Abstract
Background and Aims:
Peripheral artery disease in dialysis cases is more prone to critical limb ischemia compared to non-dialysis cases, with a significantly high rate of major amputation of the lower limbs. Lesions are distributed on the more distal side in dialysis critical limb ischemia cases. The aim of this study was to investigate the usefulness of indocyanine green angiography to determine differences in the regional circulation in the foot between dialysis and non-dialysis patients.
Materials and Methods:
The subjects included 62 cases, among which 20 were dialysis patients and 42 were non-dialysis patients. We compared the indocyanine green angiography parameters for regions of interest in the dialysis and non-dialysis groups, which included the magnitude of intensity from indocyanine green onset to maximum intensity (Imax), the time from indocyanine green onset to maximum intensity (Tmax), the time elapsed from the fluorescence onset to half the maximum intensity (T1/2), and the time from maximum intensity to declining to 90% of the maximum intensity (Td90%). These indocyanine green angiography parameters were measured at region of interest 1 (the Chopart joint), region of interest 2 (the Lisfranc joint), and region of interest 3 (the distal region of the first metatarsal bone).
Results:
In the comparison between the dialysis and non-dialysis groups, a significant difference was observed regarding Tmax, T1/2, and Td90%, especially in region of interest 3.
Conclusion:
In this study, we show that regional tissue perfusion is more deteriorated in dialysis patients compared with non-dialysis patients using indocyanine green angiography. Tmax, T1/2, and Td90% could be useful clinical parameters to compare ischemic severity of the lower limb between dialysis and non-dialysis patients.
Keywords
Introduction
Chronic kidney disease is regarded to be an independent risk factor of peripheral arterial disease (PAD), and according to international investigations on dialysis outcomes and practice patterns study, it was reported that the incidence of PAD in maintained dialysis cases is very high, at 11.5%–37.8% (average: 25.3%) (1). PAD in dialysis cases is more prone to critical limb ischemia (CLI) compared to non-dialysis cases, with a significantly high rate of major amputation of the lower limbs. The 5-year survival rate in CLI cases with dialysis is significantly lower compared to CLI cases without dialysis, and it is believed that early intervention is necessary (2). Lesions are distributed on the more distal side in dialysis cases, particularly with arterial lesions observed in the arterial arch of the foot (3). Indocyanine green (ICG) is clinically used as a source of fluorescence for near-infrared radiation (4), a marker of the renal function, and as a photosensitizer for photodynamic therapy (5, 6). Furthermore, ICG bonds with serum protein, represented by albumen, and spreads into the vascular lumen (7). Using indocyanine green angiography (ICGA), it has become possible to acquire detailed, informative images of tissue perfusion and quantitatively evaluate the condition of tissue perfusion (8).
The aim of this study was to investigate the regional perfusion at the foot for the patients with dialysis and non-dialysis with ICGA. The second aim was to evaluate the difference of peripheral perfusion between dialysis and non-dialysis patients by ICGA test compared to the conventional hemodynamical measuring method.
Materials and Methods
Patients
All protocols, surveys, and consent forms were approved by the Institutional Review Board of Tokyo Medical and Dental University Hospital. Written informed consent was obtained from all the patients. In this study, ICG fluorescent contrasting was carried out prior to treatment intervention with patients admitted to Tokyo Medical and Dental University Hospital from November 2012 to March 2014.
The study included 62 patients, among which 20 were dialysis cases (dialysis group) and 42 were non-dialysis cases (non-dialysis group). Regarding the patient background, the presence of hypertension, dyslipidemia, coronary artery disease, cerebrovascular disease, and diabetes was compared. Hypertension was diagnosed as a systolic blood pressure of >140 mmHg, a diastolic blood pressure of >80 mmHg, or a history of treatment for hypertension. Dyslipidemia was defined as a serum low-density lipoprotein cholesterol level of >140 mg/dL, a high-density lipoprotein cholesterol level of <40 mg/dL, a triglyceride level of >150 mg/dL, or a history of treatment for dyslipidemia. Coronary artery disease was defined as the presence of angina pectoris or myocardial infarction or both, as documented on coronary angiography, or according to a history of any revascularization procedures of the coronary arteries. Cerebrovascular disease was defined as a history of stroke, transient ischemic attacks, carotid artery revascularization, or cerebral hemorrhage. Diabetes was defined as cases with hemoglobin A1c (HbA1c) of 6.5% or more or cases with a medical history of diabetes. We evaluated the hemodynamical severity of the lower limbs, including the ankle brachial pressure index (ABI), and the toe brachial pressure index (TBI) and toe pressure (TP). The VasoGuard P84™ system (SciMed Ltd., Bristol, UK) was used to measure the ABI, TBI, and TP, and ICG fluorescent contrasting was carried out on the same day.
The ischemic severity of the lower limbs was classified using the Rutherford classification (9): in the dialysis group, 6 cases were classified into category 3, 4 cases into category 4, 7 cases into category 5, and 3 cases into category 6; in the non-dialysis group, 5 cases were classified into category 2, 21 cases into category 3, 13 cases in category 4, and 3 cases into category 5. Categories 1–3 were classified into an intermittent claudication (IC) group, while categories 4–6 were classified into a CLI group.
ICGA Test
Testing was carried out at a room temperature of 20 °C–25 °C with the patient resting in a supine position; 25 mg ICG (Diagnogreen™; Daiichi-Sankyo Pharmaceutical, Tokyo, Japan) was dissolved in 25 mL distilled water (Otsuka distilled water™; Otsuka Pharmaceutical, Tokyo, Japan) and 0.1 mg/kg of the mixture was injected via the cubital vein. Fluorescence photography was then immediately conducted using an infrared camera system (Photodynamic Eye™, Hamamatsu Photonics K.K., Hamamatsu, Japan). Infrared rays activate ICG and emit light weaker than 820 nm (10). A light-emitting diode of 760 nm was used as the light source to irradiate ICG, with a charge-coupled device (CCD) camera used as the detector. The camera system was placed in a position approximately 20 cm from the skin of the feet, filming was carried out for 5 min following ICG injection, and real-time fluorescence imaging was shown on a monitor and recorded using an audio video interweave system. Thereby, the time-intensity curve was measured. ICG is non-toxic and side effects rarely occur (1/40,000); no abnormalities in the hemodynamics were observed in any case, and the pulse and blood pressure were within normal ranges (11).
ICGA Image Analysis
For comparative measurements, we set three types of regions of interest (ROIs): (1) the Chopart joint, (2) the Lisfranc joint, and (3) the distal region of the first metatarsal bone, referred to in a previous report (12) (Fig. 1). In order to quantitatively evaluate ICGA, several parameters were used to evaluate and analyze regional circulation. Regarding the parameters, the time from ICG onset to maximum intensity (Tmax), the magnitude of intensity from ICG onset to maximum intensity (Imax), the time elapsed from the fluorescence onset to half the maximum intensity (T1/2), and the time from maximum intensity to declining to 90% of the maximum intensity (Td90%) (10) were evaluated (Fig. 2). An analysis of the imaging data was performed using the ROIs™ version of the U11437 software program (Hamamatsu Photonics K.K.).
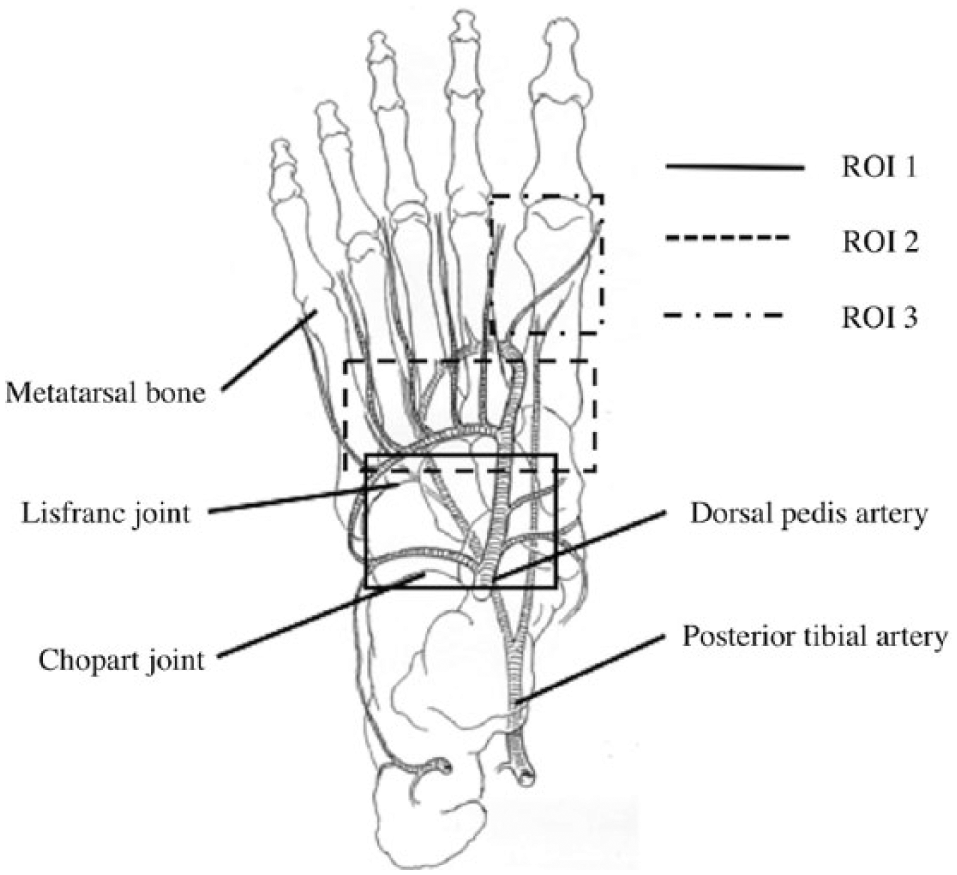
Setting of the regions of interest (ROI).
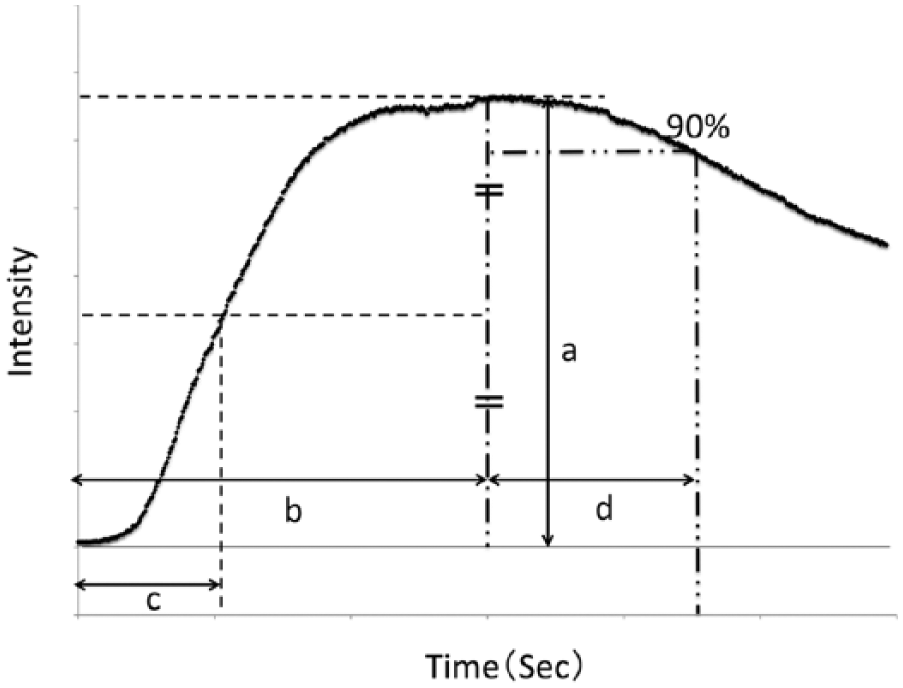
The indocyanine green angiography parameters were defined graphically: (a) Imax, (b) Tmax, (c) T1/2, and (d) Td90%.
Statistical Analysis
Measured values are presented as medians with ranges, while category variables are presented as degrees and percentages. Statistical significance was assessed using the Mann–Whitney U test for continuous variables and the chi-square test for categorical variables. A value of p < 0.05 was considered to be statistically significant. The StatView version 5 software program (Abacus Concept Inc., Berkley, CA, USA) was used for statistical processing.
Results
We included 62 patients in this study, among which 20 were dialysis cases (dialysis group) and 42 were non-dialysis cases (non-dialysis group); the median age was 66 years (range, 57–83 years) in the dialysis group and 71 years (range, 38–89 years) in the non-dialysis group. Regarding the patients’ backgrounds and comorbidities, the hemoglobin and albumen levels were significantly lower in the dialysis group than in the non-dialysis group. In comparison of the hemodynamic parameters, no significant difference was observed between the two groups regarding ABI, TBI, and TP. Moreover, no significant difference was observed between dialysis and non-dialysis cases regarding ABI, TBI, and TP in both the CLI group and IC group. Regarding the presence of comorbid disorder, the coexistence rate of ischemic heart disease was significantly higher in the dialysis group, at 60% (Table 1). Although there was no significant hemodynamic difference between the dialysis group and non-dialysis group, the dialysis group exhibited greater significant clinical severity than the non-dialysis group.
Characteristics of patients.
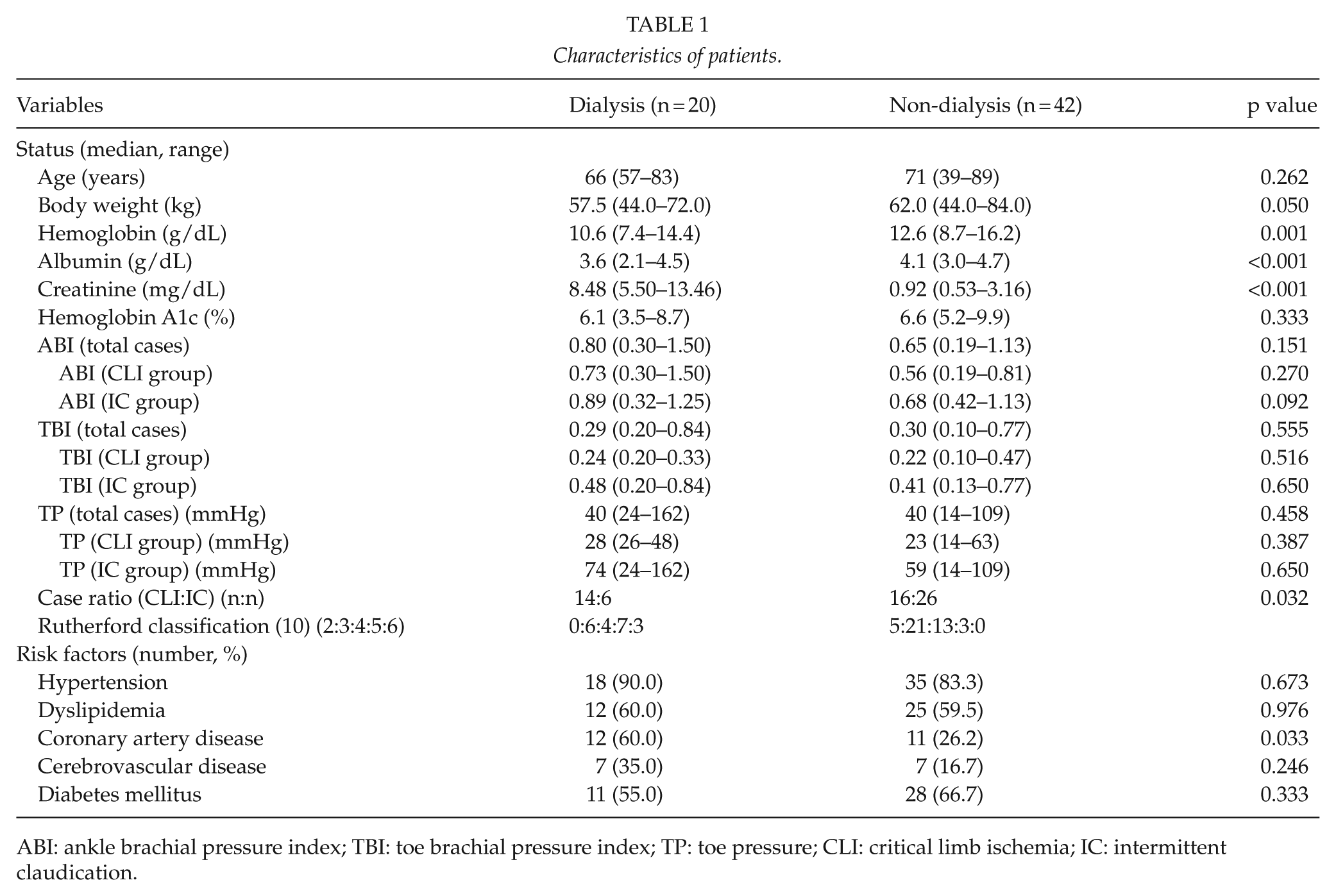
ABI: ankle brachial pressure index; TBI: toe brachial pressure index; TP: toe pressure; CLI: critical limb ischemia; IC: intermittent claudication.
We then used ICGA to compare between the CLI group (n = 30) and IC group (n = 32) (Table 2), and a significant difference was observed regarding Tmax and Td90% in all ROIs and T1/2 in ROI 2 and ROI 3. Furthermore, we classified the patients as Rutherford 4 group and Rutherford 5+6 group, and compared the ICGA parameters. We had no significant difference between the patients with or without dialysis in Rutherford 4 group, and patients with or without dialysis in Rutherford 5 and 6 groups (Table 3). In comparison between the dialysis group and non-dialysis group (Table 4), a significant difference in Td90% was observed in ROI 1 (49.3 s (range, 4.9–117.6 s) in dialysis group and 34.3 s (range, 9.6–89.4 s) in non-dialysis group), while a significant difference in Tmax (80.0 s (range, 17.2–327.6 s) and 40.6 s (range, 17.4–279.8 s), respectively), T1/2 (18.9 s (range, 8.1–95.1 s) and 15.4 s (range, 7.2–125.8 s), respectively), and Td90% (22.0 s (range, 0.7–54.8 s) and 13.4 s (0.2–60.3 s), respectively) was observed in ROI 3.
Indocyanine green angiography parameters between intermittent claudication (Rutherford 1–3 groups) and critical limb ischemia (Rutherford 4–6 groups).
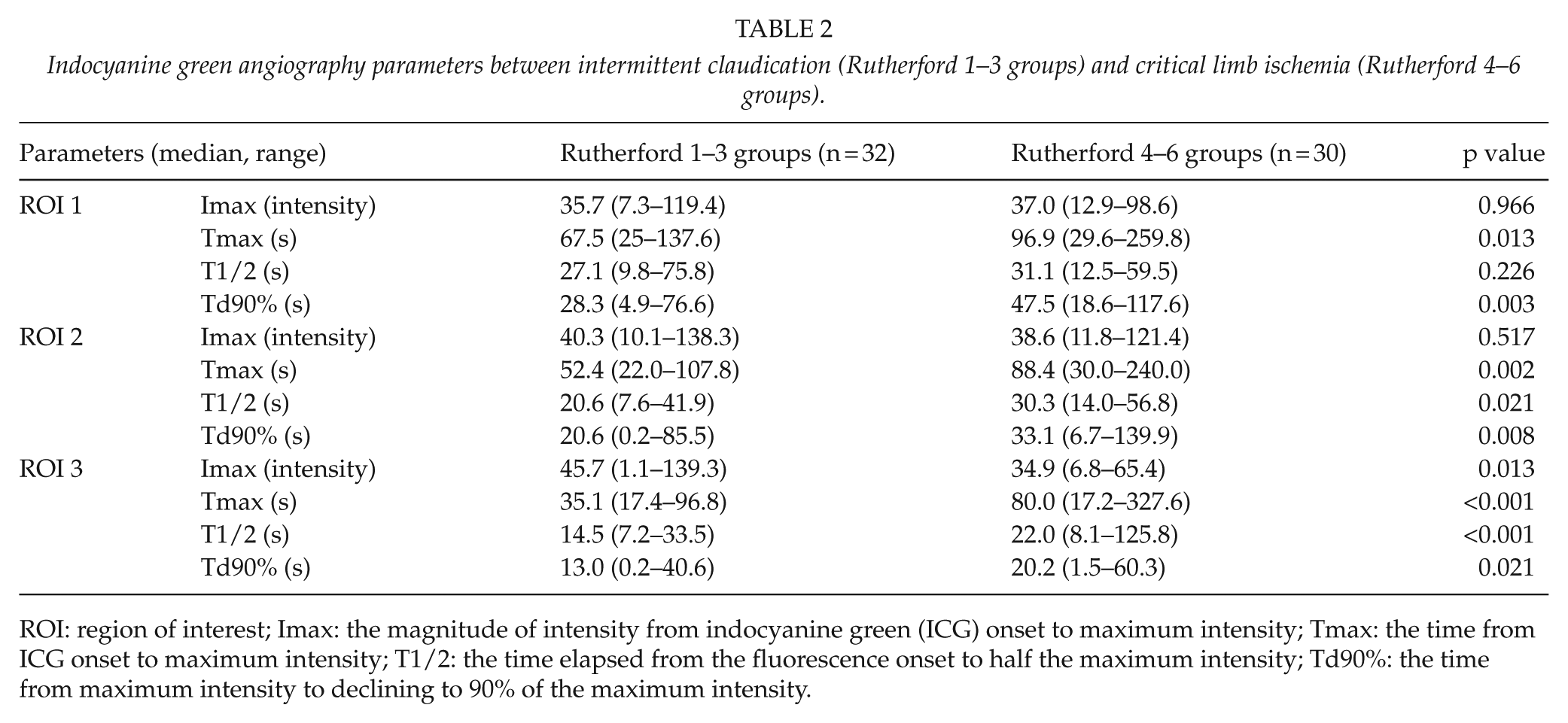
ROI: region of interest; Imax: the magnitude of intensity from indocyanine green (ICG) onset to maximum intensity; Tmax: the time from ICG onset to maximum intensity; T1/2: the time elapsed from the fluorescence onset to half the maximum intensity; Td90%: the time from maximum intensity to declining to 90% of the maximum intensity.
Indocyanine green angiography parameters of the patients with Rutherford 4 group and Rutherford 5 and 6 groups.
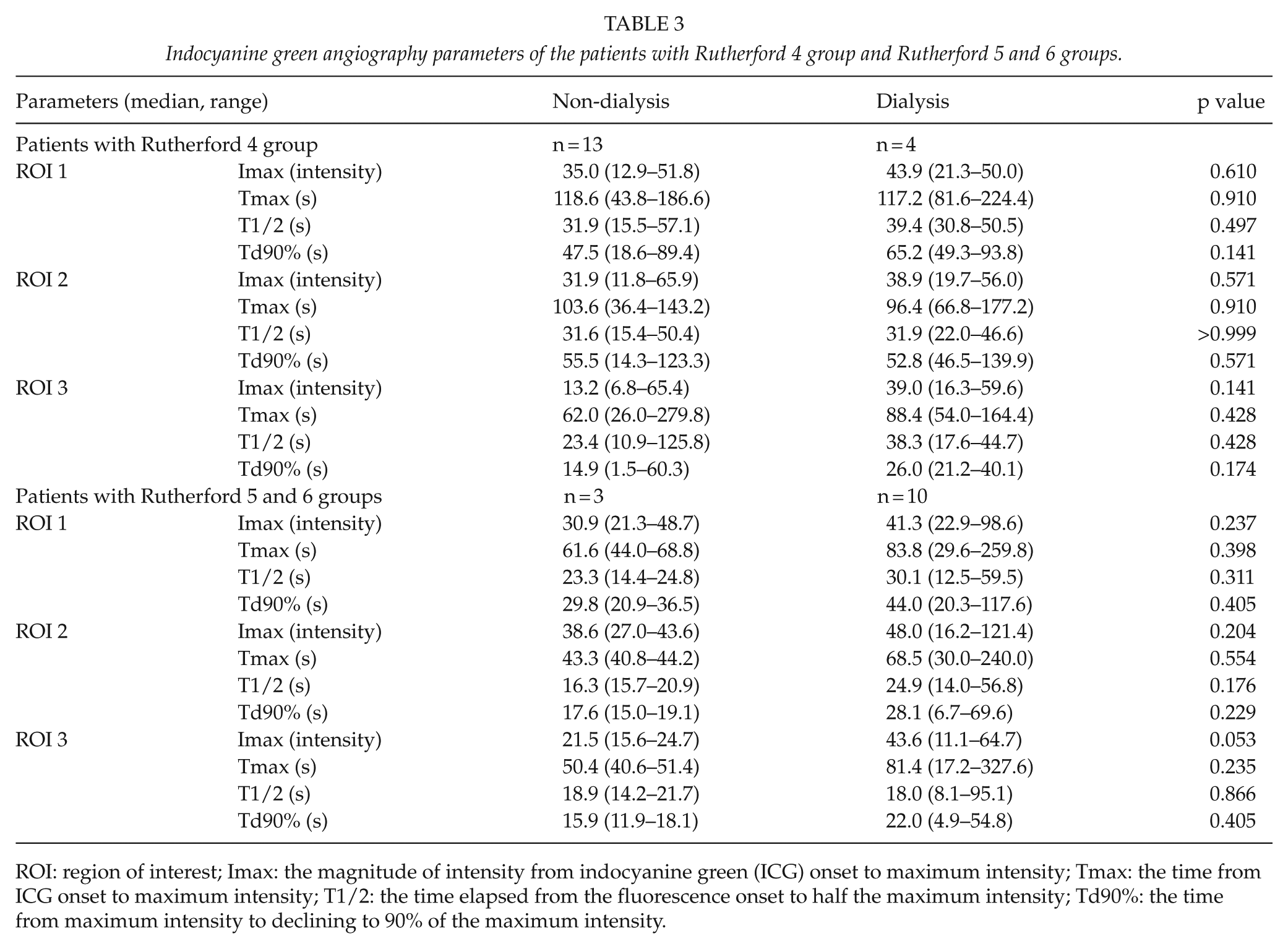
ROI: region of interest; Imax: the magnitude of intensity from indocyanine green (ICG) onset to maximum intensity; Tmax: the time from ICG onset to maximum intensity; T1/2: the time elapsed from the fluorescence onset to half the maximum intensity; Td90%: the time from maximum intensity to declining to 90% of the maximum intensity.
Indocyanine green angiography parameters between patients without dialysis and patients with dialysis.
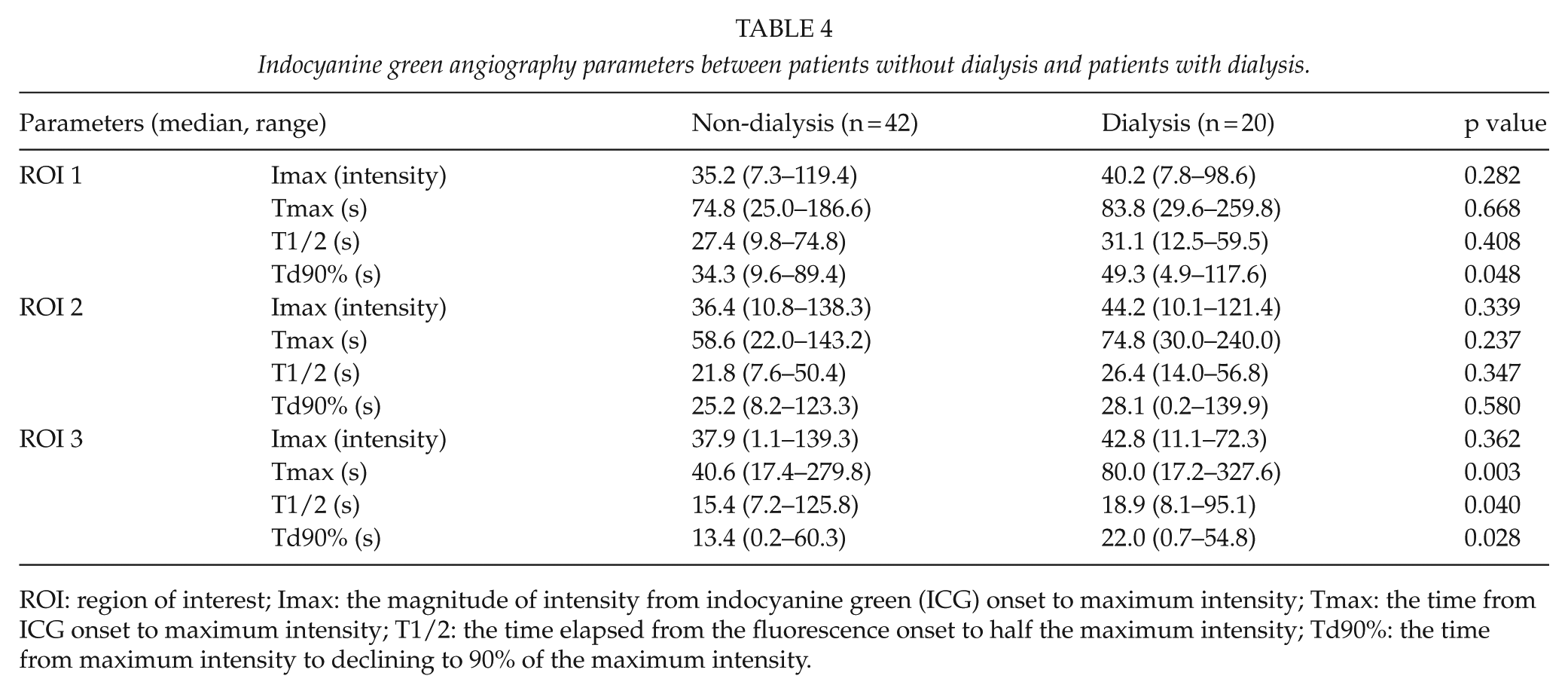
ROI: region of interest; Imax: the magnitude of intensity from indocyanine green (ICG) onset to maximum intensity; Tmax: the time from ICG onset to maximum intensity; T1/2: the time elapsed from the fluorescence onset to half the maximum intensity; Td90%: the time from maximum intensity to declining to 90% of the maximum intensity.
Discussion
In this study, the parameters of Tmax, T1/2, and Td90% were useful indices to assess the clinical ischemic severity of PAD in dialysis cases. We found that parameters such as Imax related to ICG were not correlated to the ABI, whereas the parameters of time from injecting ICG, Tmax, T1/2, and Td90% were correlated to the ABI, TBI, and TP, respectively (12–14). Our previous study reported that the intensity of ICG could depend on the distance from the camera to the skin, the patient’s skin color, and the ambient light in the testing room, then the intensity of ICG, including Imax, might be not a good parameter for assessing the perfusion using ICGA method. In our study, we could lead to similar results, which showed the significant correlation between tissue perfusion and time-related ICGA parameters, such as Tmax and T1/2 (12). Although there was no significant hemodynamic difference between the dialysis group and non-dialysis group, the dialysis group exhibited greater significant clinical severity than the non-dialysis group. Therefore, the ICGA parameter may reflect not only hemodynamic conditions, but also clinical conditions. It was previously reported that dialysis patients who suffer from severe vascular disease have micro-arteriovenous fistula (AVF) in their limbs (15, 16). We speculated that clinical differences occurred due to micro-AVFs. Similar to previous studies, the temporal parameters of ICGA, especially Td90%, were particularly useful to reflect the clinical severity. Therefore, when we compared the dialysis patients with non-dialysis patients, the ICGA parameters correlated with the hemodynamic parameters. Furthermore, ICGA might be useful to evaluate the clinical severity.
There are several limitations associated with this study. First, the sample size was small. Second, ICGA tended to overestimate the actual degree of perfusion due to the presence of inflammation (12), which can change tissue perfusion. Although these limitations must be examined in future studies, ICGA could be useful for not only evaluating the hemodynamic states of PAD patients but also comparing the clinical severity between dialysis and non-dialysis patients.
In conclusion, the parameters including Tmax, T1/2, and Td90% were shown to be useful to compare the severity of limb ischemia between dialysis and non-dialysis patients in this study. Although further studies are needed to evaluate the reliability of these parameters, especially in CLI patients, we believe that our results obtained using the quantitative analysis of ICGA will help to identify useful clinical diagnostic tools to compare differences between dialysis and non-dialysis patients.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
