Abstract
Background and Aims:
Perioperative myocardial infarction is an underdiagnosed complication causing morbidity, mortality, and considerable costs. However, evidence of preventive and therapeutic options is scarce. We investigated the incidence and outcome of perioperative myocardial infarction in non-cardiac surgery patients in order to define a target population for future interventional trials.
Material and Methods:
We conducted a prospective single-center study on non-cardiac surgery patients aged 50 years or older. High-sensitivity troponin T and electrocardiograph were obtained five times perioperatively. Perioperative myocardial infarction diagnosis required a significant troponin T release and an ischemic sign or symptom. Perioperative risk calculator was used for risk assessment.
Results:
Of 385 patients with systematic ischemia screening, 27 patients (7.0%) had perioperative myocardial infarction. The incidence was highest in vascular surgery—19 of 172 patients (11.0%). The 90-day mortality was 29.6% in patients with perioperative myocardial infarction and 5.6% in non–perioperative myocardial infarction patients (p < 0.001). Perioperative risk calculator predicted perioperative myocardial infarction with an area under curve of 0.73 (95% confidence interval: 0.64–0.81).
Conclusion:
Perioperative myocardial infarction is a common complication associated with a 90-day mortality of 30%. The ability of the perioperative risk calculator to predict perioperative myocardial infarction was fair supporting its routine use.
Introduction
Perioperative myocardial infarction (PMI) remains a severe and underdiagnosed complication causing morbidity, mortality, and substantial costs (1, 2). In-hospital mortality rates between 12% and 40% after PMI have been reported (3–6). Furthermore, a mere elevation of cardiac biomarker levels, irrespective of other ischemic features, has been shown to independently predict 30-day mortality (7, 8). PMI may also be the first manifestation of coronary artery disease (9) and has been associated with acute coronary syndrome demanding revascularisation (3, 8, 10). In addition to impaired postoperative outcome, PMI carries major economic burden. One PMI has been estimated to increase in-hospital costs by 6000€ (11).
The incidence of PMI has been investigated in several large-scale studies giving variable results between 0.3% and 36% depending on target population, study design, and the PMI definition used (4, 7, 12–15). Only 14% of these patients have typical chest pain and 53% of them exhibit clinical signs or symptoms of ischemia (3). Given the silent nature and high mortality rates of PMI, the revised global definition of myocardial infarction (MI) recommends a routine monitoring of modern cardiac biomarkers, namely, cardiac troponins, in high-risk patients, both prior to and 48–72 h after major surgery (16).
Because of the considerable prevalence and dismal prognosis of PMI, therapeutic trials on the identification and perioperative management of these patients are warranted. In order to conduct these trials, though, it is necessary to determine the true clinical picture of PMI. Patients should undergo systematic ischemia screening (17), current PMI definition must be used (16), and the perioperative cardiac risk must be accurately evaluated with a predictive risk calculator (18). Accordingly, we performed a prospective single-center study of patients undergoing unselected non-cardiac procedures aiming to define the incidence of PMI and to evaluate the predictive ability of a perioperative risk calculator to detect PMI in current clinical practice.
Methods
Ethical approval for this study (no. 11/13/03/02/2014) was provided by the Ethics Committee of the Department of Surgery in Helsinki University Hospital, Helsinki, Finland, on 30 January 2014 with a written informed consent preceding enrollment. We report the data according to the STROBE guidelines for observational studies (19).
Based on the incidence of PMI in earlier studies and desired width of confidence interval (CI) at 95% confidence level, we aimed to include 500 consecutive patients meeting the inclusion criteria. In order to ensure the representativeness of the cohort, we applied for an amendment to analyze the routine clinical data of the eligible patients, whose consent for systematic screening could not be asked, mainly due to challenges in off-hour recruitment (Fig. 1) of urgent operations. The ethical approval for an amendment was provided on 20 August 2014.
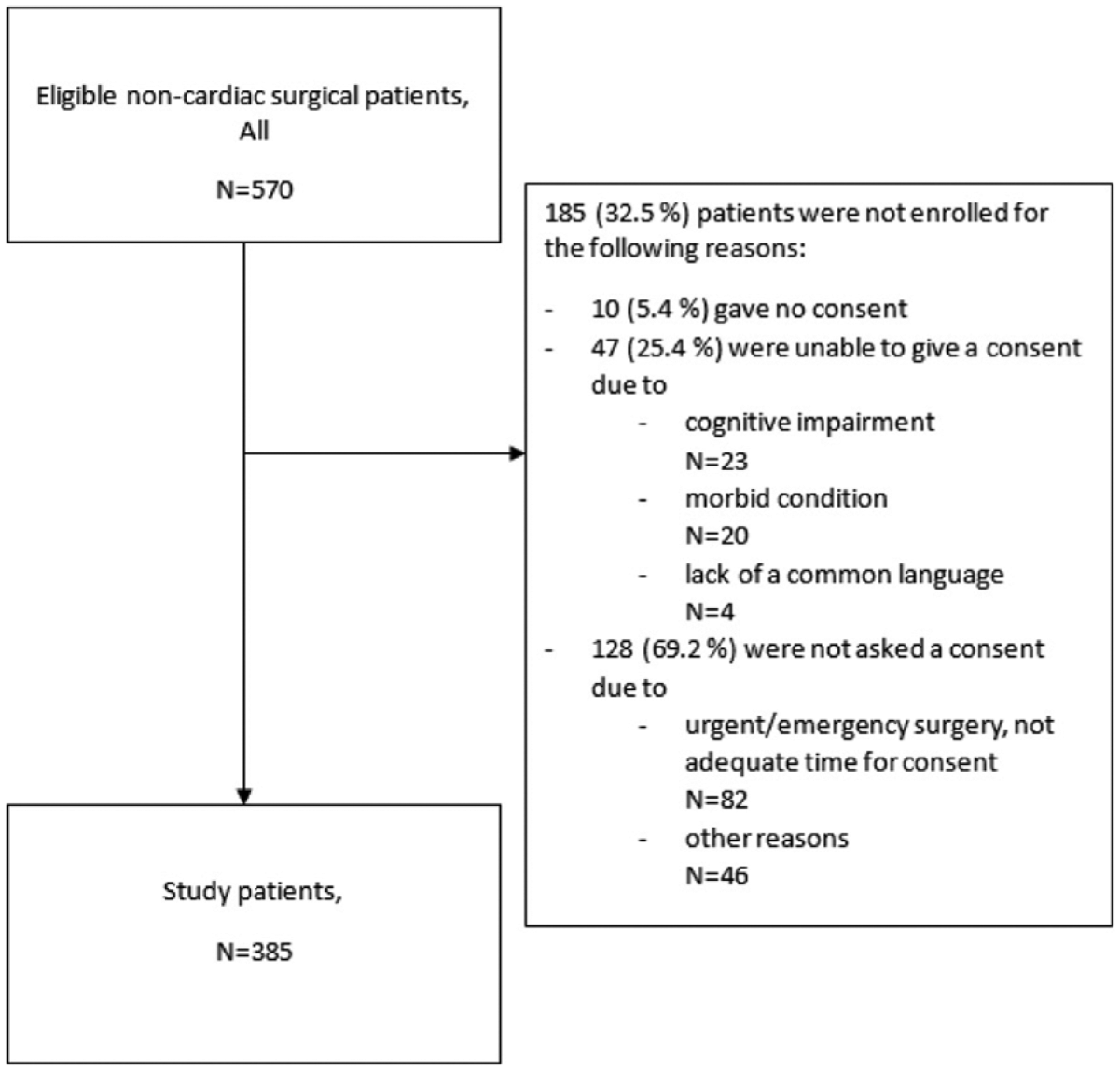
Patient flow.
Study Design
We performed a prospective study at the Meilahti Hospital, Helsinki, which provides 24/7 care for patients requiring cardiothoracic, vascular, or gastrointestinal surgery. We investigated the incidence of major postoperative cardiac events with systematic ischemia screening and the prediction of these events with a validated cardiac risk calculator (18).
Inclusion and Exclusion Criteria
Between 3 April and 19 June 2014, all consecutive non-cardiac surgery patients aged 50 years or older undergoing gastrointestinal, thoracic, or vascular surgery were considered eligible for the study. The ischemia surveillance was only conducted for the first operation for each patient. A written informed consent was obtained preoperatively from all eligible patients or their proxy. We had to exclude patients (1) who declined consent, (2) who were unable to give consent, and (3) whose consent could not be asked due to logistic reasons/emergency operation. The postoperative follow-up was conducted up to at least 90 postoperative days. Mortality dates were retrieved from the Finnish population registry center. Causes of death were determined by going through the postoperative medical records. All the deaths could be confirmed, there were no drop-outs.
Definitions
We defined perioperative period as the interval between the day preceding surgery until hospital discharge. The diagnosis of PMI was based on perioperative levels and changes of repeated high-sensitive fifth generation troponin T (hs-TnT; electrochemiluminescence immunoassay (ECLIA); Roche Diagnostics International Ltd, Rotkreuz, Switzerland) measurements and either ischemic electrocardiographic (ECG) changes or other ischemic features: (1) ischemic symptoms (chest pain, arrhythmias, and dyspnea), (2) cardiac imaging evidence of myocardial infarction, or (3) autopsy findings of acute or healing myocardial infarction. To establish the diagnosis of PMI, a rise and/or fall of TnT with at least one value above the upper reference limit (14 ng L−1) and either ischemic ECG changes or other above-mentioned ischemic features were required (16, 20, 21). A cardiologist (J.V.) analyzed the ECGs of the patients with a TnT concentration of ⩾14 ng L−1 and determined those with new ischemic changes (significant ST-elevations in two contiguous leads, significant ST-depressions in two contiguous leads, T-inversions in two contiguous leads, and a new left bundle branch block) (16). Other medical conditions potentially causing elevated TnT concentration (sepsis, acute kidney injury (AKI), and stroke) were recorded and considered when establishing the diagnosis of PMI. We defined PMI and the 90-day mortality as primary outcome measures (22). Secondary outcome measure was the performance of perioperative cardiac risk calculator.
Data Collection
To obtain patients’ medical history, characteristics, medication, physiological, and surgical data, we used electronic medical records (Uranus 8.4.3; CGI Group, Montreal, QC, Canada and Caresuite 8.2; Picis, Wakefield, MA, USA) to prospectively fill an electronic case record form (CRF) created for this study. A blood sample for TnT and an ECG was obtained five times: preoperatively, 6 h postoperatively, and on the first, second, and third morning after surgery or until discharge. We also recorded ischemic symptoms and signs (chest pain, hypotension, arrhythmias, and dyspnea). Perioperative cardiac risk evaluation using the cardiac risk calculator was performed in retrospect, thus the values had no impact on monitoring or treatment decisions. Additional ECG or TnT recordings were ordered only at the clinicians’ discretion. The study group did not interfere with the treatment of the patients. However, J.V. formulated a local recommendation on PMI management based on recent national guidelines (23). This document was distributed at the beginning of the study to all clinicians involved in the treatment of surgical patients. J.V. analyzed the ECGs blinded on patients’ 90-day survival status. In the retrospectively investigated standard care cohort, TnT analyses and ECG recordings were on the discretion of the clinician.
Statistical Analyses
We compared nonparametric data with the Mann–Whitney U-test and categorical variables with χ2 test or Fisher’s exact test. We present data as medians with interquartile ranges (IQRs) or as absolute numbers (percentage with 95% CIs). To determine the predictive ability of the cardiac risk calculator, we calculated areas under receiver operating characteristics curves (AUC) with 95% CIs. The best cut-off points for calculated cardiac risk were identified with the Youden index and these cut-off points were used to calculate sensitivity, specificity, and positive likelihood ratios (LR+). We present Kaplan–Meier survival curves for those with and without PMI using with Mantel–Cox log rank test. All analyses were performed using SPSS version 22 (SPSS; Chicago, IL, USA).
Results
During the enrollment period, 570 eligible patients underwent a non-cardiac operation. The patient flow is shown in Fig. 1. Altogether 385 patients (172 vascular, 80 thoracic, and 133 gastrointestinal) gave their consent and formed the study cohort. The demographics of the 175 patients who were unable to give consent or whose consent could not be asked are presented in an ESM Table.
Cohort with Systematic Ischemia Screening
Incidence of PMI
A significant postoperative TnT elevation was discovered in 75 (19.5%) patients and 27 (7.0%) had PMI (Table 1). All the PMIs were of ST-depression type, no ST-elevation myocardial infarctions (STEMIs) were seen. The incidence was highest in patients undergoing vascular surgery (19 of 172 (11.0%) versus 8 of 133 (6.0%) and 0 of 80 (0%) in gastrointestinal and thoracic surgical patients, respectively; p < 0.01). Patients with PMI had more cardiovascular morbidity, higher American Society of Anaesthesiology (ASA) score, higher preoperative TnT, lower nadir hemoglobin (Hb) during hospitalization, and larger vasopressor load during the day of surgery. Of note, 182 (47.3%) patients had a preoperative TnT elevation. The preoperative TnT level was significantly higher in patients who later suffered PMI (p < 0.001).
Characteristics of the ischemia surveillance patients (n = 385) stratified by the presence of PMI.
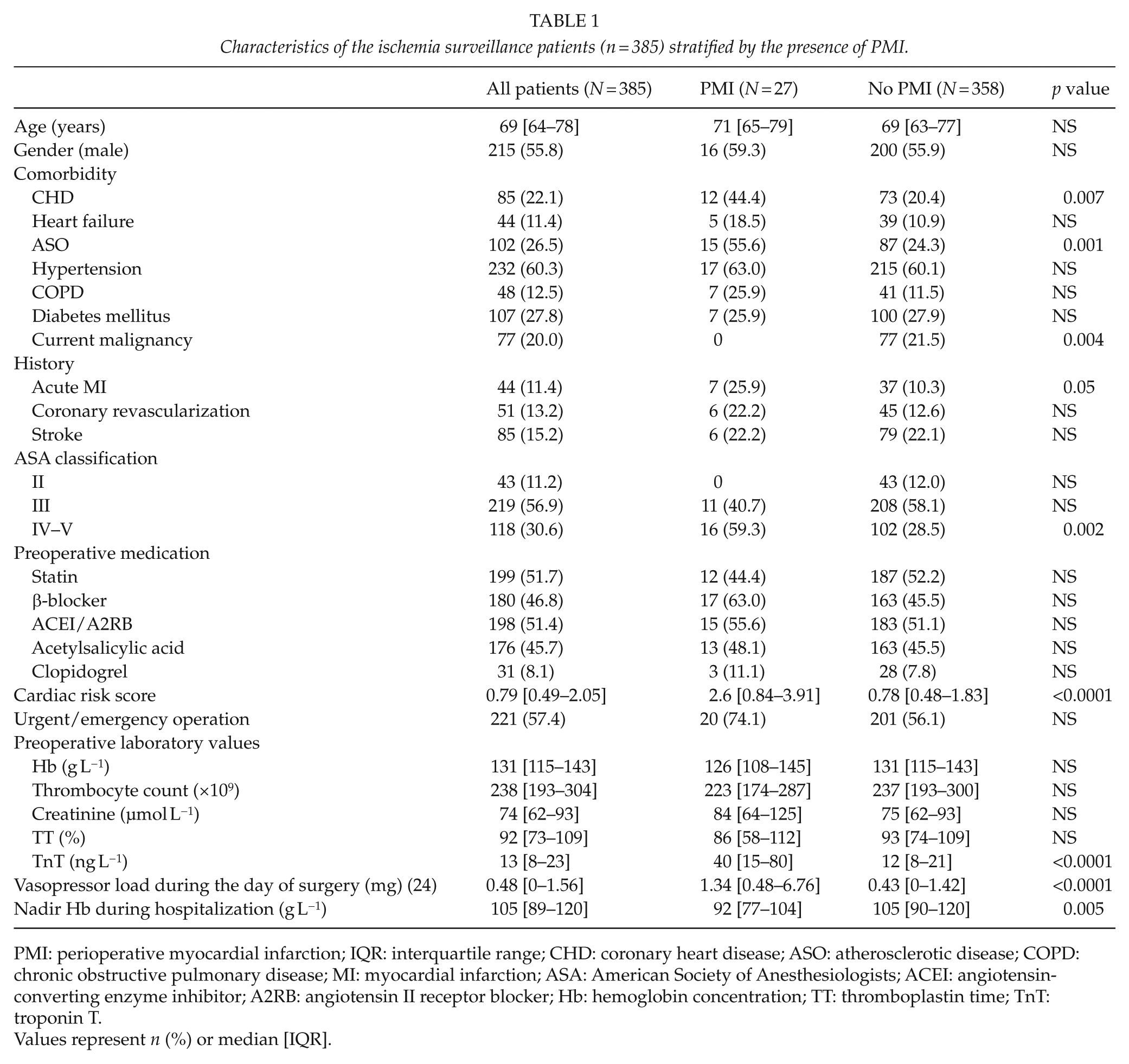
PMI: perioperative myocardial infarction; IQR: interquartile range; CHD: coronary heart disease; ASO: atherosclerotic disease; COPD: chronic obstructive pulmonary disease; MI: myocardial infarction; ASA: American Society of Anesthesiologists; ACEI: angiotensin-converting enzyme inhibitor; A2RB: angiotensin II receptor blocker; Hb: hemoglobin concentration; TT: thromboplastin time; TnT: troponin T.
Values represent n (%) or median [IQR].
The majority of PMIs (16 of 27, 59.3%) were diagnosed on the first or second postoperative day. Six patients with PMI (6 of 27, 22.2%) had ischemic symptoms. The PMI diagnosis led to a coronary angiography in four patients and three of them underwent percutaneous coronary intervention (PCI). The cardiovascular medication was optimized in four patients.
The 90-day mortality
The 90-day mortality was 7.3% (28 deaths of 385). The mortality rate was 29.6% (8 of 27) in PMI and 5.6% (20 of 358) in non-PMI patients (p < 0.001) (Fig. 2). All except one death in the PMI patients occurred during the first 23 postoperative days in contrast to the deaths of non-PMI patients distributed over the whole 90-day period. One of the eight deceased patients had acute myocardial infarction and a futile rescue-PCI, bowel ischemia was the cause of death in two patients, multiple organ failure in one patient, and one patient operated on a ruptured abdominal aortic aneurysm died with AKI and myocardial ischemia. The remaining three deceased patients had a congestive heart failure. Consequently, six of the eight deaths can be categorized to have a vascular cause. The 90-day mortality in patients with mere postoperative troponin elevation (irrespective of other ischemic symptoms) was 10.4%.
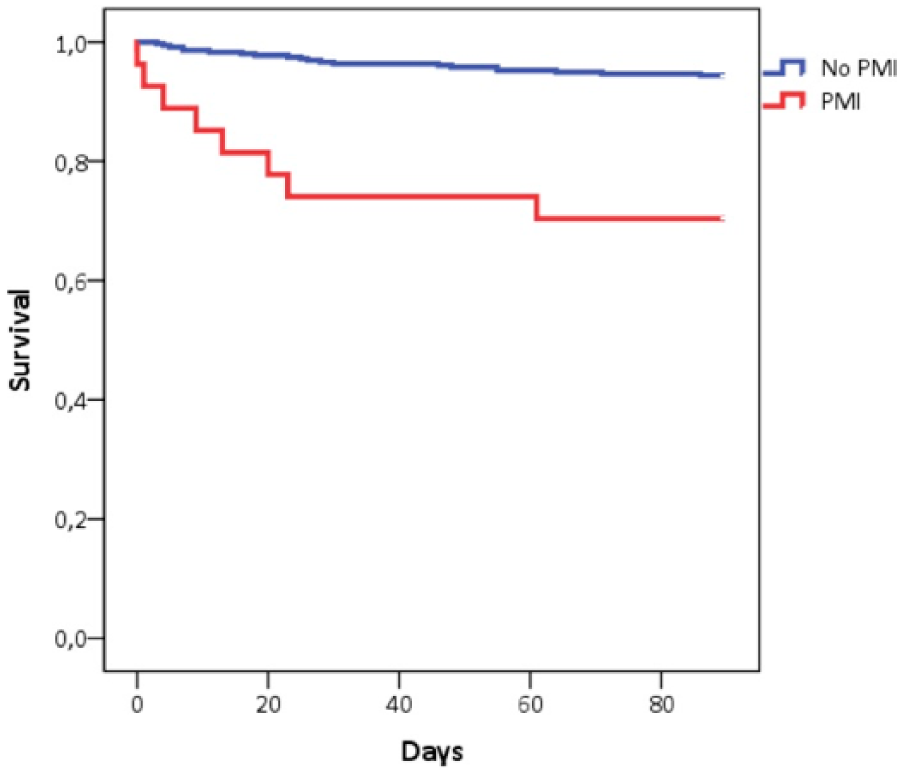
Kaplan–Maier 90-day survival curves in PMI versus non-PMI patients (n = 385).
Prediction of PMI and 90-day mortality
The perioperative cardiac risk calculator predicted PMI with an AUC of 0.73 (95% CI: 0.64–0.81). The best cut-off value was 2.55, and the sensitivity, specificity, and LR+ for this value were 0.57, 0.81, and 3.01 (95% CI: 2.02–4.49), respectively. The 90-day mortality was predicted with an AUC of 0.75 (95% CI: 0.66–0.85). The best cut-off value was 1.85, and the sensitivity, specificity, and LR+ were 0.71, 0.76, and 3.0 (95% CI: 2.23–4.04), respectively (Fig. 3A, B).
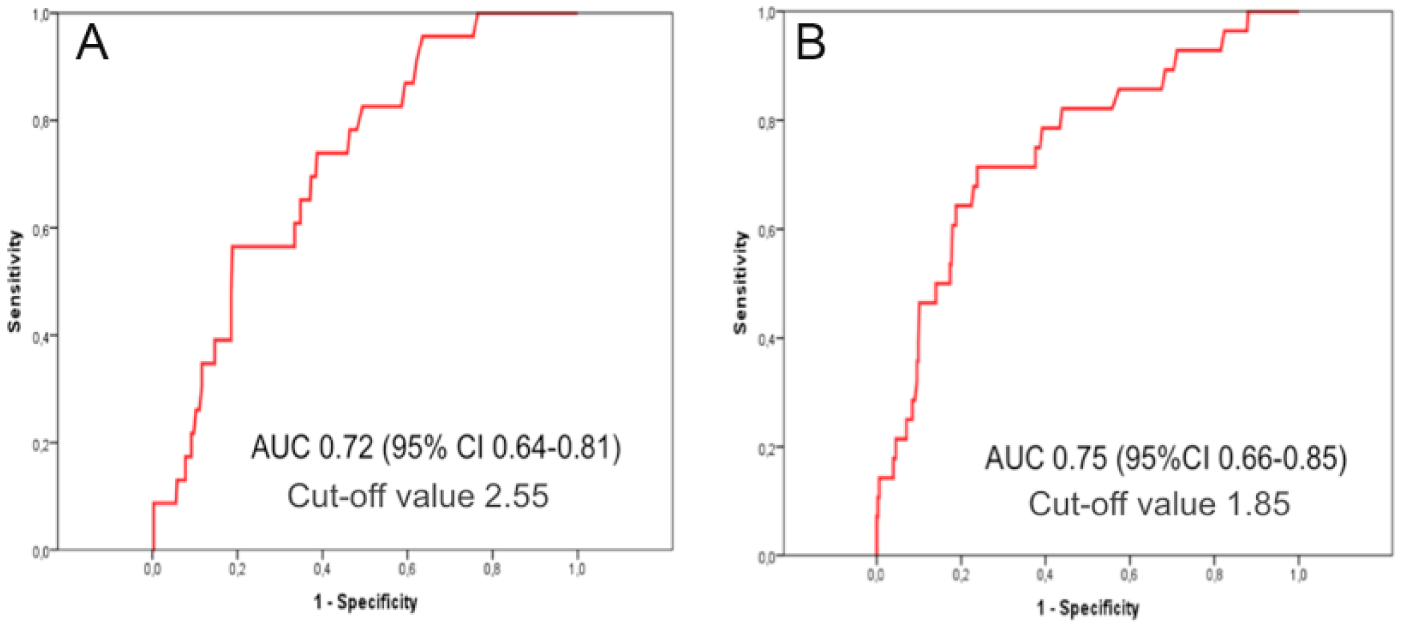
Receiver operating characteristics curves (AUC) (with 95% CI) presenting the predictive value of the perioperative cardiac risk score (A) PMI and (B) 90-day mortality (n = 385).
Cohort with Routine Perioperative Care
Patients without consent did not differ from the study patients in terms of age or comorbidities. However, they were more often operated outside office hours and underwent more urgent/emergency surgeries (ESM Table). The proportions of the different operation types were similar in the both cohorts (systematic screening and routine perioperative care).
TnT measurements ordered at the clinicians’ preference were performed in 39 (22.3%) of the 175 patients. A total of 3 patients (1.7%) had PMI and 23 patients (13.1%) died within 90 days.
Discussion
In non-cardiac surgery patients aged 50 or over, incidence of PMI was high, 7.0%, especially in vascular surgery, 11%. The 30% mortality rate of the PMI patients was considerable. The performance of the perioperative cardiac risk score in predicting PMI and 90-day mortality was fair, with an AUC of 0.73 (95% CI: 0.64–0.81) for PMI and 0.75 (95% CI: 0.66–0.85) for 90-day mortality. The best cut-off values were 2.55 and 1.85, respectively.
The incidence of PMI in our study (7%) is in agreement with earlier prospective large-scale studies (4, 7, 12–14, 25). The Perioperative Ischemic Evaluation (POISE) and Vascular events In noncardiac Surgery patients cOhort EvaluatioN (VISION) study groups have produced several retrospective and prospective analyses with TnT screening, including randomized studies showing a comparable PMI incidence from 5% to 7% (4). However, large register-based studies may show PMI incidences of less than 1% reflecting the routine diagnostic accuracy (1, 26). Among high-risk patient cohorts with ischemia surveillance, however, the incidence may exceed 10% (27). Vascular surgical patients are thought to comprise the highest risk group for perioperative cardiac morbidity and mortality (28). Clinical presentation of PMI is most often either asymptomatic or non-specific and, thus, relying on the classical symptoms alone will lead to many missed diagnoses (3). It has been repeatedly shown that register-based studies without systematic cardiac ischemia screening report significantly lower PMI incidence (1, 26). Despite the higher risk profile and mortality of the non-screened patients in this study, TnT was measured perioperatively only in 39 patients (22%) and only 3 (1.7%) PMIs were diagnosed. These findings emphasize the importance of routine TnT measuring over physician-initiated surveillance. This conclusion is in agreement with Ghaferi et al.’s (6) register-based retrospective study reporting perioperative Q wave PMI only in 0.5% of 84,730 patients undergoing general or vascular surgery. These diagnoses, however, were based on TnT measurements and ECG recordings ordered by treating physicians from symptomatic patients. Consequently, it is likely that several silent non-Q PMIs were missed in their study and are missed in routine clinical practice.
Although cardiac troponins are considered to be a specific marker for myocardial tissue damage (16), other conditions such as pulmonary embolism, sepsis, stroke, or AKI can cause substantial troponin elevations. In this study, these conditions were recorded and considered while defining the diagnosis. However, even a mere postoperative cardiac biomarker elevation irrespective of other ischemic features, myocardial injury after non-cardiac surgery (MINS) has been shown to predict both short-term (7) and long-term (29) mortality. More studies need to be conducted before this condition can be accurately defined.
The prospective European Surgical Outcomes Study (EUSOS) reported a 4% in-hospital mortality rate in adult non-cardiac surgical patients (30). In our study, the all-cause mortality in the complete cohort of 560 patients aged 50 or older was 9%. In association with PMI or MINS, previous prospective studies have reported 30-day mortality rates of approximately 10% (4, 7, 27). In our study, a high mortality rate of 29.6% was seen after TnT-surveillance-diagnosed PMI. Few studies have reported causes of death in PMI patients. In the VISION study, causes of death were divided into vascular (54%) and nonvascular (46%). Of note, that 58.2% of these myocardial injury patients would not have fulfilled the international definition of myocardial infarction (7). In our study, 75% of the PMI deaths were categorized as having a cardiovascular cause. Considering this large variability in PMI-patients’ causes of death, it can be concluded that PMI may be only a surrogate marker of disease severity with bad prognosis and not the actual cause of death (21).
Despite advancements in perioperative care, the incidence of PMI and mortality has not declined substantially during the last decades. Thus, risk stratification for PMI should be an integral part of preoperative evaluation. The perioperative cardiac risk calculator has been developed using the American College of Surgeons’ 2007 National Surgical Quality Improvement Program (NSQIP) database (n = 211,410) and validated using the 2008 patient cohort (n = 257,385) undergoing modern non-cardiac surgery (18). This calculator takes into account the severity of the disease and the nature of the surgical procedure and has been shown to be a good predictor of perioperative cardiac risk in modern non-cardiac surgery (18). In our study, the predictive value of the cardiac risk calculator was fair both for PMI (AUC of 0.73) and for 90-day mortality (AUC of 0.75). Notably, this calculator has not been earlier validated to predict mortality.
Our study has some important limitations. First, the patients were not consecutive. Although the study group also worked outside office hours, the 24/7-coverage could not be achieved. Therefore, the recruitment and consent were also performed by treating physicians and, thus, full coverage to ask written consent was not feasible. However, in our opinion, difficulties in obtaining consents for ischemia screening did not lead to a systematic selection bias in the cohort. However, we saw this as an opportunity to compare the intensively screened patients to patients treated and diagnosed according to normal hospital protocols. Due to differences in the recruitment and data collection, statistical methods could not be utilized and the comparison of the different strategies remained descriptive. Second, we cannot report the exact incidence of perioperative ischemic ECG changes in this cohort of non-cardiac surgical patients because only patients with elevated TnT concentration (⩾14 ng L−1) had further analysis of their ECG.
Finally, performing a study may improve the diagnosis and treatment of other similar patients as well (Hawthorne effect). The systematically screened and routine patients were treated on the same wards by the same physicians and nursing staff. Thus, it may be assumed that even more PMIs would have remained undetected in missed patients without the study, and the true advantage of systematic screening may be bigger than observed.
The strengths of our study include a relatively large prospective sample of patients undergoing a broad range of non-cardiac surgical procedures. We consider that the study cohort represents an operative patient population at a tertiary care university hospital. The best available fifth generation hs-TnT was utilized in all samples, and the definition of PMI used was established. Furthermore, to our knowledge, no previous study has evaluated the predictive power of the perioperative cardiac risk calculator in a prospective non-US cohort.
Conclusion
To conclude, PMI is still a common complication especially in elderly patients undergoing non-cardiac vascular surgery, and it is associated with substantial 90-day mortality. Given that the perioperative cardiac risk calculator predicted both PMI and 90-day mortality, we would like to recommend its routine use and perioperative ischemia screening.
Footnotes
Acknowledgements
Preliminary data of this study were presented as an electronic poster at the Euroanaesthesia 2015 Congress, Berlin, Germany.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by clinical research funding TYH 2014239 from the University of Helsinki and Helsinki University Hospital (V.P., L.V.), and grants from the Sigrid Juselius Foundation (V.P.) and the Finnish Society of Angiology (L.V.).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
