Abstract
Background and Aims:
Sentinel node biopsy is a standard method for nodal staging in patients with clinically localized cutaneous melanoma, but the survival advantage of sentinel node biopsy remains unsolved. The aim of this case–control study was to investigate the survival benefit of sentinel node biopsy.
Materials and Methods:
A total of 305 prospective melanoma patients undergoing sentinel node biopsy were compared with 616 retrospective control patients with clinically localized melanoma whom have not undergone sentinel node biopsy. Survival differences were calculated with the median follow-up time of 71 months in sentinel node biopsy patients and 74 months in control patients. Analyses were calculated overall and separately in males and females.
Results:
Overall, there were no differences in relapse-free survival or cancer-specific survival between sentinel node biopsy patients and control patients. Male sentinel node biopsy patients had significantly higher relapse-free survival (P = 0.021) and cancer-specific survival (P = 0.024) than control patients. In females, no differences were found. Cancer-specific survival rates at 5 years were 87.8% in sentinel node biopsy patients and 85.2% in controls overall with 88.3% in male sentinel node biopsy patients and 80.6% in male controls and 87.3% in female sentinel node biopsy patients and 89.8% in female controls.
Conclusion:
Sentinel node biopsy did not improve survival in melanoma patients overall. While females had no differences in survival, males had significantly improved relapse-free survival and cancer-specific survival following sentinel node biopsy.
Introduction
The goal of sentinel node biopsy (SNB) in patients with clinically local cutaneous melanoma is accurate nodal staging. The histopathological status of the sentinel node (SN) is the single most important indicator of survival. SN status separates melanoma population prognostically into high-risk and low-risk subgroups providing a logical basis on which options for selective lymphadenectomy, adjuvant therapies, and tailored follow-up schedules can be based. The routine use of SNB is supported by established guidelines in both the United States and Europe (1–4).
Despite the prognostic value, there is no definitive evidence regarding the survival benefit of SNB. Few retrospective studies have demonstrated significant survival benefit favoring the use of SNB (5). Prospective randomized Multicenter Selective Lymphadenectomy Trial 1 (MSLT-1) failed to demonstrate overall survival benefit in a selected subgroup of patients with intermediate-thickness melanoma (6).
We conducted a single-institution case–control study comparing prospective SNB patients with historical control patients who had not undergone SNB. The preliminary results showed no impact on cancer-specific survival associated with SNB (7). This report presents new results with a longer follow-up time, 71 months in prospective SNB patients and 74 months in the control patients. The primary aim was to examine whether there is a relapse-free survival (RFS) or cancer-specific survival benefit associated with SNB procedure. The secondary aim was to compare the outcome between genders.
Materials and Methods
SNB Patients
From October 2001 to December 2006, consecutive patients with clinically localized (American Joint Committee on Cancer (AJCC) stage I–II) invasive (Clark level II–V) cutaneous melanoma were prospectively enrolled into this study at the Department of Plastic and General Surgery, Turku University Hospital, Turku, Finland. The study protocol was approved by the Institutional Review Board of the Turku University Hospital and an informed consent was obtained from each patient.
All patients underwent lymphoscintigraphy before the surgical procedure. Technetium-99m-labeled colloidal albumin or nanocolloid was injected intradermally at two to four points at the margins of the primary melanoma or the biopsy scar. The injected dose was approximately 70 MBq in the volume of 0.2–0.3 mL. After lymphoscintigraphy, SNB was performed within the next 20 h. For the localization of SNs, a blue dye injection and gamma detecting probe were used. All blue-stained and radioactive lymph nodes were defined as SNs and excised. After the excision of SNs, the melanoma or the biopsy scar was excised according to current guidelines.
Excised and formalin-fixed SNs were first cut coarsely into longitudinal slices. The slices were embedded in paraffin, further sectioned, and stained with hematoxylin and eosin (HE). If no metastatic melanoma cells were identified in the HE-stained sections, further sections were cut and immunohistochemical staining with Melan-A and/or S-100 antibodies was performed.
Completion lymph node dissection (CLND) was offered to SN-positive patients. After surgery, all patients were referred for further follow-up in the Department of Oncology and Radiotherapy, Turku University Hospital. For SN-positive patients, the follow-up schedule consisted of an initial screening by whole-body computed tomography and/or positron emission tomography and clinical examination every 3–6 months during the first 5 years. Routine chest X-ray and blood tests including liver chemistry were performed annually. The most recent follow-up information of all patients was updated from the electronic patient records.
Control Patients
For comparison, a retrospective cohort was collected from January 1983 to September 2001. The data of consecutive patients with stage I–II melanoma were collected from the original patient records of Turku University Hospital. Patients were excluded if elective lymph node dissection (ELND) was performed or if the Breslow thickness of the primary lesion remained undetermined. The cause and time of death were obtained from the patient records and from the autopsy reports. Complementary data were obtained from Statistics Finland’s Archive of Death Certificates with the permission of the Ministry of Social Affairs and Health of Finland.
Definitions and Study Endpoints
The false-negative rate of the SNB procedure was calculated as the rate of nodal recurrences in SN-negative patients over the number of all true nodal-positive patients. RFS was defined as time from the initial treatment until the first recurrence. Recurrence was defined according to the site of the first relapse. Cancer-specific survival was defined as the time from initial melanoma treatment until the disease-specific death of metastatic melanoma. The primary study endpoint was melanoma-related death. The secondary study endpoint was the recurrence of melanoma.
Statistical Analyses
In statistical analyses, categorical variables were analyzed by the χ2 test and continuous data by the nonparametric Mann–Whitney U test. The starting point for all survival analyses was the initial melanoma treatment. RFS and melanoma-specific survival curves were constructed by the Kaplan–Meier method and SNB and control group differences were analyzed by the log rank test. The ticks along the curves in the survival plots represent censored observations. Deaths from other causes or unknown outcome were marked as censored observations for cancer-specific survival. Age-adjusted survival analyses were performed using the Cox proportional hazards regression model; hazard ratios (HRs) with 95% confidence intervals (CIs) were calculated. The confounding effect of prognostic factors on group difference in survival was tested by interaction effect between confounding variable and group. Because the maximum follow-up time of SNB patients was 9.8 years, the follow-up time was cut off at 10.0 years in control patients and all events after this time point were censored. A P value of less than 0.05 was considered statistically significant.
Results
SNB
A total of 305 patients underwent lymphoscintigraphy consisting of 145 males (47.5%) and 160 females (52.5%). Lymphoscintigraphy was successful in 297 (97.4%) study patients, in 142 males (97.9%) and in 155 females (96.9%). Histopathologically, 50 patients (16.8% of successfully mapped patients) were SN-positive. A total of 30 males (21.1%) and 20 females (12.9%) were SN-positive. The mean number of metastatic SNs was 1.7 in men and 1.2 in women. The size of each micrometastasis was classified by Starz’s centripetal thickness, that is, tumor penetrative depth (d), measured as the maximum distance of the tumor cells from the interior margin of the lymph node capsule. The S classification was SI (d ⩽ 0.3 mm) in 9 patients (5 males, 4 females), SII (0.3 mm < d ⩽ 1.0 mm) in 16 patients (9 males, 7 females), and SIII (d > 1.0 mm) in 24 patients (15 males, 9 females). There were no significant differences between male and female patients (P = 0.826). In one male patient, the S classification was undetermined. Forty-nine SN-positive patients underwent CLND and 10 of them (20%) had additional nonsentinel metastases. The median follow-up time of the patients was 71 months (mean, 64; range, 2–117). There were eight same-basin nodal recurrences in SN-negative patients, which yielded a false-negative rate of 13.8%, a sensitivity of 86.2%, and a negative predictive value of 96.8%. SN was metastatic in five patients with thin melanoma (Breslow thickness ⩽ 1.0 mm).
Control Patients
A total of 611 patients were included consisting of 302 males (49.0%) and 314 females (51.0%). The median follow-up time was 74 months (mean, 75; range, 2–120). Regional lymph node relapse occurred in 71 patients (11.5%), and 67 nodal-positive patients (10.9%) underwent therapeutic lymph node dissection (TLND). The baseline characteristics of the study patients are presented in Table 1.
Clinical and histopathological characteristics of study patients.
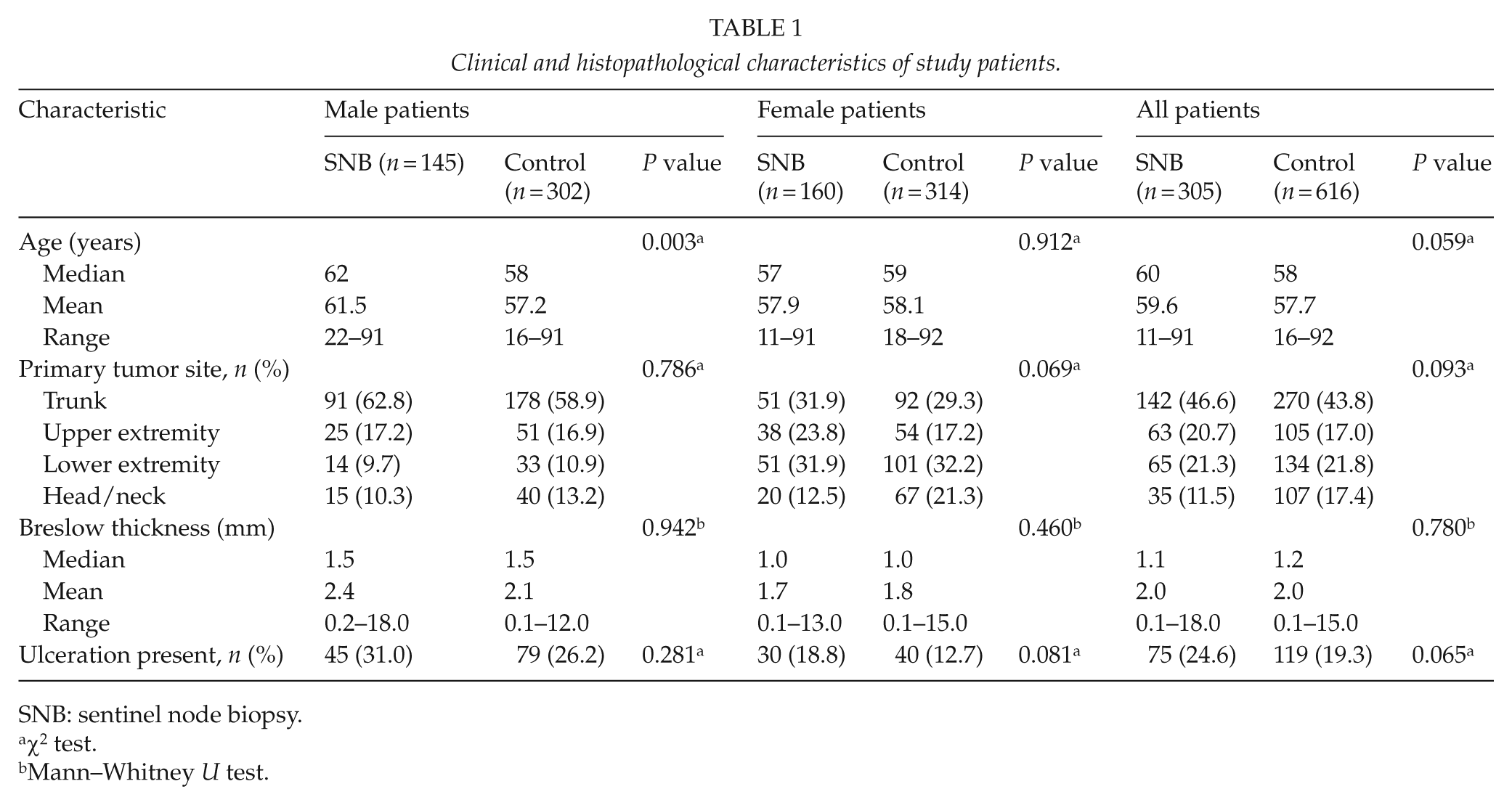
SNB: sentinel node biopsy.
χ2 test.
Mann–Whitney U test.
Survival Analyses
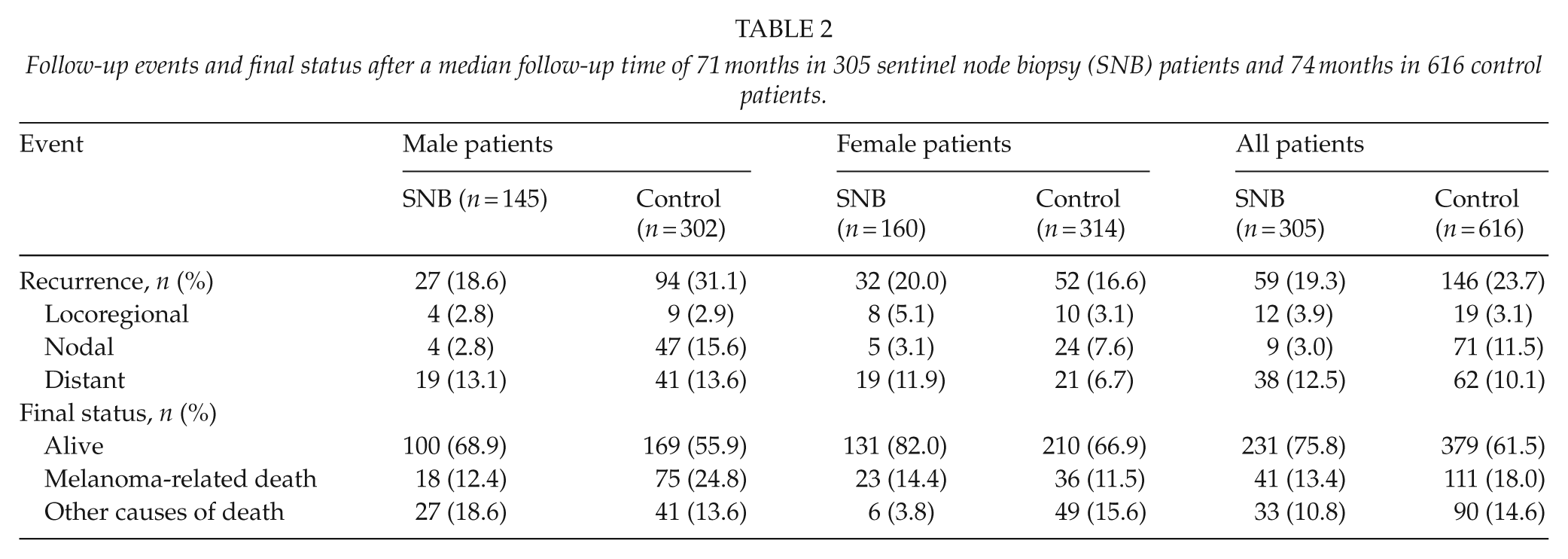
The follow-up events are presented in Table 2. There were no significant differences in RFS (age-adjusted HR, 0.81; 95% CI, 0.60–1.10; P = 0.171) or cancer-specific survival (age-adjusted HR, 0.76; 95% CI, 0.53–1.09; P = 0.139) between SNB and control groups (Fig. 1). The confounding effect of Breslow thickness, ulceration status, age, gender, and the anatomic location of primary tumor on group difference in survival was analyzed. Categorically, only gender status was found to affect RFS and cancer-specific survival (gender by study group interaction effect, P = 0.002 and P = 0.003, respectively). In females, there were no significant differences in RFS (age-adjusted HR, 1.39; 95% CI, 0.90–2.15; P = 0.143) or cancer-specific survival (age-adjusted HR, 1.37; 95% CI, 0.81–2.32; P = 0.243) between the study groups (Fig. 2), but in males, SNB patients had significantly higher RFS (age-adjusted HR, 0.51; 95% CI, 0.33–0.80; P = 0.003) and cancer-specific survival (age-adjusted HR, 0.48; 95% CI, 0.28–0.82; P = 0.007) than control patients (Fig. 3). Cancer-specific survival rates at 5 years were 87.8% (standard error, SE, 2.0%) in all SNB patients and 85.2% (SE, 1.5%) in all controls, 87.3% (SE, 2.8%) in female SNB patients and 89.8% (SE, 1.8%) in female controls, and 88.3% (SE, 2.8%) in male SNB patients and 80.6% (SE, 2.4%) in male controls.
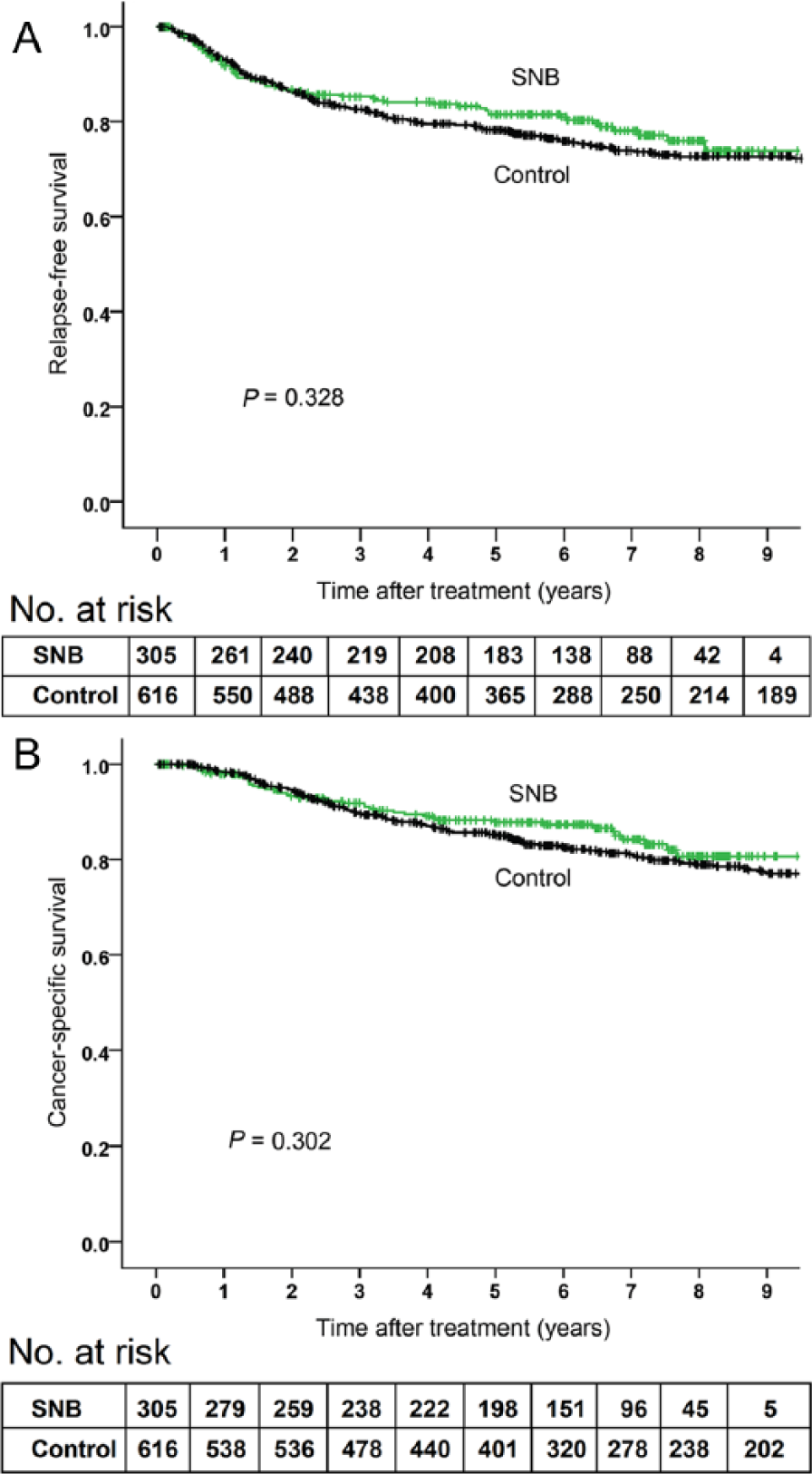
A) Relapse-free survival and B) cancer-specific survival in 305 sentinel node biopsy (SNB) patients and 616 control patients. P values are calculated by log rank test.
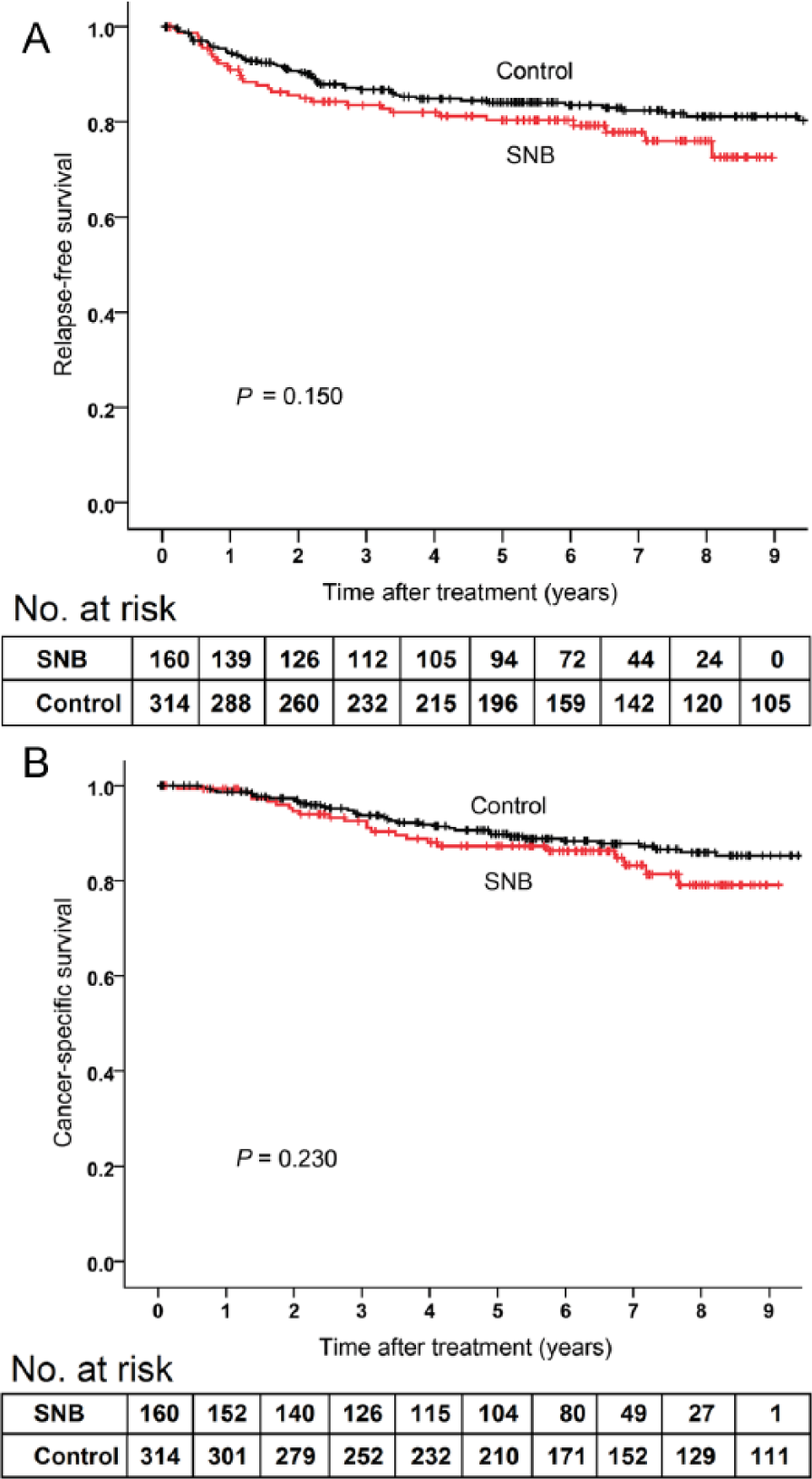
A) Relapse-free survival and B) cancer-specific survival in 160 female sentinel node biopsy (SNB) patients and 314 female control patients. P values are calculated by log rank test.
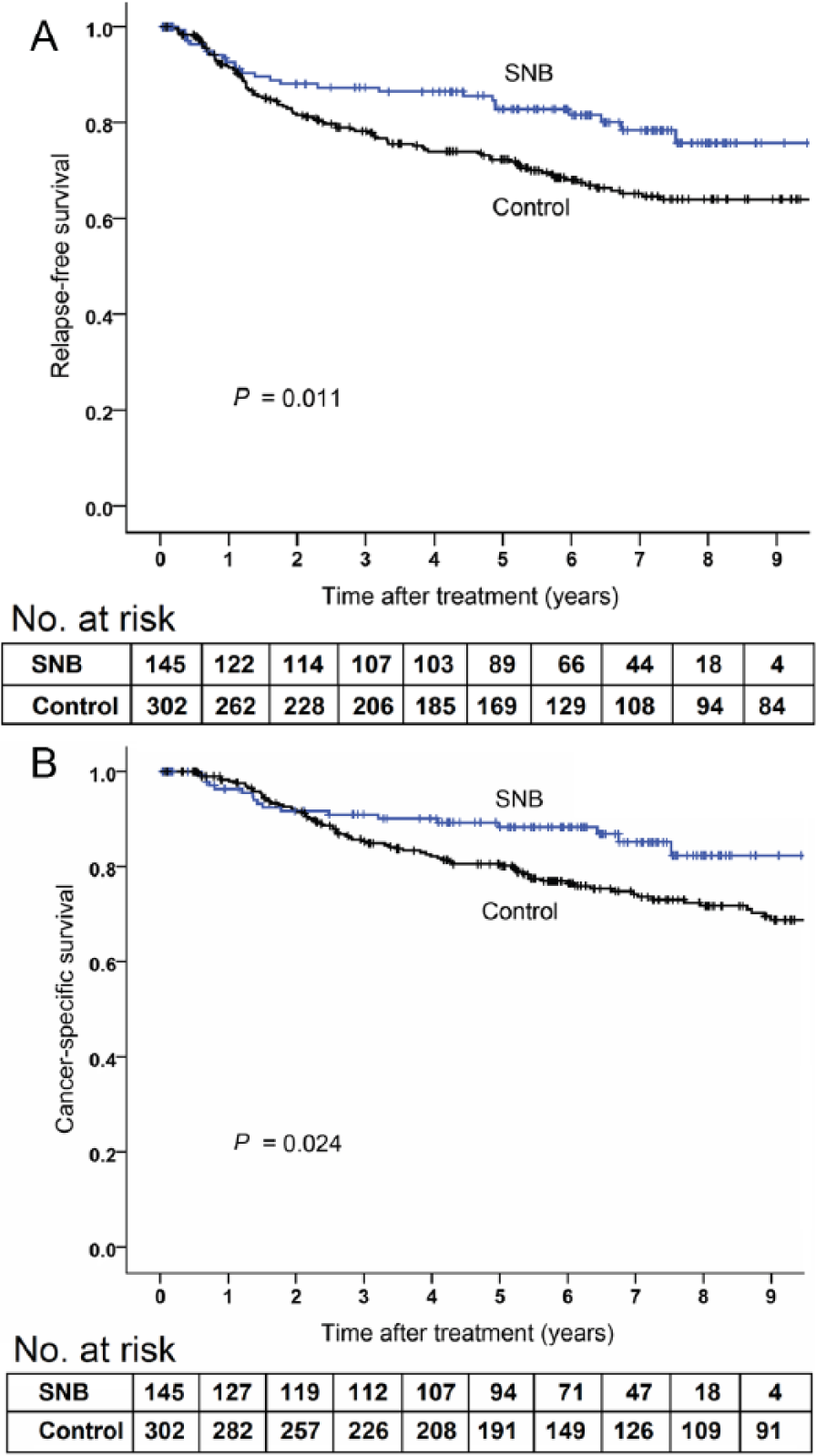
A) Relapse-free survival and B) cancer-specific survival in 145 male sentinel biopsy (SNB) patients and 302 male control patients. P values are calculated by log rank test.
Follow-up events and final status after a median follow-up time of 71 months in 305 sentinel node biopsy (SNB) patients and 74 months in 616 control patients.
Discussion
In this study, no differences existed between the study populations regarding RFS and cancer-specific survival, but interestingly, we found a disparity between genders. Women who underwent SNB showed no trends for increased survival, but in men, there were significant differences both in RFS and cancer-specific survival between SNB patients and their controls.
In prior studies, the impact of gender status on the survival benefit of SNB was not determined. Scoggins et al. (8) showed in a prospective multi-institutional study, consisting of 3324 patients, that gender is an independent predictor of survival. Men were more often sentinel-positive than women, but multivariate analysis showed that gender itself does not predict SN positivity. Men survived less than women both in sentinel-negative and sentinel-positive patients. In this study, however, the impact of SNB on survival was not investigated (8).
Morton et al. (6) reported results of MSLT-1 with no difference in overall survival between SNB patients and controls, but improved survival was found between nodal-positive patients. Between genders, no comparison analyses were reported.
Three German case–control studies reported on survival benefit following SNB (5, 9, 10). Satzger et al. (5) compared 377 retrospective pre-SNB patients with 296 SNB patients and associated with SNB, they found a better RFS, distant metastases free survival, and overall survival. Kretschmer et al. (9) compared 314 SNB-positive patients with 623 patients undergoing delayed TLND and found survival benefit favoring SNB patients. A study by Leiter et al. (10) compared 440 non-SNB patients with 439 SNB patients and found improved RFS in SNB patients, but no difference in overall survival. Again, none of these studies reported any gender differences.
In contrast, several studies have investigated gender disparity in the prognosis of melanoma, but independently of SNB. In a retrospective registry-based study by Mervic et al. (11), the data of 7338 melanoma patients were evaluated and gender was found to be an independent prognostic factor in melanoma (P = 0.002), but patients with positive SNs were excluded. Some other studies have also confirmed male gender to be an independent adverse prognostic factor (12–14). De Vries et al. (15) calculated that men have a relative risk of 1.85 (95% CI, 1.65–2.10) to die from melanoma. Women have a 30% independent relative survival advantage over men in stage I/II melanoma (13), and this benefit is present at all stages of disease progression (16).
The biological basis of this prognostical gender disparity in melanoma remains hypothetical. Sex hormones may play a role because melanocytes express estrogen receptors (ERs) (17) and estrogens may increase the number of melanocytes and modify their melanin content (18). Increased systemic estrogen levels are found to be related to hyperpigmentation and nevus enlargement during pregnancy (19). According to some retrospective studies, pregnancy may be a protective factor in melanoma (20, 21). Estrogen receptor β (ERβ) has a tumor-suppressive effect on a variety of human cancers (22) and is found to be the predominant ER type in melanocytic lesions (23, 24) and highly expressed in dysplastic nevi and melanoma in situ but not during melanoma progression (24). Compared to men, malignant melanocytic lesions in females are found to have more intense ERβ immunostaining (24). There are also studies suggesting that anti-estrogenic agent tamoxifen might be beneficial in the treatment of metastatic melanoma (25).
Furthermore, a strong gender-related factor is the anatomical location of melanoma, which is related to different amounts and patterns of sun exposure. In our study, the proportion of melanomas found in the trunk area was 30.2% in females and 60.2% in males. Melanomas rise through more than one biologic pathway and whether anatomic locations are associated with these heterogeneous pathways in disease development is unknown (26). Melanoma arising in sun-exposed areas, such as trunk, presents frequent mutations in BRAF gene, which is strongly associated with melanocortin-1 receptor (MC1R) polymorphisms. The expression of this gene can be modulated by sex hormones (27) and melanocyte regulation in the trunk may have hormone-related features since tumor distribution is different between males and females (28).
Since the introduction of SNB technique in 1992 (29), the survival benefit of SNB remains unsolved. The reason may be the lack of statistical power in many SNB trials, as in this study with low number of endpoint events. The gender-related factors also present a novel angle to this issue. While women have an independent survival advantage over men, it is surprising that SNB procedure may be curative in a subgroup of men but not in women. This separation may be related to gender-related patterns of disease progression, as men have a higher incidence of regional lymphatic metastases. In patients without nodal metastases, SNB provides only prognostic information but no therapeutic benefit. Also, in our study, the proportion of nodal-positive SNB patients was higher in men (23.4%) compared with women (15.6%). We suggest that a subgroup of men develops a stepwise model of disease progression and that the eradication of the first nodal micrometastasis may interrupt the whole metastatic cascade. In contrast, in females, simultaneous lymphatic and hematogenic dissemination may be the predominant pattern of dissemination explaining the non-existent survival effect. The biological basis of this gender-dependent hypothesis remains an enigma.
Our study should be interpreted based on its retrospective nature. It is unclear whether gender disparity is associated only with SNB procedure or also with other unregistered factors. The patients of each study group were treated during two different time periods. The first control patient was operated in 1983 and the last SNB patient in 2007. There are many issues that make this a challenging comparison because melanoma staging and treatment was dramatically different in 1983 than in 2007. During this long period, there may have been complex environmental and epidemiological changes related to both melanoma and human health. Gender status may have attributed to these factors.
The study cohorts matched well based on Breslow thickness and other baseline factors, but some unreliability may be associated with tumor ulceration. In the 1980s, the importance of ulceration was not recognized. In this study, original specimens were not reviewed, but every histopathological report was read meticulously. In most cases, it was clear whether the erosion of the epidermis was present or not, but in some cases, this information remained uncertain. Mitotic rate was not routinely determined over the course of this comparison between groups.
Another limitation is the low number of endpoint events and therefore differences in survival between genders may be statistically significant but coincidental. After a stratified subgroup analysis, there were only 27 recurrences and 18 melanoma-related deaths in male SNB patients. In some elderly men, the cause of death was clinically based on other comorbidities and no autopsy was performed. Although no recurrent melanoma was observed in them, undetected metastatic disease may be the underlying cause of death in elderly patients.
Conclusion
In the whole patient group, we were not able to find an improvement in survival outcomes related to SNB. However, in a secondary analysis, sorted by gender status, males had significantly improved RFS and cancer-specific survival after SNB, which females lacked. This result should be validated by further independent studies.
Footnotes
Acknowledgements
The authors thank Robert M. Badeau, PhD, for the linguistic revision of this manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by a grant from the University Hospital of Turku (EVO).
