Abstract
Objectives:
Since the early 1990s, low long-term survival rates following pancreatic surgery for pancreatic ductal adenocarcinoma have challenged us to improve treatment. In this series, we aim to show improved survival from pancreatic ductal adenocarcinoma during the era of centralized pancreatic surgery.
Methods:
Analysis of all pancreatic resections performed at Helsinki University Hospital and survival of pancreatic ductal adenocarcinoma patients during 2000–2013 were included. Post-operative complications such as fistulas, reoperations, and mortality rates were recorded. Patient and tumor characteristics were compared with survival data.
Results:
Of the 853 patients undergoing pancreatic surgery, 581 (68%) were pancreaticoduodenectomies, 195 (21%) distal resections, 28 (3%) total pancreatectomies, and 49 (6%) other procedures. Mortality after pancreaticoduodenectomy was 2.1%. The clinically relevant B/C fistula rate was 7% after pancreaticoduodenectomy and 13% after distal resection, and the re-operation rate was 5%. The 5- and 10-year survival rates for pancreatic ductal adenocarcinoma were 22% and 14%; for T1-2, N0 and R0 tumors, the corresponding survival rates were 49% and 31%. Carbohydrate antigen 19-9 >75 kU/L, carcinoembryonic antigen >5 µg/L, N1, lymph-node ratio >20%, R1, and lack of adjuvant therapy were independent risk factors for decreased survival.
Conclusion:
After centralization of pancreatic surgery in southern Finland, we have managed to enable pancreatic ductal adenocarcinoma patients to survive markedly longer than in the early 1990s. Based on a 1.7-million population in our clinic, mortality rates are equal to those of other high-volume centers and long-term survival rates for pancreatic ductal adenocarcinoma have now risen to some of the highest reported.
Introduction
Pancreatic surgery has a high morbidity rate, and even very high-volume hospitals report high postoperative mortality. In-hospital, 30-day, or 60-day mortality in high volume centers for pancreatic-head resections ranges from 0.9% to 8.1% (1–5). In a very high-volume center in Heidelberg, Germany, with a case load of more than 2000 pancreaticoduodenectomies, the in-hospital mortality rate was 3.9% (5). Overall morbidity after pancreaticoduodenectomy has even been 59% with 27% of the cases being serious adverse events (6, 7). Post-operative pancreatic fistula is one of the most serious complications and may lead to death.
Overall long-term survival (>5 years) of pancreatic ductal adenocarcinoma (PDAC), ranges from 0%–18% (4, 8–10). In a debate over the accuracy of a histological diagnosis of PDAC one suggestion is that diagnosis of all long-time survivors needs re-evaluation by experienced pancreatic pathologists (8).
Only a few reports from high-volume centers have indicated longer survival for a certain very small proportion of PDAC patients (2, 4, 9). These cases contribute 5%–6% of all patients undergoing surgery for PDAC (2, 4).
The earlier high postoperative mortality, complication rates, and very low long-term survival rates for PDAC patients after pancreatic surgery have led to a demand for improved treatment for this devastating disease. Internationally, centers for pancreatic surgery have been established with multidisciplinary treatment. By this approach, diagnostic accuracy of PDAC, postoperative mortality, and long-term survival for PDAC patients can be improved markedly (11–14). In southern Finland, all pancreatic resections were centralized to Helsinki University Hospital, with its 1.7 million population basis from the year 2000.
Aim
The aim of this study was to evaluate the outcome of patients undergoing pancreatic surgery in general as well as the possible survival benefits of PDAC patients in our hospital during its centralization.
Patients And Methods
From the Helsinki University Hospital database, we identified all patients undergoing pancreatic surgery other than necrosectomy between January 2000 and September 2013. Survival data came from patient records and from the Finnish population registry on September 2014 followed by analysis of cause of death (COD) from pancreatic cancer or unrelated cause. Information on age, gender, American Society of Anesthesiologists (ASA) Physical Status Classification, tumor-histology, grade, Union for International Cancer Control (UICC) size of the tumor, nodes, and metastasis (TNM) stage, lymph-node status, margin-resection status (R, considered R0 when the clear resection margin is >1 mm), type of resection, complications, adjuvant therapy, and tumor-marker levels came from patient records. Analysis of postoperative pancreatic fistula rates was according to the international study group of post-operative pancreatic fistula (ISGPF) criteria (15).
Our pancreaticoduodenectomy operation technique was developed during the 1990s to become a standardized procedure by the year 2000. For malignant disease, operations began with laparoscopy to exclude peritoneal metastases. When none were detected, a transverse laparotomy followed. Pancreatic resection was according to the non-touch en-block principal including distal gastrectomy. Resection included right-sided omentectomy, excision of the right cranial peritoneal leaf of the transverse colon exposing the caval vein and renal veins. Excision of the lymph nodes of the hepaticoduodenal ligament, around the right side of the celiac axis, and excision of the aorto-caval notch caudally to the inferior arterial mesenterial axis, as well as along the anterior and right lateral side around the origin of the superior mesenteric artery were followed. If the tumor had infiltrated the superior mesenteric or portal vein, resection was the choice. Pancreatico-jejunal anastomosis was routinely performed end-to-side with a double-layer duct-to-mucosa suture line. All the patients were referred to adjuvant therapy. Neoadjuvant therapy was administered when the tumor was radiologically borderline resectable (16).
All histological specimens were re-evaluated and the diagnosis for PDAC was carried out by two experienced pancreatic pathologists. All slides from 2000 to 2006 were re-evaluated by both our local pancreatic pathologist (S.N.) and a German pancreatic pathologist (J.L.). Centralization of surgery over time led to centralization of pathology, as well, and specimens from 2007 and later were re-evaluated locally (S.N.). Pancreatic malignancies other than PDAC were excluded from survival analysis.
This study complies with the Declaration of Helsinki and was approved by the Surgical Ethics Committee of Helsinki University Hospital, and the National Supervisory Authority of Welfare and Health.
Statistics
Life tables were calculated according to Kaplan–Meier survival analysis, and the log rank test served for comparisons. Pancreatic cancer-specific overall survival was calculated from date of operation to death. Cox proportional hazards regression served for multivariate analysis. The Cox model assumption of constant hazard ratios (HRs) over time was tested by including the time-dependent covariate separately for each testable variable, when the assumption was valid. Variables included in the analyses were T-classification, N-classification, disease-free margin R0, lymph-node ratio (LNR) below 20%, adjuvant treatment, grade, perivascular invasion, carbohydrate antigen 19-9 (CA19-9) <75 kU/L, and carcinoembryonic antigen (CEA) <5.0 µg/L. Backward conditional stepping removed insignificant variables at p < 0.1. Results are age- and sex-adjusted. All statistical analyses were performed with SPSS (v21; IBM, New York, NY, USA). Statistical significance was p < 0.05.
Results
From January 2000 to September 2013, 853 patients underwent pancreatic surgery, (Table 1). Of the 853, 581 (68%) patients underwent pancreaticoduodenectomy, 195 (21%) distal pancreatic resection, 28 (3%) total pancreatectomy, and 49 (6%) other pancreatic procedures such as middle pancreatic resection, enucleation, or uncinate resection.
Patients and operations (n = 853).

BMI: body mass index.
Post-Operative Outcome
Of the 853, 13 (1.5%) patients died post-operatively. The in-hospital mortality after pancreaticoduodenectomy was 2.1% (Table 2.) Clinically relevant grade B or C fistulas occurred in 7% after pancreaticoduodenectomy and in 13% after distal resection (Table 2). Of the 44 (5%) patients who were re-operated, 40 (91%) were after pancreaticoduodenectomy. There occurred 17 (39%) pancreatic leaks (grade C fistulas), 10 (23%) biliary leaks and peritonitis, 13 (30%) hemorrhages, and four other re-laparotomies.
Postoperative complications after the pancreatic resection.
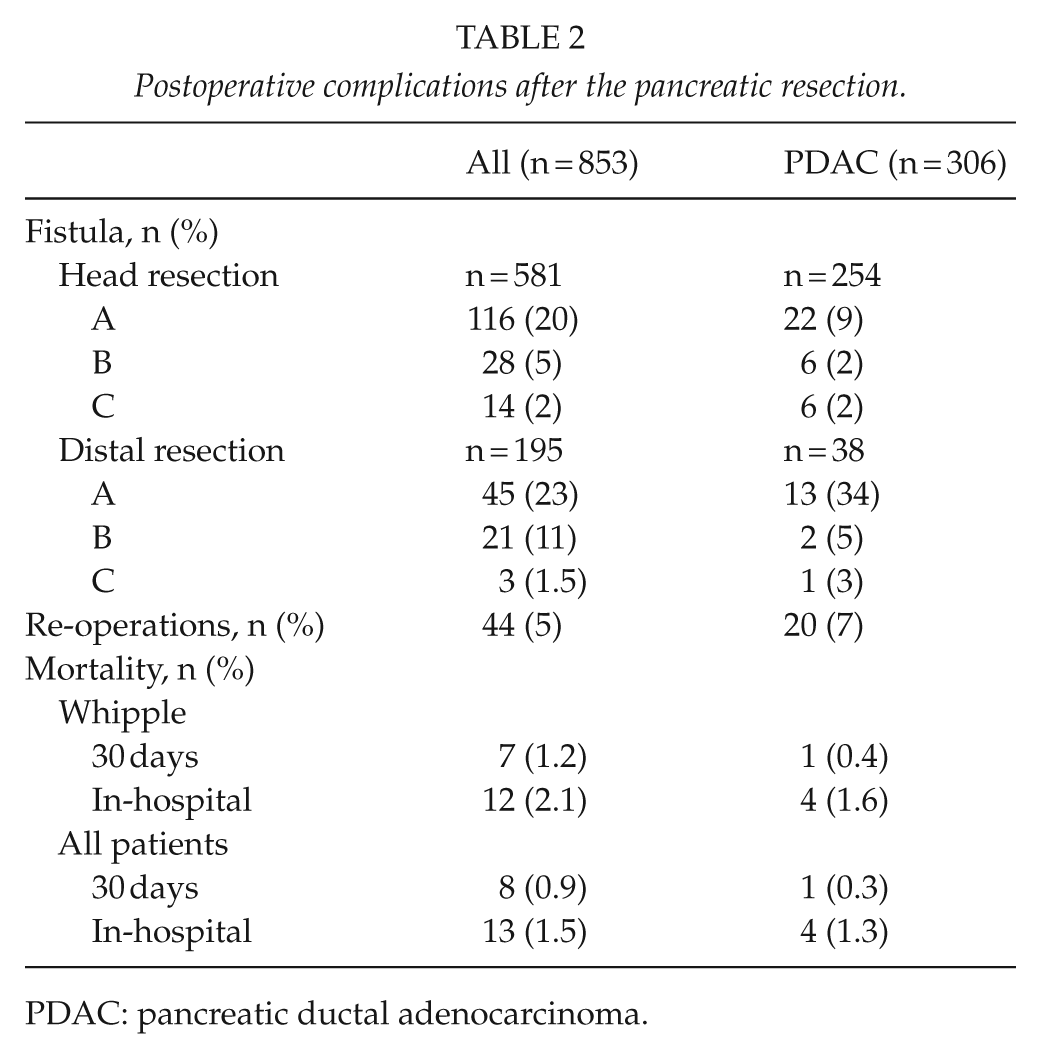
PDAC: pancreatic ductal adenocarcinoma.
Histological diagnosis was PDAC for 309 patients. After excluding three cases with a diagnosis not congruent, 306 patients remained for final analysis (Table 3). Of the PDAC patients, 52 (17%) received neoadjuvant therapy, consisting of either gemcitabine, gemcitabine combined with cisplatin, oxaliplatin, or radiation therapy. We had 151 (53%, with information being available of 285 patients) patients who received adjuvant therapy, mainly gemcitabine or its combination with cisplatin, kabesitabine, or radiation therapy. The 23 (8%) patients who turned out to have metastasis at imaging after surgery but before receiving adjuvant therapy, all received palliative oncological therapy.
Clinical and pathological parameters of the 306 PDAC patients.

CA19-9: carbohydrate antigen 19-9; CEA: carcinoembryonic antigen; ASA: American Society of Anesthesiologists; SEM: scanning electron microscopy; BMI: body mass index.
Long-Term Outcome
The overall 1-, 3-, 5-, and 10-year survival rates for the 306 patients radically operated on for PDAC were 74%, 36%, 22%, and 14% (Table 4). In univariate analysis, longer survival was significantly associated with CA19-9 <75 kU/L (p = 0.001), CEA <5.0 µg/L (p = 0.001), smaller tumor (T1-2) (p<0.001), lack of lymph-node metastasis (p = 0.001) or LNR <20% (p = 0.001), higher histological differentiation grade (p = 0.004), lack of perivascular invasion (p = 0.001), and having adjuvant therapy (p = 0.016) (Table 5; Fig. 1). Lack of perineural invasion was associated with a tendency for better survival (p = 0.055) (Table 5). Neither age, gender, and ASA class nor neoadjuvant therapy was significantly associated with survival.
Survival of 306 patients undergoing surgery for PDAC in Helsinki from January 2000 to September 2013.

Note: p-values calculated with the log rank test versus other patients at 5 years (i.e. not R0N0 or not T1-2, R0N0, Fig. 1H).
Factors associated with survival in PDAC patients in univariate analysis (n = 306).
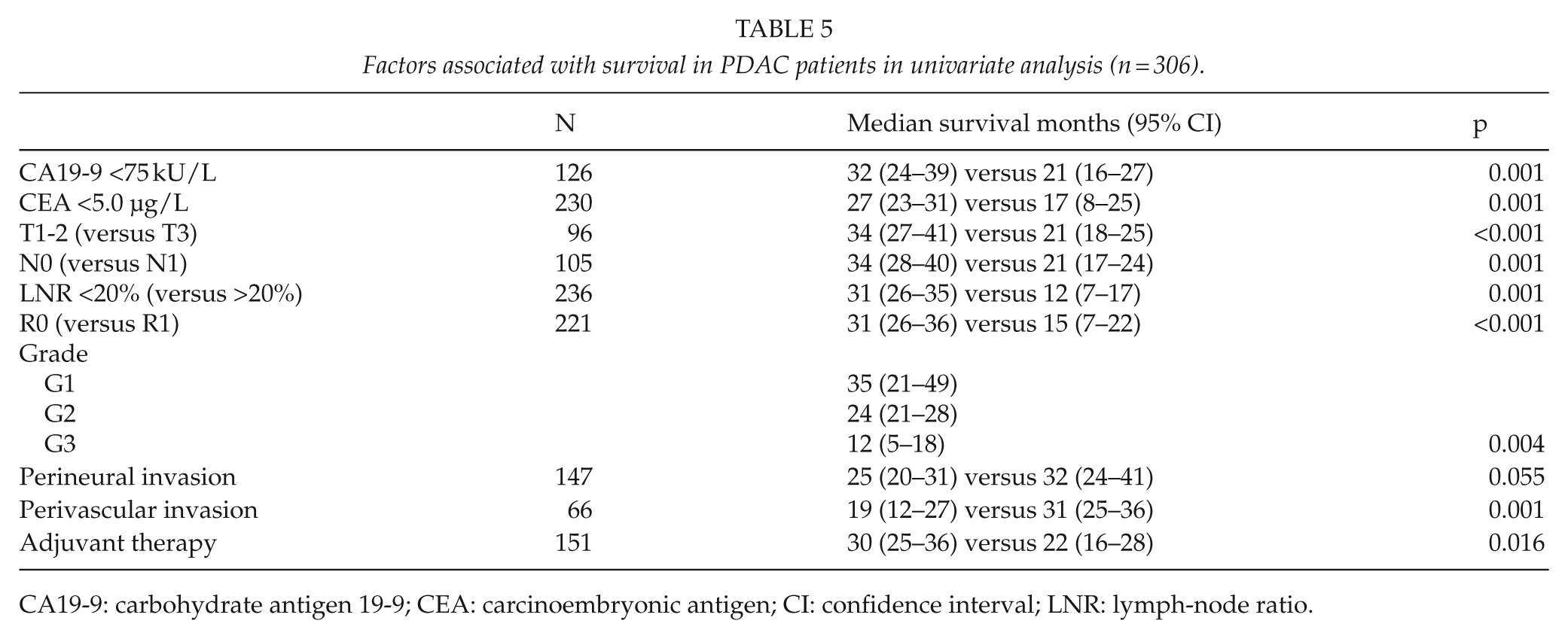
CA19-9: carbohydrate antigen 19-9; CEA: carcinoembryonic antigen; CI: confidence interval; LNR: lymph-node ratio.
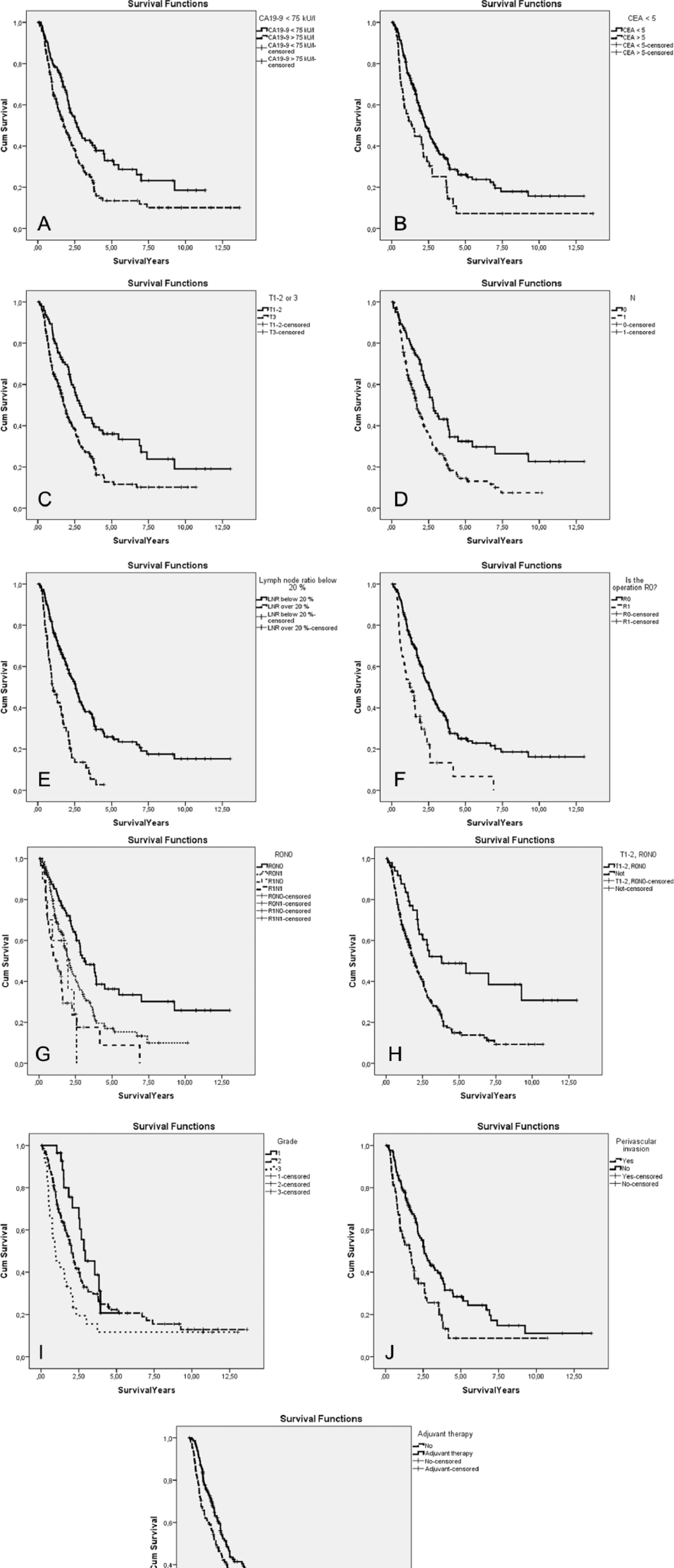
Association of A) preoperative CA19-9 <75 kU/L (p = 0.001), B) preoperative CEA <5.0 µg/L (p = 0.001), C) tumor stage T1-2 (p < 0.001), D) N-stage (p < 0.001), E) LNR < 20% (p < 0.001), F) disease-free margins (R0) (p < 0.001), G) lymph node status and resection margin (p < 0.001), H) small tumor stage (T1-2), no lymph-node metastasis (N0) and clear resection margins (R0) (p < 0.001), I) histological differentiation grade (p = 0.004), J) perivascular invasion (p = 0.001), and K) adjuvant therapy compared to observation alone (p = 0.016) on survival after the pancreatic resection in PDAC patients.
When the R0N0 and tumor stage T1-2 patients (n = 49, 16%) were analyzed separately, their survival rates for five and 10 years were 49% and 31% (median survival 46 versus 23 months, p < 0.001) (Table 3). More than half the N1 patients (111, 57%) received adjuvant therapy and benefitted significantly from it (median survival 25 versus 13 months, p = 0.021).
In multivariate analysis, patients who received post-operative adjuvant treatment had a significantly decreased HR, while patients with N1 status or patients with R1 had significantly poorer HRs than did patients with N0-status, radical operation (R0), and who had received no adjuvant treatment. Patients with CA19-9 >75 kU/L, CEA >5.0 µg/L, and LNR >20% also had significantly increased HRs (Table 6). Multivariate analyses were adjusted for age and sex.
Multivariate analysis of 306 patients undergoing surgery for PDAC in Helsinki January 2000 to September 2013.
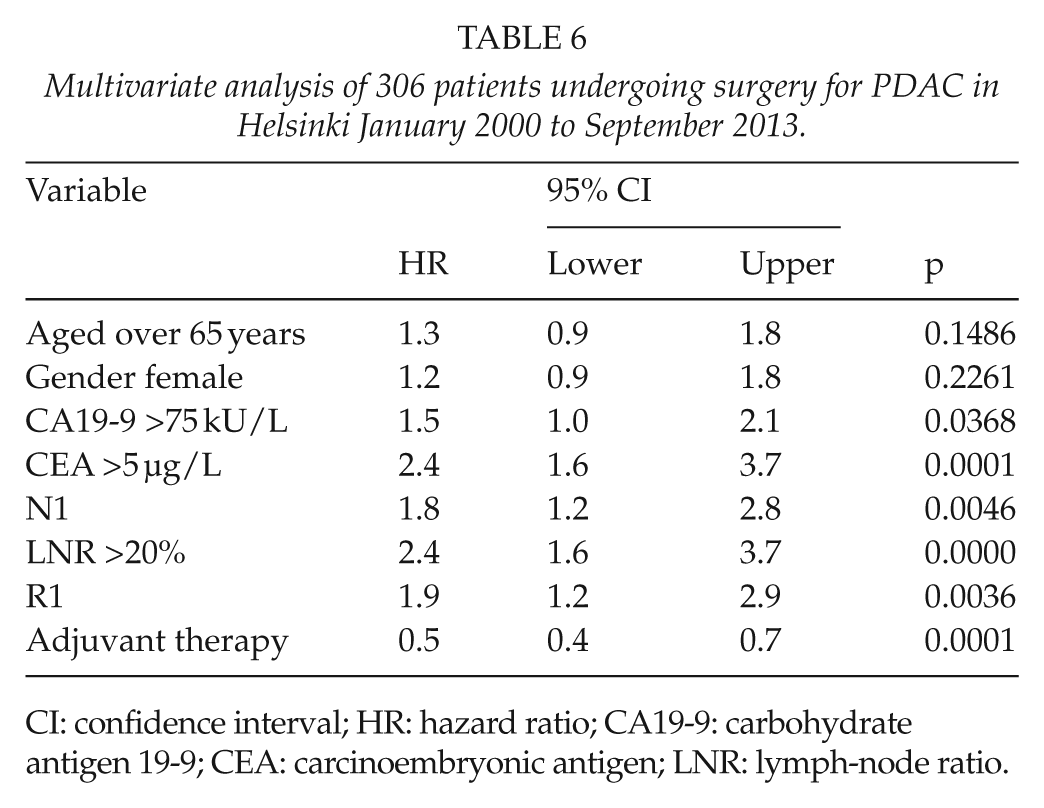
CI: confidence interval; HR: hazard ratio; CA19-9: carbohydrate antigen 19-9; CEA: carcinoembryonic antigen; LNR: lymph-node ratio.
Discussion
Since the beginning of 2000, the population base of our hospital has increased to 1.7 million. Larger hospital volume and improved surgical treatment may explain our very low post-operative in-hospital mortality rate nowadays of 2.1% after pancreaticoduodenectomy, which is at the same level as reported from very-high-volume international pancreatic centers. For example, Johns Hopkins Hospital in the USA reported 1%–2% postoperative mortality rates among almost 1500 pancreaticoduodenectomies, and Heidelberg in Germany, an in-hospital mortality of 3.9% after more than 2000 pancreatic resections (2, 5). In one Japanese pancreatic surgery database with 8575 pancreaticoduodenectomies, the in-hospital mortality was 2.8% (17).
In our hospital, clinically relevant ISGPF grade-B or -C fistulas occurred in 7% of pancreaticoduodenectomies. Johns Hopkins Hospital reported a 5% fistula rate (2). The variation has been high (5%–16%) among centers (5, 17). After distal resections fistula rates vary even more (0%–40%) (5, 18). In our hospital, the grade-B or -C fistula rate after distal resection was 13%. Several attempts have been made to reduce the number of fistulas by differing surgical techniques and with somatostatine analogs. A Cochrane analysis recommended octreotide for high-risk patients only (19). Prophylactic pasireotide in a recent randomized single-center study reduced the number of clinically significant fistulas to half (20). Pasireotide has been in prophylactic use in our center since that publication, but following the period of the current study. Our re-operation rate was 5% at the same level as in other large series reporting a 3%–6% re-operation rate (9, 21)
In our series, the overall 5-year survival of PDAC was 22% and in a subgroup of patients with T1-2, N0, and R0 tumors (49 patients, 16% of all) the 5-year survival was a very high 49% and even the 10-year survival was 31%. In 2006, at Johns Hopkins Hospital, the overall 5-year survival for pancreatic head cancer was 18%. When the tumor was <3 cm, and well or moderately differentiated, radically operated (R0), and lymph nodes were free of cancer, 5-year survival improved to 43% (2). Of 1175 pancreaticoduodenectomies for pancreatic head cancer, the patients with these features numbered 56 (4.8%) (2). In Heidelberg, the survival, among 1071, of an almost similar group of 62 patients (5.8%) (tumor grading Tis-T2), the 5-year survival was 54% (4). In our series, Tis was considered high-grade dysplasia, and these patients were not included in PDAC survival analysis. One explanation for our notably good survival results may be that an increasing number of patients are operated on at an earlier stage of the disease. In our series, tumor stage was T1-2 in 96 (32%) of the patients or the lymph nodes were free of metastasis in 105 (35%) patients compared to other centers with a 76%–78% N1 prevalence (2, 4). Several other favorable tumor characteristics such as CA19-9 <75 kU/L, CEA <5.0 µg/L, higher cancer-differentiation grade, and lack of perivascular invasion favored longer survival in both univariate and multivariate analysis, as could be expected. Our results stress the need for an earlier diagnosis of PDAC.
The role of extended lymphadenectomy has been under debate. In gastric cancer, the increased survival after D2 lymph node dissection encouraged us to change the surgical technique toward the same principle also in PDAC patients (22, 23). In some studies, extended lymhadenectomy has led to an increased number of R0 resections associated with better survival. Whether extended lymphadenectomy improves overall survival remains beyond the scope of our study. Thus far, five prospective randomized studies show no survival benefit after extended lymphadenectomy. Conversely, after extended lymphadenectomy, one meta-analysis showed a decrease in two- and 3-year survival (4, 24–29).
Covering the years 1990–1996, a former one Finnish study showed that only one patient in Finland with PDAC survived clearly longer than 5 years (8). A high number of misdiagnoses among long-term survivors were falsely listed in the tumor registry. Our current data are the results of the era of centralization of pancreatic surgery. Moreover, pathology has been centralized, and with the increase in cases an increase in experience in that field was gained. High-volume centers can achieve high diagnostic experience meaning that only limited outside reference evaluation is necessary. This was supported by two independent pathologists confirming the diagnosis of PDAC between 2000–2006 (S.N. and J.L.). In addition, the classification of pancreatic tumors (World Health Organization (WHO)) has been standardized and is in general use as well due to the increasing numbers of pancreatic-resection specimens.
In the Carpelan-Holmström study (8) no patients (data from 1990–1996) received oncological adjuvant treatment, although now it has become routine after pancreatic cancer surgery. In the 21st century, prospective randomized European Study Group for Pancreatic Cancer (ESPAC) 1-3 and Charité Onkologie 001 (CONCO-001) trials have shown that patients receiving adjuvant chemotherapy survive significantly longer (30, 31). Our retrospective series showed a similar result in favor of adjuvant therapy. Neoadjuvant therapy has in some series improved surgical results, raising the number of R0 resections. Further randomized controlled studies are necessary (32, 33).
Conclusion
Pancreatic surgery is safe in high-volume centers. Our center’s low hospital mortality for pancreaticoduodenectomy (2.1%) is similar to that reported from other high volume centers. Our long-term survival for PDAC is now one of the highest reported. Early detection and multidisciplinary treatment of PDAC is essential for better survival. During the era of centralization of pancreatic (cancer) surgery, more patients have lived markedly longer than did those operated on in the early 1990s.
Footnotes
Acknowledgements
Mary and Georg C. Ehrnrooth Foundation, the Sigrid Juselius Foundation, and Helsinki University Hospital Research Funds are acknowledged for supporting this study. None of the foundations have been involved in the study design.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
