Abstract
Background:
Compliance with guidelines for treating patients with peripheral artery disease (PAD) lags compliance for treating patients with coronary artery disease. We assessed the gap between guidelines and practice for patients with PAD who underwent lower extremity revascularization (LER) at our institution from 2007 to 2010.
Methods:
Quality of care (QoC) was calculated by measuring provider performance on four indicators (antiplatelet therapy, dyslipidemia management, control of hypertension, and diabetes) derived from the ACCF/AHA PAD guidelines. The QoC score was calculated at the time of admission and at time of discharge for each patient, and reflects the proportion of indicated treatments received.
Results:
Patients (n = 734, mean age 70±11, female 51%) were followed for a mean of 2.0±1.4 years (range 0–5.7) following LER. The indication for LER was claudication (24.8%), rest pain (16.7%), and tissue loss (58.4%). The percentage of patients with a perfect QoC score increased significantly during hospital admission (11% to 21%, p < 0.001). Significant multivariate predictors of perfect QoC score included race/ethnicity, Charlson score, severity of LE ischemia, and observation period (admission, discharge). Multivariate analysis demonstrated that age>75 years, heart failure, chronic kidney disease, rest pain, and tissue loss—but not compliance with four guideline-based therapies—were associated with decreased freedom from the composite endpoint of major amputation, repeat revascularization, and death.
Conclusions:
Although adherence to guidelines improved over time, we found a significant gap between guidelines and practice for this cohort of patients at increased risk for adverse cardiovascular events.
Keywords
Introduction
Peripheral artery disease (PAD) affects more than 10 million people in United States and is associated with a high morbidity and mortality.1,2 Patients with PAD are at increased risk for myocardial infarction and stroke; those with symptomatic PAD—especially critical limb ischemia—are at increased risk for adverse limb events including major revascularization and limb loss.3,4
The American College of Cardiology Foundation (ACCF) and American Heart Association (AHA) management guidelines for patients with PAD established quality of care (QoC) measures for managing the multiple risk factors in this high-risk population.5–7 The guidelines emphasize the importance of treating patients with antiplatelet and lipid-lowering medication, as well as optimizing control of hypertension and diabetes. Subsequently, performance measures, which are used to describe cardiovascular quality metrics with attributes suitable for public reporting and for explicit comparison of care among institutions and health care providers—were created for PAD patients.
Compliance with these guidelines has been variable in multiple large, randomized control trials8–10 and at least one large registry report. 11 The gap between ACCF/AHA guidelines and practice for PAD remains larger than the gap for other cardiovascular diseases (CVDs).12,13 However, the relationship between these QoC measures and outcomes in patients with PAD is not as well established as it is for other CVDs.
We assessed the gap between ACCF/AHA guidelines and practice, as well as the predictors of freedom from major amputation, repeat revascularization, and death for patients with PAD who underwent lower extremity revascularization (LER) at our institution.
Methods
Study population
The cohort was composed of patients ≥ 40 years old who underwent surgical, endovascular, or hybrid revascularization for LE arterial occlusive disease at Montefiore Medical Center from January 1, 2007 to December 31, 2010. Patients were included if they were admitted to the hospital for a limb-related cause (Appendix 1) and underwent LER for claudication, ischemic rest pain, or tissue loss as defined by International Classification of Diseases, Ninth Revision (ICD-9) or Current Procedural Terminology (CPT) codes (Appendix 2) during admission. Exclusion criteria included previous revascularization on the index limb, upper extremity procedures (e.g. dialysis access), or a procedure for acute limb ischemia (e.g. embolectomy) (Figure 1). Thus, the focus for this study was on patients treated for chronic limb ischemia. Our unit of statistical analysis was patient, rather than limb. For patients who underwent bilateral LER, we included the earliest revascularization procedure in the dataset. The study was approved by the Montefiore Medical Center Institutional Review Board.
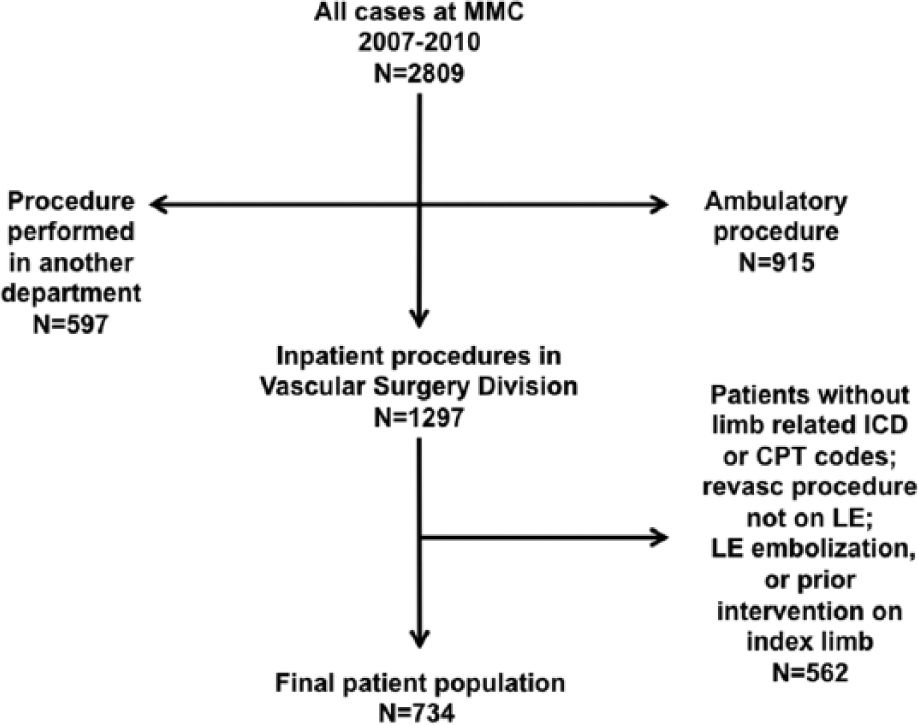
Flow diagram showing patient inclusion and exclusion.
Definitions and measures
Demographic information, hospital admission, and discharge data were abstracted from the medical record. Hemoglobin A1C (HgbA1C) and lipid profile were included provided they were measured within one year prior to the index admission. Diabetes was defined as HgbA1C > 7.0% or on therapy. The cutoff of 7.0% for diagnosing diabetes reflects the prevailing standard employed during the observation period of this study. 6 Chronic kidney disease (CKD) was defined as glomerular filtration rate < 60 ml/min/1.73 m2 calculated according to the Modification of Diet in Renal Disease (MDRD) equation. The Charlson Comorbidity Index score, a predictor of long-term morbidity and mortality, was computed based on ICD-9 codes for co-morbid conditions at the index admission.
Patients’ medications at the time of admission were abstracted from the nurse’s admission assessment and the physician admission note. Following discharge, patients were followed in the vascular surgery clinic. We tracked readmission for repeat revascularization or amputation. Mortality data were obtained from death in-house or from the Social Security Index (SSI).
QoC
QoC was evaluated by measuring provider performance on four indicators derived from the ACCF/AHA guidelines and practice for PAD.5,7 A QoC score was calculated at time of admission and at time of discharge for each patient. Indicators of care assessed were: being prescribed an antiplatelet agent (Class I recommendation, all patients), control of dyslipidemia (Class I recommendation, all patients), hypertension (Class I recommendation, all patients), and diabetes (Class IIa recommendation, diabetics only).6,14 Definitions for the four QoC measures are shown in Table 1. For the QoC measure “control of dyslipidemia,” we opted to use the less stringent definition from the 2010 performance guidelines that calls for use of a statin or low-density lipoprotein (LDL) < 100 mg/dl. 7 For diabetics, we calculated the diabetes QoC component only at discharge since HgbA1C reflects glycemic control for the preceding three months (i.e. it does not reflect any change in therapy that may have occurred during the index admission). For LDL and HgbA1C, we utilized values obtained within one year of the index admission date. For each patient, the number of indicated treatments was the denominator, and the number of treatments received the numerator in a QoC score, reflecting the proportion of indicated treatments each patient received. For diabetics, the maximum denominator was four points, whereas for nondiabetics the maximum was three points. The overall score ranges from 0 to 1 (Table 1). The composite outcome was major amputation (any amputation at or above the ankle), repeat revascularization, and all-cause mortality.
Quality of care measures.

LDL: low-density lipoprotein; HgbA1C: hemoglobin A1C.
Statistical methods
Analysis of variance (ANOVA) was used to compare the means by severity of LE ischemia, and chi-square for dichotomous variables. McNemar test was used to compare medications on admission and discharge. A paired t-test was used to compare QoC measures between admission and discharge. A generalized linear mixed model (GLMM) was estimated to evaluate factors related to perfect QoC (QoC = 100%). Predictors included year of discharge, observation period (admission, discharge), age > 75 years, gender, race/ethnicity, cardiology consult, and severity of LE ischemia. To determine if improvement in care varied over time, a term modeling the interaction between observation period and time was included in the model.
Univariate and multivariate Cox proportional hazard models were used to identify predictors of the composite endpoint. Covariates that were significant in the univariate models were entered into a multivariate Cox regression. To assess the relationship of QoC received to amputation-free survival, a multivariate Cox proportional hazard model was performed. Covariates included in the model were age (≥75 years) and disease severity (claudication, rest pain, tissue loss).
Statistical analysis was performed in SAS 9.3 (SAS Institute, Cary, NC) and SPSS (IBM Corp. Released 2012. IBM SPSS Statistics for Windows, Version 21.0. Armonk, NY: IBM Corp.). p values of 0.05 were used to determine statistical significance in univariate and multivariate analyses.
Results
Patient characteristics
Demographic characteristics and clinical history are presented in Table 2. Patients (n = 734, mean age 70 ± 11, female 51%) were followed for a mean of 2.0 ±1.4 years (range 0–5.7) following LER. The indication for LER was claudication (24.8%), rest pain (16.7%), and tissue loss (58.4%). The cohort was composed of non-Hispanic whites (25.6%), African Americans (32.3%), Hispanics (35.1%), and others (7.0%). Patients presenting with rest pain and tissue loss were more likely to be older, African American, diagnosed with heart failure, diabetes, and CKD. Increasing severity of LE ischemia was associated with a higher Charlson score (p < 0.0001). Open surgery—aortobifemoral bypass, infrainguinal bypass, or extra-anatomic bypass—was performed on 55.9%; endovascular repair was performed on 38.1%; and hybrid reconstruction, a combination of open and endovascular repair, was performed on 5.8% of patients. Patients with more advanced ischemia were more likely to undergo surgical repair (p = 0.0002).
Baseline patient characteristics by severity of lower extremity ischemia.
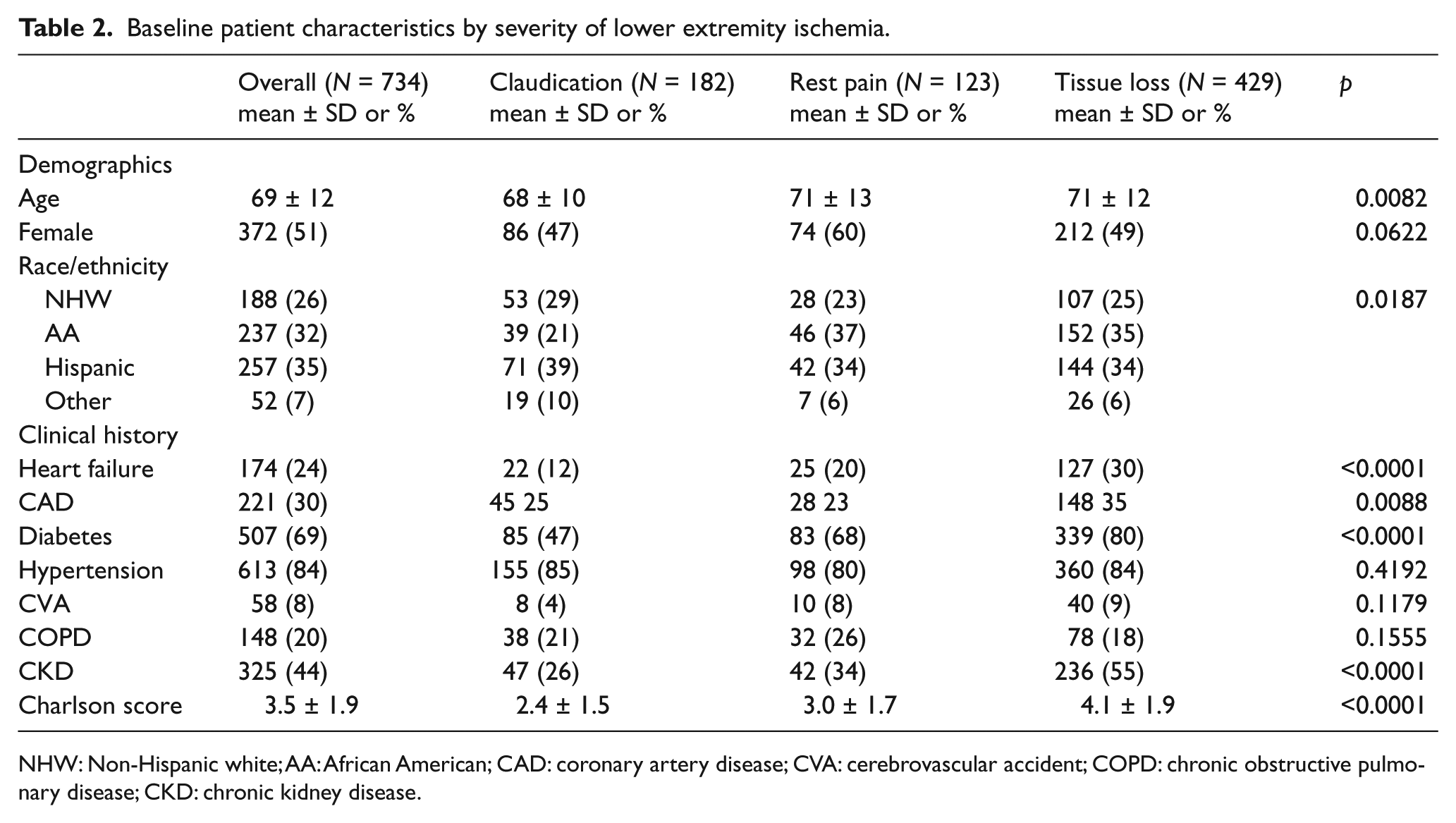
NHW: Non-Hispanic white; AA: African American; CAD: coronary artery disease; CVA: cerebrovascular accident; COPD: chronic obstructive pulmonary disease; CKD: chronic kidney disease.
QoC
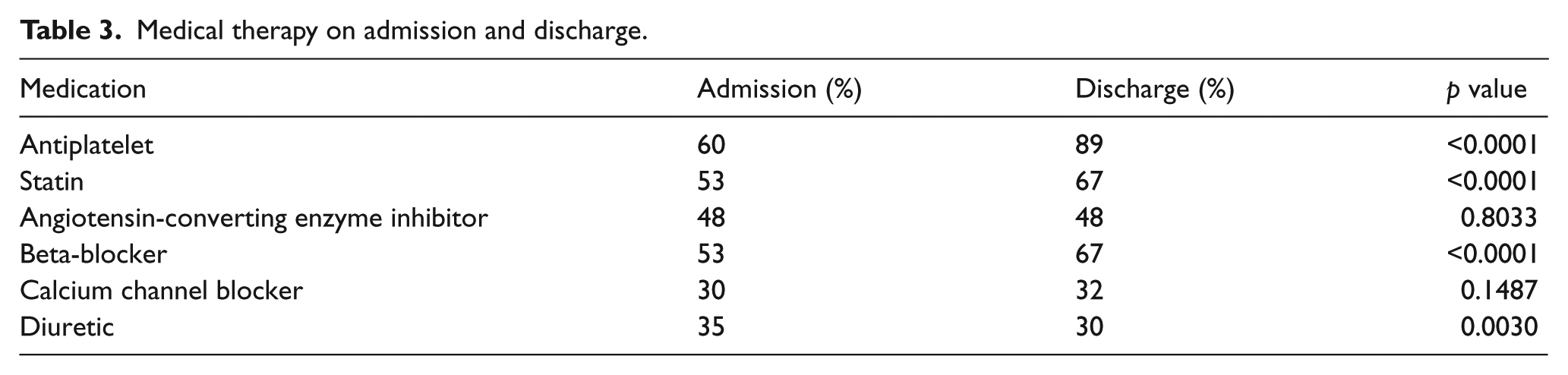
On admission, 60% of patients were taking an antiplatelet agent (Table 3). Univariate analysis did not reveal significant predictors for antiplatelet use on admission or discharge. At discharge, the percentage of patients receiving an antiplatelet agent increased to 89% (p < 0.0001). Antiplatelet use on admission and on discharge remained fairly constant over the study period (Table 4).
Medical therapy on admission and discharge.
Antiplatelet use, control of dyslipidemia, systolic blood pressure at or below JNC VII goal, and control of diabetes (HbgA1C < 7.0%) on admission and discharge from 2007 to 2010.
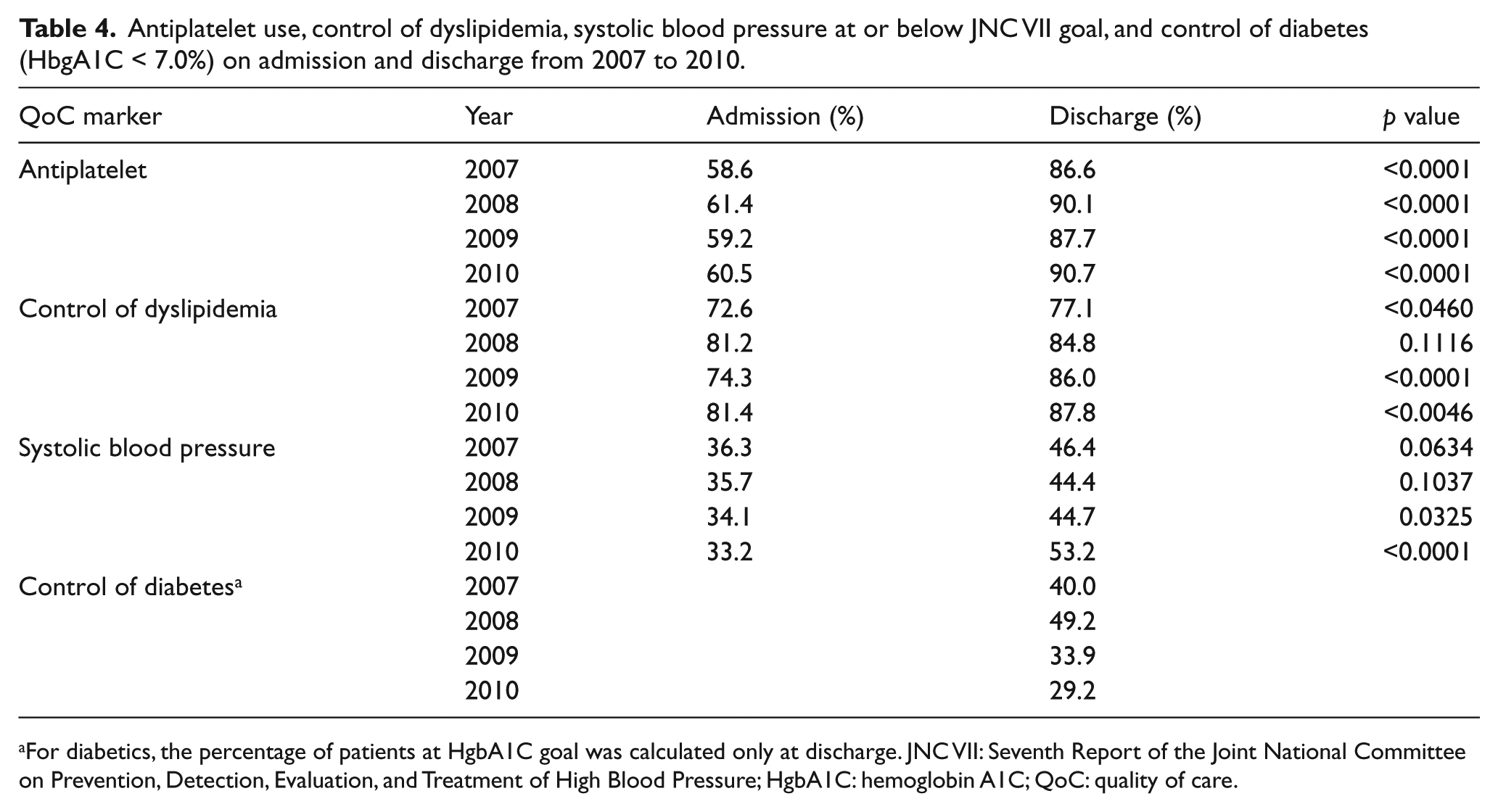
For diabetics, the percentage of patients at HgbA1C goal was calculated only at discharge. JNC VII: Seventh Report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure; HgbA1C: hemoglobin A1C; QoC: quality of care.
On admission, 53% of patients were taking a statin. Of those who were not on a statin, 24% had an LDL < 100 mg/dl. Thus the total percentage of patients whose dyslipidemia was in control on admission was 77%. Univariate predictors of meeting the statin QoC measure on admission included Charlson score, and claudication as the indication for revascularization. Multivariate predictors of statin use on admission included Charlson score (odds ratio (OR) 1.3, 95% confidence interval (CI) 1.1–1.4) and indication for revascularization (claudication OR 1.9, 95% CI 1.2–3.0). Discharge statin use increased to 67% (p < 0.0001). Patients who were evaluated by a cardiologist during admission were more likely to be started on a statin than patients who were not evaluated (p < 0.05). Of those patients not taking a statin, 17% had an LDL < 100 mg/dl. Thus, the total percentage of patients whose dyslipidemia was under control upon hospital discharge was 84%. Univariate predictors of meeting the dyslipidemia QoC measure on discharge included lower Charlson score and indication for revascularization. Female gender approached but did not reach significance (p = 0.08). Multivariate predictors included Charlson score (OR 1.2, 95% CI 1.05–1.32) and indication for revascularization (claudication OR 2.0, 95% CI 1.1–3.6). Usage of statins on discharge increased significantly from 2007 to 2010 (p = 0.0005).
At time of hospital admission, 35% of patients met the Seventh Report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure (JNC VII) systolic blood pressure (SBP) goal. At time of discharge, 47% of patients were at SBP goal (p < 0.0001). In the 12 months prior to admission, 90% of diabetics underwent HbgA1C testing. At discharge, 38% of diabetics achieved target HgbA1C.
The distribution of patients with a perfect QoC score is shown in Figure 2. The percentage of patients with a perfect QoC score increased significantly during hospital admission (11% to 20.7%, p < 0.001). Significant multivariate predictors of perfect QoC score included race/ethnicity, Charlson score, severity of LE ischemia, and observation period (admission, discharge). Thus, patients had an increased likelihood of a perfect QoC score at discharge, or if they were claudicants. Hispanic patients were less likely than non-Hispanic whites or African American patients to have perfect scores; a higher number of comorbid conditions was associated with a decreased likelihood of a perfect QoC score. Age, year of discharge, gender, and evaluation by a cardiologist were nonsignificant in the multivariate model, as was the interaction between observation period and time, which was dropped from the model.
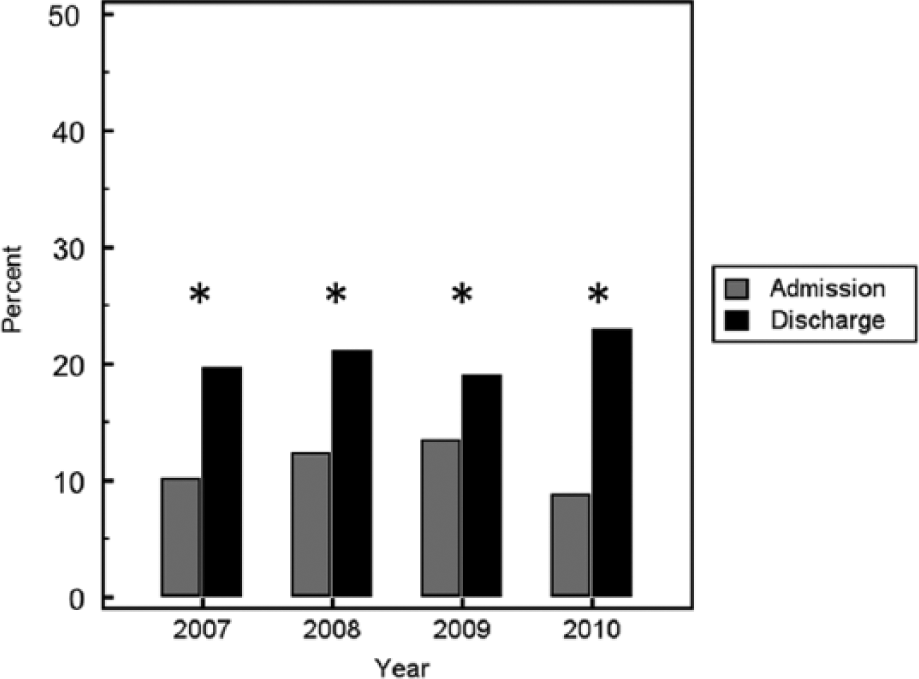
Bar graph depicting the percentage of patients with a perfect quality of care (QoC) score on admission and at discharge from 2007 to 2010. For each year, the percentage of perfect QoC scores was higher for discharge than on admission (*, p < 0.0001). The trend over time was significant as well.
Outcomes
Overall outcomes declined according to severity of LE ischemia—claudication, rest pain, or tissue loss—for all endpoints measured, including amputation (2%, 14%, 17%, respectively; p < 0.0001), all-cause mortality (10%, 35%, 44%, respectively; p < 0.0001), and amputation-free survival (89%, 56%, 47%, respectively; p < 0.0001) (Figure 3). Univariate analysis showed the following variables were associated with decreased event-free survival: age > 75 years, heart failure, CKD, rest pain, and tissue loss. Neither the QoC score, nor any of its four components, was associated with the composite endpoint. Multivariate analysis demonstrated that age > 75, heart failure, CKD, rest pain, and tissue loss were associated with decreased event-free survival (Table 5).
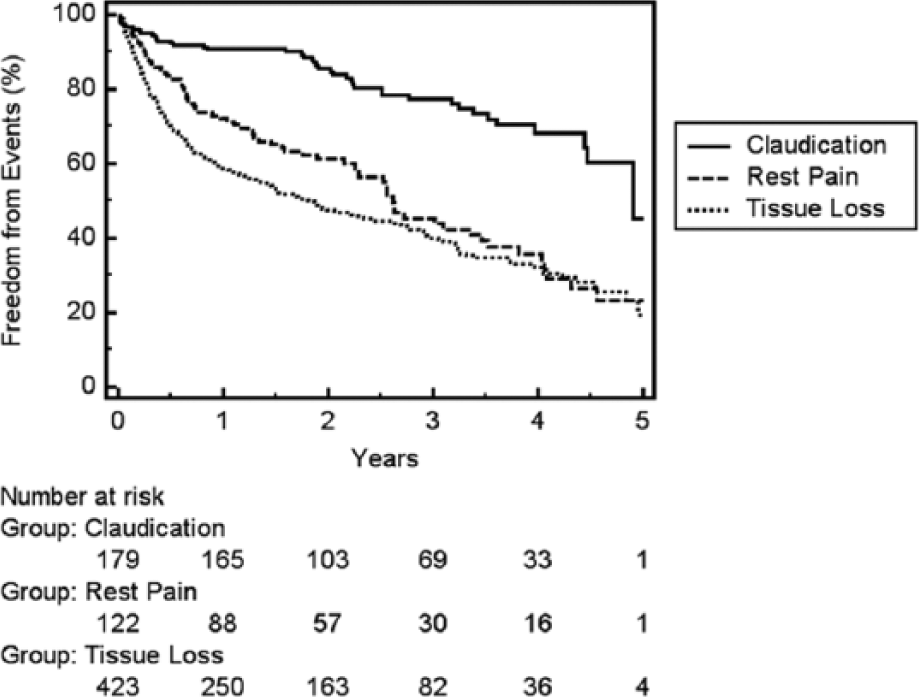
Kaplan-Meier plot for the composite endpoint of freedom from repeat revascularization, major amputation, and death. The difference between claudicants and patients with rest pain or tissue loss was significant (p < 0.0001).
Adjusted correlates associated with decreased event-free survival.
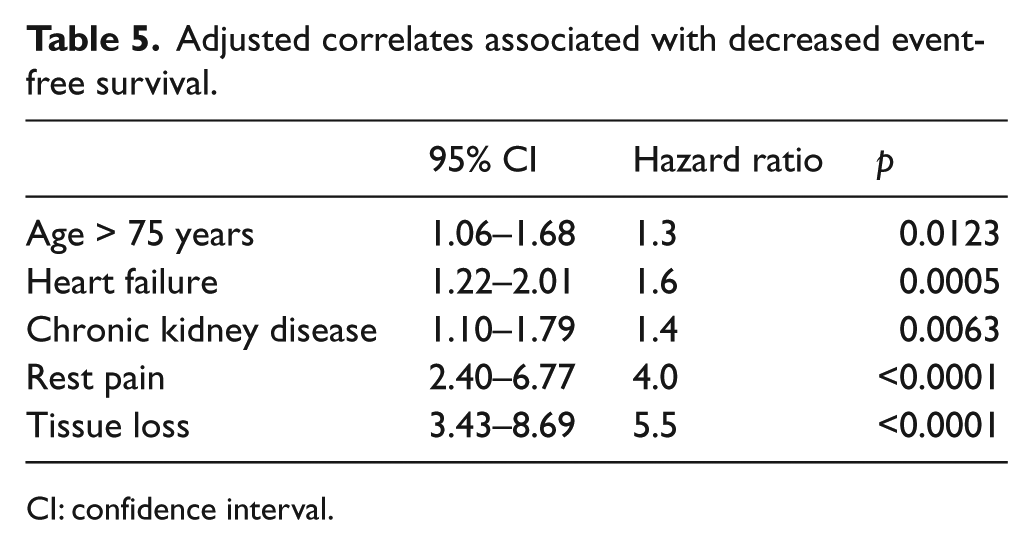
CI: confidence interval.
Discussion
Despite evidence that optimizing medical therapy in patients with PAD is associated with reduced mortality, 15 there remains a significant gap between guidelines and practice. The PARTNER study 16 was one of the first to reveal that patients with PAD receive less intensive risk factor modification than patients with CVD. Compared with patients with CVD, fewer patients with PAD were treated for dyslipidemia (56% vs 73%), received an antiplatelet agent (54% vs 71%), or were treated for hypertension. This pattern was also observed in a cohort of 1733 patients with isolated PAD identified by an administrative database but not confirmed by noninvasive testing. 17 More recently, a retrospective observational study demonstrated lower and less aggressive use of statins in patients with PAD versus those with coronary artery disease (CAD). 13
The use of risk factor modification in patients with symptomatic PAD who undergo LER is also low.8,11,18 Conte and colleagues examined the medical therapy provided to 1403 patients who underwent infrainguinal bypass for critical limb ischemia as part of the PREVENT III trial. 8 On admission, 67% were taking an antiplatelet and 46% a lipid-lowering agent. On discharge, antiplatelet use increased to 80%, while the percentage of patients on lipid-lowering therapy remained unchanged. Compliance with optimal medical therapy was more common in a cohort of 1357 patients who underwent endovascular intervention for stable claudication from 2007 to 2009 in the Blue Cross Blue Shield of Michigan Cardiovascular Consortium Peripheral Vascular Intervention Database. 11 On admission, 85% were taking an antiplatelet agent versus 92% on discharge; statin use increased modestly from 76% on admission to 81% on discharge. Appropriate therapy was used less often in minority patients and those diagnosed with chronic obstructive pulmonary disease (COPD), and more often in patients with diabetes, CAD, and prior peripheral revascularization.
In the present cohort, we identified a significant gap between guidelines and practice in the community that improved following admission for LER. At time of discharge, the percentage of patients with a perfect QoC score increased from 11% to 20.7%. Antiplatelet use and control of blood pressure and diabetes remained fairly constant, but statin use increased markedly during the observation period. Increased adherence to guidelines was driven by the vascular surgery admitting team, as cardiology consults were obtained largely to assist with pre-operative cardiac risk stratification rather than assessing medication regimens. A similar trend was observed by Armstrong et al., who assessed compliance with four guideline-based therapies: smoking cessation, and being placed on an aspirin, a statin, and an angiotensin-converting enzyme inhibitor. 19 From 2006 to 2011, compliance with all four therapies increased from 27% to 38%. 19 Although our dataset was not designed to address the question of why compliance with guidelines improved, we speculate that general awareness about the possible benefits of antiplatelets and statins in patients with PAD was increasing. Various strategies have been proposed to improve provider compliance and decrease further the gap between guidelines and practice including provider education, 20 use of health information technology including the electronic medical record, computerized provider order entry, and decision support, 21 financial incentives such as pay-for-performance, and penalties for nonperformance. 22
Consistent with other studies, we found that advanced age, heart failure, CKD, and more advanced LE ischemia were associated with the composite outcome of decreased event-free survival. 23 In our cohort, patient outcome did not appear altered by use of appropriate, guideline-based medical therapy. Our findings are in contrast to other studies that have shown a decrease in mortality, 24 major adverse cardiac and cerebral events, 19 and major adverse limb events15,19,25 with guideline-based medical therapy. Patients in the Vascular Study Group of New England cohort who were treated with an antiplatelet and statin demonstrated improved 30-day and five-year survival compared with patients not receiving these agents. 24 Only 26% of patients in that series underwent arterial bypass (the remainder underwent carotid intervention or repair of abdominal aortic aneurysm), and the degree of LE ischemia was not reported. Ardati and colleagues found that users of aspirin and statins had decreased peripheral intervention, limb salvage surgery, and amputation at six months, but no difference in death, myocardial infarction, and stroke. 11 Patients in that study were treated for claudication and followed for a relatively short duration. Armstrong and colleagues reported that PAD patients with claudication (44%) or critical limb ischemia (56%) who underwent angiography and met all four guideline-based therapies experienced decreased major adverse events compared with patients who receive fewer than four recommended therapies. 19 In the present cohort, > 75% of the patients underwent revascularization for critical limb ischemia. Thus, a greater percentage of patients in our series had rest pain and tissue loss and all underwent endovascular or surgical revascularization.
Patients who do not receive appropriate medical therapy until their PAD reaches an advanced stage may experience poorer outcomes. For example, a study of process of care in 796 PAD patients (ankle-brachial index (ABI) < 0.9) from the Veterans Administration found that glucose control was protective against death or a limb event. However, dispensing a PAD-specific medication (e.g. cilostazol or pentoxyphylline), achieving adequate blood pressure, and LDL control were associated with an increased risk of death. 26 Delayed recognition and treatment of PAD until it is so advanced that it produces LE symptoms may mitigate the potential beneficial effects of therapy.
Our study is subject to the limitations inherent in a retrospective study. The QoC score provides a snapshot of care—and compliance with guidelines—on admission and on discharge following LER. We did not exclude patients with reported intolerance to statins (statin-induced myopathy, elevated liver function tests, or intolerance to statin documented in the medical record) or to aspirin (history of hypersensitivity, concurrent warfarin therapy, history of coagulopathy or gastrointestinal (GI) bleeding). Although we expect that such exclusions would be limited, this may have lowered the QoC score for some patients. The QoC hypertension measure reflects blood pressure control in an inpatient setting, rather than the outpatient setting that is addressed by the JNC VII guidelines. Inpatient blood pressure measurements may be less accurate than in the outpatient setting and may reflect situational factors (acute pain, anxiety) that can result in blood pressure elevation. 27 The QoC indicator for glycemic control uses HgbA1C, a measure of glycemic control over three months. Thus, even if a patient failed to meet target A1C on admission but was prescribed a more aggressive diabetic regimen prior to discharge, the diabetes QoC measure would be unchanged. For many patients, the index admission may have represented their initial PAD diagnosis, and therefore they may not have been treated according to guidelines. Finally, we were unable to capture data on compliance with medical regimen on discharge, which may have reduced our ability to assess whether perfect QoC score is associated with better outcomes.
Although adherence to guidelines improved over time, we found a significant gap between guidelines and practice from 2007 to 2010. Clinicians caring for patients admitted for LER should take the opportunity to optimize therapy for this cohort of patients with markedly increased risk of adverse cardiovascular events.
All authors had access to the data and a role in writing the manuscript.
Footnotes
Appendix
Procedure codes for population selection.
| Procedure | ICD-9 code | CPT code |
|---|---|---|
| Surgical revascularization | 38.08, 38.18, 38.38, 38.48, 38.68, 38.88, 39.25 39.29, 39.49, 39.56, 39.57, 39.58, 39.59, 39.99 | 35556, 35566, 35571, 35583, 35585, 35587, 35621, 35623, 35641, 35646, 35654, 35656, 35661, 35665, 35666, 35671, |
| Endovascular revascularization | 00.55, 39.50, 39.79, 39.90, | 35474, 35470, 35492, 35493, 35495 |
ICD-9: International Classification of Diseases, Ninth Revision; CPT: Current Procedural Terminology.
Declaration of conflicting interest
None declared.
Funding
This work was supported by the American Heart Association (grant/award number: 13BGIA16720014), and the National Institutes of Health (grant/award numbers: K23 HL096893, R01 HL63090, andT32 HL007812).
