Abstract
Objectives
We aimed to evaluate the association of interleukin-6 (IL-6) expression levels with stroke.
Methods
According to the set search strategy, we systematically screened relevant studies using PubMed and extracted study results regarding IL-6 from the literature for comprehensive quantitative analysis to explore the relationship between IL-6 level and stroke risk.
Results
This study included 15 publications with a total of 1696 participants, with 975 cases in the case group and 721 cases in the control group. Meta-analysis showed that IL-6 levels were significantly higher in the stroke population than those in the control group (standardized mean difference = 1.22, 95% confidence interval = 0.79–1.64). Subgroup analysis showed that there was no significant difference in heterogeneity for IL-6 detection methods between the two groups (I2 = 0, P = 0.47). The difference in heterogeneity test results regarding geographic region was statistically significant (I2 = 89.7%, P < 0.01). The results of heterogeneity testing for mean participant age were also statistically significant (I2 = 84.3%, P = 0.01).
Conclusion
The present study results showed that IL-6 may be significantly associated with stroke development.
Introduction
According to the 2019 Global Burden of Disease, Injury and Risk Factor Study, stroke was the second leading cause of death and disability worldwide in 2019, with more than 5.5 million people dying from the disease each year.1,2 More than 50% of survivors have severe disabilities, including loss of cognitive, language, and motor functions. 3 Many studies have shown that inflammatory factors are important in stroke progression and prognosis, with a concomitant increase or decrease in inflammatory factors at the onset of stroke, followed by a more pronounced progression of cerebral infarction and worse prognosis. 4 Currently, inflammation is attracting attention as a potential target for reducing stroke risk. Interleukin-6 (IL-6) is an important inflammatory mediator that stimulates leukocyte aggregation and thrombosis, leading to atherosclerosis and ultimately to stroke. Therefore, IL-6 may be strongly associated with stroke.5,6 Clarifying the mechanism between IL-6 levels and stroke will be beneficial to the development of anti-inflammatory treatment to prevent stroke. In this study, we conducted a systematic review of published research data on stroke and IL-6 using meta-analysis to increase the sample size and reduce the chance error to better explain the association between IL-6 and stroke. Our findings will be useful for the early prevention and treatment of stroke as well as in clinical practice.
Methods
Study procedures
This meta-analysis was conducted following the recommendations of the PRISMA 2020 guidelines. 7 Two investigators (X.H. & M.Z.) independent selected the articles for study inclusion and performed data extraction. Any discrepancies were resolved by consensus. This trial was not prospectively registered but it was retrospectively in registered INPLASY: registration number INPLASY202450089. Ethics approval and patient consent were waived owing to the nature of this systematic review and meta-analysis.
Search strategy
We searched the PubMed database for studies investigating the association between IL-6 and stroke, published between 1 January 1990, and 16 June 2023. We used a combination of the keywords stroke and interleukin 6; the detailed search strategy is provided in Table S1.
Eligibility criteria
Publications selected in the initial search were screened and assessed for eligibility according to the following inclusion criteria: (1) studies on the association between IL-6 and stroke; (2) case–control studies and cohort studies; (3) participants in the case group were diagnosed with stroke, and the control group comprised non-stroke patients including other hospitalized patients or individuals undergoing health checkups; (4) measurement criteria: serum IL-6 level, with the mean and standard deviation reported or the original data reported, which could be used to calculate the mean and standard deviation; (5) the original documents are all published documents. We excluded the following: (1) duplicated studies, (2) animal experiments, (3) non-English articles, (4) reviews, case reports, conference abstracts, and articles where the relevant data were unavailable.
Data extraction and quality assessment
Three authors independently extracted the following data from each eligible study: first author’s surname, year of publication, type of study, types of stroke, blood collection time, number of patients and controls, number of men, mean participant age, and interleukin-6 levels. The methodological quality of each study was independently assessed by three authors using the Newcastle–Ottawa Scale (NOS) for case-control studies. The NOS scale has a maximum score of 9, and studies with scores ≥7 are considered to be of high quality.
Statistical analyses
In this meta-analysis, statistical analyses were performed using the “meta” package in R 4.1.3 (www.r-project.org). We calculated the standardized mean difference (SMD) and 95% confidence interval (CI) to estimate potential associations between IL-6 and stroke, with P < 0.05 defined as statistically significant. I2 was used to evaluate inter-study heterogeneity. If the random effects model I2 was greater than 50%, the DerSimonian and Laird method was used for the analysis. Otherwise, a fixed-effects model (Mantel–Haenszel) was used for analysis. We also performed subgroup analyses based on participants’ geographic region and age, as well as the method used for IL-6 detection. The stability of the results was verified in sensitivity analysis. Publication bias was assessed using funnel plots and Egger regression tests. The Egger test is a statistical tool for quantitative evaluation of funnel plot asymmetry. P < 0.05 is considered statistically significant, indicating publication bias.
Results
Search results
Figure 1 summarizes the flow chart of the literature search and study inclusion/exclusion. After initially screening 1300 articles identified in the literature search, we finally included 15 articles. Based on the inclusion criteria, we included 975 patients with stroke and 721 without stroke in this meta-analysis.
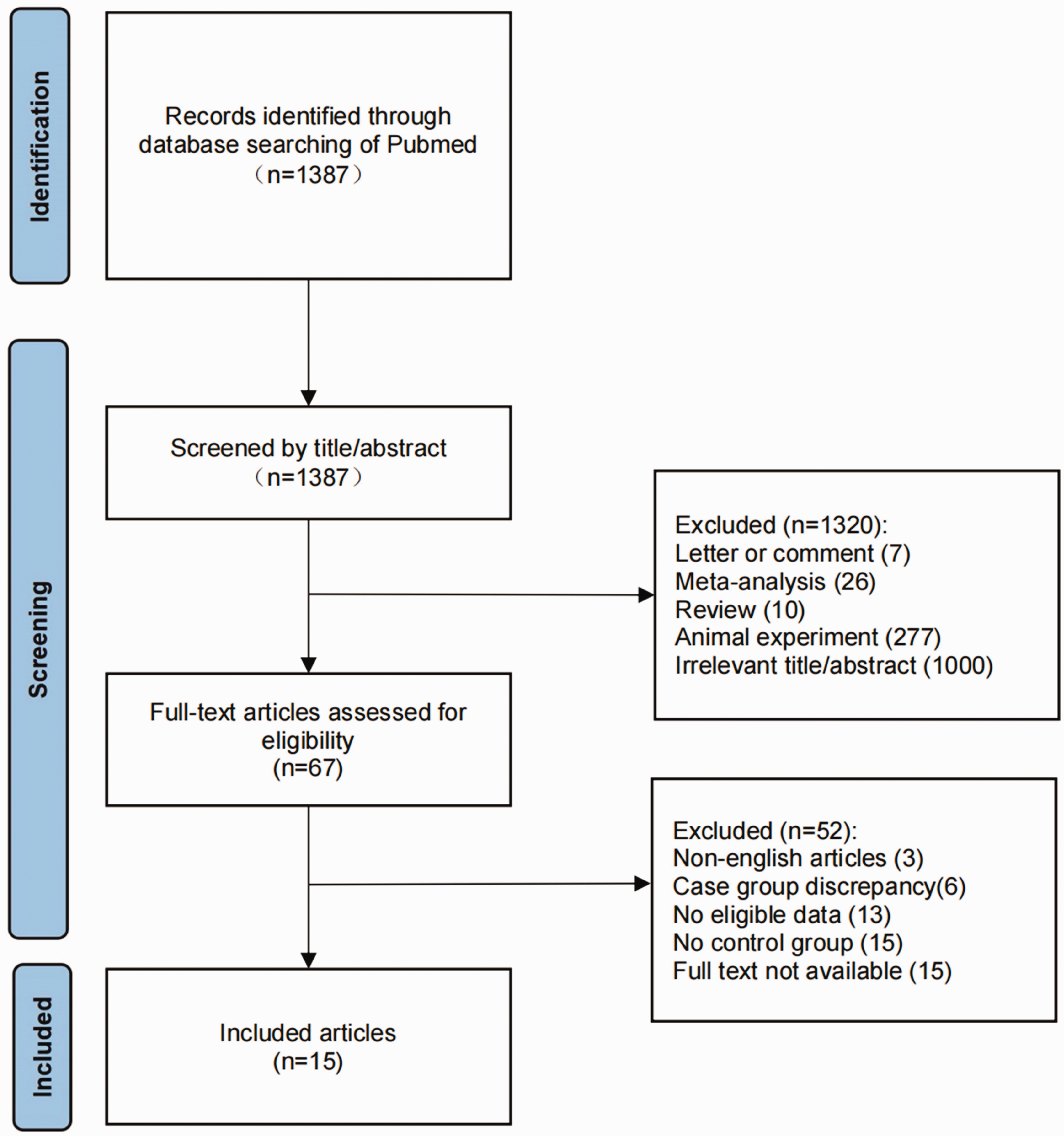
Flow chart of study retrieval and inclusion.
Study characteristics
Table 1 summarizes the basic characteristics of the studies included in the meta-analysis, including author, year of publication, cases/controls, age, sex, and NOS score. The mean participant age was 66 (35–68) years in the case group and 65 (61–71) years in the control group. There were 537 (55.08%) male participants in the case group and 352 (48.82%) male participants in the control group. Additional information on the study inclusion can be found in Table S2.
Features of the studies included in meta-analysis of the association between interleukin 6 and stroke risk.

NOS, Newcastle–Ottawa Scale.
Results of meta-analysis
The results of the 15 included studies showed I2 = 89%, P < 0.01, with significant statistical heterogeneity; therefore, a random effects model was used for meta-analysis. The results showed that IL-6 levels in patients with stroke were significantly higher than those in the control group; the difference was statistically significant (SMD = 1.22, 95% CI = 0.79–1.64), indicating that IL-6 levels were closely related to stroke (Figure 2).
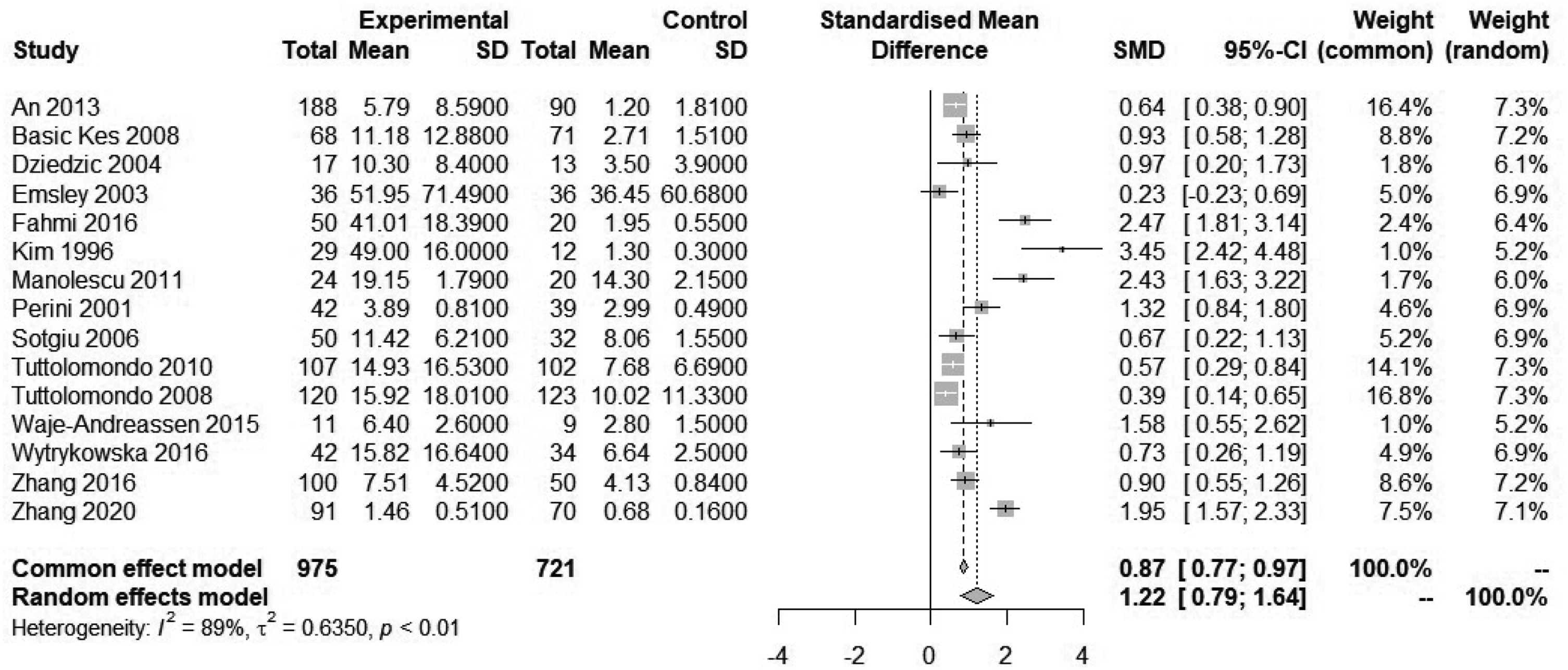
Forest map of the association between interleukin 6 (IL-6) and stroke risk.
Subgroup analysis
The 15 included studies were grouped according to detection method, region, and age for meta-analysis; the results are shown in Table 2.
Subgroup analysis of the association between stroke and interleukin 6.

SMD, standardized mean difference; CI, confidence interval; ELISA, enzyme-linked immunosorbent assay; CLIA, chemiluminescence immunoassay.
Based on subgroup analysis for IL-6 detection methods, there was no heterogeneity between the two groups (I2 = 0%), indicating that the assay method did not affect the results of the meta-analysis.
According to geographic region-based subgroup analysis, the heterogeneity among the three groups was high (I2 = 89.7%), suggesting that the source of the sample may affect the results of meta-analysis.
In age-based subgroup analysis, the heterogeneity test results were significantly different (age ≤65 years: I2 = 77%, P < 0.01, age >65 years: I2 = 90%, P < 0.01). At the same time, the proportion of participants with average age ≤65 years (SMD = 1.90) was significantly greater than the proportion with average age >65 years (SMD = 0.86), suggesting a younger-age trend in stroke.
Publication bias
We used funnel plots to evaluate potential publication bias (Figure 3). The shape of the funnel plots was asymmetrical (Egger’s P < 0.001), indicating possible publication bias.

Funnel plot of the association between interleukin 6 (IL-6) and stroke risk.
Sensitivity analyses
In the comparison of IL-6 levels, after removing the greatest-weighted data (An et al. 2013 8 ) (SMD = 1.26, 95% CI: 0.81–1.72) or the least-weighted data (Waje et al. 2005 9 ) (SMD = 1.20, 95% CI: 0.75–1.65), the results were basically consistent with those before removing these data, indicating that the combined results are basically reliable and stable (Figure 4).
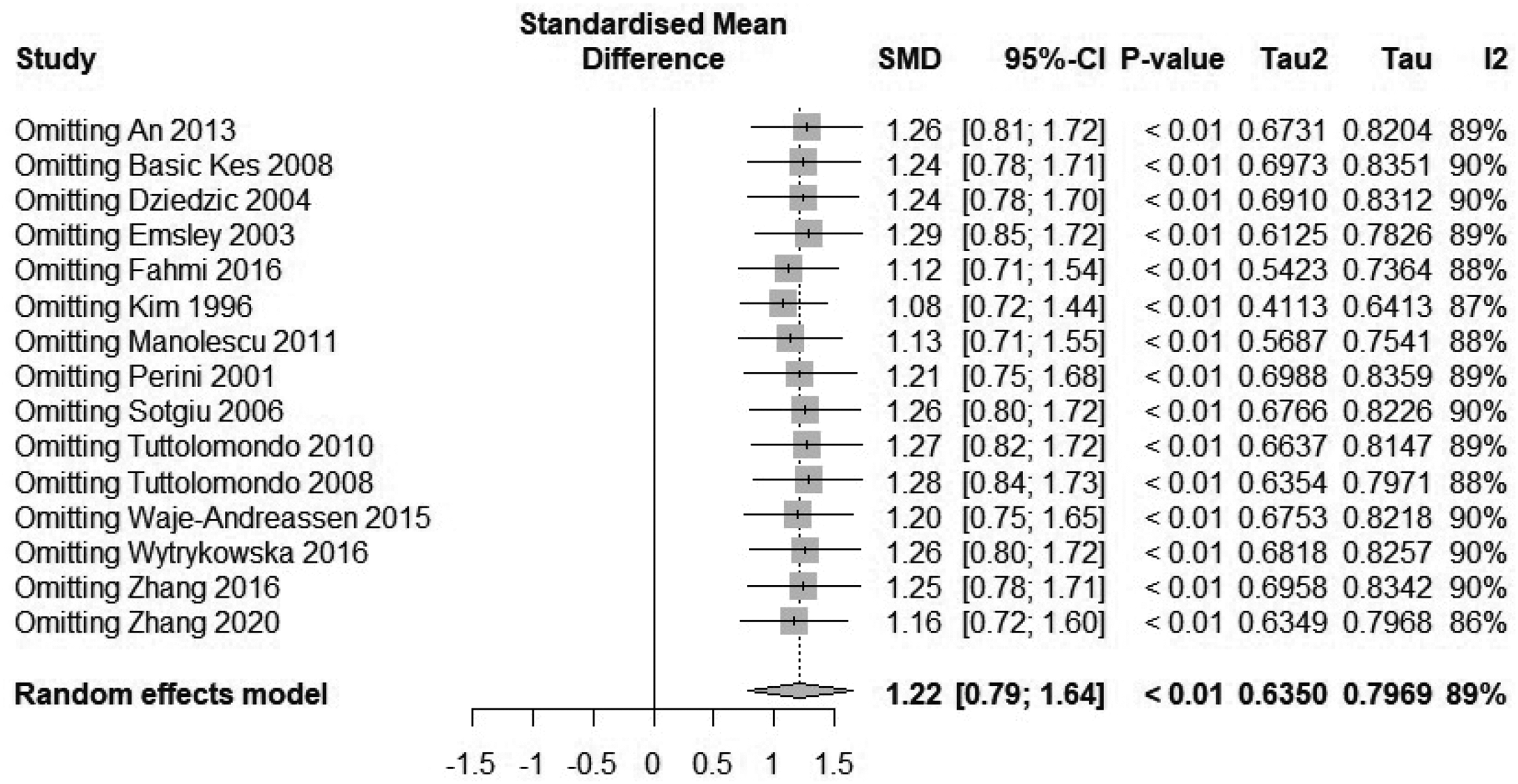
Sensitivity analysis of the correlation between interleukin 6 (IL-6) and stroke risk.
Discussion
In the present study, we evaluated 15 independent studies involving 1696 individuals, including 975 cases and 721 controls. Meta-analysis revealed an association between IL-6 concentrations and stroke, with higher concentrations in the case group than those in the control group, suggesting an important role of IL-6 in the progression of stroke.
Stroke is a neurological disorder characterized by blockage of blood vessels. Blood clots can form in the cerebral arteries, blocking blood flow and causing blood vessels to rupture, which leads to bleeding. 10 It has been found that the immune inflammatory response plays a key role in stroke pathology and is involved in the onset, development, and prognosis of patients with stroke. Immunological stress and chronic inflammation occur in the endothelial cells, releasing multiple inflammatory factors while increasing endothelial cell adhesion and promoting the adhesion and infiltration of inflammatory cells. This leads to the accumulation of lipids and foam cells, which in turn leads to the development of arterial obstruction. 11 The most direct cause of ischemic stroke is the rupture of atherosclerotic plaques. 12 The inflammatory state of peripheral blood is an important predictor of stroke. 13 Therefore, it is particularly important to test for inflammatory markers in people at risk for stroke.
IL-6 is an inflammatory factor found in a variety of tissues, which is produced by activated microglia, astrocytes, neurons, and peripheral immune cells. 14 IL-6 expression levels are low in serum, cerebrospinal fluid, and brain tissue under normal conditions, and overexpression is closely associated with the development and progression of inflammation, autoimmune diseases, and malignancies. 15 In the inflammatory response, IL-6 not only promotes accelerated lipid deposition and foam cell formation, it also stimulates vascular endothelial-cadherin phosphorylation, which causes vascular damage, induces cytotoxic T-lymphocyte activation, and promotes antibody production by B cells, thereby contributing to increased inflammation.16–18 The direct result of immune and inflammatory responses drives the formation and development of atherosclerotic plaques. 19 The main etiological pathogenesis of stroke involves the inflammatory response process in the cerebral vasculature. Thus, IL-6 may be associated with the risk of stroke development.20,21
Heterogeneity was high in this study (I2 = 89%, P < 0.01). In the subgroup analysis, for the different IL-6 detection methods, a high degree of heterogeneity was found within subgroups and no heterogeneity was identified between subgroups. This may be owing to the inclusion of fewer studies using chemiluminescence immunoassay, affecting the results of subgroup analysis. The different race and ethnicity of participants in different geographic regions may be a source of heterogeneity. However, the few studies conducted in African populations after stratification made it difficult to meet the requirements of subgroup analysis, which is a limitation of this study that should be addressed in the future. The different mean participant ages may also influence heterogeneity, with a significantly greater proportion of patients with stroke having a mean age <65 years (SMD = 1.90) than those with mean age >65 years (SMD = 0.86), suggesting a trend toward stroke at younger ages. There are many risk factors of stroke, including TOAST (Trial of ORG 10172 in Acute Stroke Treatment) classification and atrial fibrillation. The main cause, cardiogenic cerebral embolism, is also associated with inflammation, indicating that the relationship between IL and stroke subgroups warrants further study.22,23 The present findings may also be influenced by other heterogeneities, such as the timing of serum collection for IL-6 detection or TOAST typing; however, the specific source of heterogeneity in this meta-analysis could not be identified using the available information.
In this study, publication bias was found among studies using the Egger’s test. The reasons for heterogeneity and publication bias were analyzed by combining the studies. First, it is possible that the included case–control studies involved cases sourced from outpatients or inpatients, which may have been subject to selection bias. Second, the comparability between groups of important confounders across studies was unclear, which may have contributed to confounding bias. Finally, the small number of studies included in this study, the lack of inclusion of Chinese and other foreign-language literature, and the limited quality of the included English-language publications may have affected the reliability of the present results, and thus, the strength of the conclusions regarding the findings.
Apart from the analytic heterogeneity and publication bias described above, our study has several additional limitations. First, the limited number of studies and the fact that most were published prior to 2017 may have led to biased results. Second, the small sample size of included studies may limit the generalizability of our findings. Finally, we performed meta-analysis of all strokes without distinguishing whether they were acute or chronic and without TOAST typing, which remains to be further analyzed.
Currently, typical secondary strategies to prevent stroke are focused on hypertension, diabetes mellitus, and dyslipidemia whereas preventing inflammation is usually neglected. Current research suggests that IL-6 is associated with stroke. Therefore, stroke risk may be reduced by controlling levels of the inflammatory factor IL-6. Several studies have shown that lifestyle changes such as exercise, 24 weight loss, 25 and smoking cessation 26 can reduce IL-6 levels and help to prevent stroke. As for the treatment of stroke, Ridker 27 reported that atherosclerotic thrombosis can be treated by inhibiting the expression and activity of IL-6 using anti-cytokine drugs, thereby reducing brain tissue damage. In addition to this, Lambertsen et al. 28 suggested that specific inhibition of the IL-6 trans-signaling pathway using soluble glycoprotein 130 may be a promising therapeutic tool to investigate in future stroke research.
Conclusion
The present meta-analysis of 15 publications initially showed a possible association between IL-6 and stroke. However, this study has considerable heterogeneity and limitations. Further studies with larger samples and higher quality are needed to further investigate the association between IL-6 and stroke risk.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605241274626 - Supplemental material for Association between interleukin-6 levels and stroke: a systematic review and meta-analysis
Supplemental material, sj-pdf-1-imr-10.1177_03000605241274626 for Association between interleukin-6 levels and stroke: a systematic review and meta-analysis by Xinyi Huang, Manman Zhang, Jiaojiao Wang and Fuyong Hu in Journal of International Medical Research
Footnotes
Author contributions
Xinyi Huang conducted the study, performed a literature search, and determined the final included studies based on inclusion and exclusion criteria. Xinyi Huang, Manman Zhang, and Jiaojiao Wang extracted data from the included studies and assessed the study quality. Xinyi Huang analyzed the data and prepared a draft of the manuscript. Xinyi Huang and Fuyong Hu reviewed and revised the manuscript. All authors reviewed the findings and read and approved the final manuscript.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This work was supported by the Natural Science Foundation for the Higher Education institutions of Anhui Province [grant no. 2022AH051533]; and the Anhui Provincial Quality Engineering Project [grant no. 2021jyxm0942].
Supplementary material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
