Abstract
Background
Definitions of massive transfusion following injury help identify patients at the greatest risk of death. However, these definitions primarily use blood component therapy. The use of whole blood (WB) transfusion protocols has seen a resurgence, with evidence of improved outcomes compared to component therapy. Therefore, our aim was to define and stratify patients into low, intermediate, and high risk for death based on volume of blood products transfused utilizing a WB-first resuscitation strategy.
Methods
Patients that received at least 1 unit of whole blood following injury between January 2016 and November 2021 were identified. Receiver operating characteristic (ROC) curves to predict death based on volume of blood products transfused were constructed. Patients were stratified to low, intermediate, and high risk of death based on positive likelihood ratios.
Results
There were 785 patients identified to have received at least 1 unit of WB following injury during the study period. Based on ROC curve analysis, the best predictor of death was volume of whole blood plus packed red blood cells (PRBC) in the first hour (AUC 0.66,
Discussion
The combination of WB + PRBC volume within the first hour following injury is the best predictor of death. Further, volumes of WB + PRBC transfused within the first hour can be used to stratify patients’ risk of death.
Level of Evidence
Level IV.
Study Type
Prognostic and Epidemiological.
Key Takeaways
This is the first study to have identified thresholds that differentiate patients at high, intermediate, and low risk of death based on a whole blood first approach to massive transfusion in trauma. Volume of whole blood plus packed red blood cells given in the first hour after arrival was most predictive of mortality. Patients that received greater than 5100 mL of combined product had the highest risk of death within 24 hours and 28 days.
Introduction
One of the most common causes of preventable death following trauma is uncontrolled hemorrhage.1-3 As such, significant research has focused on mitigating the deleterious effects of trauma-induced coagulopathy (TIC), 4 while also developing optimal resuscitation strategies to improve mortality and utilize fewer blood products.5-8 Much of this work has focused on resuscitation strategies using blood components; however, more recent work has evaluated the use of whole blood (WB) as the primary resuscitative blood product. This work has shown a survival benefit compared to component therapy.9-14 Furthermore, there has been recent work demonstrating an association between earlier WB resuscitation and increased survival in hemorrhagic shock.15,16
Massive transfusion protocols (MTPs) offer a proven benefit in resuscitation of patients in hemorrhagic shock. Traditional massive transfusion definitions have included the quantity of packed red blood cell (PRBC) units over a span of time to identify patients at greatest risk of death.17,18 However, these models of massive transfusion take several hours to define. The traditional definition of 10 U PRBC transfused within a 24-hour period is plagued with bias and limitations and is not a clinically relevant surrogate measurement for transfusion needs and early mortality from hemorrhage.19-22 Incorporating WB into MTP, specifically a higher whole blood ratio, is associated with a decreased probability of mortality in trauma patients. 23 Further, several rapid prediction models have been developed and evaluated to help identify patients at the highest risk of death from hemorrhage, as early as within the first hour.20,24-26 To date, only a few of these predictive models have stratified patients at variable risk of death based on volumes of blood products transfused, and none of them have utilized WB within their prediction model.20,24
Most definitions of massive transfusion and predictors of death following hemorrhage have been created using blood components. Currently, there is no accepted definition of massive transfusion incorporating WB as the first or primary resuscitative blood product. As the utilization of WB resuscitation protocols become more common, newer definitions of massive transfusion or risk of death from hemorrhage are needed to help identify and study patients at the greatest risk of death. Utilizing outdated massive transfusion definitions has not considered the rapid evolution and changes in transfusion practices over the past several years with increased use of WB and as a result, what constitutes massive transfusion in patients receiving WB remains undefined. Several models exist that incorporate earlier time intervals with certain transfusion thresholds and quantifying risk of death in support of newer massive transfusion definitions; however, much of this is based on blood component resuscitation. Therefore, our objective is to begin to re-define massive transfusion using a WB-first resuscitation protocol by correlating volumes of product transfused with risk of death.
Methods
This is a retrospective cohort study of a prospectively collected database. Data was collected in consecutive trauma patients that received WB as part of their resuscitation from January 2016 to November 2021 at Atrium Health Wake Forest Baptist Hospital, a tertiary referral center affiliated with Wake Forest School of Medicine. Prior to 2018, our institutional resuscitation practice was component based using a ratio of 1:1:1 (plasma:red blood cells: platelets); however, as plans to transition to a WB-first strategy evolved, patients prior to 2018 occasionally received WB dependent on supply availability. Starting in 2018, our institutional massive transfusion resuscitation protocol was formally transitioned to WB-first where patients >16 years old and females not of childbearing age would receive at least 2 units WB prior to transitioning to component therapy. Our protocol started including women of childbearing age in 2022; however, this was after our data collection period. All whole blood is leukocyte reduced low-titer (1:200) type “O.” Our lab uses a 26-day shelf life, but to prevent waste, whole blood is converted to components on prior to expiration if it remains unused. The average volume of blood components at our institution is as follows: Whole blood is 500 mL, PRBC is 300 mL, plasma is 300 mL, an apheresis pack of platelets is 325 mL, and a 5-unit bag of cryoprecipitate is 90 mL. This study was approved by the institutional review board (IRB) #00058468.
Clinical data was abstracted from the trauma registry and blood bank registry. Demographic and physiologic data collected included age, sex, Glasgow Coma Scale (GCS) on arrival, systolic blood pressure (SBP) in the emergency department, heart rate (HR) in the emergency department, mechanism of injury, traumatic brain injury (TBI), Injury Severity Score (ISS), and volume of blood products administered at 1, 6, and 24 hours from time of arrival. Comparisons between patients with and without severe TBI (sTBI) were also performed.
As there are differences between patients with and without sTBI, ROC curves were constructed for volumes of blood product transfused, considering the presence or absence of sTBI.10,24,27 Severe TBI was defined as a Head Abbreviated Injury Scale (AIS) greater than or equal to 3. Several models were tested to determine the best predictor of early mortality and were evaluated with and without the presence of sTBI to avoid confounder from death due to traumatic brain injury. Receiver operating characteristic (ROC) curves to predict death based on volume of blood products transfused (within 1, 6, and 24 h from time of arrival) were constructed. Interval likelihood ratios are defined as the slope of the ROC curve between any 2 points on the ROC curve (points A and B) 28 and calculated as (Sensitivity of Point A – Sensitivity of Point B)/(Specificity of Point B -Specificity of Point A). The positive likelihood ratio (+LHR) in this setting modifies the pretest probability of death, and a + LHR >1 increases the post-test probability of death, with a greater + LHR increasing the post-test probability. 29 We used the +LHR to stratify patients into groups. A high risk of death was defined as the lowest volume of least number of the combination of WB + PRBC volume that corresponded to a + LHR for death of >10x.24,29 An intermediate zone was defined a + LHR for death of 5x (or as close to 5x as could be obtained). The low-risk group was the cohort receiving fewer WB + PRBC volumes than the intermediate risk group.
IBM SPSS Statistics version 27 (IBM; Armonk, NY) and GraphPad Prism version 10.0.2 (GraphPad Software, Inc; La Jolla, CA) were used for statistical analysis. Data is presented as calculated medians with interquartile ranges. All tests were two-tailed with significance set at
Results
Characteristics of all Trauma Patients and Comparison of all Patients With and Without Severe TBI Who Received Whole Blood as Part of Their Resuscitation. SBP = Systolic Blood Pressure; HR = Heart Rate; GCS = Glasgow Coma Scale; TBI = Traumatic Brain Injury; INR = International Normalized Ratio; NISS = New Injury Severity Score; ICU = Intensive Care Unit; LOS = Length of Stay
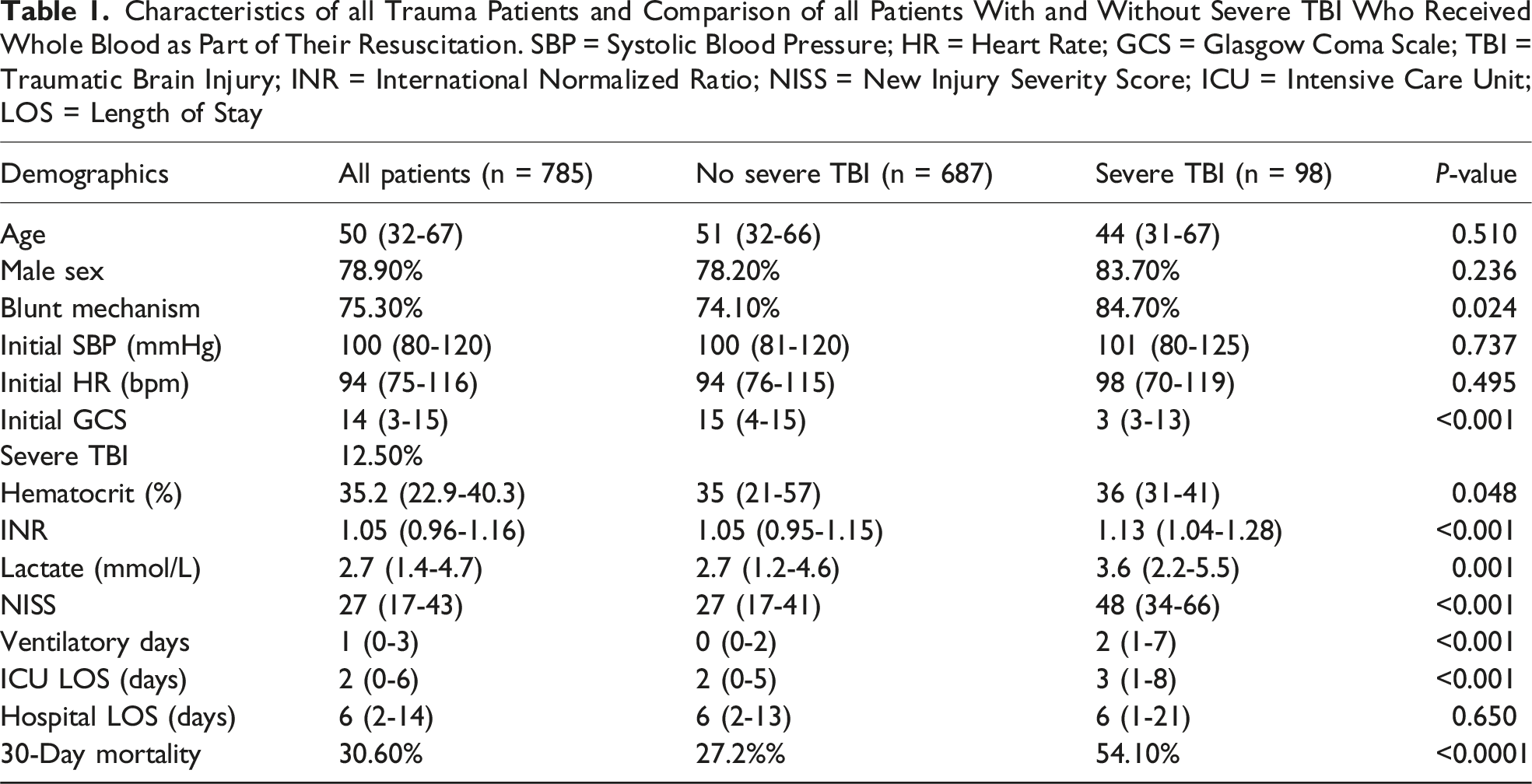
Volume of Blood Products Received at Set Time Intervals Among all Patients as Well as Those With and Without a Severe TBI
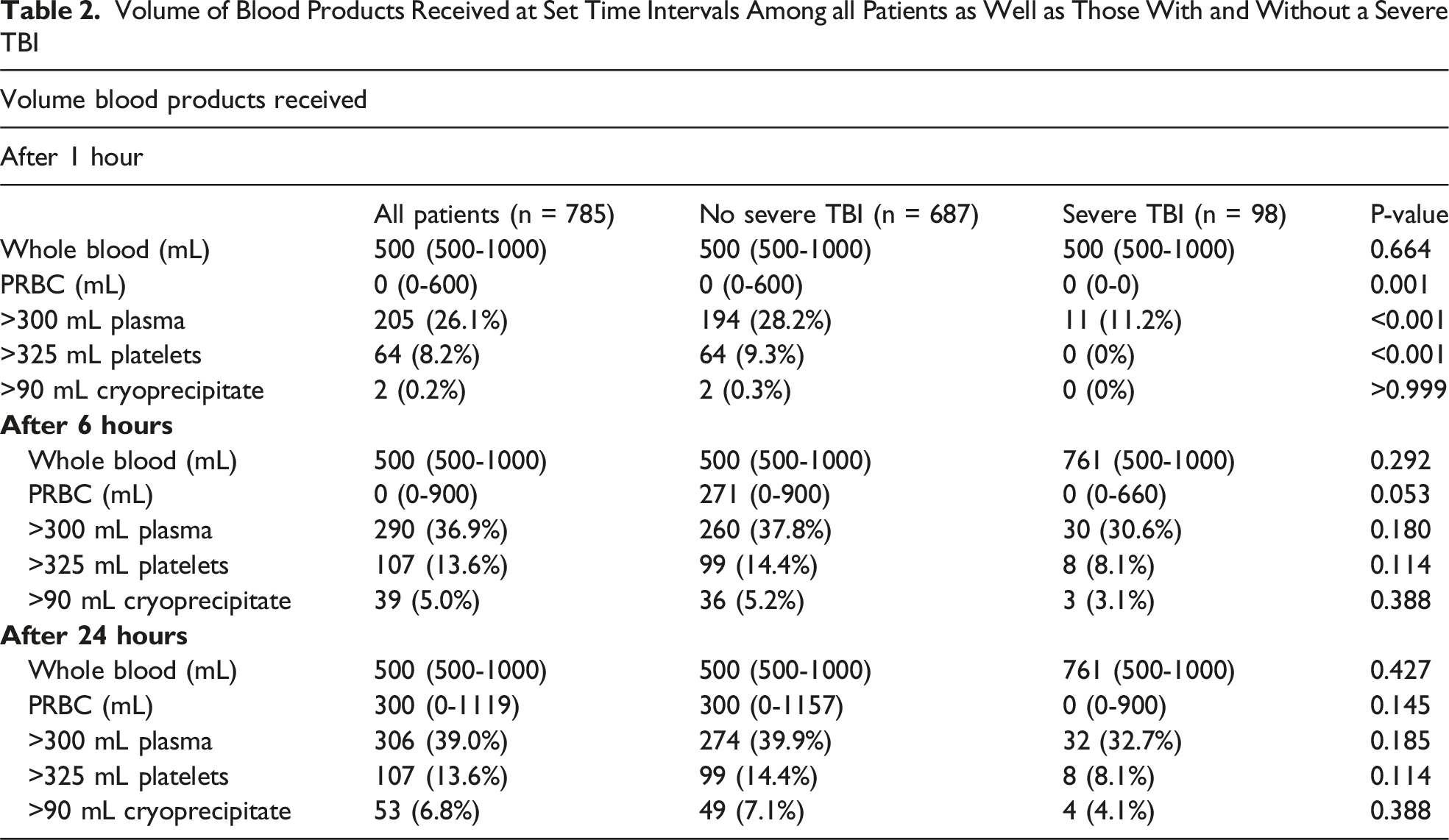
Area Under the Receiver Operating Curve (AUROC) for Various Blood Volume Transfusions Within Hours 1, 6, and 24 Evaluating a Primary Outcome of 24-Hour Mortality. Area Under the Curve (AUC) was Determined for Patients With Inclusion and Exclusion of Traumatic Brain Injury (TBI)
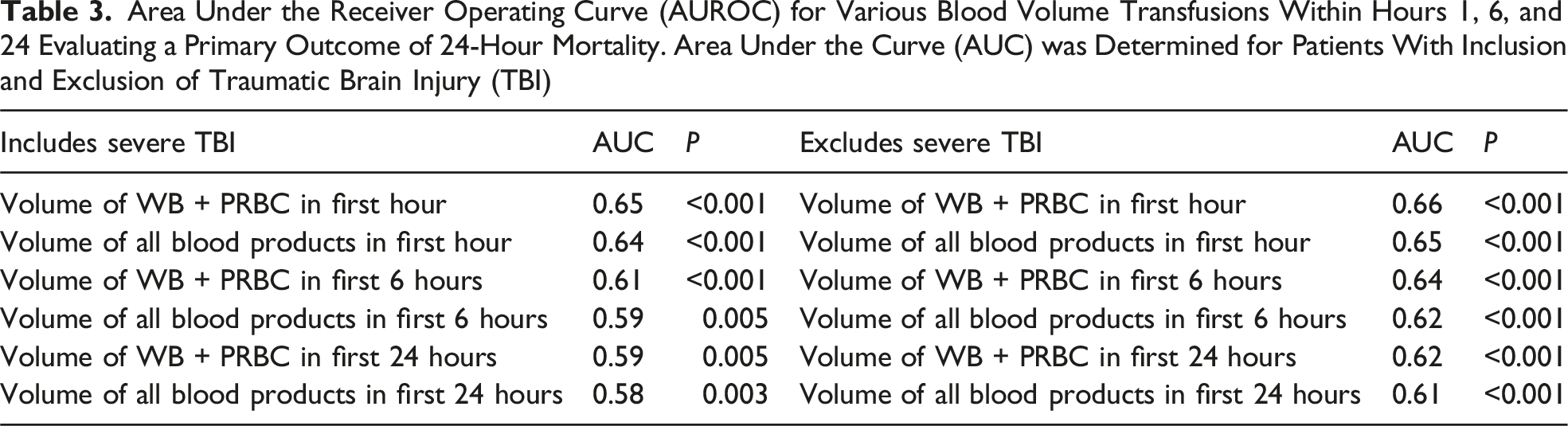
Definition of Low, Intermediate, and High Risk of Death Groups With Positive Likelihood Ratios (Pos LHR) and Mortality, Excluding TBI Patients
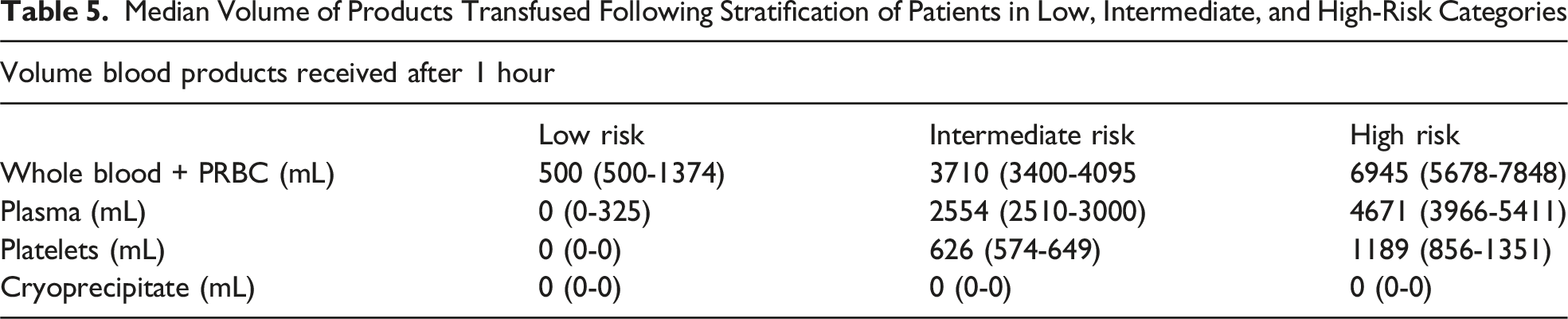
Median Volume of Products Transfused Following Stratification of Patients in Low, Intermediate, and High-Risk Categories
Discussion
In this study, patients were stratified based on volume of WB and PRBC transfused at different time intervals to identify patients at high, intermediate, and low risk of death. These thresholds provide the ability to identify patients at greatest risk of death within the first hour of presentation in order to help define MTP utilizing an upfront WB resuscitation protocol. Further analysis of these thresholds was used to determine 28-day mortality (Table 4). Massive transfusion following traumatic injury carries a high mortality rate and has been demonstrated within the literature from 60 to 80%.24,26 Previous work has largely evaluated the use of individual component therapy in defining massive transfusion. Incorporating a patient’s risk of death based on volume of product transfused can help further quantify what the meaning of massive transfusion is with whole blood resuscitation strategies. The identification of such thresholds has shown utility in predicting risk of death and may be utilized as tools to help inform care decisions, guide goals of care discussions, as well as provide a definition for future research endeavors.
Goal-directed therapy and ratio-based transfusion protocols have led to improvements in patient outcomes following trauma.5,7,8,18 These have primarily focused on transfusion or resuscitation protocols based on individual blood component therapy. However, there has been a resurgence in the use of WB over the last several years that first began with utilization of WB during the conflicts in Iraq and Afghanistan.30-33 Further, there are several differences in the military vs civilian application of WB, primarily the fact that the military use of WB may consist of fresh warm WB from a “walking blood bank” compared to cold-stored WB in the civilian sector.33,34 Recently, several civilian trauma centers have evaluated outcomes in patients who received WB instead of strict blood component therapy with positive outcomes.9-16,23
Prior to recent studies illustrating the benefits of WB, massive transfusion was primarily goal-directed or ratio-based.5-8,18 Using blood components, several models have been developed for early prediction and quantifying risk of death from hemorrhage.20,24-26 These models have effectively replaced historic massive transfusion definitions, as these historic definitions take several hours to define and are often plagued with bias.5,16,19-22 In 2008, the Denver group re-defined massive transfusion as >10 units of PRBC or death within the first 6 hours. 18 This definition removed the limitation of survivor bias. 18 Other, more recent, studies have evaluated risk of death over a shorter time. Nunns et al stratified risk of death from hemorrhage into low, intermediate, and high risk (massive transfusion) based on one-hour PRBC transfusion requirements. 24 They found a transfusion of >4 units of PRBC within the first hour was associated with a high need for concomitant platelet and cryoprecipitate transfusion as well as a 51% mortality. 24 Redefining MTP within the one-hour time frame with the use of WB may prove beneficial for resource allocation and identifying patients earlier who are at a high risk of death.
Two other models have been developed to identify patients at risk of death following injury, the Resuscitation Intensity (RI) and Critical Administration Threshold (CAT). The RI was defined as total products in the first 30 minutes (PRBC, plasma, and crystalloid) while the CAT score was positive if the patient received a ≥3 U RBC during any one-hour period in the first 24 hours arrival.25,26 Both RI and CAT are more applicable than traditional definitions of massive transfusion at predicting risk for early mortality. 25 The limitation of all these models is that they define risk of death using blood components therapy only and were not developed using WB resuscitation protocols. Resuscitation strategies have rapidly evolved with utilization of WB and even these newer prediction models may be outdated as they do not consider WB in their definition. Further, a recent evaluation of massive transfusion in patients that received WB defined a WB massive transfusion score as a predictor of poor outcomes. 31 To our knowledge, our model, which identifies low, intermediate, and high risk of mortality, is the first to stratify risk of death using transfused WB volumes.
Several models were tested to determine the best predictor of early mortality (Table 3). Further, these models were evaluated with and without the presence of sTBI, given that patients with sTBI had a significantly higher risk of mortality but also received less blood products (Table 1). Removal of TBI from the model allowed for a small improvement in the performance with an AUROC to 0.66. AUROC curves were also developed for total volumes of all blood products, including platelets, plasma, and cryoprecipitate. However, the performance of volume of WB + PRBC was more predictive of mortality compared with other components. We were able to define patients at the lowest risk of death as those that received less than 3.4 L of WB + PRBC within the first hour. The patients with high risk of death were those that received more than 5.1 L of WB + PRBC.
The retrospective nature of this study is a limitation, and as such, data is only as accurate as the trauma registry and blood bank collection. Prospective analysis is needed to validate our definitions to a larger population. Transfusion practices across institutions differ, and these definitions may not apply to facilities without access to a WB-first approach. Similarly, while some emergency medical services began to have the capability of providing pre-hospital blood products, we were unable to collect this data to include in this study but it remains an area of future study. Unfortunately, changes in the blood bank collection software have prevented us from accessing blood product utilization data prior to 2016. Similarly, as the blood bank collection software transitioned as well as migration of our medical system, this has led challenges in acquiring and capturing data from more recent patients. During the study period, transfusion practices also began to evolve at our institution, whereas women of childbearing age were initially excluded from receiving WB, in 2022, our resuscitation practice included this patient population; however, this was after our data collection period. This may introduce the possibility of selection bias within our study population. Within the study population, all patients receiving WB were included, which introduces a risk of survivorship bias. Furthermore, the AUC for predicting death was only 0.66 and this is far from an excellent predictor of mortality. However, as the use of WB becomes more common, it is an important starting point for further research on MTP in the WB era.
In conclusion, this is the first study to have identified thresholds that differentiate patients at high, intermediate, and low risk of death based on a WB-first approach to MTP. These thresholds provide the ability to identify patients at greatest risk of death within the first hour of presentation and further define what massive transfusion means in the WB era of resuscitation. Further research is needed to validate these thresholds over a broad range of facilities that implement a WB-based resuscitation protocol for injured patients.
Footnotes
Author Contributions
Literature search: JC, AMN, and GRS
Study design: JC, AMN, HC, CG, MAM, NM, JH, and GRS
Data collection: AMN and GRS
Data analysis: JC, AMN, and GRS
Data interpretation: JC, AMN, and GRS
Writing: JC, AMN, and GRS
Critical revision: JC, AMN, MA, HC, CG, MAM, NM, JH, and GRS.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
