Abstract
Background and Aims:
Fluid resuscitation of severely injured patients has shifted over the last decade toward less crystalloids and more blood products. Helsinki University trauma center implemented the massive transfusion protocol in the end of 2009. The aim of the study was to review the changes in fluid resuscitation and its influence on outcome of severely injured patients with hemodynamic compromise treated at the single tertiary trauma center.
Material and Methods:
Data on severely injured patients (New Injury Severity Score > 15) from Helsinki University Hospital trauma center’s trauma registry was reviewed over 2006–2013. The isolated head-injury patients, patients without hemodynamic compromise on admission (systolic blood pressure > 90 or base excess > –5.0), and those transferred in from another hospital were excluded. The primary outcome measure was 30-day in-hospital mortality. The study period was divided into three phases: 2006–2008 (pre-protocol, 146 patients), 2009–2010 (the implementation of massive transfusion protocol, 85 patients), and 2011–2013 (post massive transfusion protocol, 121 patients). Expected mortality was calculated using the Revised Injury Severity Classification score II. The Standardized Mortality Ratio, as well as the amounts of crystalloids, colloids, and blood products (red blood cells, fresh frozen plasma, platelets) administered prehospital and in the emergency room were compared.
Results:
Of the 354 patients that were included, Standardized Mortality Ratio values decreased (indicating better survival) during the study period from 0.97 (pre-protocol), 0.87 (the implementation of massive transfusion protocol), to 0.79 (post massive transfusion protocol). The amount of crystalloids used in the emergency room decreased from 3870 mL (pre-protocol), 2390 mL (the implementation of massive transfusion protocol), to 2340 mL (post massive transfusion protocol). In these patients, the blood products’ (red blood cells, fresh frozen plasma, and platelets together) relation to crystalloids increased from 0.36, 0.70, to 0.74, respectively, in three phases.
Conclusion:
During the study period, no other major changes in the protocols on treatment of severely injured patients were implemented. The overall awareness of damage control fluid resuscitation and introduction of massive transfusion protocol in a trauma center has a significant positive effect on the outcome of severely injured patients.
Keywords
Introduction
Uncontrolled bleeding remains the leading cause of potentially preventable deaths after severe trauma (1–3). Hemorrhagic shock and exsanguination account for about 50% of deaths in the first 24 h after injury (4, 5). About one-third of bleeding trauma patients present with coagulopathy on admission (6), which is associated with a fourfold or greater increase in mortality (7, 8).
The strategy of fluid resuscitation in severely injured patients requiring massive transfusion (MT) has been changing over the last decade. High volume fluid resuscitation with crystalloids has been replaced with damage control resuscitation (DCR). This DCR approach consists of avoidance of hypothermia, permissive hypotension (systolic blood pressure (sBP) targeted at 90 mmHg in bleeding patients without head injury), the immediate administration of blood products (packed red blood cells (PRBC), fresh frozen plasma (FFP), and platelets), avoidance of crystalloids and colloids, and damage control surgery or angioembolization to treat the cause of bleeding (9–13).
Along with the evolvement of DCR, many trauma centers have implemented pre-defined protocols of MT for trauma patients suffering major hemorrhage, which can be defined as “the loss of a patients’ total blood volume in less than 24 h” (14) or, more practically in the acute trauma setting, “an estimated blood transfusion volume of over four units in the initial 2–4 h” (9). The term “massive transfusion” is mostly used for ≥10 U PRBC within 24 h (15), alternatively ≥10 U PRBC within 6 h (16). DCR and massive transfusion protocols (MTPs) have been associated with improved survival in severely injured patients (7, 17–19).
The MTP was implemented at the end of 2009 in the Helsinki University trauma center. The aim of the present study was to review the changes in fluid resuscitation and its influence on outcome of severely injured patients with hemodynamic compromise treated at the single tertiary trauma center.
Material and Methods
Trauma Unit and Trauma Registry of Helsinki University Hospital
In Southern Finland, the treatment of severe blunt injuries of adult patients (>16 years) is centralized to Helsinki University trauma center (HU trauma center), which is one of Europe’s largest trauma centers treating 400–450 severely injured (Injury Severity Score (ISS) > 15) adult patients annually (children under the age of 16 and patients with penetrating body injuries are treated at a different location within the Helsinki University Hospital). The catchment area of the designated trauma center includes approximately 2 million inhabitants. So far, the HU trauma center is the only hospital with a trauma registry in Finland, the Trauma Registry of Helsinki University Hospital (TR-THEL). All trauma admissions to the HU trauma center from 1 January 2006 onwards have been reviewed by three trauma nurses, and all patients with New Injury Severity Score (NISS) over 15 have been entered into the registry.
MTP in the Trauma Unit of Helsinki University Hospital
MTP was implemented in the HU trauma center at the end of 2009. The activation criteria were defined as sBP < 90 mmHg and/or no radial pulse according to pre-notification call or sBP < 90 mmHg and/or no radial pulse or suspicion of massive hemorrhage on arrival at the HU trauma center. The first emergency transfusion package (ETP) contains four units of PRBC (O−) and four units of FFP (AB). From the second ETP onwards, also one unit of platelets (which contains platelets from four donors) is included in addition to four units of PRBC and four units of FFP (PRBC:FFP:platelets ratio 1:1:1). The MTP in the HU trauma center is shown in Table 1.
Massive Transfusion Protocol in Helsinki University trauma center (for adult patients).
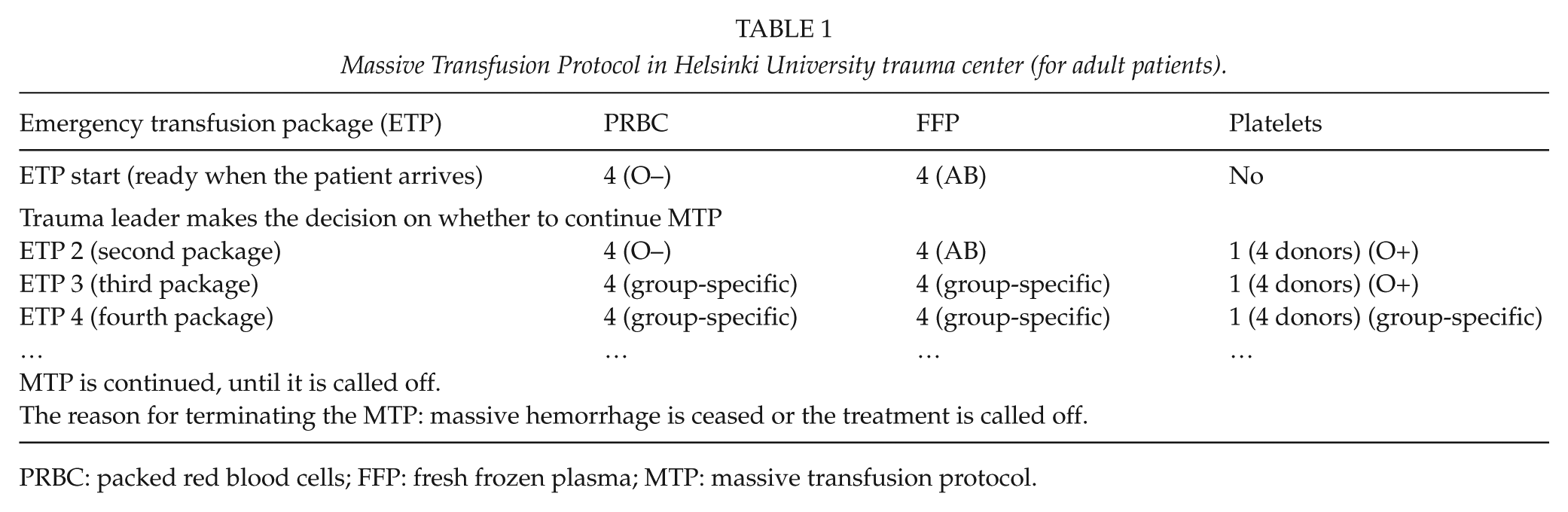
PRBC: packed red blood cells; FFP: fresh frozen plasma; MTP: massive transfusion protocol.
Patient Inclusion
We included all patients with severe injury (defined as an NISS > 15) treated in emergency room (ER) and entered into the TR-THEL between 1 January 2006 and 31 December 2013. Patients dead on arrival, transferred in from another hospital, with burn injury, with isolated head injury, without hemodynamic compromise on arrival (defined as sBP > 90 mmHg or base excess (BE) > –5.0 mmol/L on arrival), or with missing data for Revised Injury Severity Classification II (RISC II) calculations were excluded.
Data Analysis
The study period was divided into three phases: 2006–2008 (pre-protocol), 2009–2010 (the implementation of MTP), and 2011–2013 (post MTP). The subgroups of the patients were formed accordingly.
The primary outcome measure was death in hospital within 30 days from admission. For each subgroup, expected mortality (using the prognoses derived from the RISC II (20)) and Standardized Mortality Ratio (SMR) were calculated. SMR is defined as the ratio of the observed mortality rate divided by the expected mortality rate. SMR values over 1 (higher observed than predicted mortality) indicate a poorer than average quality of care, and SMR values under 1 (higher predicted than observed mortality) a better than average care. The SMR is presented with its 95% confidence interval.
For each subgroup, prehospital fluids and timings, the first recorded values of partial thromboplastin time (PTT), BE and hemoglobin on admission, and the amounts of crystalloids, colloids, PRBC, FFP, platelets, and total volume of fluids given in the ER were calculated. The volumes were adjusted to nearest 10 mL. Furthermore, the blood products’ (PRBC, FFP, and platelets) relation to crystalloids and FFP’s relation to PRBC were calculated. Patients who were not given any PRBC in the ER were identified.
For the statistical analyses, SPSS statistical software was used (IBM Corp., Version 20.0. Armonk, NY, USA). Differences between the pre-phase and the post-phase were evaluated with Pearson’s Chi-squared test and Mann-Whitney U-test for categorical and continuous measurements, respectively. Statistical comparisons with the intermediate phase were not performed. Categorical variables are presented as percentages with number of cases, and continuous variables are presented as mean, standard deviation. Median values have been added where appropriate. The study protocol was approved by TR-THEL’s administrative board.
Results
A total of 354 patients were included (Fig. 1). The numbers of the patients in the subgroups were 147 (pre-protocol), 86 (at implementation of MTP), and 121 (post MTP). There were no relevant differences in patient characteristics between the groups (Table 2). A slight trend of decreasing volume of prehospital-administered fluids was noticed. The prehospital time (minutes from dispatch call to arrival to hospital) and the coagulation status on arrival (the first measured PTT, BE, and hemoglobin) remained steady (Table 3). Also, the distribution of Abbreviated Injury Scale (AIS) ≥ 2 injuries in different body regions was observed to be comparable during the study period (Fig. 2).
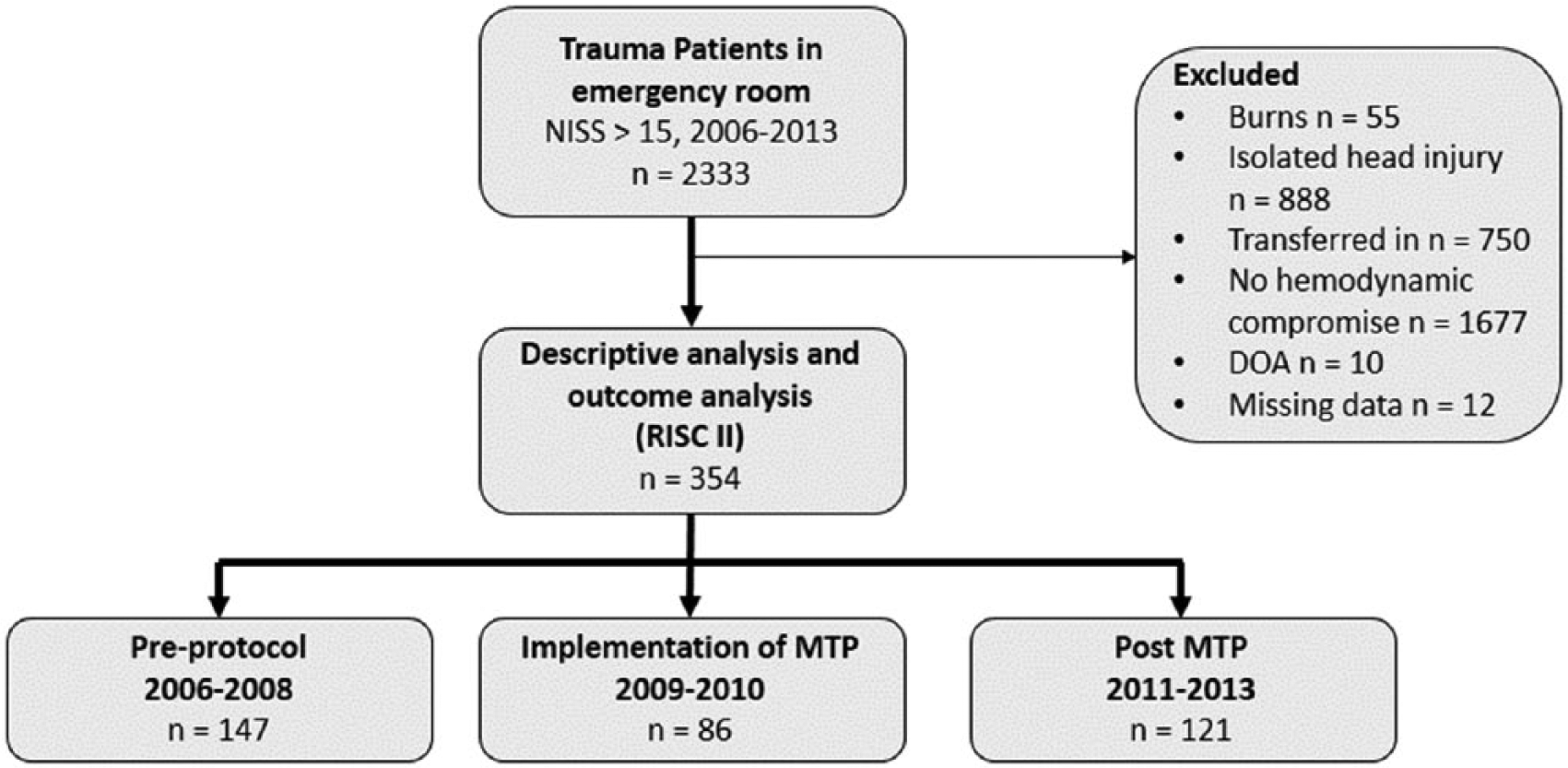
Flow diagram of included and excluded patients in outcome analysis (multiple reasons for exclusion could apply).
Patient characteristics. Continuous values are presented as mean/median (SD).
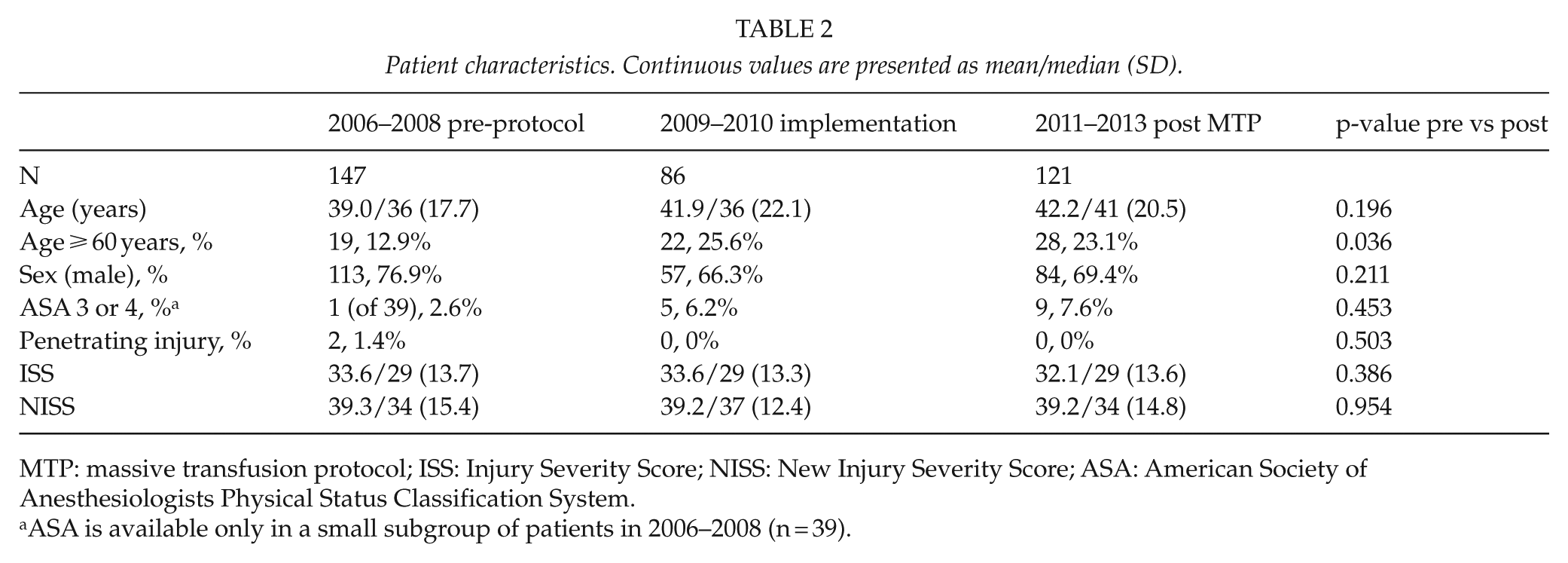
MTP: massive transfusion protocol; ISS: Injury Severity Score; NISS: New Injury Severity Score; ASA: American Society of Anesthesiologists Physical Status Classification System.
ASA is available only in a small subgroup of patients in 2006–2008 (n = 39).
Prehospital fluids and time (minutes from dispatch call to arrival to hospital), first measured partial thromboplastin time (PTT), base excess and hemoglobin on arrival. Mean/median (SD).
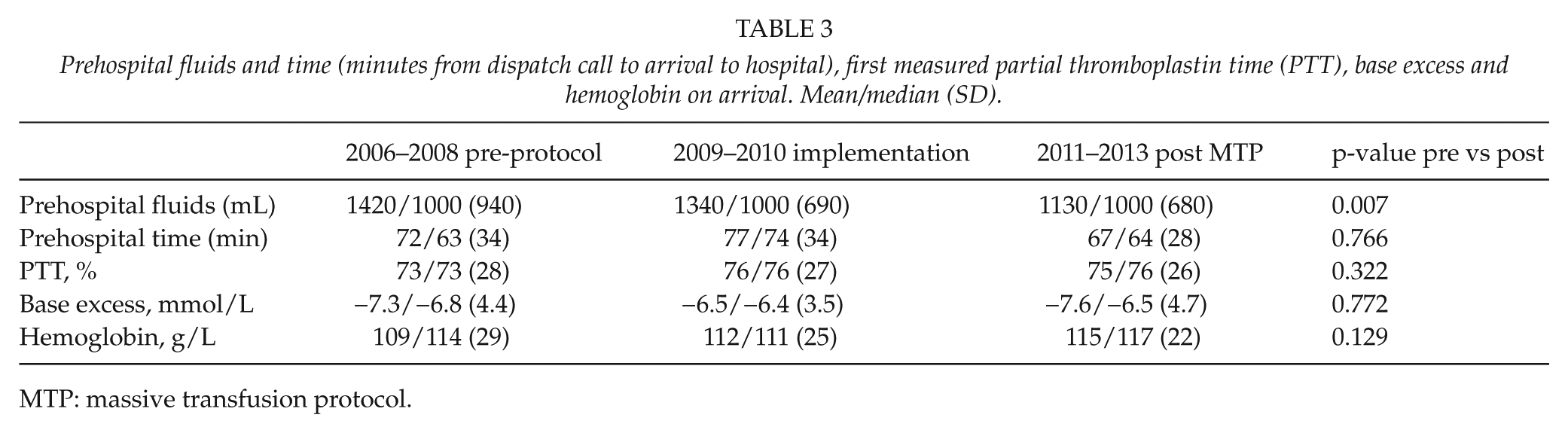
MTP: massive transfusion protocol.
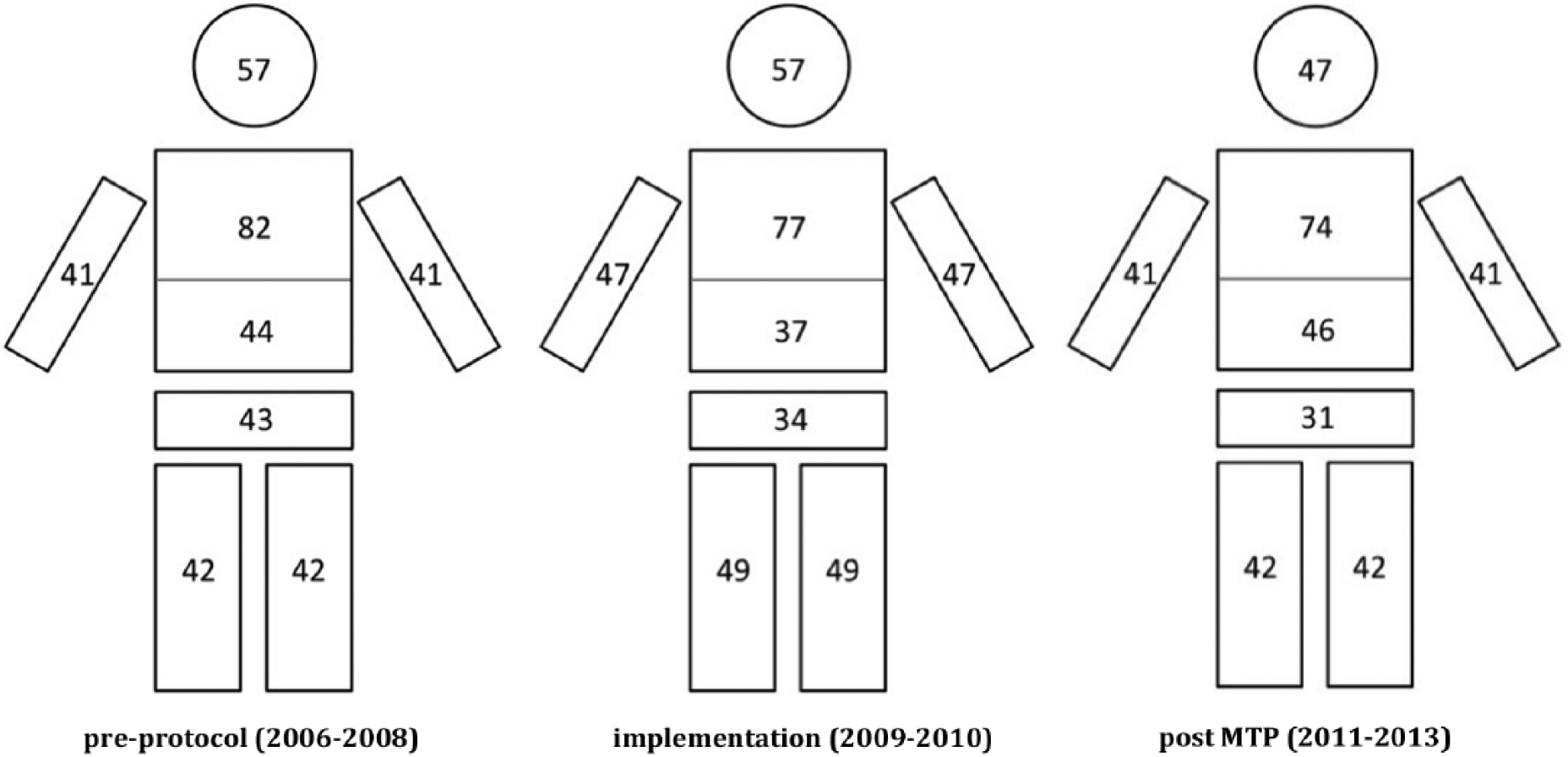
Injury distribution. Percentage of register’s patients having injuries (AIS ⩾ 2) in head, thorax, abdomen, pelvis, or upper/lower extremity is shown.
There was a slight trend of decreasing ER time (the time from arrival to hospital to departure from the ER) during the three time periods. The percentage of patients undergoing emergency bleeding-control operations decreased during the study period. The amount of crystalloids used in the emergency department decreased from 3870 mL (pre-protocol), 2390 mL (implementation), to 2340 mL (post MTP). The use of colloids was relatively minimal in general and also showed a decreasing trend during the study period (370, 330, and 140 mL, respectively). The overall blood products’ (PRBC, FFP, and platelets together) relation to crystalloids increased from 0.36, 0.70, to 0.74, respectively (Table 4). The percentage of patients receiving no PRBC in the ER was 47.9 (70/147, pre-protocol), 45.9 (39/86, implementation), and 51.2 (62/121, post MTP) (Table 5).
In-hospital fluid resuscitation, ER time, emergency operations/angioembolization for hemorrhage. Mean/median (SD).
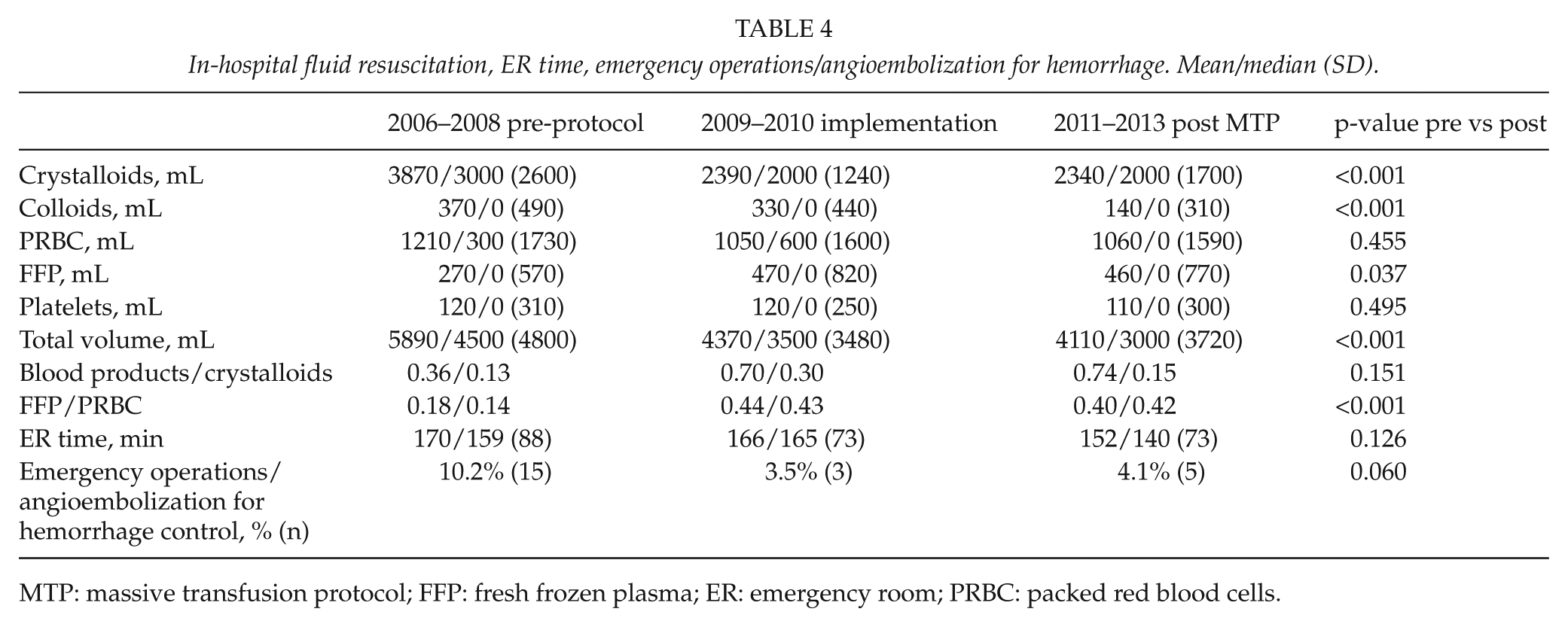
MTP: massive transfusion protocol; FFP: fresh frozen plasma; ER: emergency room; PRBC: packed red blood cells.
The percentage of patients requiring red blood cell transfusion in the emergency department.

MTP: massive transfusion protocol; PRBC: packed red blood cells.
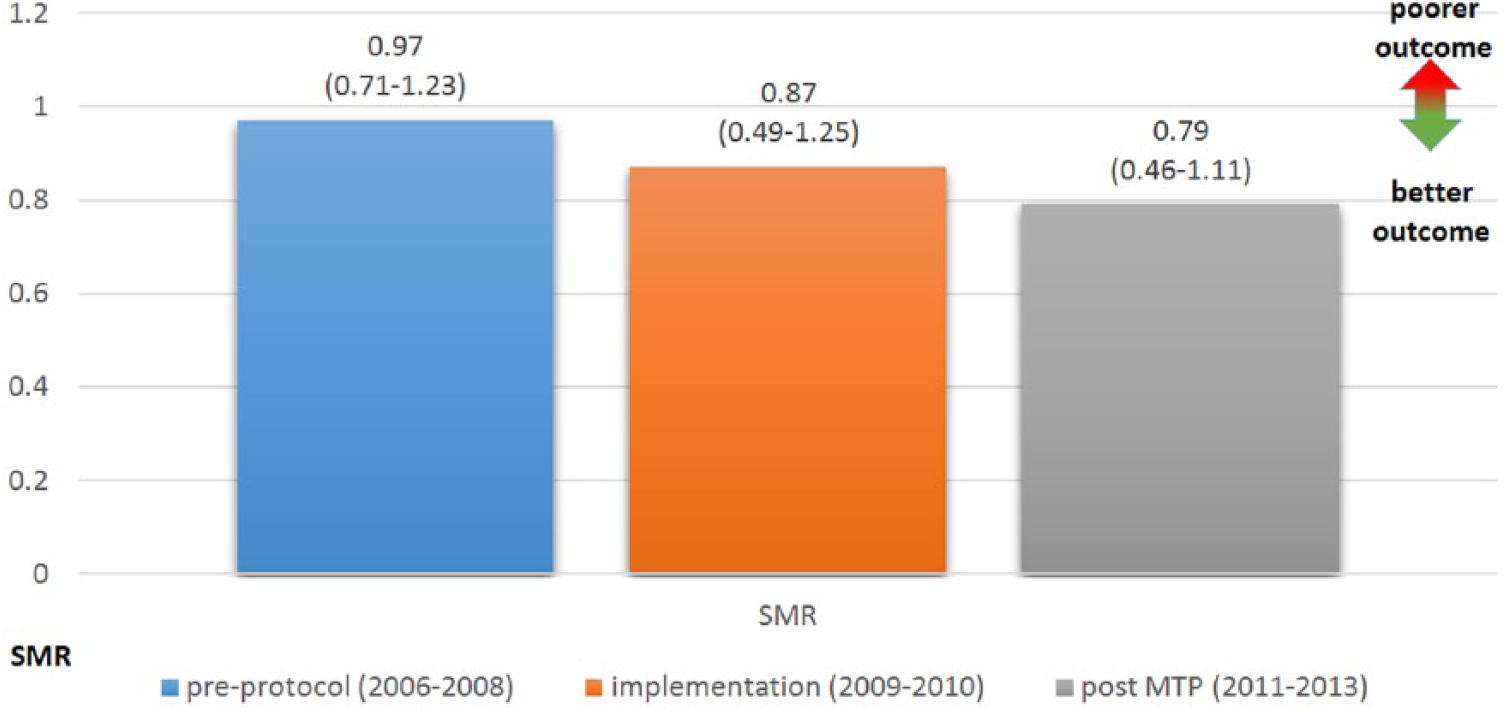
There was a decrease in three steps in the observed crude mortality (26.5% (39/147), 18.6% (16/86), and 15.7% (19/121), respectively). More importantly, the SMR values (the ratio between observed and expected mortality) also decreased in three steps, indicating better survival during the study period from 0.97 (pre-protocol), 0.87 (implementation), to 0.79 (post MTP) (Table 6).
Observed 30-day mortality, expected mortality based on the RISC II (Revised Injury Severity Classification) prognosis, and Standardized Mortality Ratio (SMR) with 95% confidence interval.
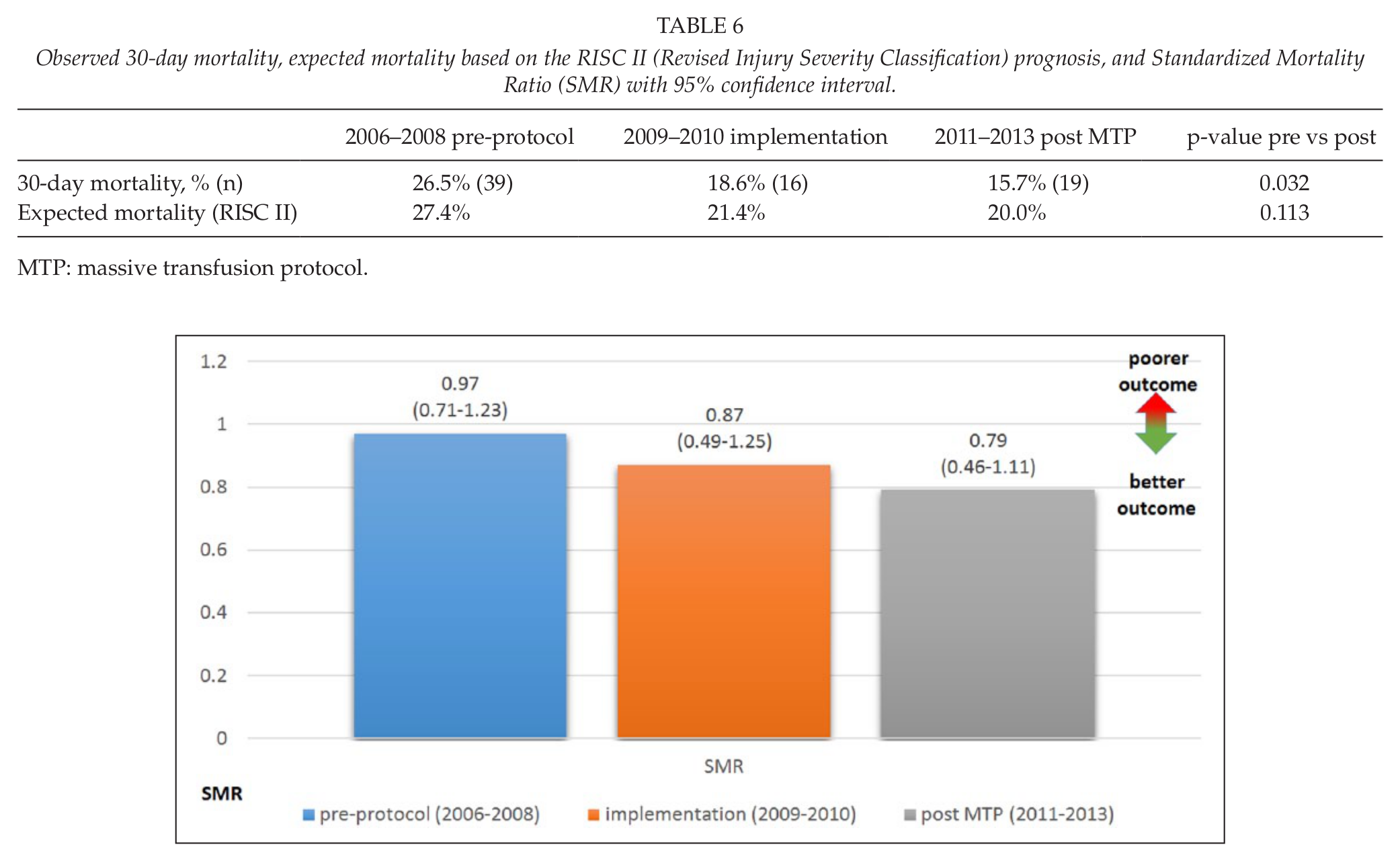
MTP: massive transfusion protocol.
Discussion
We reviewed the evolvement of fluid resuscitation and its effect on mortality at the single tertiary trauma center, and noted better survival of hemodynamically unstable patients along with the implementation of MTP. During the study period, blood products’ relation to crystalloids and FFP’s relation to PRBC increased and the volume of fluids administered prehospital decreased indicating adaptation of the principles of damage control fluid resuscitation in the treatment of bleeding in severely injured patients.
In the HU trauma center, the MTP was implemented in the end of 2009. However, the education and discussion concerning MTP had already started months before and this likely has influenced the decisions made in the fluid resuscitation of hemodynamically compromised patients before the official implementation of the MTP; this is why we decided to also cover the patients from the beginning of 2009 in the “implementation”-subgroup. It also allows for the full adaptation of a novel major implementation; thus we decided to consider the year 2010 as continued “implementation.”
The treatment of a severely injured patient is multifactorial. Thus, evolvements in all the fields of care, resources, and facilities have an impact. In the presenting center, no other major changes on the protocols in the treatment of severely injured patients emerged during the study period. Some changes, however, were made in the infrastructure of the emergency department: a new computed tomography (CT) scan with faster imaging capacity and easier access from the shock room was inaugurated in October 2008, and a new blood and fluid warmer with capacity for MT was deployed in January 2009 as a part of the MTP implementation. In addition, regular mortality and morbidity meetings on the treatment of severely injured patients were commenced in November 2008. These changes might also partly explain the better survival of the severely injured patients over the years.
Studies based on conventional fluid resuscitation guidelines investigating the effect of plasma:PRBC ratios on mortality have been recognized to involve survivorship bias to favor a positive outcome for high FFP:PRBC ratio. It has been considered that patients who died early never had the chance to receive sufficient FFP to match the number of PRBC units transfused (21). In our study, after implementing the MTP in the HU trauma center, already the first ETP includes four units of FFP and four units of PRBC (1:1 ratio) and, thus, the possibility of survivorship bias is reduced. Plasma administration has been associated with a substantial increase in complications, in particular Acute Respiratory Distress Syndrome (ARDS), with no improvement in survival in patients who do not require MT (<10 U PRBC within 12 h of admission) (22). This is why some trauma centers start FFP transfusion only after six PRBCs to make sure the patient really needs MT.
This study has several limitations. First, as in all registry studies, the quality of the data must be considered. In TR-THEL three dedicated and trained trauma register nurses collect and code the data from a single trauma center into a hospital registry, which could be considered to limit the amount of erroneous data input. Second, we were limited to using in-hospital mortality as the primary end point. The effect of the evolvement of fluid resuscitation in correcting the possible trauma-induced coagulopathy was not examined. In a recent international multicenter clinical study, it was noted that the DCR and MTP strategies applied in three trauma centers did not consistently correct coagulopathy during hemorrhage (23). Third, the included patients (with possible hemodynamic compromise) were defined as sBP < 90 mmHg on arrival and/or first measured BE < –5.0 mmol/L in the ER. While sBP of 90 mmHg is commonly used to define both hypotension and shock (9), it may well also be present without severe bleeding. Furthermore, though low BE is associated with marked bleeding (24), low values can also be present for example in the case of hypoventilation or heavy alcohol load in the blood. This is reflected in the percentage of the patients (around 50%) who were not given any PRBC in the ER despite the definition of “hemodynamic compromise” used in this study, that is, “shock” was due to some other reason than bleeding. Fourth, the evolution of DCR to include permissive hypotension may reflect in sBP on arrival. The amount of patients with a similar “bleeding profile” may arrive to the ER with lower sBP than before the adaptation of the concept of permissive hypotension in the prehospital system.
The fluid resuscitation of trauma patients with major hemorrhage has evolved over the years. The adaptation of DCR strategies and implementation of MTP have improved survival of these patients. Much of this improvement has been attributed to increased plasma and platelet to PRBC ratios (25, 26). In addition to this, the better results over the years in the HU trauma center might also partly be due to improved knowledge and skills in damage control surgery and angioembolization in the case of uncontrolled bleeding. However, the number of patients needing emergency operations or angioembolization to control massive bleeding decreased during the years. This might be due to a decreased number of less severe bleedings not manifesting into severe ones because of enhanced coagulation status. Another reason could be the lower absolute number of injuries leading to major hemorrhage. However, the coagulation status on arrival and the percentage of thoraco-abdominal or extremity AIS ≥ 2 injuries remained steady during the study period; only the percentage of patients sustaining pelvic injuries slightly decreased. This might indicate enhanced fluid resuscitation as the reason for diminished need for bleeding-control interventions.
We noted a relatively low number of trauma patients with major hemorrhage over 8 years. Thus, ongoing education and simulation concerning DCR strategies is needed in order to keep up and to further improve the results. Also, along with studies indicating improved survival with the use of antifibrinolytic medication (27 –30), tranexamic acid has become routine in prehospital treatment and will be added in the MTP of the HU trauma center. The future direction in the treatment of trauma patients with major hemorrhage is toward goal-directed hemostatic resuscitation with thromboelastometry to guide the transfusion. Thromboelastometry was implemented in 2014 in the treatment protocol in the HU trauma center—the effect in the outcome results will be reviewed. Monitoring the effectiveness of created guidelines or changes made in the treatment protocols is an essential part of quality control of modern hospitals treating trauma patients.
Conclusion
The overall awareness of damage control fluid resuscitation and introduction of MTP in a trauma center has a significant positive effect on the outcome of severely injured patients.
Footnotes
Acknowledgements
The authors wish to thank Kirsi Willa for technical assistance and Satu Tirkkonen, Pirkko Tonder and Markku Kytönen for data input.
Declaration of Conflicting Interests
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
