30-day and overall mortality in CS population. Interestingly, this cytokine significantly increases in patients who required RRT. Furthermore, different studies suggested an association between the magnitude of the early inflammatory response and organ dysfunction, that is, plasma levels of IL-6 2 h after CPB correlated with cardiorespiratory dysfunction (19). Consistently, Wei et al. (20) reported a correlation between plasma levels of IL-6 at 5 min after cross-clamp release and myocardial injury. IL-6 is considered to be a major mediator of the acute phase response to CPB and in case of thoracic aorta and valve surgery (21). In 2011, Kim et al. (21) examined the relationship between early biomarker appearance (NGAL and multiplex cytokines) and organ injury at different time points in 38 CS patients. Among the 22 inflammatory biomarkers assayed, 14 had a statistically significant increase in the first 24 h compared to baseline. Peak levels occurred at 1 h in all biomarkers (exception for IL-1α and IL-12p70). Interestingly, IL-6, IL-10, IL-5, IL-18, and RANTES reached peak levels at 1 h postclamp release and remained elevated through 24 h. In particular, they observed that IL-6 and IL-10 levels were very significantly elevated. Furthermore, patients with higher IL-6 levels had a significantly longer ICU and in-hospital stay, more pulmonary failure, and resource utilization (21). Unfortunately, these authors did not analyze biomarker levels at 48 h.
We examined biomarkers at 48 h after CS; in fact, the inflammatory response immediately after CS may represent transient red cell injury and not reflect a generalized inflammatory response in the patient. Collecting samples after 48 h, we avoid the spurious inflammatory response and the effects of the hemodilution related to the CPB pump prime on the biomarkers concentration (12, 13). For this reason, the observed persistent elevation in inflammation in the first 48 h might be a good, powerful predictor of clinical outcomes, such as mortality and adverse renal outcome. With regard to biomarkers, hemodilution, decreasing blood viscosity, improving regional blood flow, changing plasma protein proportion, and changing blood volume may alter biomarkers’ evaluation.
We did not observe a significant difference in PCT and IL-6 levels among patients with different types of surgery and different ECC time. However, these data disagreed with Sponholz et al.’s (18) observations for PCT and Laffey et al.’s (22) comments for IL-6. Sponholz performed a systemic review of the literature with the aims of describing the evolution of serum PCT in CS patients and characterizing PCT variations in this population. Several factors, in fact, may influence the evolution of PCT levels after CS. These authors reported that uncomplicated CS induced a postoperative increase in serum PCT levels. Furthermore, the peak PCT levels are reached within 24 h postoperatively and return to normal levels within the first week. They declared that this increase seemed to be dependent on the specific surgical procedure and on intraoperative events (18). In particular, Franke et al. (23) observed higher PCT levels in patients after off-pump coronary artery bypass grafting (n = 42) than in those (n = 24) after off-pump coronary artery bypass surgery. Unfortunately, these authors did not analyze inflammatory biomarkers at 48 h, but they reported that IL-6 returned to baseline value on the third postoperative days. With regard to IL-6, no difference was found depending on surgery type (23). Moreover, Laffey et al. (22) reported that CS and the use of ECC lead to a more pronounced activation of cytokines and a strong inflammatory effect than other surgical procedures.
Interestingly, we observed an increased level of PCT and IL-6 in patients with higher transfusion need. Senay et al. (24) reported that the influence of red cell transfusion on the inflammatory response during CPB is very important; furthermore, the increase in inflammatory response during CPB leads to postoperative complications and is a risk factor for mortality and morbidity in CS patients (25). In addition, acute lung injury and adverse effects of fresh frozen plasma transfusion have been well described and reported (26, 27). Transfused blood may have immunomodulatory and proinflammatory effect; in this contest, Jiwaji et al. (28) reported in a randomized controlled trial that leukoreduced blood transfusion does not increase circulating soluble markers of inflammation, such as cytokines, L-selectin, and human neutrophil elastase.
In this study, we observed the utility and power of IL-6 and PCT to predict mortality and renal outcome in this small group; so, we speculated that these two biomarkers could be an effective tool in CS population: recently, the usefulness of biomarkers on stratifying mortality risk in the critical care setting has been assessed, either for a single biomarker or a biomarker panel. In our opinion, this study contributes to biomarker implication in clinical practice: both PCT and IL-6 were increased after CS, and these points are particularly relevant in the contest of the new concept of “renal angina.” This term has been proposed as an empirical clinical concept to better determine which patients are at highest risk for AKI; it is a combination of patients’ risk factors and subtle changes in sCr and urine output as a methodology to predict evolving AKI (29). In addition, the combination of renal angina concept with biomarkers could be the best and promising approach for the early and reliable detection of AKI (29). Unfortunately, although PCT and IL-6 are strictly related to AKI, ROC curve for both biomarkers were not so elevated to define them as “kidney angina biomarkers.”
We acknowledge certain limitations in our study; first of all, the small sample size. This is not a limitation in absolute terms, but related to the number of AKI, progression in CKD stage, and mortality, which is crucial for robustness of the statistical analysis. However, as the number of subgroup patients is small, the statistical power decreased. Furthermore, due to comparatively small sample size and the different subgroup analysis performed, the information is limited. Second, we only determined PCT and IL-6 levels at one time point. Serial measurements of plasma levels of PCT and IL-6 will be needed.
We hypothesized that a combination of PCT for predicting renal outcome and IL-6 for predicting mortality in an inflammatory multi-biomarker panel as opposed to a single biomarker should be taken as “add on-value” rather than a “unique-predictive” data. Furthermore, there is growing interest in defining the role of biomarkers in CS, and studies on PCT and IL-6 pharmacokinetics and clinical applications with day-by-day variations are still necessary to better understand the application of these two biomarkers in CS. Further study on the inflammatory process in CS population may provide clues into the possible therapies to reduce morbidity in this population.
These findings provide a further rationale for ongoing clinical studies focused on biomarkers and CS population. Earlier and correct identification of patient risk for organ injury, such as kidney, may allow for earlier intervention and reduce resource utilization. Further prospective studies on larger number of patients are needed to determine optimal CS techniques to reduce and prevent inflammation and CSA-AKI. Exciting developments on biomarkers’ front are forthcoming and clinical practice may soon be changing. Optimized biomarkers’ assessment can significantly impact time to therapeutic intervention and associated outcome.
Footnotes
Acknowledgements
The authors express their gratefulness to the medical, nursing, and secretarial staff from the Cardiac Surgery Department, St. Bortolo Hospital, Vicenza. In particular, they express their gratefulness to Dr Hinna Danesi, Dr Salvador, and Dr Mucino-Bermejo for their contribution to the management of patients and data collection. The authors are grateful to Fiorenza Ferrari for her contribution to the revision of the paper.
Author Contribution
A.B. and G.M.V contributed equally; A.B. contributed to the conception of the study, interpretation of data, drafting the article, data collection and management, and laboratory analysis; G.M.V. contributed to the conception of the study, interpretation of data, and drafting the article; M.d.C. performed the laboratory analysis; D.G. contributed to the statistical analysis and revising the paper; M.C. performed the laboratory analysis; and C.R. provided intellectual content of critical importance to this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by Associazione Amici del Rene Vicenza (A.A.R.VI) ONLUS.
