Abstract
Background and Aims:
Hemorrhage is a rare but dreaded complication after pancreatic surgery. The aim of this study was to examine the incidence, risk factors, management, and outcome of postpancreatectomy hemorrhage in a tertiary care center.
Materials and Methods:
A retrospective observational study was conducted on 500 consecutive patients undergoing major pancreatic resections at our institution. Postpancreatectomy hemorrhage was defined according to the International Study Group of Pancreatic Surgery criteria.
Results:
A total of 68 patients (13.6%) developed postpancreatectomy hemorrhage. Thirty-four patients (6.8%) had a type A, 15 patients (3.0%) had a type B, and the remaining 19 patients (3.8%) had a type C bleed. Postoperative pancreatic fistula Grades B and C and bile leakage were significantly associated with severe postpancreatectomy hemorrhage on multivariable logistic regression. For patients with postpancreatectomy hemorrhage Grade C, the onset of bleeding was in median 13 days after the index operation, ranging from 1 to 85 days. Twelve patients (63.2%) had sentinel bleeds. Surgery lead to definitive hemostatic control in six of eight patients (75.0%). Angiography was able to localize the bleeding source in 8/10 (80.0%) cases. The success rate of angiographic hemostasis was 8/8. (100.0%). The mortality rate among patients with postpancreatectomy hemorrhage Grade C was 2/19 (10.5%), and both fatalities occurred late as a consequence of eroded vessels in association with pancreaticogastrostomy.
Conclusion:
Delayed hemorrhage is a serious complication after major pancreatic surgery.Sentinel bleed is an early warning sign. Postoperative pancreatic fistula and bile leakage are important risk factors for severe postpancreatectomy hemorrhage.
Introduction
Hemorrhage after pancreatic resection, although uncommon, is a potentially fatal complication that requires timely diagnosis and treatment (1–6). The pathogenesis of postpancreatectomy hemorrhage (PPH) is multifactorial. Early PPH within the first 24 h after surgery is generally caused by technical failures of inadequate hemostasis and perioperative coagulopathy and usually necessitates immediate relaparotomy. Late PPH after the first postoperative day has a more complex pathogenesis and may be related to erosion of peripancreatic vessels secondary to pancreatic fistula formation or ulceration at the site of an anastomosis (7). Despite the relatively recent International Study Group of Pancreatic Surgery (ISGPS) definition of PPH (8), the optimal operative and interventional treatment strategy for PPH is controversial. Because of the rarity and clinical heterogeneity of severe PPH, randomized trials have not been possible to perform. Therefore, treatment algorithms and management must still rely on data that become available from observational studies in order to guide clinical decision-making.
Herein, we report our experience in the management of PPH at our institution. The aim of this study was to investigate the incidence, risk factors, management, and outcome of PPH after major pancreatic resections in a tertiary care center specialized in pancreatic diseases. One of the unique aspects of this study is that pancreatic head resections were performed with pancreatic reconstruction via pancreaticogastrostomy (PG). This is both good and bad in that it creates a new source of bleeding (anastomotic) which is more easily accessible for endoscopic management. It also draws the pancreatic anastomosis away from the gastroduodenal artery, where bleeding is more problematic when following pancreaticojejunostomy (PJ) anastomosis.
Methods
This was a retrospective cohort study of 500 consecutive patients undergoing major elective pancreatic resections at the Department of Surgery, Skåne University Hospital, Lund and Malmö, Sweden, during the period from January 2000 to August 2015 for pancreatic or periampullary tumors. For the purposes of this study, total pancreatectomy, multivisceral resections, necrosectomies, and enucleations were excluded. The study protocol was approved by the human ethics committee at Lund University. Follow-up was recorded using medical charts and the local patient administrative database (Patient Administrative Support in Skåne (PASiS)).
Surgical Technique
The standard operation for tumors of the pancreatic head included classic Whipple procedure with a limited distal gastrectomy (9). Abdominal access was achieved through a bilateral subcostal incision. After exclusion of extrapancreatic metastatic disease, a Kocher maneuver was performed to mobilize the duodenum and head of the pancreas followed by assessment of the superior mesenteric vein. This was followed by cholecystectomy and portal vein dissection. The pancreas was divided at the level of the neck. The distal stomach and duodenum were then dissected, enabling complete removal of the specimen. Pancreatic anastomosis was performed by anastomosing the pancreatic remnant to the posterior gastric wall midway between the lesser and greater curvature. Finally, the remaining reconstruction was completed by performing an end-to-side hepaticojejunostomy and a side-to-side gastrojejunostomy. Distal pancreatectomy was performed for tumors in the body and tail of the pancreas and in suspected malignant cases included splenectomy. All patients received thrombotic prophylaxis with low-molecular-weight heparin (enoxaparin sodium, 40 mg/day) preoperatively and postoperatively until discharge.
PPH
PPH was defined according to the ISGPS guidelines considering the following three parameters: (1) time of onset, (2) location, and (3) severity (8). Early PPH was defined as bleeding occurring within 24 h and late PPH if the bleeding started after 24 h postoperatively. The location of bleeding was categorized as intraluminal or extraluminal. Severe hemorrhage required more than 4 U of packed cells within 24 h, a decrease in hemoglobin of more than 4 g/dL, or a need for relaparotomy or interventional angiography to stop the bleeding. PPH Grade A had little or no clinical implication, while PPH Grades B and C lead to critical worsening of the patient’s clinical condition necessitating further treatment. PPH Grade C was defined as potentially life-threatening bleeding.
Sentinel Bleed
Sentinel bleeding was defined as minor blood loss via surgical drains or the gastrointestinal (GI) tract with an asymptomatic interval of at least 12 h until development of hemorrhagic shock (5, 10–12).
Postoperative Pancreatic Fistula
Postoperative pancreatic fistula (POPF) represents a delayed healing of the pancreatic–enteric anastomosis or a leakage from the pancreatic ducts. POPF was defined by a drain output of any measurable volume of fluid on or after postoperative day 3, with an amylase content greater than three times the upper normal serum value (13). POPF Grade A is a biochemical fistula that has no clinical implication. POPF Grades B and C require active treatment such as prolonged or additional drainage, parenteral/enteral nutrition, antibiotics, somatostatin analogues, or reoperation.
Statistical Analysis
Statistical analyses were performed using the Stata MP statistical package version 13.1 (2013) (StataCorp LP, College Station, TX). Independent predictors of PPH Grades B and C were identified using logistic regression analyses. Any variable from the univariable test with a P value <0.100 was selected as a candidate for the multivariable analysis. In the iterative process of variable selection, covariates were removed from the model if they were non-significant and not a confounder, as described by Hosmer–Lemeshow (14), resulting in a main effect model. Odds ratios (ORs) are presented with 95% confidence intervals.
Missing values were imputed using the chained-equations multiple imputation (MI) technique as described by White et al. (15). The imputation method was predictive mean matching. The number of iterations for each chain was 10, and the number of imputed data sets was 10.
Results
The clinical characteristics of the patient cohort are summarized in Table 1. Median follow-up time was 2.6 (range 0–15.6) years. Median age was 66 years and M:F ratio was 238:262. A total of 396 patients underwent Whipple’s pancreaticoduodenectomy, and 104 patients underwent distal pancreatectomy. Mean operative duration was 453 min. Median estimated blood loss was 600 mL. One hundred and sixteen patients (26.4%) received intraoperative blood transfusion. The indications for pancreatectomy were adenocarcinoma of the pancreas or periampullary region in 318 patients (63.6%), and the remaining patients were operated for cystic pancreatic lesions (72 patients), neuroendocrine tumors (47 patients), chronic pancreatitis (34 patients), periampullary adenoma (13 patients), metastatic renal cell cancer (4 patients), gastric and duodenal ulcers (3 patients), acinic cell carcinoma (1 patient), pancreatoblastoma (1 patient), signet ring cell carcinoma of the ampulla of Vater (1 patient), familiar adenomatous polyposis of the duodenum (1 patient), leiomyosarcoma of the duodenum (1 patient), lymphoma (1 patient), sarcoma (1 patient), metastatic colonic cancer (1 patient), and metastatic non–small cell lung cancer (1 patient). The 30-day mortality rate was 0.6%, and the 90-day mortality rate was 1.6%.
Clinical details of patients undergoing pancreatic resection.
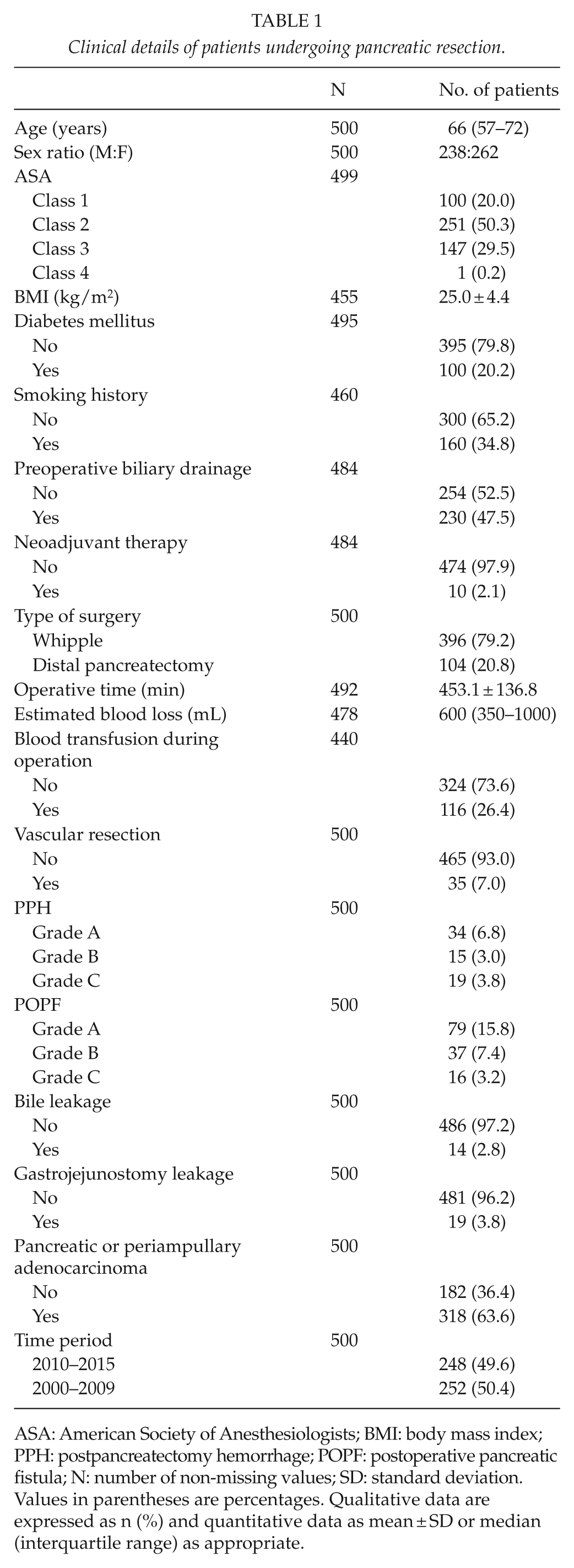
ASA: American Society of Anesthesiologists; BMI: body mass index; PPH: postpancreatectomy hemorrhage; POPF: postoperative pancreatic fistula; N: number of non-missing values; SD: standard deviation.
Values in parentheses are percentages. Qualitative data are expressed as n (%) and quantitative data as mean ± SD or median (interquartile range) as appropriate.
Incidence of PPH
Sixty-eight patients (13.6%) developed hemorrhagic complications. Thirty-four patients (6.8%) had PPH Grade A, 15 patients (3.0%) had PPH Grade B, and 19 patients (3.8%) had PPH Grade C.
Risk Factors for Severe Hemorrhage
Significant predictors of PPH Grades B and C on univariable analysis were operative time (P = 0.018), POPF Grades B and C (P < 0.001), bile leakage (P < 0.001), and gastrojejunostomy leakage (P < 0.001) (Table 2). On multivariable analysis, the only independently significant predictors of PPH Grades B and C were POPF Grades B and C (OR 9.47, 95% confidence interval 4.30–20.88; P < 0.001) and bile leakage (OR 4.83, 95% confidence interval 1.32–17.71; P = 0.017) (Table 3).
Univariable logistic regression analysis of potential predictors of PPH Grades B and C.
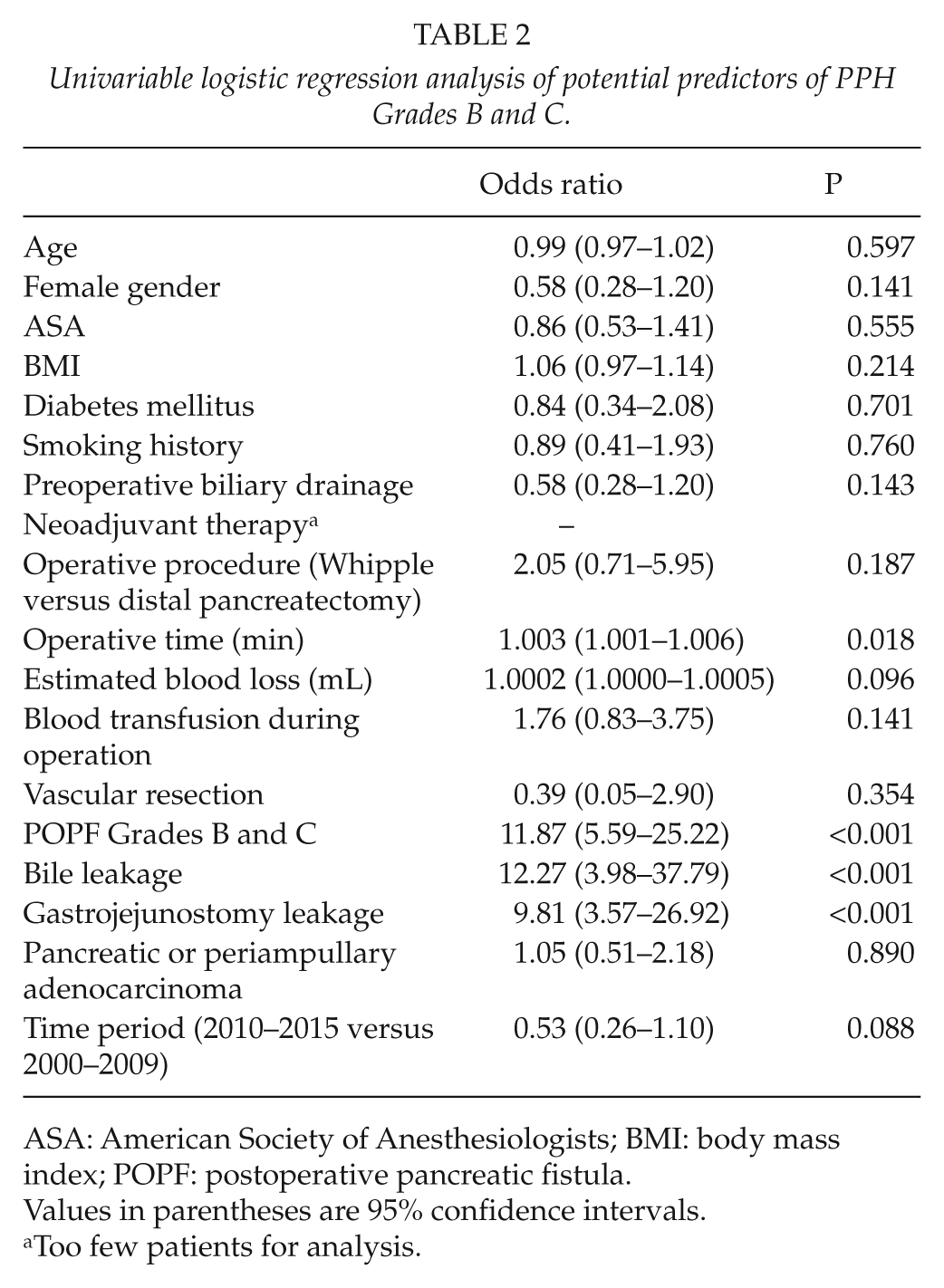
ASA: American Society of Anesthesiologists; BMI: body mass index; POPF: postoperative pancreatic fistula.
Values in parentheses are 95% confidence intervals.
Too few patients for analysis.
Multivariable logistic regression analysis of independent significant risk factors associated with PPH Grades B and C.
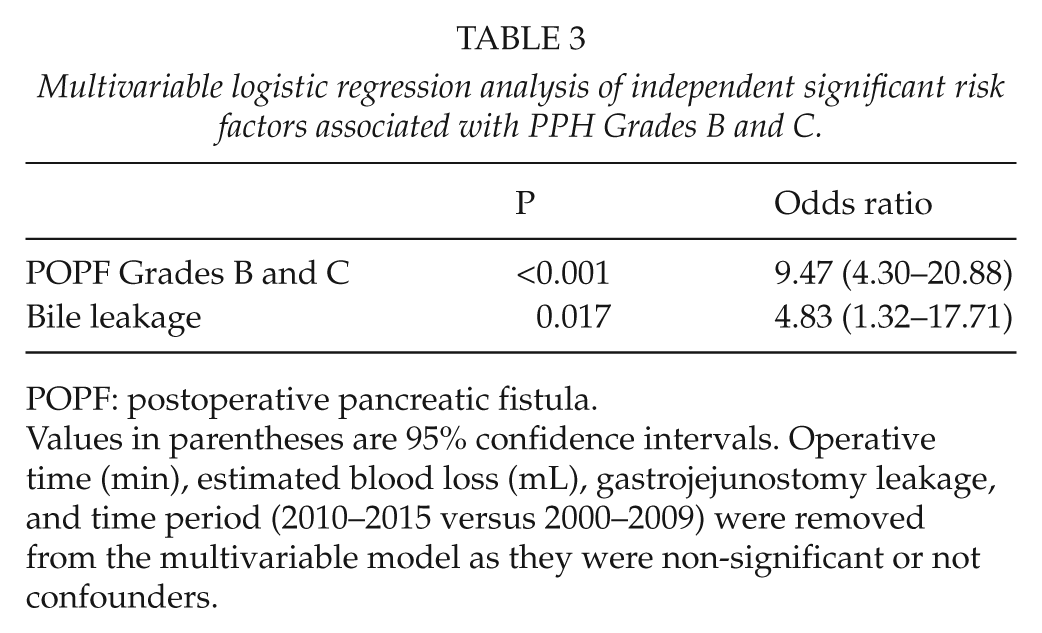
POPF: postoperative pancreatic fistula.
Values in parentheses are 95% confidence intervals. Operative time (min), estimated blood loss (mL), gastrojejunostomy leakage, and time period (2010–2015 versus 2000–2009) were removed from the multivariable model as they were non-significant or not confounders.
A multivariable analysis was made restricted to only patients with pancreatic head resection. The same two variables, namely, POPF Grades B and C and bile leakage, remained significant in the multivariable model. However, the ORs changed slightly for POPF Grades B and C (OR 12.04, 95% confidence interval 5.12–28.28; P < 0.001) and bile leakage (OR 4.41, 95% confidence interval 1.12–17.40; P = 0.034).
Management of Life-Threatening Hemorrhage (PPH Grade C)
The median time from time of surgery to the onset of grade C bleed was 13 days, ranging from 1 to 85 days. Three patients were readmitted due to late bleeding. Of the 10 patients who had POPF, 6 patients (60.0%) had their drains still in place at the time of the bleeding episode. Twelve out of 19 PPH Grade C patients (63.2%) had sentinel bleeds. For intraluminal PPH, upper GI endoscopy was performed in 10 patients, revealing the bleeding focus in 6 patients (60%). Angiography was able to localize the bleeding source in 8/10 (80.0%) cases. Pseudoaneurysms were the cause of bleeding in 8 patients, including the proper hepatic artery (2 patients), the gastroduodenal artery (2 patients), the right hepatic artery (1 patient), the common hepatic artery (1 patient), the inferior pancreaticoduodenal artery (1 patient), and a small jejunal artery (1 patient). The remaining patients had bleeding at the PG (7 patients), pancreatic cut surface (1 patient), diffuse liver bleed (1 patient), omental bleed (1 patient), and upper GI bleed of unknown origin (1 patient). Management included surgery in 5 patients, angiographic coiling/stenting in 4 patients, surgery complemented by angiographic intervention in 2 patients, endoscopic and angiographic intervention in 2 patients, endoscopic and surgical treatment in 1 patient, endoscopic treatment in 1 patient, and 4 patients were managed conservatively with resuscitation (Table 4). Fig. 1 shows successful angiographic stenting of a pseudoaneurysm in the proper hepatic artery (patient no. 13).
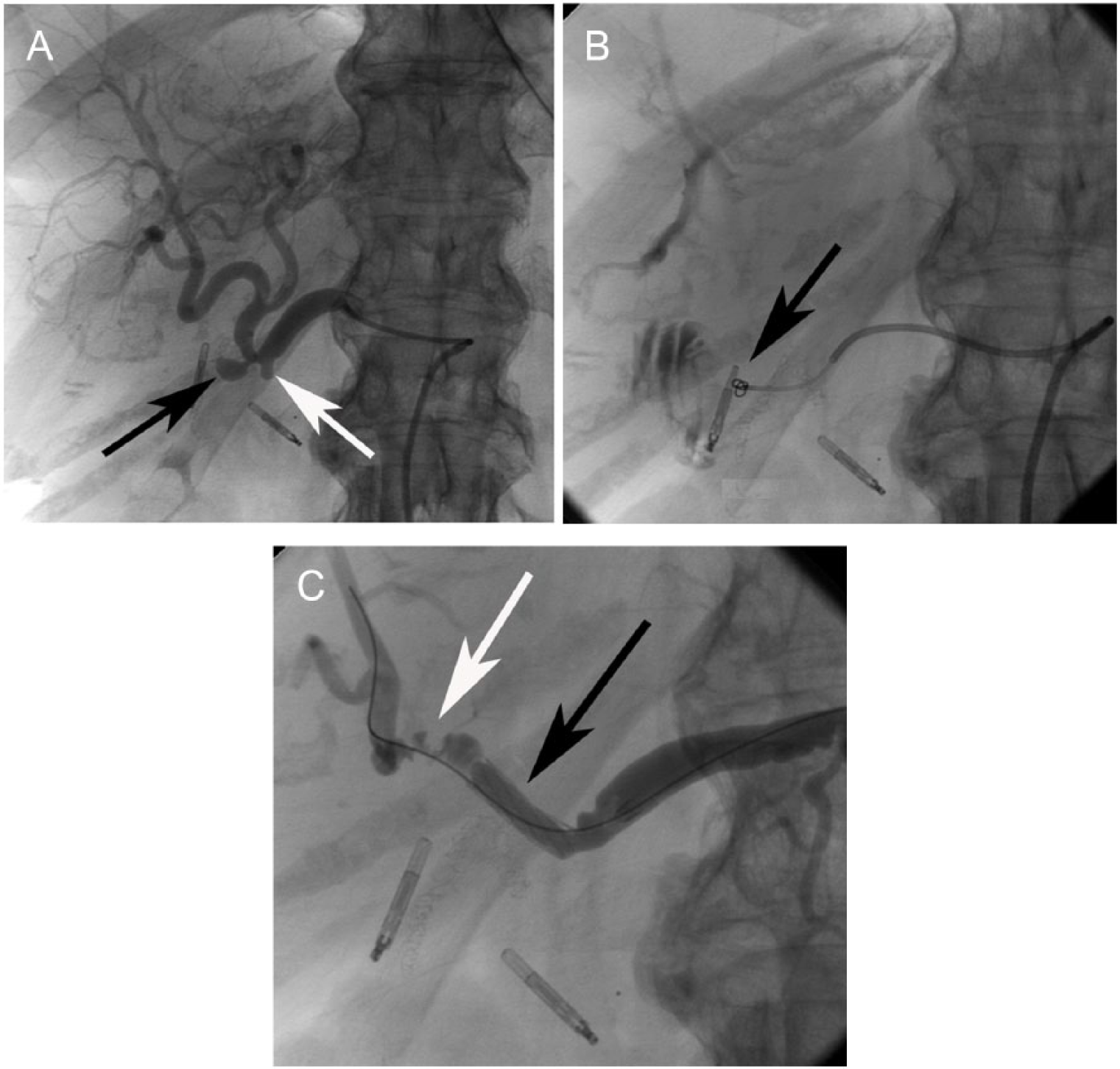
Patient no. 13. Angiography 60 days after Whipple’s pancreaticoduodenectomy. The procedure was performed in general anesthesia. Through a 7-French introducer placed in the right femoral artery, a 5-French glide-cobra catheter (Terumo, Västra Frölunda, Sweden) was advanced with the tip in the celiac trunk. Angiography verified the 8 mm × 5 mm pseudoaneurysm in the proper hepatic artery. Following catheterization of the pseudoaneurysm with a 2.7-French Progreat Microcatheter (Terumo), two 4-mm microcoils were deployed. However, due to rupture of the pseudoaneurysm, the coils were dislodged to the duodenum, and leakage of contrast was also seen. After measurement, a 17-mm Jostentgraft (Abbott Scandinavia AB, Solna, Sweden) was placed and dilated to 5 mm to exclude the pseudoaneurysm. Final angiography showed exclusion of the pseudoaneurysm, good contrast flow but also arterial spasm distal to the device. A) Pseudoaneurysm from the proper hepatic artery (black arrow) and ligated gastroduodenal artery (white arrow). B) Contrast and migrated coil (arrow) in the duodenum from the ruptured pseudoaneurysm. C) Stentgraft successfully positioning excluding the pseudoaneurysm (black arrow) and spasm in the right hepatic artery (white arrow).
Management of patients with potentially fatal bleeding (PPH Grade C).
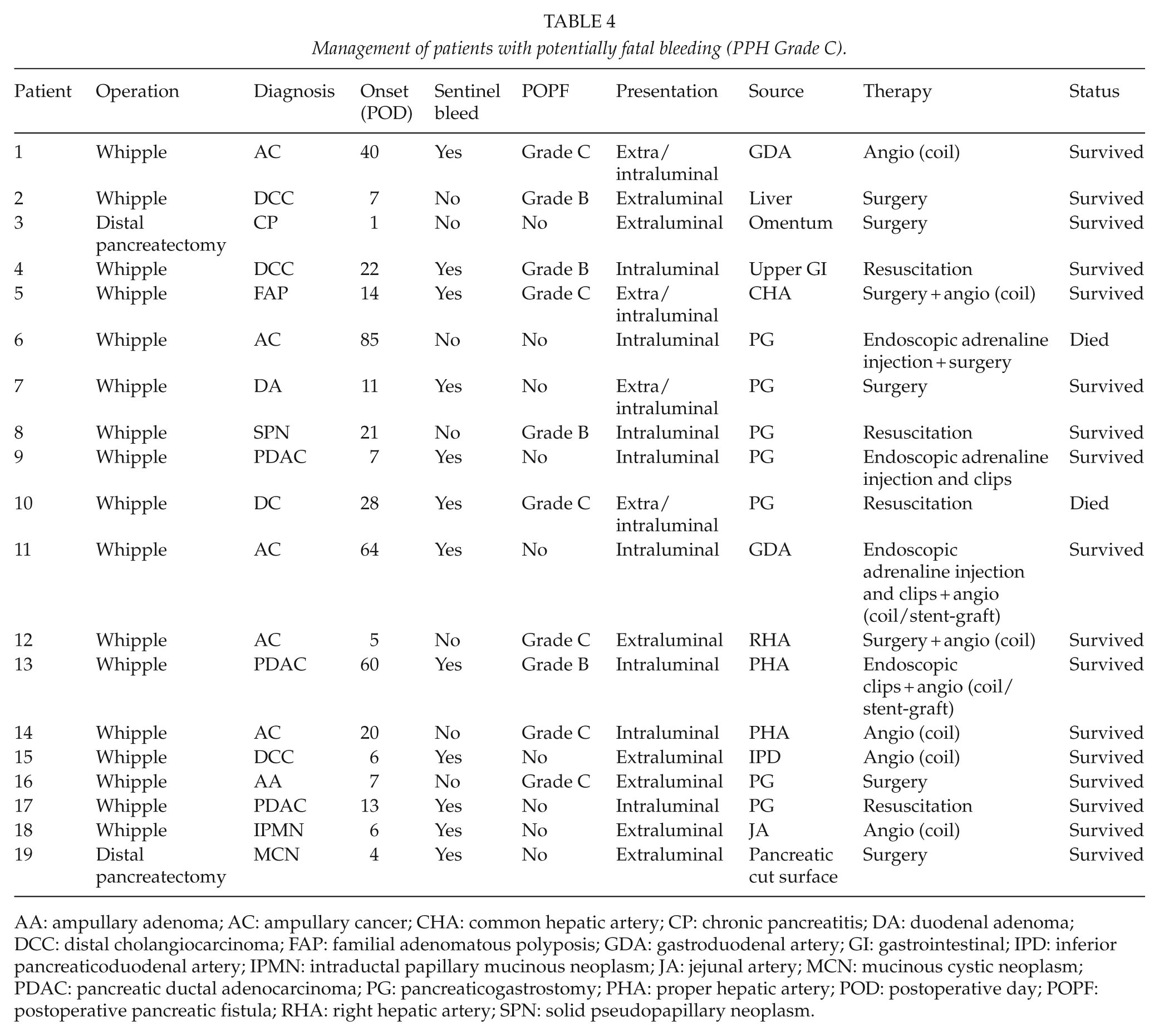
AA: ampullary adenoma; AC: ampullary cancer; CHA: common hepatic artery; CP: chronic pancreatitis; DA: duodenal adenoma; DCC: distal cholangiocarcinoma; FAP: familial adenomatous polyposis; GDA: gastroduodenal artery; GI: gastrointestinal; IPD: inferior pancreaticoduodenal artery; IPMN: intraductal papillary mucinous neoplasm; JA: jejunal artery; MCN: mucinous cystic neoplasm; PDAC: pancreatic ductal adenocarcinoma; PG: pancreaticogastrostomy; PHA: proper hepatic artery; POD: postoperative day; POPF: postoperative pancreatic fistula; RHA: right hepatic artery; SPN: solid pseudopapillary neoplasm.
Impact of Hemorrhage on Mortality
The mortality rate was 2/19 (10.5%) in patients with PPH Grade C. One patient presented with hematemesis 85 days after the index operation and underwent upper GI endoscopy where a squirting artery was identified in the vicinity of the PG. It was injected with adrenaline but the bleeding started anew and was too extensive to allow for sclerotherapy. A laparotomy was subsequently performed where an eroded vessel, associated with the suture line of the PG, was oversewn. The patient had a long postoperative recovery but was eventually discharged after 2 weeks. However, the patient was readmitted a few days later with hemorrhagic shock and died from acute GI bleeding. The other patient presented with massive bleeding through the nasogastric (NG) tube and abdominal drain 28 days postoperatively, but due to old age (83 years) and uncertain radicality of resection, only conservative measures were undertaken. At autopsy, a small, eroded vessel of about 5 mm in diameter was identified, and located behind the stomach, and was deemed to be the site of bleeding.
Discussion
The exact incidence of hemorrhagic complications after pancreatic surgery is not known. Until recently, the lack of uniform definitions of PPH has been a challenge. We found that the overall incidence of PPH was 13.6% in our series using the ISGPS criteria. Grade C PPH occurred in 3.8% of patients and was associated with a mortality rate of 10.5%. These figures compare well with previous series. In the literature, the frequency of PPH Grades A to C using the ISGPS criteria varies from 3% to 29% (1, 4, 6, 16–19), with a mortality rate of 7%–31% for PPH Grade C (1, 4, 6, 16, 18, 20).
The distinction between early and late PPH has a crucial impact on therapeutic management. Early PPH is believed to be caused by technical failures in terms of inadequate hemostasis in the operative field, bleeding from the suture line of the gastroenteric or enteroenteric anastomosis, or bleeding from the transection surface of the pancreatic anastomosis (5). Its management usually involves relaparotomy. On the other hand, late PPH has a more complex pathophysiology and requires a multimodality approach for its management. One of the most important mechanisms leading to late PPH is the erosion of blood vessels due to postoperative leakage of a pancreatic or biliary anastomosis. Subsequent intra-abdominal abscess formation may also erode major vessels. Furthermore, the placement of drains may cause mechanical pressure on the vessel wall or facilitate infectious spread along the drain leading to vessel wall damage (2). In our series, most life-threatening bleedings (Grade C) were late. The median onset was 13 days postoperatively, with an upper range of 85 days, and three patients had to be readmitted due to bleeding, indicating that late PPH could occur even after patient’s discharge from the hospital.
The risk factors for hemorrhagic complications have not been studied well. In this study, we found that POPF and bile leakage were independent risk factors for developing severe PPH. POPF is the most consistently reported risk factor for PPH according to previous studies, while bile leakage has not been correlated to increased bleeding risk in prior studies. Darnis et al. (1) reported that POPF, type of resection (Whipple versus other), advanced age, and nutritional risk index were risk factors for PPH. Feng et al. (19) found that male gender, pancreatic duct diameter, end-to-side invagination PJ, POPF, and intra-abdominal abscess were the independent risk factors for late PPH after pancreaticoduodenectomy. In a recent study, Wellner et al. (4) performed a risk factor analysis of PPH Grade C after major pancreatic resection. Nine factors were found to significantly predict the risk of PPH Grade C. Among these factors were advanced age, male sex, body mass index, portal venous resection, multivisceral resection, and POPF, while centralization of care, intraoperative transfusion, and preoperative biliary drainage were significant protective factors against bleeding complications. In our series, we used a PG as the preferred method of pancreatic anastomosis. The study by Wellner et al. (4) reported that although PG was associated with more bleeding episodes than with PJ, it was independently associated with reduced mortality due to PPH. It was suggested that PPH and underlying POPF and septic conditions tend to resolve better with PG, and furthermore, completion pancreatectomy can be more easily performed with a PG than a PJ.
Endoscopy was frequently undertaken for patients presenting with intraluminal bleeding, but the success rate was not high as frequently no therapeutic target was identified, and the endoscopic intervention often had to be complemented with other treatments to achieve definite control of the bleeding, such as surgery or angiographic intervention, which is in line with previous studies (4, 5, 21). Angiography was able to localize the bleeding source in 8/10 (80.0%) cases in our series. The success rate of angiographic hemostasis was 8/8 (100%). False-negative angiography could be due to the intermittent bleeding (7). Surgery leads to definitive hemostatic control in 6 of 8 patients (75%) in our series, while 2 patients re-bled and had to undergo angiographic intervention to control the bleeding. Thus, angiographic intervention should be the preferred treatment option in hemodynamically stabile patients, as supported by previous studies (2). Our experience is that angiographic coiling and covered stents are equally effective, although recent studies prefer covered stents to reduce the risk of compromising hepatic artery blood flow (22). Other centers are also moving from surgery to interventional radiology in their therapeutic algorithms for PPH (18).
The recognition of sentinel bleed is crucial for the management of late PPH. A sentinel bleed describes the presence of a minor bleeding either from the GI tract or surgical drains that precedes major PPH, usually due to pseudoaneurysms or erosive vessels. We found that 63% of patients with PPH Grade C had sentinel bleeds. Data from previous studies suggest that between 30% and 100% of patients with late PPH present with sentinel bleeds (5, 10, 12, 20). The presence of a sentinel bleed after pancreatic surgery should lead to an emergency computed tomography (CT) angiography or immediate angiography to localize the source of bleeding.
This study was limited by the retrospective nature of the analysis and the long accrual period during which some aspects of care and vigilance may have changed. We adjusted for known differences between comparison groups, albeit unknown or unmeasured differences in baseline clinical characteristics and treatments of our population may have influenced our results. Missing values were handled by MI technique, which represents the most appropriate method available to date.
In conclusion, severe PPH is an uncommon, yet potentially fatal complication after major pancreatic surgery. Most severe bleeding episodes occur late in the postoperative course and sometimes even after the patient has been discharged from the hospital. The identification of a sentinel bleed should lead to early angiographic assessment before the occurrence of massive PPH. Angiographic coiling and stenting are effective measures to treat pseudoaneurysmal bleeding. Relaparotomy remains a valid treatment option for hemodynamically unstable patients. As POPF and bile leakage are the most important risk factors for severe PPH, future strategies to prevent anastomotic leakage may also reduce the frequency of PPH after pancreatic surgery.
Footnotes
Declaration Of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
