Abstract
Introduction:
Pseudomyxoma peritonei is a rare disease causing peritoneal carcinomatosis. In patients with extensive carcinomatosis, curative treatment is unachievable. Palliative debulking therapy is the only treatment in relieving symptoms. We report our results from palliative debulking surgery at a national pseudomyxoma peritonei center in Denmark.
Methods:
From January 2007 to October 2012, we performed 27 palliative operations for pseudomyxoma peritonei with debulking at our institution. All patients were evaluated and found eligible for palliative treatment only. Patients were prospectively registered, while perioperative data were collected retrospectively from patient records.
Results:
The majority of patients (n = 25) received an omentectomy (93%) as the primary procedure. In total, 17 (63%) received additional surgery. Median operative time was 88 min (range: 33–160 min). Median stay at the recovery ward was 6 h (2–288 h). Median hospital stay was 8 days (4–105 days). In all, 23 (85%) patients had no in-hospital complications, whereas 4 patients experienced complications (15%). Two of the complications were mild (Clavien-Dindo grade II), and two experienced severe complications (grade III and IV). Thirty-day mortality was 0%. Median survival was 3.0 years (0.2–6.2 years).
Conclusion:
Palliative debulking is a safe procedure with an acceptable morbidity and mortality offering immediate disease control.
Introduction
Pseudomyxoma peritonei (PMP) is a rare disease with an incidence of 1–2 per million per year (1–4). The primary tumor most often originates from the appendix and is characterized by a mucin-producing potential (4). When this results in perforation of the appendiceal barrier, mucin spreads into the abdominal cavity. The patients often present with suspected acute appendicitis, an ovary mass in females, a new-onset hernia in males or with increased abdominal distension which, in time, may cause obstructive bowel disease (1–6). The disease may also be diagnosed as an incidental finding on radiographic imaging (Fig. 1) (6).
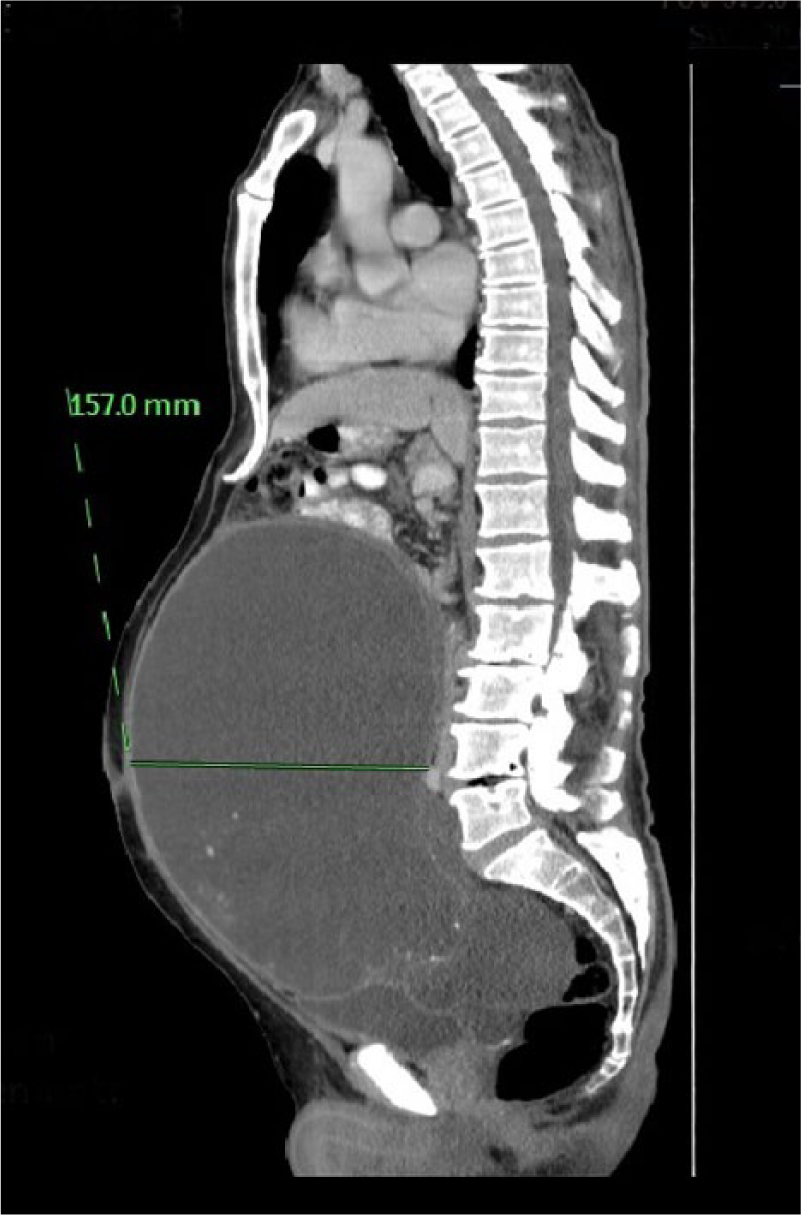
Patient suffering from a large tumor mass. The patient was treated with palliative debulking surgery. The picture is printed with permission from the Department of Radiology, Aarhus University Hospital THG, Denmark.
Crytoreductive surgery (CRS) combined with heated intraperitoneal chemotherapy (HIPEC) is considered to be the only curable treatment for this life-threatening disease (1–3, 5, 7, 8). CRS consists of absolute macroscopic tumor removal. Residual disease should be less than 2.5 mm. If complete cytoreduction is not possible due to extensive disease, then tumor debulking combined with palliative systemic chemotherapy is an option (1, 5). Little is known, however, about outcomes following palliative debulking therapy, as other studies often mix patients receiving debulking surgery, complete CRS, and, in some cases, HIPEC (1, 3, 7). Our aim is, thus, to focus on the results from palliative debulking surgery only. Results are from a single institution, which is the national PMP referral center of surgery in Denmark.
Methods
All PMP patients in Denmark are referred to our institution for evaluation and surgical treatment. All patients are registered prospectively. Patients with PMP receiving palliative debulking surgery from January 2007 to October 2012 were included in this study.
PMP Patients Selected for Surgery
Contraindications for complete CRS and HIPEC consisted of physiologic age above 75 years, severe comorbidity (American Society of Anesthesiology (ASA) score above 2), extraperitoneal disease, invasive growth into retroperitoneal space or abdominal wall (except previous incision port sites), massive disease involvement of the small bowel or its mesentery, more than one stenosis of the small bowel due to carcinomatosis or disease involvement of the hepatic pedicle or pancreas (9). Palliative debulking surgery was offered to those patients in whom complete CRS and HIPEC were contraindicated, yet experienced pain or symptoms of bowel obstruction with increasing abdominal distension, mucinous ascites or omental cake.
Preoperative Evaluation
Patients were evaluated preoperatively by contrast-enhanced multi-sliced (positron emission tomography (PET)) computed tomography (CT) scan of the chest, abdomen, and pelvis, as well as gastroscopy and colonoscopy and, if necessary, preoperative laparoscopy, as described elsewhere (9, 10). Treatment was planned at multidisciplinary team meetings (9). The tumor extent was registered according to the Dutch 7 Region Count (Dutch score) at surgery in all patients (11). All patients were graded preoperatively according to the ASA score.
Surgery
All palliative procedures were undertaken using a midline incision. The goal was to diminish tumor burden as well as reduce future mucin production. Most cases involved resection of the greater omentum (resected below the transverse colon) and sometimes tumor debulking as well. This often consisted of appendectomy, ileocaecal resection or right-sided hemicolectomy, but any major tumor or metastasis might be resected, if it did not constitute too great a surgical risk. Surgery was performed by one of three surgeons experienced in performing CRS and HIPEC. Perioperatively, all patients received low-molecular-weight heparin as thrombo-prophylaxis and an epidural for analgesia.
Postoperative Course
Postoperative data were collected retrospectively from patient records, pathology reports, and the anesthesia and recovery ward charts. Postoperative in-hospital complications occurring either at our institution or local hospital were graded according to the Clavien-Dindo classification. The classification has been described elsewhere (12), but briefly restated here: grade 0 refers to no complications, and grade I constitutes a deviation from the normal postoperative course which is self-resolving. Grade II refers to complications requiring medical treatment. Grade III refers to complications requiring surgery, endoscopy, or interventional radiology. Grade IV is a life-threatening condition requiring intensive care. Grade V refers to complications resulting in death. Complications investigated were reoperation for bleeding, reoperation for gastrointestinal leakage, explorative laparotomy, vacuum-assisted closure, wound infection (superficial or deep), intra-abdominal abscess, sepsis, pleural effusion (requiring drainage), acute kidney injury (requiring dialysis), acute respiratory distress syndrome, pneumonia, deep venous thrombosis, pulmonary embolism, urinary tract infection, and intestinal fistula (internal or external). Pathology specimens were re-evaluated by a single pathologist with special interest in PMP and graded according to the World Health Organization (WHO) classification (13). Patients with high-grade tumors were referred to the oncologic department at their primary hospital for palliative chemotherapy.
Statistics
Continuous variables are given as median and range. Patients receiving debulking surgery twice are included twice. The calculation of survival data was specified from the date of operation and performed using the Kaplan–Meier method (14). Patients were followed until death or 20 February 2014, whichever came first.
Results
In this study, 27 patients with a median age of 70 (39–81) years were included. Of them, 20 (74%) patients were men. Median follow-up was 28 months (86–2259 days). Anesthesia reports were available for 21 patients only (median operation time and stay at the recovery ward). Palliative surgery was planned preoperatively in 22 patients. Five patients were deemed amenable to CRS and HIPEC, but tumor extent was found too extensive at surgery, and palliative surgery was performed instead. These five patients have partially been included in a previous study (15).
Operative Data
According to the Dutch score, 25 patients had involvement of all seven regions (11). In two patients, the spread of the disease was not classified. A total of 26 patients had extensive disease with involvement of the small bowel and its mesentery as a sign of late-stage disease. The majority of patients (n = 25) received an omentectomy (93%) as the primary procedure; 17 (63%) received additional surgery, most often an appendectomy (44%). Other procedures performed were ileocaecal resection (7%), right hemicolectomy (4%), small bowel resection (4%), stoma (4%), salpingectomy (4%), oophorectomy (7%), peritonectomy (4%), and splenectomy (4%). One patient was operated twice. One patient was operated subacutely due to bowel obstruction. The remaining two patients (7%) had tumor debulking (4 and 7.5 kg of mucin mass, respectively) but no organ resection.
Median operative time was 88 min (33–160 min). Only one patient received fresh frozen plasma within the first 24 h (7 units). Nine patients received transfusion of packed red blood cells within the first 24 h (1–4 units). See Table 1 for further details.
Pre- and perioperative data.
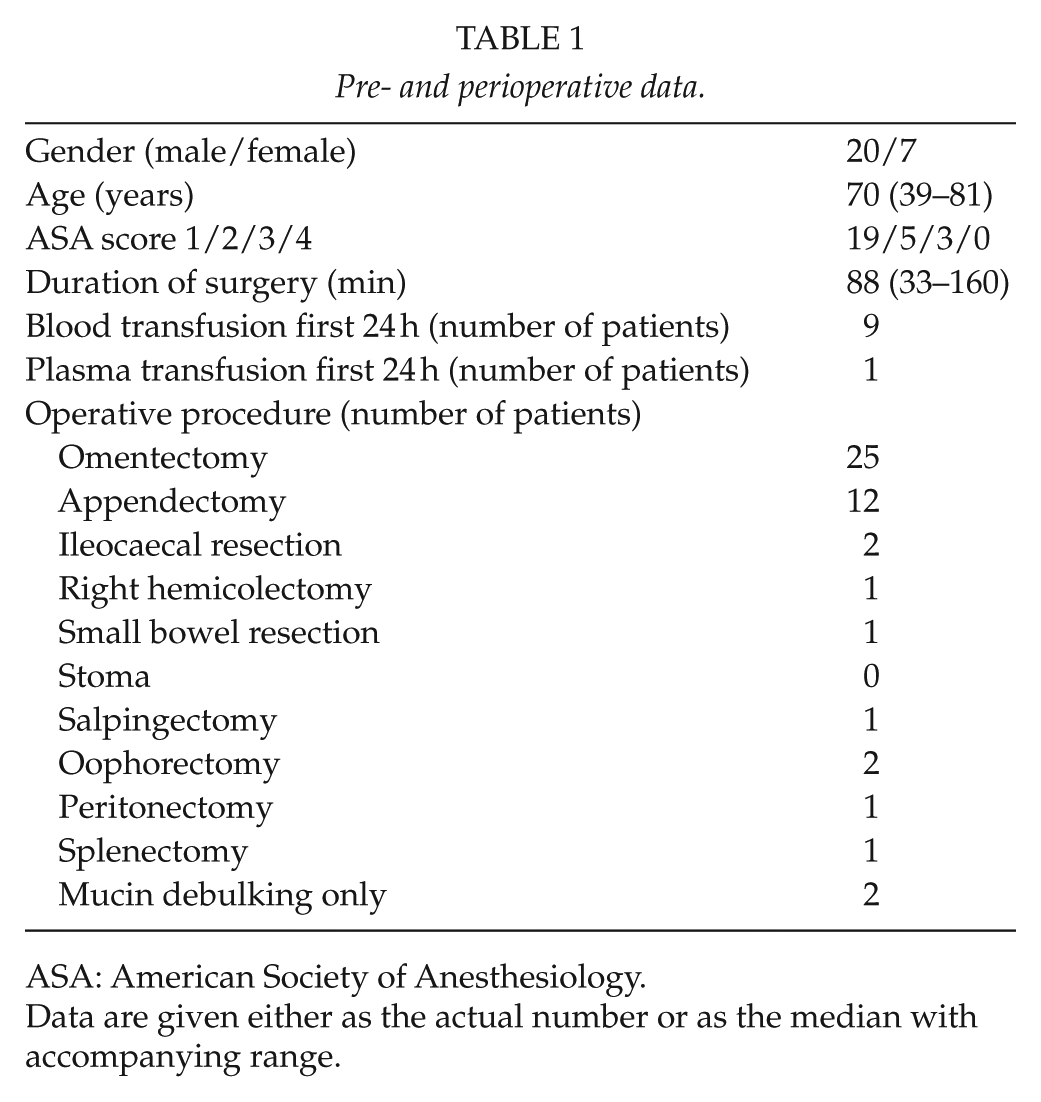
ASA: American Society of Anesthesiology.
Data are given either as the actual number or as the median with accompanying range.
Postoperative Course
Median stay at the recovery ward was 6 h (2–20 h). Median hospital stay was 8 days (4–105 days). In all, 23 (85%) patients had no in-hospital complications, whereas 4 patients experienced complications (15%). Two of the complications were mild (Clavien-Dindo, grade II). One patient was re-operated after transfer to a local hospital. The patient was septic and with abdominal pain. At explorative laparotomy, an intra-abdominal abscess was found and evacuated (grade III). Another patient was re-operated at our institution due to gastric perforation following omentectomy (grade IV). This patient experienced postoperative sepsis and respiratory failure and was the only patient in the present series admitted to the intensive care unit (12 days). The patient was discharged from hospital after 105 days. The overall 30-day mortality was 0% (Table 2).
Postoperative data.
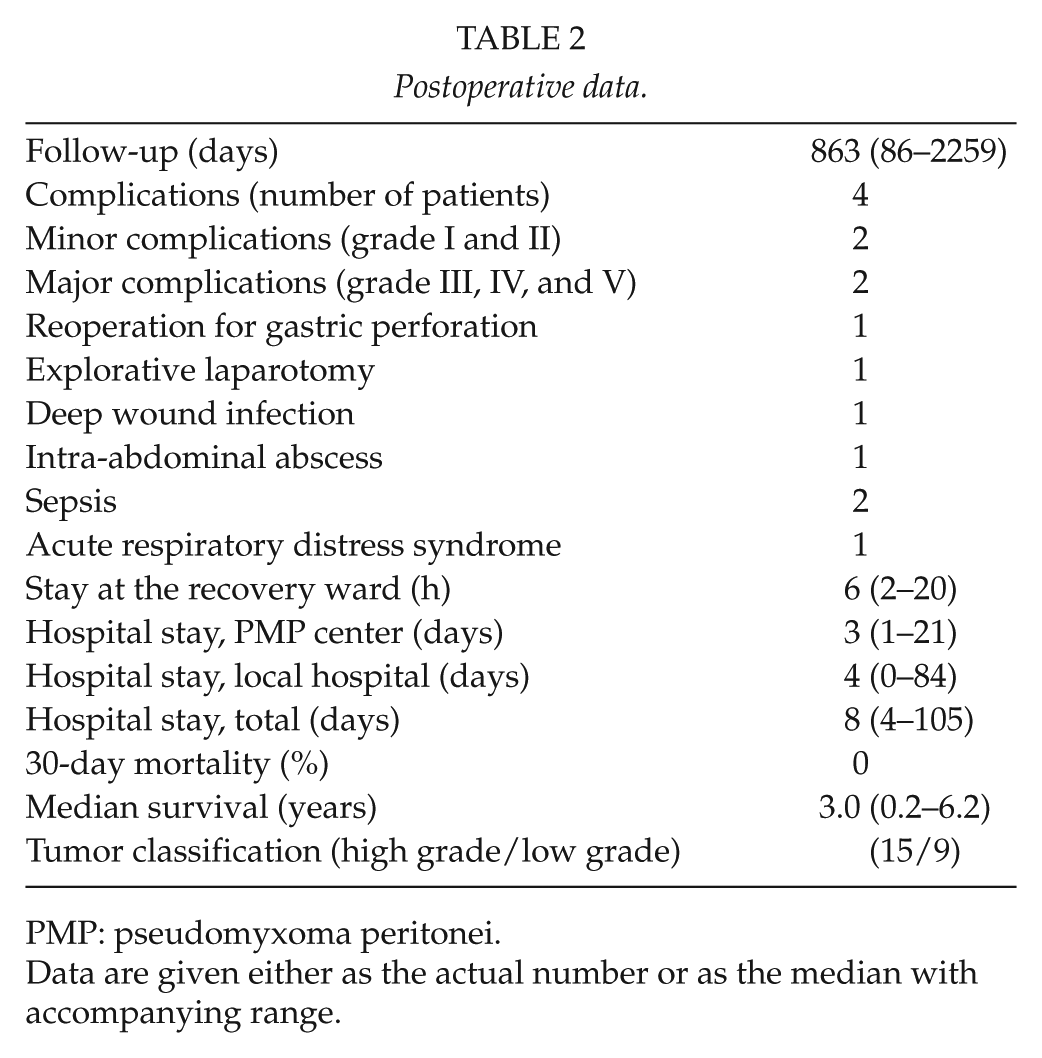
PMP: pseudomyxoma peritonei.
Data are given either as the actual number or as the median with accompanying range.
In total, 20 (74%) patients received postoperative chemotherapy with a median delay from the time of surgery to initiation of chemotherapy of 53 (23–898) days. Various chemotherapeutic regimens were used, always in combination, except for capecitabine. The drugs most often used were capecitabine, bevacizumab, irinotecan, and fluorouracil, and the most used combination included irinotecan/fluorouracil/bevacizumab. All patients receiving chemotherapy were diagnosed with either a “high-grade” tumor (n = 3), an “intermediary grade” tumor (n = 7) or the tumor was not classified (n = 10). Two patients did not receive chemotherapy though diagnosed with a “high-grade” tumor. One patient refused further treatment, whereas the reason for not receiving chemotherapy was unknown for the other patient.
Overall survival is depicted in Fig. 2, where median survival was 3.0 years (0.2–6.2 years). As of 20 February 2014, there were 10 patients still alive.
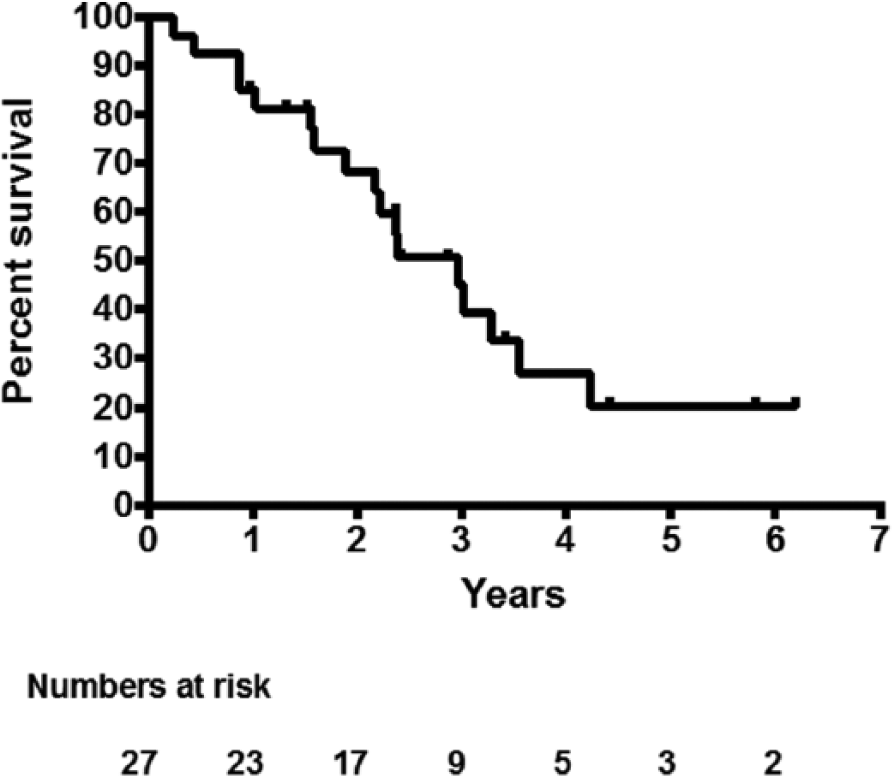
Kaplan–Meier overall survival plot.
Pathology
In the original pathology reports, 12 tumors were not classified, eight were classified as “intermediary grade,” and 5 as “high grade.” Only one tumor was classified as “low grade.” However, re-evaluation by a dedicated pathologist demonstrated 15 high-grade tumors and 9 low-grade tumors. All, but two tumors were of appendiceal origin, whereas one was of ovarian and one was of unknown origin. Two specimens were lost for re-evaluation.
Discussion
This single-center retrospective study shows that debulking surgery as a palliative measure in PMP is a feasible method in order to control the disease. The procedure is generally well tolerated and there were few complications. Only two patients experienced a major complication, that is, Clavien-Dindo grade III or IV. This is of course associated with the relative low ASA score of the present patients. There were only three patients classified as ASA 3 and there were no ASA 4 patients. It should also be added that procedures were performed by a small group of dedicated surgeons and limited to those cases in which large and accessible specimens could be removed with anticipated technical success. This is, of course, also reflected in the low operative times.
We experienced a postoperative 30-day mortality of 0%. This is difficult to compare to other studies, as the study population of other studies is often a mix of patients receiving debulking surgery, complete CRS, and, in some cases, HIPEC. Postoperative oncologic treatment is not often detailed or even mentioned. Nevertheless, Miner et al. (6) reported a 30-day mortality of 4% in patients receiving incomplete cytoreduction; however, HIPEC was performed in selected cases. Median survival in this group was 4.2 years compared to 3.0 years in our study. It is well known that survival is highly dependent on the histopathology of the disease (high- versus low-grade pseudomyxoma) (6, 16). One study, however, is noted for publishing data on palliative surgery separately. Dayal et al. reported retrospectively their results on 748 patients receiving treatment for PMP. Of these patients, 205 received palliative debulking surgery only. They found a 30-day mortality of 1% and a median survival of 2.7 years (8). This is very similar to our results.
Most authors agree that HIPEC can be used as a supplementary treatment in resectable PMP. Conversely, there is no consensus regarding HIPEC as a palliative treatment of PMP combined with tumor debulking (1–3, 16, 17). Chua et al. showed in a very large study (n = 2298) that HIPEC was an independent prognostic factor for an improved progression-free survival in patients with PMP. However, the patients were treated with both complete and incomplete cytoreduction, and these data cannot be applied to patients receiving palliative surgery alone. Furthermore, HIPEC had no impact on overall survival (16). Järvinen et al. (18) reported their result with serial debulking achieving both incomplete and complete cytoreduction. They actually showed an acceptable 5-year survival in this mixed population of 67% almost similar to other authors using HIPEC. 10-year survival, however, was only 31% which is a good deal lower compared with the HIPEC treatment. It should furthermore be noted that as the number of previous operations increases, new debulking procedures become more difficult. Youssuf et al. presented a large study with 456 patients operated for an appendiceal tumor. Complete CRS was not possible in 152 patients, and therefore, only major tumor debulking was performed. They performed an additional HIPEC procedure in selected patients, but this number was not reported. They do state, however, that it is a procedure that should be implemented with care, due to a significant morbidity and reoperation rate (1). HIPEC is described as beneficial in the treatment of malignant ascites in two studies. Garofalo et al. described 14 patients treated with laparoscopic HIPEC due to malignant ascites from miscellaneous cancers (gastric cancer, colorectal cancer, ovarian cancer, breast cancer, and peritoneal mesothelium). The ascites appeared to be controlled on follow-up CT scans (19). Ba et al. (20) described 16 cases of gastric cancer treated with laparoscopic HIPEC. Although symptoms were relieved, 13 patients died within 9 months. It should be mentioned, however, that the prognosis in PMP is more favorable than carcinomatosis from various other gastrointestinal cancers, which is why these results cannot be applied to PMP. As the literature on the subject is sparse, our institution has not yet implemented HIPEC in treatment of incurable PMP. Laparoscopy, however, may have a role in the palliative treatment of PMP as reported by Kelly et al. (21). They reported their results on 10 patients undergoing 17 laparoscopic debulking procedures. They had very limited complications and all procedures were performed as same-day surgeries. This method, however, is limited to debulking of mucinous ascites only and not for solid mass debulking.
In our study, one patient was operated on twice; only mucinous mass was removed at the second operation. Serial debulking procedures have previously been common in treating PMP (6, 22). Miner et al. (6) presented a study with an average operation rate of 2.2, ranging from 1 to as many as 6 operations per patient. They describe that complete cytoreduction is possible even if they did not succeed at the first operation. It was, however, significantly more common after the first and second time, and operations performed more than three times were only palliative in order to control symptoms. The durability of symptom control tended to decrease after each operation and was significantly shorter after the third and fourth operation (6). In the present series, the majority of patients had advanced disease with massive involvement of the small bowel and its mesentery. There is no consensus on the matter, and indication for surgery must be individualized, taking into account the state of the patient, including co-morbidities and accessibility of the tumor burden. Intuitively, the effect of debulking probably decreases with the number of procedures, while the risk of complication increases.
Of our patients, 74% received chemotherapy. The time from operation to chemotherapy was 53 days, albeit with a wide range (23 to 898 days). Different approaches regarding initiation of chemotherapy were used, but our oncologist associates generally favored implementation within 3 months. In a few cases, chemotherapeutic treatment was postponed until symptom recurrence. The chemotherapeutic agent of preference was capecitabine, which was also the only drug used as mono-therapy. There is no consensus on the treatment of PMP. In the study from Chua et al. (16), the most important treatment increasing survival significantly was surgery with complete cytoreduction (22).
Our study is the first study to report on debulking surgery from a single national PMP center in a palliative cohort only. It strengthens our study, further, that patients are registered prospectively using the same selection criteria during the entire period. Additionally, pathology was investigated by a single pathologist with special interest in PMP. A limitation in our study, however, was that perioperative data were collected retrospectively, which might underestimate the number of complications. Finally, the number of patients included was relatively low, rendering subgroup analyses and comparisons difficult.
Conclusion
We conclude that palliative debulking surgery is a feasible method with an acceptable complication rate and a low postoperative morbidity and mortality. We aimed to keep surgery as minimal as possible, only removing accessible pathology in order to minimize operative risk. Further studies are needed in order to fully investigate the potential gain from HIPEC treatment in palliative PMP surgery.
Footnotes
Declaration Of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
