Abstract
Background:
It is a widely held belief that intraoperative tumor perforation in colon cancer impairs survival and causes local recurrence, although the prognostic importance remains unclear.
Aim:
The aim of this study was to assess the effect of unintended intraoperative tumor perforation on postoperative mortality and long-term survival.
Material and Methods:
This national cohort study was based on data from a prospectively maintained nationwide colorectal cancer database. We included 16,517 colon cancer patients who were resected with curative intent from 2001 to 2012.
Results:
Intraoperative tumor perforation produced a significantly impaired 5-year survival of 40% compared to 64% in non-perforated colon cancer. Intraoperative tumor perforation was an independent risk factor for death, hazard ratio 1.63 (95% confidence interval: 1.4–1.94), with a significantly increased 90-day postoperative mortality of 17% compared to 7% in non-perforated tumors, p < 0.001. We showed that tumor fixation, emergency operations, and laparotomies were associated with an increased risk of intraoperative tumor perforation.
Conclusion:
This nationwide study demonstrates that intraoperative tumor perforation in colon cancer is associated with statistically significant reduced long-term survival and increased postoperative mortality.
Keywords
Introduction
Complications to colorectal cancer include tumor perforation, obstruction, and massive bleeding. Tumor perforation is a rare complication in colorectal cancer, with a reported incidence range of 1.6%–3.7% (1–6), and is believed to contribute to tumor cell spillage, implantation (7), higher rates of local and peritoneal recurrence (3, 4, 7), and poorer survival (2–5, 7, 8). Tumor perforation is associated with locally advanced cancer and usually occurs following tumor necrosis and an adjacent inflammatory reaction, although it could also occur as an iatrogenic injury during surgery. Tumor perforation can be described in the following three forms: free tumor perforation with purulent/feculent peritonitis, contained tumor perforation with abscess formation or fistulous connection to an adjacent organ or structure, and intraoperative tumor perforation with tumor perforation during operation or transected tumors (9). The distinction between free tumor perforation, contained tumor perforation, and intraoperative tumor perforation is important because the pathogenesis, management, and outcomes might be very different.
Most existing literature address free tumor perforation or spontaneous form with a reported incidence of 3.3%–7.0% in colon cancer (10–12). Studies have shown that free tumor perforation in colon cancer is associated with an increased 30-day postoperative mortality rate of 15.4%–19.0% (9, 11–13), a decreased 5-year survival of 14.0%–33.0% (10–12, 14), and an increased recurrence rate of 22.9% (13). Limited literature exists regarding intraoperative tumor perforation in colon cancer, and its prognostic importance has not yet been clearly defined.
The aim of this study was to examine the prognostic effect of intraoperative tumor perforations after curative-intent resection of colon cancer and to identify independent risk factors for death.
Material and Methods
The study design is a retrospective cohort study with data based on the prospectively maintained database of the Danish Colorectal Cancer Group (DCCG) (15).
We extracted data for all patients with primary colon cancer treated by surgical resection with curative intent between 1 May 2001 and 16 June 2012 from the DCCG database (n = 16,781). We excluded patients with rectal cancer, unresectable tumors, no surgery performed, Union for International Cancer Control (UICC) stage IV, bowel perforation, perforation due to endoscopic procedures, and non-curative surgeries. After crosschecking, we also excluded “unknown” tumor perforation status and patients lost to follow-up. To ensure all rectal cancers were excluded, we reviewed the pathology reports of the cases with procedure codes of “conventional abdominoperineal resection” and “resection of the rectum,” and we excluded additional 13 cases of rectal cancers. Fig. 1 summarizes the process of exclusion.
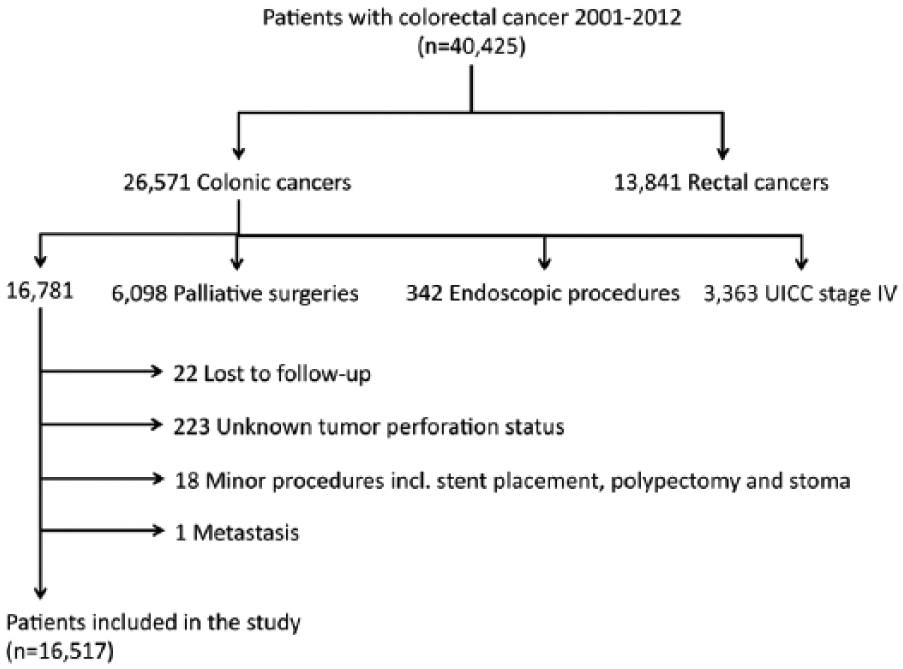
Excluded patients.
We crosschecked the data with the Danish National Patient Registry (16) and the Danish Pathology Data Bank (17) to ensure consistency and to add tumor perforations that had been wrongly classified. We searched for “perforation” in the Danish Pathology Data Bank among patients with a tumor perforation status recorded as “non-perforated” or “unknown,” resulting in 403 possible tumor perforations. We evaluated the pathology reports of these patients and requested and read the operation reports for the cases still not determined. This crosschecking revealed 124 additional cases of tumor perforation, which equated to 10% more than the 1225 tumor perforations that the surgeons had registered in the database.
Definitions
Colon cancer was defined as an adenocarcinoma located between 15 cm from the anal verge and up to the ileocecal valve, including the appendix.
Tumor perforation was defined as perforation at the site of the primary tumor, excluding bowel perforations close to the tumor or invasive growth. The pathologist identified tumor perforations as a transmural defects in the tumor, extending to either the surgical resection margin or through the peritoneal surface.
Intraoperative tumor perforation was defined as unintended tumor perforation during the operation. Intraoperative tumor perforation includes tumor perforation caused by mobilization of the tumor, incision into the tumor, or a transected tumor. Transected tumors were nonradical resected tumors. The surgeons reported intraoperative tumor perforations in the operation report and in the DCCG’s proforma.
Free tumor perforation was defined as feculent/purulent peritonitis. Contained tumor perforation was defined by abscess formation or a fistulous connection to an adjacent organ or structure.
A potentially curable tumor was defined as a potentially resectable tumor with no metastasis (UICC I–III). The definition of radical resection was an R0 resection with circumferential resection margins >1 mm. Nonradical resection was defined as micro- or macroscopic tumor growth at the resection margin, R1 or R2 resections.
In the database, laparotomies included converted laparoscopic procedures. We divided the 21 hospitals in Denmark into low-volume, medium-volume, and high-volume hospitals according to the number of colectomies per year. Low-volume hospitals performed <50, medium-volume hospitals performed 50–80, and high-volume hospitals performed >80 operations per year on average during the study period.
Data Source
The DCCG database is a nationwide clinical quality database. The database was established in 2001 by the DCCG (15) and is publicly financed. All patients in Denmark with a first-time diagnosis of colorectal adenocarcinoma are recorded. The database is prospectively maintained, and surgeons, pathologists, and oncologists register the data. The patient completeness of the database is 98.6%, and 97.8% of all surgical proforma have been recorded (15).
Denmark
In Denmark, people are identified by a unique civil registry number, which has been given to every Danish citizen by the Central Population Register since 1968 (18).
Great improvements have occurred during the past 10 years in chemotherapy, the surgical approach, and the centralization of colon cancer treatment. The adjuvant chemotherapy agents Oxaliplatin and 5-FU (flourouracil)/Leucovorin were implemented in 2005 for node-positive colon cancers (UICC III) and in 2009 for high-risk UICC stage II conditions, including tumor perforations. The laparoscopic approach in colon cancer was gradually implemented during the 2000s, resulting in use of laparoscopy in 6% of colorectal operations in 2005 and in 31% in 2008 (19). In the centralization of colorectal treatment in 2007, five regions replaced 13 counties, and the number of hospitals performing colorectal cancer surgery decreased from 49 to 20. In 2006, Denmark began training and certifying colorectal surgeons through a 2-year training program. Before 2006, 64% elective and 49% emergency colon cancer operations were performed by surgical gastroenterologists, and the remaining operations were performed by general surgeons and non-specialists (19, 20).
Statistical Methods
Statistical calculations were performed using SAS system version 9.4 (SAS Institute, Cary, NC, USA), except for Kaplan–Meier curves and the survival rates, which were calculated with SPSS software version 19 (IBM corporation). Survival analyses were conducted according to the Kaplan–Meier survival method, and pairwise comparisons between the groups were conducted using log-rank tests. The overall median survival time is presented with 95% confidence intervals (95% CI). The overall 30- and 90-day mortality and overall 5-year survival were obtained with life tables and compared using the χ2 test.
Cox regression analysis was performed to identify the independent risk factors for death. A test for proportional hazards was performed by visual inspection of the log(−log(survival)) versus log of survival time graph. Proper assumptions for the Cox model such as independency between the study subjects and independent censuring were all considered satisfied. Multivariate logistic regression was performed to identify risk factors for intraoperative tumor perforation versus non-perforated tumor using a backward stepwise approach. We performed a multivariate analysis on variables indicated as significant in the univariate logistic regression. The variables age, gender, UICC stage, surgeon specialization, hospital volume, and Charlson’s score were not significant in the univariate logistic regression. All the tests were two-sided, with p-values less than 0.05 considered statistically significant.
Results
Between 1 May 2001 and 16 June 2012, a total of 26,571 colon cancer patients were registered in the DCCG Database. A total of 16,517 colon cancer patients were included in the analysis (Fig. 1). Of the included patients, 15,178 (91.9%) had non-perforated tumors, and 1339 (8.1%) had tumor perforation, which included 467 (2.8%) free tumor perforation, 642 (3.9%) contained tumor perforation, and 230 (1.4%) intraoperative tumor perforation cases.
Patient, Tumor, and Operation Details
Table 1 shows the patient and tumor characteristics. The variable body mass index (BMI) was excluded due to a high amount of missing data (up to 61%). Intraoperative tumor perforation and contained tumor perforation cases were more likely to be fixated tumors. Table 2 shows the operative data. Most operations were elective, except in the free tumor perforation group. In the intraoperative tumor perforation group, 90% of operations were laparotomies, 76% were elective operations, and 67% were registered as radical resections. The intraoperative tumor perforation group did not have lower proportions of specialized surgeons than the other groups. Non-specialists operated on only 7% of non-fixated intraoperative tumor perforation cases, and only 5% were operated on at low-volume hospitals.
Patients and tumor characteristics.
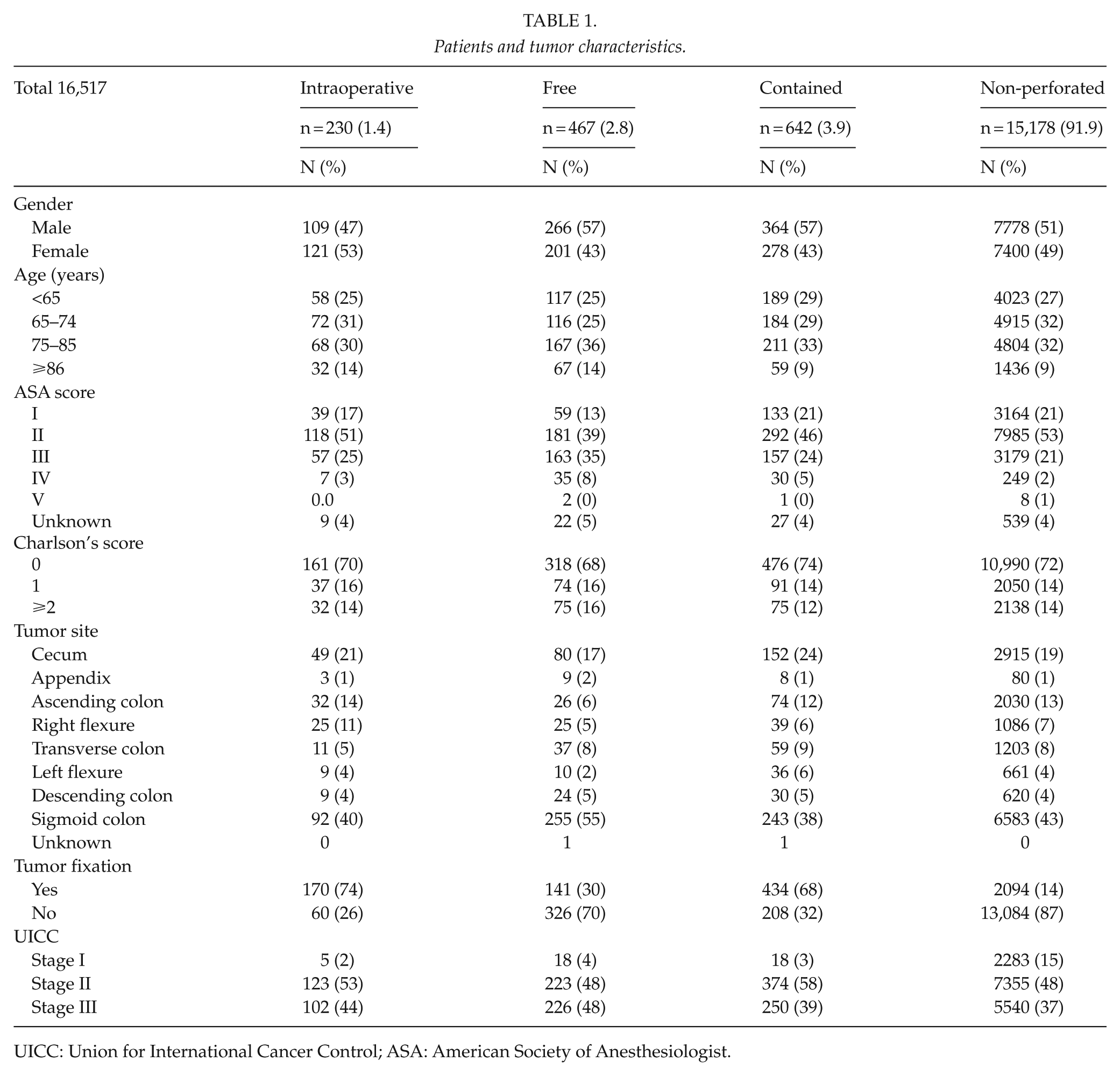
UICC: Union for International Cancer Control; ASA: American Society of Anesthesiologist.
Operative data.
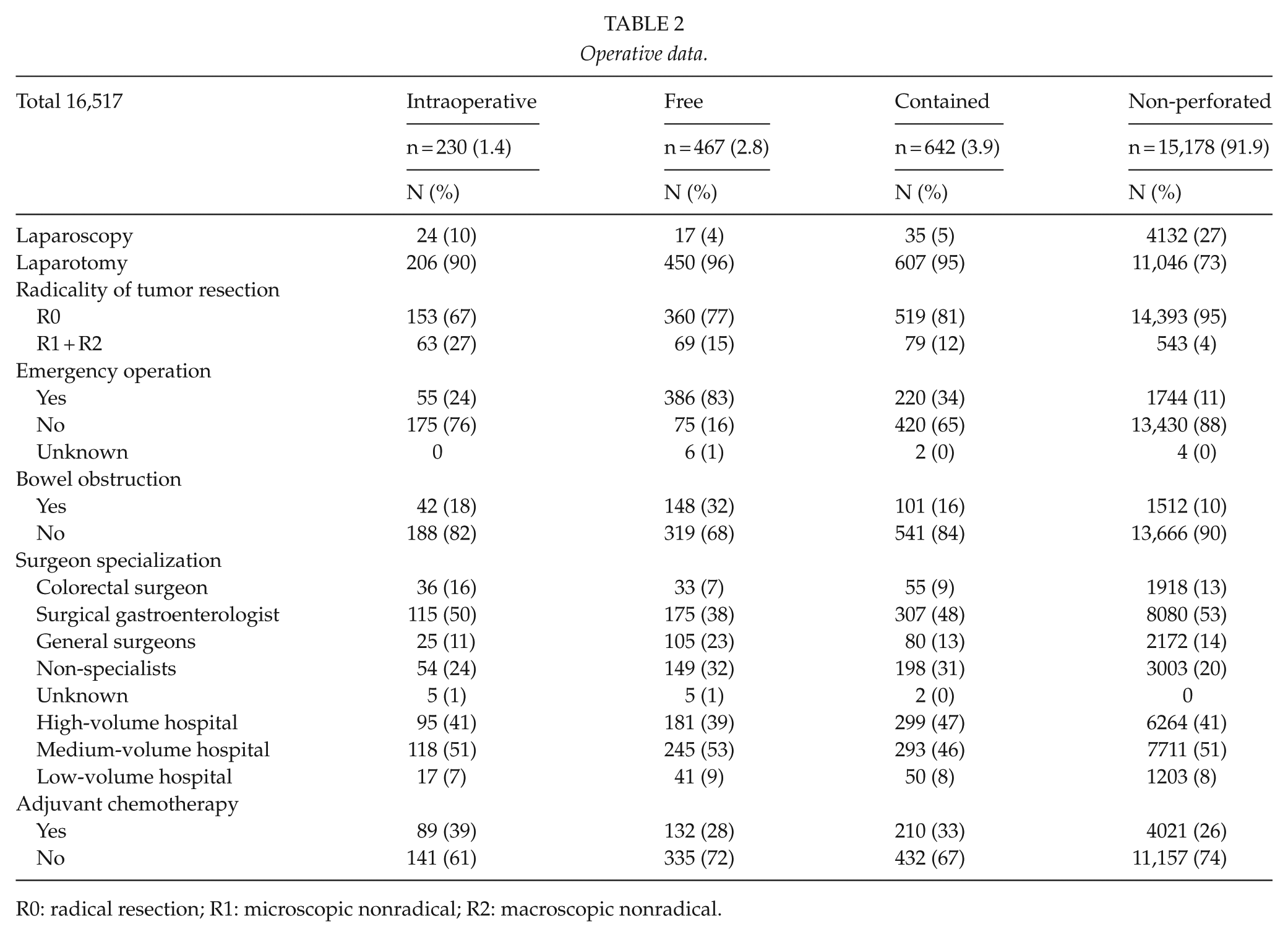
R0: radical resection; R1: microscopic nonradical; R2: macroscopic nonradical.
The frequency of intraoperative tumor perforation decreased significantly over the study period, from 137 (60%) before 2007 to 93 (40%) after 2007, p < 0.019.
After the implementation of adjuvant chemotherapy for high-risk tumors in 2009, the frequency of adjuvant chemotherapy increased significantly in every group from 34% to 52% in the intraoperative tumor perforation group, from 29% to 48% in the contained tumor perforation group, from 25% to 38% in the free tumor perforation group, and from 24% to 34% in the non-perforated tumor group, p < 0.001. The proportion of adjuvant chemotherapy in the tumor perforation groups after 2009 was highest in medium-volume hospitals (44%–55%) and lowest in low-volume hospitals (9%–15%).
Survival and Postoperative Mortality
Table 3 shows the survival and mortality rates. The overall 5-year survival rates were 40% for the intraoperative tumor perforation group, 28% for the free tumor perforation group, 42% for the contained tumor perforation group, and 64% for the non-perforated tumor group, overall log-rank p < 0.001. The 5-year survival remained decreased for the intraoperative tumor perforation cases after controlling for the 90-day mortality (48%) or when excluding the nonradical resected cancers (51%). Fig. 2 illustrates the overall survival for the four groups and the significantly reduced survival in the three perforated groups, most prominently in the free tumor perforation group. Fig. 3 illustrates the overall mortality for the four groups. The free tumor perforation group had the highest 30-day mortality of 26%, followed by the intraoperative tumor perforation group with 13%, the contained tumor perforation group with 10%, and the non-perforated tumor group with 5%, p < 0.001.
Data on survival and mortality.
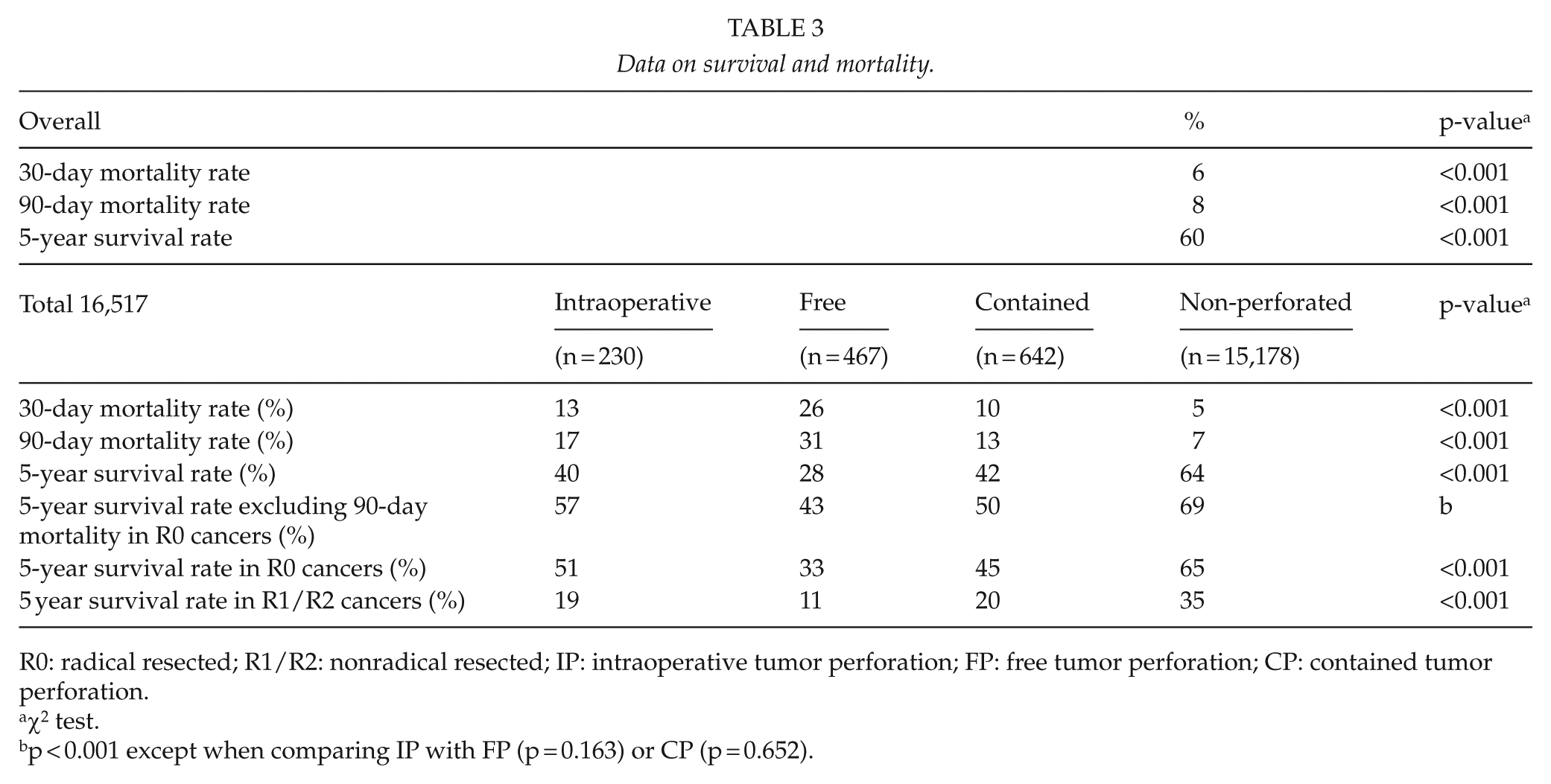
R0: radical resected; R1/R2: nonradical resected; IP: intraoperative tumor perforation; FP: free tumor perforation; CP: contained tumor perforation.
χ2 test.
p < 0.001 except when comparing IP with FP (p = 0.163) or CP (p = 0.652).
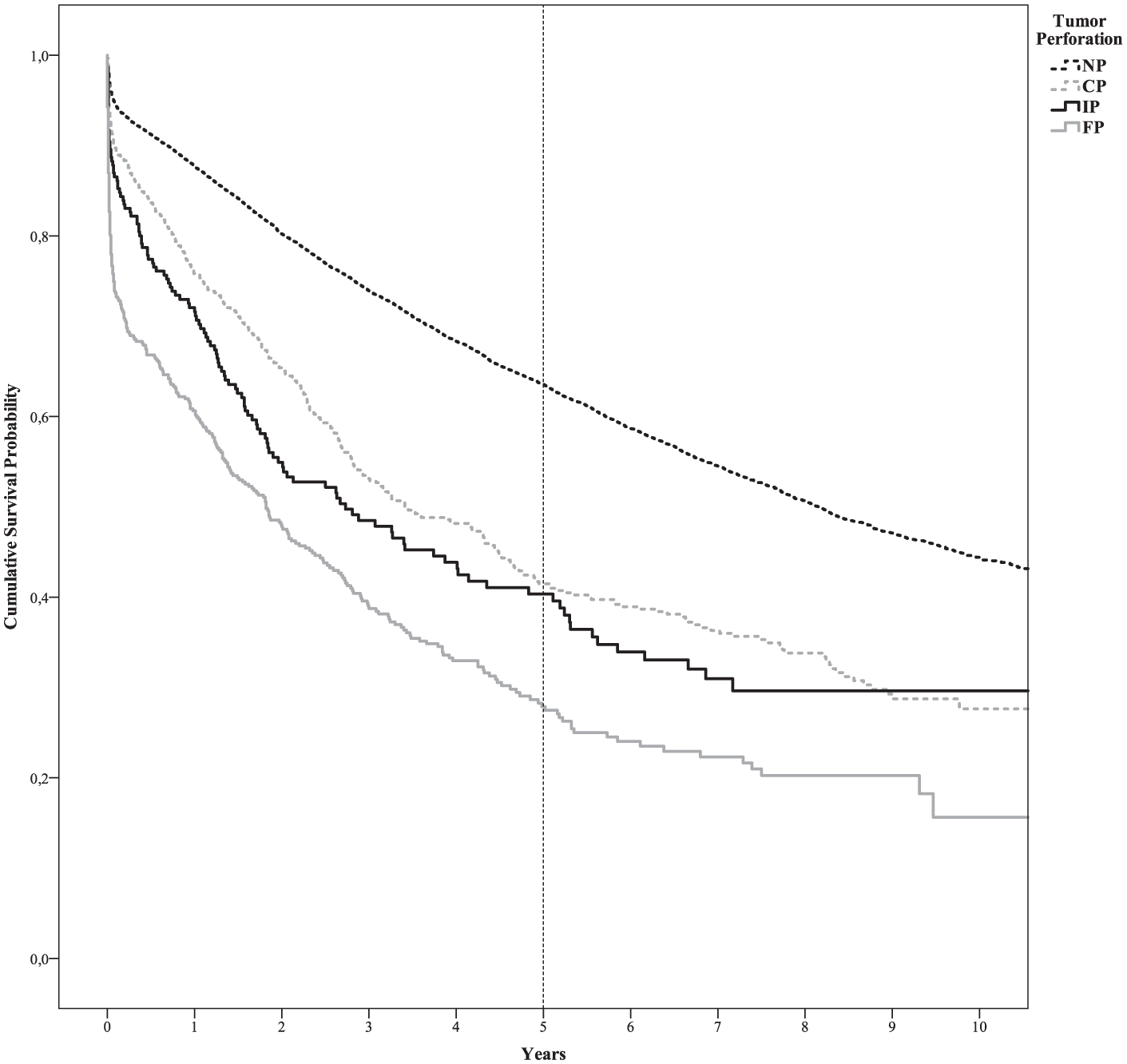
Kaplan–Meier curves of the cumulative survival probability in the four groups. The 5-year survival rates were 64% for the non-perforated tumor group, 42% for the contained tumor perforation group, 40% for the intraoperative tumor perforations group, and 28% for the free tumor perforation group (overall log-rank p < 0.001). Pairwise comparisons showed significant differences, except when comparing the contained tumor perforations with the intraoperative tumor perforations group (p = 0.118).
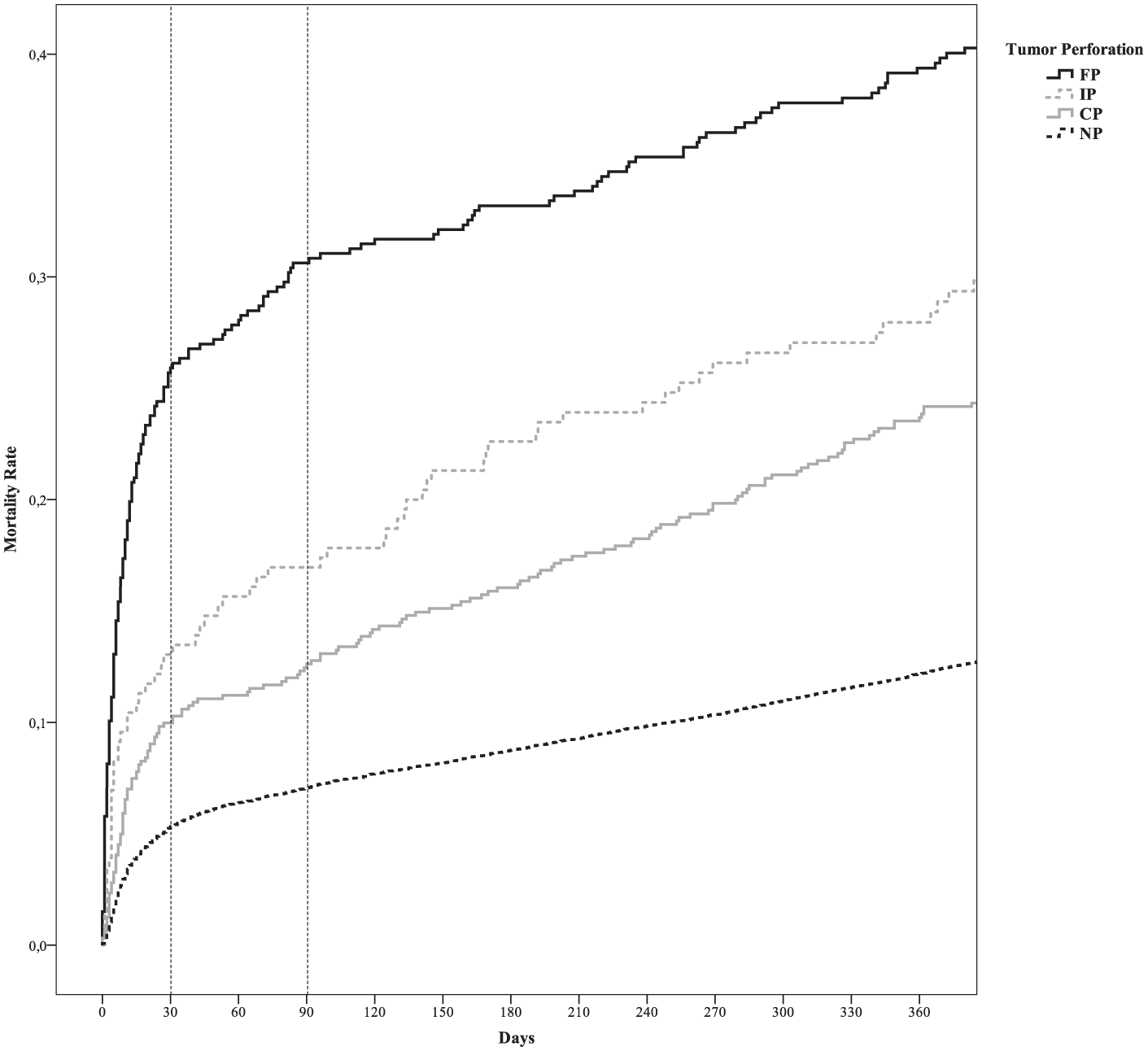
One-survival Kaplan–Meier analysis of overall mortality. The overall 30- and 90-day mortalities were 5% and 7% for the non-perforated tumor group, 10% and 13% for the contained tumor perforations group, 13% and 17% for the intraoperative tumor perforations group, and 26% and 31% for the free tumor perforations, respectively. Overall log-rank p < 0.001. Pairwise comparisons showed significant differences, except when comparing the contained tumor perforations with the intraoperative tumor perforations group (p = 0.118).
The Cox regression analysis showed that all types of tumor perforations, tumor fixation, and nonradical operations were independent risk factors for death. The multivariate logistic regression showed that tumor fixation, emergency operations, and laparotomies were risk factors for intraoperative tumor perforation (Table 4).
Cox regression analysis of factors associated with death and multivariate logistic regression of risk factors associated with intraoperative tumor perforation.
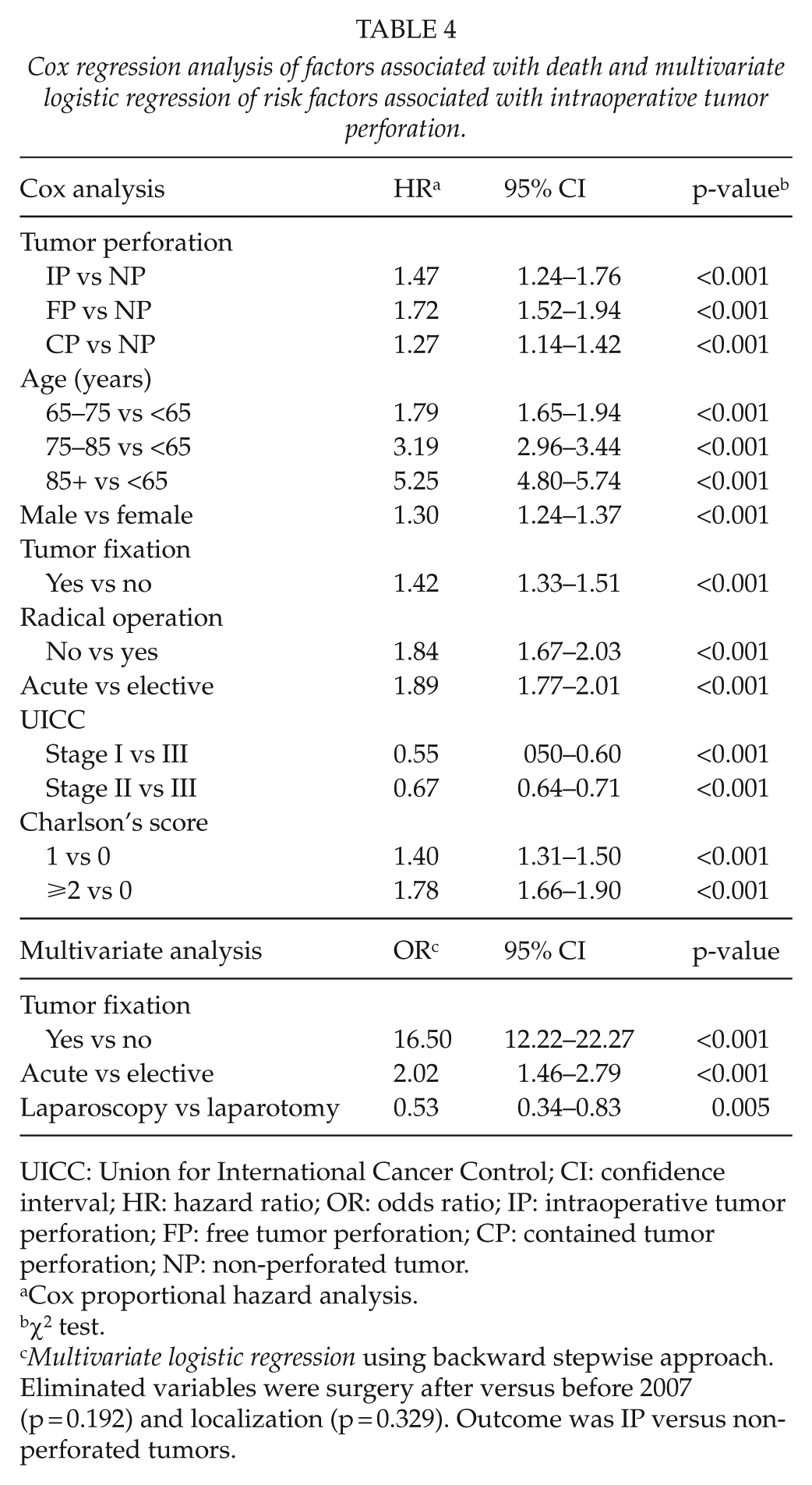
UICC: Union for International Cancer Control; CI: confidence interval; HR: hazard ratio; OR: odds ratio; IP: intraoperative tumor perforation; FP: free tumor perforation; CP: contained tumor perforation; NP: non-perforated tumor.
Cox proportional hazard analysis.
χ2 test.
Multivariate logistic regression using backward stepwise approach. Eliminated variables were surgery after versus before 2007 (p = 0.192) and localization (p = 0.329). Outcome was IP versus non-perforated tumors.
Discussion
The aim of this study was to assess the prognostic impact of intraoperative tumor perforation after curative-intent resection of colon cancer and to identify independent risk factors for death.
The main finding in this nationwide study of 16,517 colon cancer patients was a significantly impaired 5-year survival of 40% after intraoperative tumor perforation compared to 64% in non-perforated tumor cases. This study showed that intraoperative tumor perforation was an independent risk factor for death with a significantly increased 30-day mortality of 13% (compared to 5% in non-perforated tumor cases) and a 90-day mortality of 17% (compared to 7% in non-perforated tumor cases).
The incidence of tumor perforation in this study was 8%, which is higher than the reported range of 3.3%–7.0% for colon cancer (10–12, 21). This finding might be due to our thorough crosschecking process of data from multiple sources, which might have increased the numbers of tumor perforations.
The impaired 5-year survival in the tumor perforation cases might have been caused by different factors. First, some studies have suggested that the impaired survival in tumor perforation is exclusively due to the high postoperative mortality (4, 11). However, other studies (9, 12) show that the 5-year survival is still decreased in the tumor perforation group after controlling for the postoperative mortality. Our results supported this finding because the 5-year survival remained decreased at 48% for intraoperative tumor perforation cases compared to 68% in non-perforated tumor cases after excluding the 90-day mortality.
Second, the impaired survival could be due to local recurrence. This factor was not investigated in this study, although an older study from 1984 (7) showed an increased 5-year local recurrence rate of 80% in intraoperative tumor perforation cases compared to 10% in non-perforated Duke’s B stage colon cancer. However, these results were based on a small single-institution study.
Finally, studies have shown that nonradical resection is one of the strongest predictors of impaired long-term survival, which emphasizes the need to aim for complete resection with en-bloc multi-visceral resection involving en-bloc removal of the tumor and adjacent infiltrated organs (21, 22).
In this study, 27% of the intraoperative tumor perforation cases were registered as transected/nonradical resected tumors and were a risk factor for death. We showed a decreased 5-year survival of 19% in intraoperative tumor perforation cases registered as nonradical resected compared to 51% in radical resected intraoperative tumor perforation cases, p < 0.001. However, the surgeons reported the radicality until 2010, which might lead to misclassifications.
This study showed that tumor fixation, emergency operations, and laparotomies increased the risk of intraoperative tumor perforation. Computed tomography (CT) scanning has been shown to have high accuracy and a low rate of overstaging in the identification of fixated and locally advanced tumors (23, 24). Methods to identify the remaining 26% of non-fixated intraoperative tumor perforation cases remain unknown. However, the fixation does not fully explain the poor prognosis of intraoperative tumor perforation cases because both intraoperative tumor perforation and tumor fixation were shown as independent risk factors for death in the Cox regression.
Because intraoperative tumor perforation often occurs during elective operations, the surgeon’s expertize might be an important factor. A review by Iversen et al. and a Cochrane review by Iversen et al. (25, 26) and Archampong et al. (27) demonstrated that the surgeon’s sub-specialty as well as increasing experience significantly improved short- and long-term outcomes. Additionally, Iversen et al. demonstrated a significant relationship between high hospital volume and improved short- and long-term outcomes. However, Archampong was unable to find a similar significant effect when case-mix adjustment was taken into account, and the cut-off point varied considerably among the studies. In this study, neither the degree of specialization nor the hospital volume was found to be significant risk factors for intraoperative tumor perforation. Nevertheless, different confounders might be related to the outcome of high-volume hospitals. First, subspecialized colorectal surgeons are more likely to work in high-volume hospitals and have a higher caseload. Second, high-volume hospitals are more likely to have proper multidisciplinary teams, assistance from other specialties such as subspecialized pathologists and access to chemotherapy and radiotherapy. Third, the most complex elective cases are often sent to high-volume specialized centers.
A strength of this study is the large, consistent, and reliable dataset secured by three factors. First, the dataset includes every colon cancer patient from all Danish departments. Second, the dataset is a compilation of data from three different population-based national registries with exact data on death. Third, all data on tumor perforation have been manually crosschecked. In addition, the study has a homogeneous cohort created by excluding rectal cancer, metastasis, and non-curative operations in an attempt to avoid potential confounders. However, the study has some limitations. The registrations are based on daily practice and assessment in individual departments. Nevertheless, the patient completeness is 99.7% in the database, and the surgical departments are requested to correct any logical errors or missing data. As in other clinical databases, a possibility exists that rates of adverse intraoperative events might have been underestimated, misclassified, or incorrectly reported. However, we have mitigated this risk by crosschecking the pathology reports, which led to inclusion of an additional 10% of tumor perforation cases. When evaluating our survival results, the introduction of the laparoscopic approach, the centralization of surgery in 2007 in Denmark and the implementation of adjuvant chemotherapy in 2009 should be considered.
This nationwide study demonstrates that intraoperative tumor perforation in colon cancer is associated with statistically significant reduced long-term survival and increased postoperative mortality.
Footnotes
Acknowledgements
The authors thank the Danish Colorectal Cancer Group and data manager Dr Steffen Høgskilde, who extracted data from the databases of the DCCG, Danish Pathology Registry, and National Patient Registry. The Danish Data Protection Agency has approved this project. This work was presented in part at the annual meeting of Danish Society of Surgery, Copenhagen, Denmark, 13–15 November 2014.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by grants from Covidien to finance the data extraction and crosschecking of the data.
