Abstract
Background and Aims:
Ulcerative colitis increases the risk of developing colorectal cancer. Colonoscopic surveillance is recommended although there are no randomized trials evaluating the efficacy of such a strategy. This study is an update of earlier studies from an ongoing colonoscopic surveillance program.
Material and Methods:
All patients with ulcerative colitis were invited to the surveillance program that started in 1977 at Örnsköldsvik Hospital, located in the northern part of Sweden. Five principal endoscopists performed the colonoscopies and harvested mucosal sampling for histopathological evaluation. Some 323 patients from the defined catchment area were studied from 1977 to 2014. At the end of the study period, 130 patients, including those operated on, had had total colitis for more than 10 years.
Results:
In total, 1481 colonoscopies were performed on 323 patients during the study period without any major complications. In all, 10 cases of colorectal cancer were diagnosed in 9 patients, of whom 1 died from colorectal cancer. The cumulative incidence of colorectal cancer was 1.4% at 10 years, 2.0% at 20 years, 3.0% at 30 years, and 9.4% at 40 years of disease duration, respectively. The standardized colorectal cancer incidence ratio was 3.01 (95% confidence interval: 1.42–5.91). Major surgery was performed on 65 patients; for 20 of these, the indication for surgery was dysplasia or colorectal cancer. Panproctocolectomy was performed in 43 patients.
Conclusion:
This study supports that colonoscopic surveillance is a safe and effective long-term measure to detect dysplasia and progression to cancer. The low numbers of colorectal cancer-related deaths in our study suggest that early detection of neoplasia and adequate surgical intervention within a surveillance program may reduce colorectal cancer mortality in ulcerative colitis patients.
Introduction
It is well known that patients with longstanding ulcerative colitis (UC) have an increased risk of developing colorectal cancer (CRC) (1, 2). In patients with UC, CRC is diagnosed approximately 10 years earlier, compared to patients with sporadic CRC; more than 25% of UC cancer patients are afflicted before the age of 40 (3).
The earlier strategy to avoid CRC development was prophylactic panproctocolectomy (PPC) after 8–10 years of disease. This procedure changed during the 1970s to the concept of colonoscopic surveillance in most inflammatory bowel disease centers. This concept is based on the assumption that CRC in UC is preceded by epithelial dysplasia in colorectal mucosa (4, 5). By harvesting biopsies at regular intervals throughout the colorectum, it may be possible to select patients at high risk of CRC and selectively recommend prophylactic PPC. Surgery could also be performed in a more favorable time for cure, as endoscopists might also detect CRC in earlier stages. Nevertheless, there are reports that this strategy is ineffective, and no randomized clinical trial data on surveillance compared to prophylactic surgery, or watchful waiting, are available (6–8). However, some data indicate that surveillance programs are beneficial for the patient in respect to mortality from CRC (9–12). Moreover, even if there is no clear and undisputed evidence of the efficacy of surveillance, several authorities and guidelines advocate such a strategy (13–16).
This report is a major update from an ongoing surveillance program of unselected patients with UC from a defined catchment area (9, 17, 18). We are now adding 14 more years of observation to the study in the aim of evaluating this surveillance program.
Materials and Methods
Study Population
All known patients with UC from the catchment area of Örnsköldsvik Hospital have been invited to attend an ongoing colonoscopic surveillance program, launched in 1977. The patients recruited to this program were found by scrutinizing charts from the Department of Surgery, Internal Medicine and Pediatrics as well as by repeated inquiries to the general practitioners in the area from 1961 to 1983. In addition to this historical search, patients were continually enrolled into the program. The catchment area is well defined, but the population has declined from 65,000 to 59,000 inhabitants (2014). Follow-up of the study ended at 14 November 2014. Total colitis (TC) was defined as morphologically altered mucosa concordant to colitis to or beyond the hepatic flexure, while left-sided colitis (LC) constituted cases with lesser extent of inflammation; patients with ulcerative proctitis only were excluded. End-points were PPC (indicating no cancer risk), death from any cause, relocation of the patient from the area, and termination of the study. The regional ethical review board at Umeå University approved the study.
Colonoscopies
Mainly five different endoscopists performed all colonoscopies. The first colonoscopy was made when the patients entered the study or at time of diagnosis. Repeat colonoscopies were performed at surveillance, initially after 10 years’ duration of the disease. In 1988, the protocol was altered to initiate surveillance after 6 years of disease. In patients with TC, colonoscopies were performed biannually until 20 years of disease and then annually. The protocol has principally been equivalent for patients with LC although some patients with clinically quiescent disease, who also were repeatedly negative for dysplasia in histological sampling, have been investigated every fourth year in their second decade and biannually in their third decade of disease. At colonoscopy, two samples were obtained from flat mucosa at six defined locations (cecum, ascending, transverse, descending, sigmoid colon, and rectum) for histopathologic evaluation; thus, the number of biopsy samples amounted to 12 in general. In addition to these sites, particular attention was paid to areas of mucosal irregularity and plaque-like lesions that might represent macroscopic areas of dysplasia (dysplasia-associated lesion or mass (DALM)).
Dysplasia was diagnosed and classified principally by two pathologists over the full study period, as high-grade dysplasia (HGD), low-grade dysplasia (LGD), and indefinite for dysplasia according to an international classification (19). Progression of dysplasia was defined as the subsequent occurrence of HGD, DALM, or CRC, after a previous finding of LGD. Repeated findings of LGD and diagnosis of HGD, DALM and CRC were indications for surgery, besides the most common indication of therapy-resistant disease (TRD).
Interval cancers were defined as invasive cancer diagnosed after the first negative screening examination, but before the next recommended surveillance examination, in accordance to guidelines (20).
Statistical Analyses
The statistical calculations were performed on the cohort, as it was constituted 14 November 2014. We performed survival analysis in order to evaluate time to CRC development in relation to UC onset and diagnosis of dysplasia; we also analyzed time to CRC or HGD combined. Censoring was made for any other end-point, detailed above. We also calculated the relative risk for development of CRC, expressed as the standardized incidence ratio (SIR), which was defined as the ratio between the observed and the expected number of cases in the cohort compared to the background population. Stratification was performed for extent of colitis. All statistical analyses were performed in the statistical softwares SPSS and R, versions 23 and 2.15, respectively.
Results
Patients
Some 323 patients were enrolled in the surveillance program, whereof 14 (4.3%) changed diagnosis during their course of disease to predominantly Crohn’s disease, resulting in 309 patients with confirmed diagnosis of UC. A total of 1481 colonoscopies were performed on all of the above patients without major complications. Clinical data on the study cohort are available in Table 1. Of note, more than two-thirds of the patients suffered from TC, whereas the remainder was made up of LC (Fig. 1).
Clinical data in 323 patients who underwent colonoscopic surveillance for ulcerative colitis.
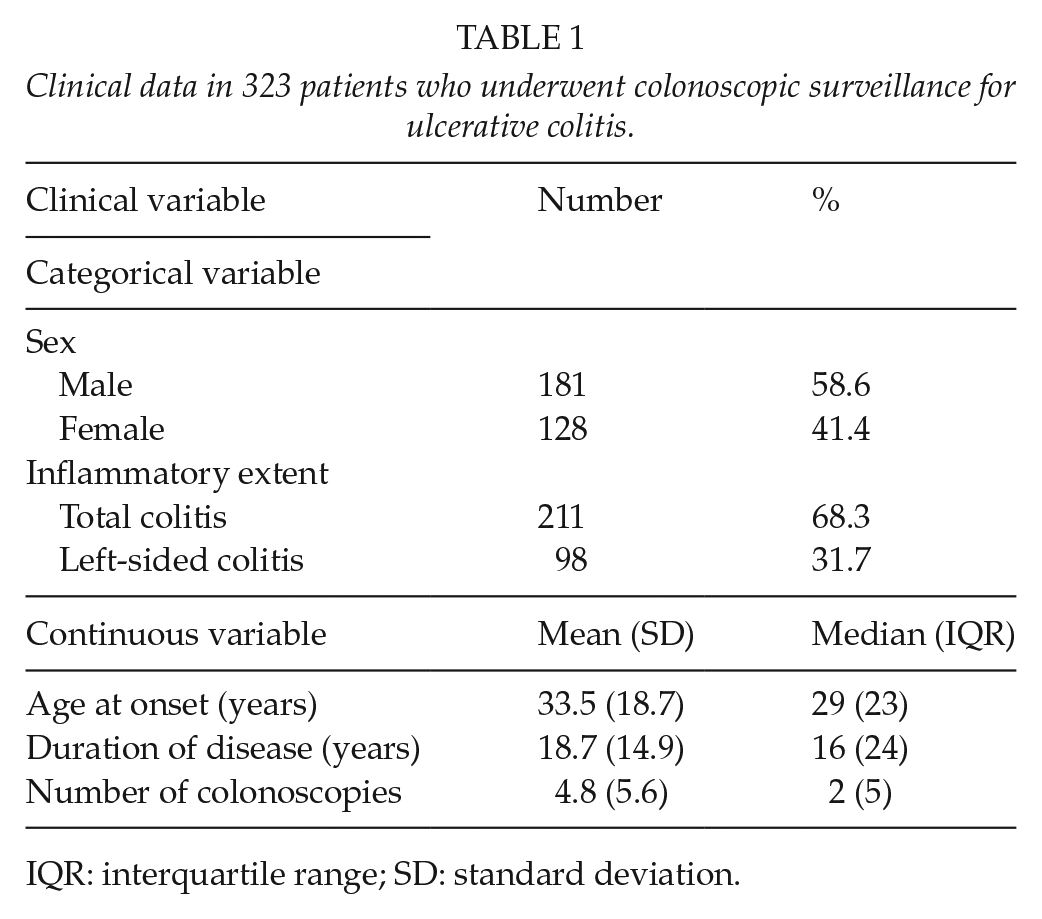
IQR: interquartile range; SD: standard deviation.
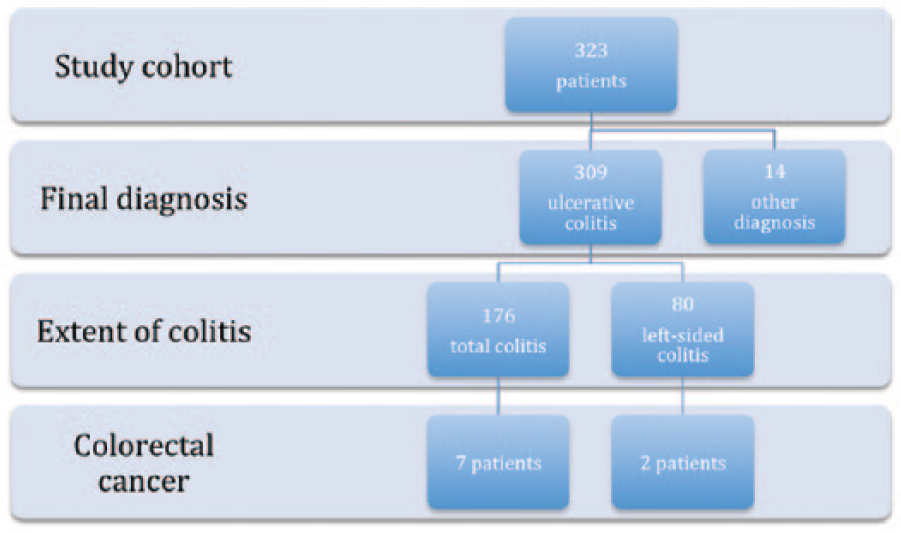
Flowchart showing the course for patients with colorectal cancer at the end of the study.
During the study period, 68 patients died, of whom 53 from intercurrent disease or advanced age. In all, 2 died from CRC, 1 from UC, 1 from pouch perforation, and 11 from unknown reasons. In all, 33 patients moved out of the catchment area, and 19 were excluded because of poor compliance. Totally, 17 patients left the program because of advanced age and 11 patients due to intercurrent disease. Some 233 patients entered, and 151 left the program. At the conclusion of the study, 130 patients, including those who had undergone partial colorectal resection before or after 10 years of disease and those who had undergone PPC after 10 years of disease, had TC for more than 10 years. The study comprised 5777 person-years.
Cancer Risk
Progression from LGD to a more complex morphologic aberration was seen in 21 of 49 (43%) patients. These patients were monitored up to 16 years (median: 3 years) before any aggravation in dysplasia; eventually, indication for surgery was dysplasia and cancer in 12 and 3 patients, respectively, while 6 patients were not operated on (one patient had a non-diagnosed CRC at autopsy). Time to CRC development, stratified by diagnosis of LGD and HGD, is shown in Fig. 2, while time to development of any dysplasia or cancer in all patients is shown in Fig. 3; furthermore, time to development of HGD or cancer is displayed in Fig. 4.
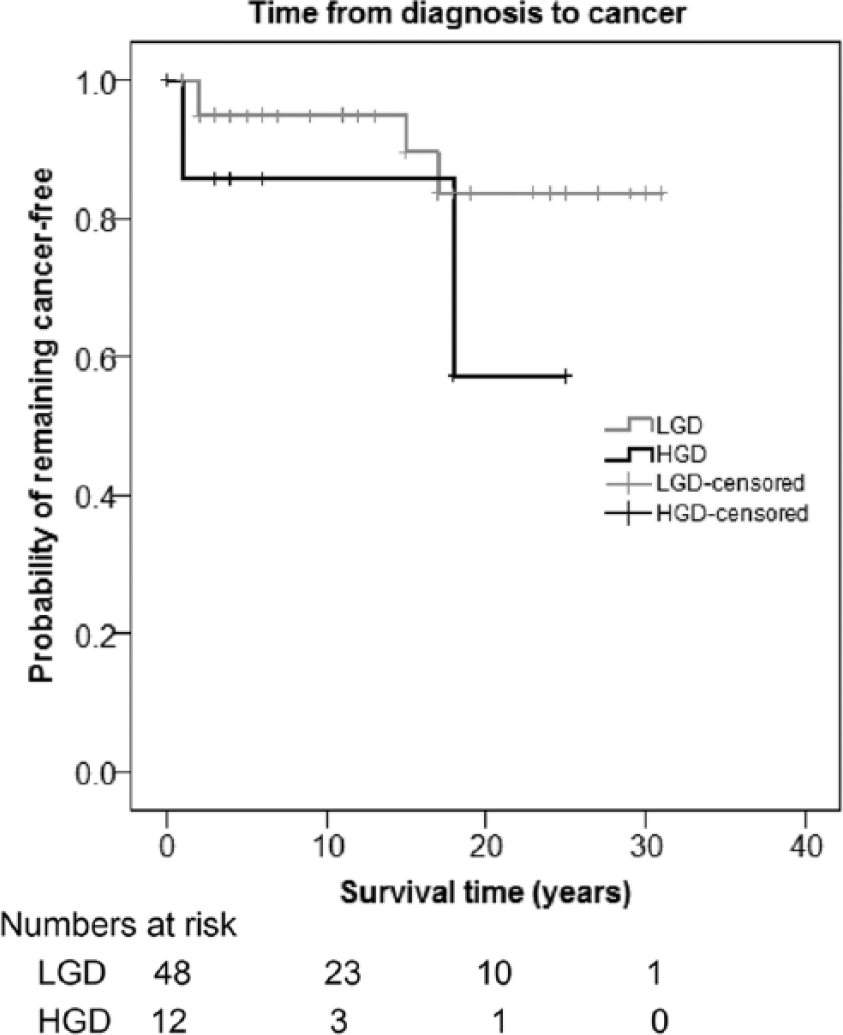
Time to development of colorectal cancer (CRC) after diagnosis of low-grade dysplasia (LGD) or high-grade dysplasia (HGD).
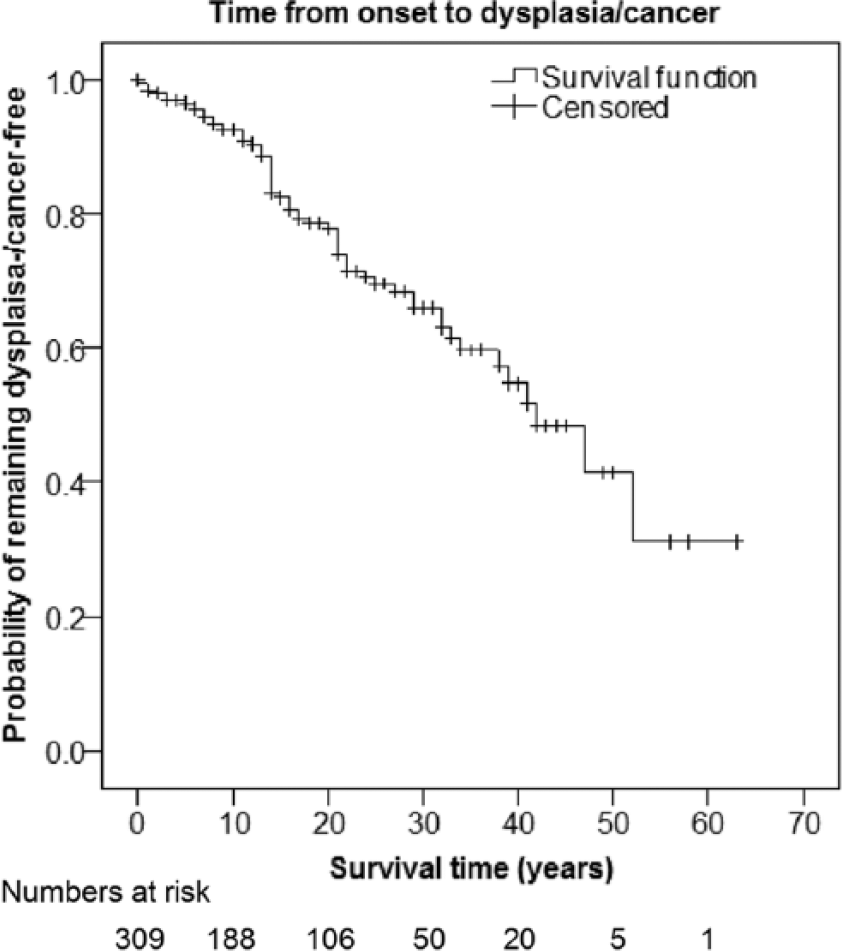
Time to development of any dysplasia or cancer from ulcerative colitis (UC) onset.
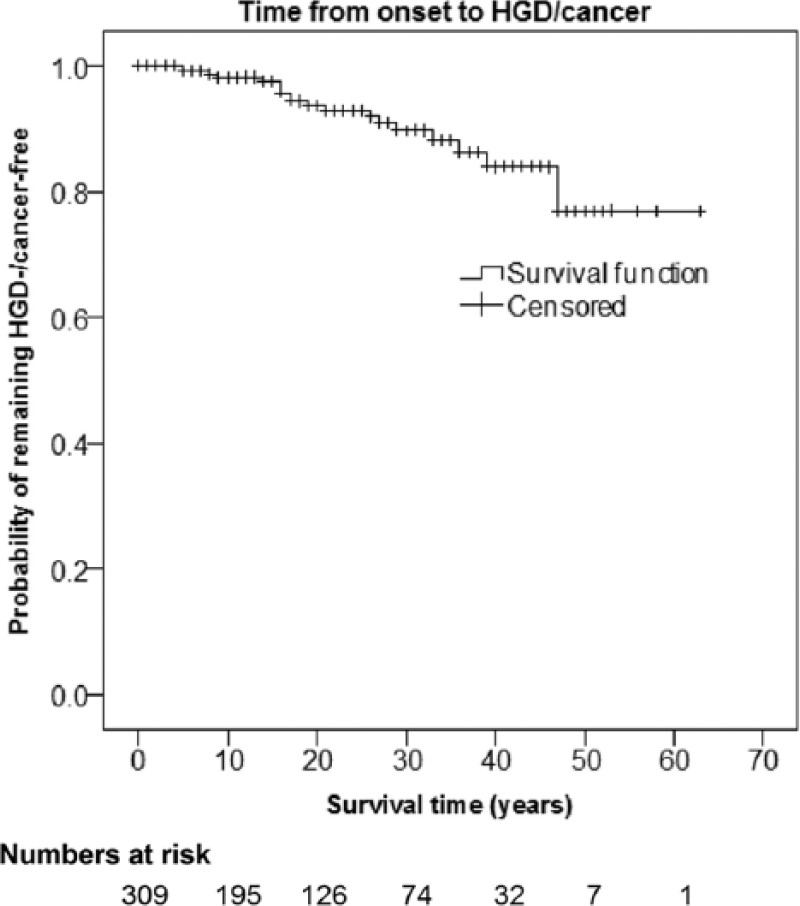
Time to development of high-grade dysplasia (HGD) or colorectal cancer (CRC) from ulcerative colitis (UC) onset.
In all, 10 CRCs were found in 9 patients, as one patient was diagnosed post-mortem with synchronous transverse and rectal tumors, after having refused treatment for fatal acute colitis. Only one patient died because of metastatic CRC; this patient was diagnosed with cecal cancer 9 years after the initial colonoscopy and before the first planned surveillance colonoscopy (during a period when surveillance started after 10 years of disease). Interval cancers affected one out of eight patients, while it was not possible to evaluate interval cancer status in one case.
The cumulative CRC incidence is depicted in Fig. 5. After 10, 20, 30, and 40 years of disease, the cumulative incidence was 1.4%, 2.0%, 3.0%, and 9.4%, respectively.
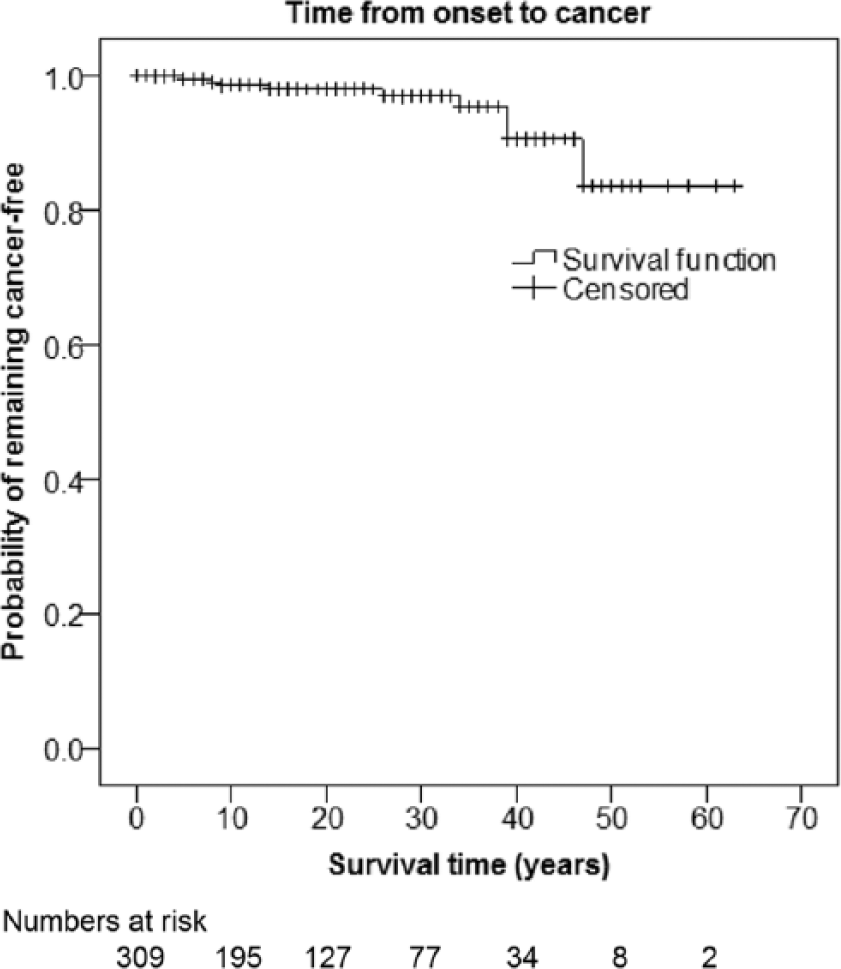
Time to colorectal cancer (CRC) development from ulcerative colitis (UC) onset.
The SIR was calculated at 3.01 (95% confidence interval (CI): 1.42–5.91); the observed CRC incidence was 9 while the expected number of cases amounted to 2.7 during the period. The corresponding SIRs for TC and LC were 3.50 (95% CI: 1.41–7.20) and 2.23 (95% CI: 0.27–8.0), respectively.
Surgical Resections
Some 87 major colorectal operations were performed on 65 patients, of whom 57 had TC and 8 LC. Indications for surgery were severe TRD, 40 patients; repeated findings of LGD in flat mucosa, 2 patients; findings of HGD, 4 patients; DALM, 7 patients; or CRC, 7 patients. One patient was operated on for colonic lymphoma, one due to a spontaneous colonic perforation and another patient because of primary sclerosing cholangitis; the latter patient underwent surgery in 1981 in the belief at the time that PPC would slow the disease progress. The indications for surgery for two patients were unknown. The patients were informed of the different surgical procedures, that is, PPC with ileal pouch anal anastomosis (IPAA), PPC with permanent ileostomy, and colonic and/or rectal resection. Some of the patients rejected surgery as long as there was no established CRC. Even if CRC was diagnosed, some patients did not want to undergo PPC and therefore a colonic and/or rectal resection was performed (21).
At the end of the study, 43 patients had been operated on with PPC, 19 with colonic resection (18 colectomies and 1 subtotal colectomy) and 3 with rectal resection. There were unexpected findings of one case of CRC, one case of HGD, and one case of LGD in the operative specimens.
Discussion
In this population-based cohort study of colonoscopic surveillance for UC, we found that patients under surveillance risked developing CRC of 1.4% at 10 years, 2.0% at 20 years, 3.0% at 30 years, and 9.4% at 40 years after disease onset; compared to the background population, there was a threefold increased risk of developing CRC in these UC patients. We also observed that 43% of patients that developed LGD eventually progressed during follow-up.
The main strength of this study is the long period of follow-up, comprising 38 years of surveillance. The patient cohort derives from a defined catchment area with low population mobility, which provides population-based data, thus alleviating selection bias. Moreover, patient compliance has been excellent, as only 19 out of 323 patients abstained from surveillance. Staff turnover among the endoscopists as well as pathologists has also been minimal, ensuring uniform reporting and registration. Weaknesses of the study include the conventional technique used during colonoscopy, as chromoendoscopy was not routinely available; several studies indicate that chromoendoscopy is superior regarding the yield of dysplasia in targeted biopsies (22–24). Furthermore, no detailed data on medical anti-inflammatory treatment are available, which may otherwise modify dysplasia development in UC patients; in the early years of the program, sulfasalazine medication was provided for relatively asymptomatic patients, while this was replaced later on with 5-aminosalicylate drugs and, in some cases, azathioprine—very few patients were treated with monoclonal antibodies, for example, infliximab. To add, the study sample size is admittedly small, making statistical inference difficult, as shown by the wide CIs.
Colonoscopic surveillance does not eliminate the risk of advanced CRC, as shown in the St Mark’s cohort study, in which 13 patients developed advanced cancers during surveillance (3). In the current cohort, progression from LGD amounted to 43%, a similar figure to the 39% found in the former study (3). The interval cancer risk was low in our population, defined according to recent guidelines (20); however, if advanced cancer (stage III or worse) is included in the interval cancer definition, numbers accumulate to five out of eight evaluable cases, which is more concordant to earlier research (3).
We found an increased risk of CRC in our cohort relative to the background population with an SIR of 3.01 (95% CI: 1.42–5.91). Most population-based studies support the notion that UC is a risk factor for CRC (2, 25), but results are not consistent (26, 27). The corresponding CRC incidence rate was 1.56/1000 person-years in this study, which is in accordance with the results from the most recent meta-analysis performed by Castano-Milla et al. (28). In the latter analysis, it was also shown that the CRC risk has declined in the last decades, possibly due to better surveillance and improved medical management (13–16). This study supports this contention, as also the cumulative incidence rates of CRC stratified by decade of disease duration show only a moderate CRC risk, in contrast to the earlier meta-analysis performed by Eaden et al. (1).
While this study was not aimed at elucidating risk factors for CRC development, there was support in our data for the established risk factors long duration of disease and extensive involvement.
Today, there are hardly any authorities that argue against surveillance in UC; nevertheless, there are indications that the implementation of surveillance programs meets resistance (29). The results in this study support the findings of an elevated risk of CRC in patients with UC and that colonoscopic surveillance is an effective method of diminishing lethal CRC. It also provides data that surveillance programs are feasible even over time, with a high grade of compliance.
During the study period, there has been a conservative approach toward surgery in the hospital. Patients with findings of LGD have been followed for a long time, with more frequent colonoscopies rather than prompt surgery. As less than half of the LGD population eventually progressed to more advanced dysplasia, the natural history of LGD may be less malignant than previously thought, corroborating earlier data (30). It also seems safe to respond to the finding of LGD with mere adaptation of the surveillance intervals.
In conclusion, this study supports that colonoscopic surveillance is a safe and effective long-term measure to detect dysplasia and progression to cancer. The low numbers of CRC-related deaths in our study suggest that early detection of neoplasia and adequate surgical intervention within a surveillance program may reduce CRC mortality in UC patients.
Footnotes
Acknowledgements
The authors are grateful to Örnsköldsvik Hospital gastroenterologists Matts Andersson, MD, and Silvia Alaoui, MD, for their invaluable effort in providing colonoscopic surveillance. They would also like to thank statistician Erling Englund for his sound advice.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The regional ethical review board at Umeå University approved the study.
Funding
The Cancer Research Foundation in Northern Sweden, Research Council Västernorrland County, and the Swedish Cancer Society have provided financial support.
