Abstract
Background and Aims:
Curative resection is the treatment of choice for gastric cancer. Although it has been concluded that D2 lymphadenectomy plus para-aortic nodal dissection does not improve survival rate in curable gastric cancer, it is unclear whether D2 plus para-aortic nodal dissection has a benefit in some groups of patients. We conducted a retrospective study in our hospital, in which we compared D2 with D2 plus para-aortic nodal dissection lymphadenectomy for gastric cancer in subgroups of each clinical characteristic in terms of long-term survival after surgery.
Material and Methods:
We selected 1792 patients who had undergone the treatment with curative intent between 1990 and 2007, 1344 in the D2 group and 448 in the D2 plus para-aortic nodal dissection group. Each procedure was verified by pathological analyses. The primary end points were 5-year overall survival.
Results and Conclusions:
Median follow-up periods were 50 months for patients assigned to D2 group and 54 months for patients assigned to D2 plus para-aortic nodal dissection group. Overall 5-year survival was not significantly higher in patients assigned to D2 plus para-aortic nodal dissection surgery compared to those assigned to D2 surgery (31.2% (95% confidence interval: 19.8%–42.6%) vs 26.6% (95% confidence interval: 20.3%–32.9%); log-rank p = 0.433). D2 plus para-aortic nodal dissection surgery should only be used for curable gastric cancer of T3-4 and N2 stage and should not be used for T1 disease and total gastrectomy.
Introduction
Gastric cancer is still the most common cause of cancer-related deaths worldwide and is a major clinical problem because of the poor prognosis and the lack of treatment methods. Nowadays, surgical resection is the major treatment method for gastric cancer; however, the efficacy of nodal dissection is still under debate. The more extended the surgery, the greater the risk of operation-related morbidity and mortality; nodal dissection increases morbidity, as has been previously reported (1). It has been reported that the mortality for gastrectomy surgery in the West often exceeds 5%, and sometimes, even gets close to 16% in some studies (2 –4); only Japanese studies have reported a lower than 2% mortality rate (5). Besides the morbidity being higher or lower, there are also reports showing that lymph-node dissection did not adversely influence quality of life (QOL) (6) and that operation-related morbidity did not influence survival (7, 8).
Because the 5-year overall survival rate of patients with para-aortic nodal metastases can be as high as 20% after systematic dissection (9), extensive surgery has been performed in Japan since the 1980s for stage T2b, T3, and T4 gastric cancers. In this article, we conducted a single-institutional study and report the long-term survival data for the two surgical groups of D2 and D2 plus para-aortic nodal dissection (PAND), and we compared the subgroups of each clinical characteristic in terms of long-term survival after surgery. We demonstrated that even without significantly more operative complications and mortality, D2 plus PAND surgery does not have an overall survival benefit greater than that of D2 surgery except for those with stage T3-4 and N2 disease, who are more suitable for D2 plus PAND surgery.
Methods
Patients
We selected 1792 histologically confirmed gastric cancer patients who underwent a radical operation at the Fourth Affiliated Hospital of the Harbin Medical University, China, between 1990 and 2007. Of these patients, 1344 were allocated to D2 group and 448 to D2 plus PAND group. The inclusion criteria were as follows: (1) patients with histologically proven, potentially curable gastric adenocarcinoma and physical fitness suitable for elective operation of either type of lymphadenectomy; (2) patients with curative D2 or D2 plus PAND operations were performed; (3) patients for whom a complete medical record was available; (4) patients of every period of diagnosis and every surgeon are roughly equal; and (5) patients who never received neoadjunctive therapies or any kind of adjunctive therapy. After confirming the inclusion criteria of a patient, the surgeon contacted the research group to receive a randomly generated assignment of the patient to standard D2 lymphadenectomy alone or D2 lymphadenectomy plus PAND. The surgeon then performed the assigned operation.
All patients were followed up by letter or telephone interviews. The last follow-up was December 2012. Clinical findings, surgical findings, pathological findings, and every follow-up were collected and recorded in the database. The study protocol was approved by the Ethics Committee of Harbin Medical University.
Surgical Procedures and Classifications of Gastric Cancer
Surgery was performed at the Fourth Affiliated Hospital of the Harbin Medical University. Surgical procedures and pathological assessment were standardized in accordance with the Japanese Classification of Gastric Carcinoma, first English edition (9). All patients in the study underwent standard total or distal subtotal gastrectomy, depending on the location and macroscopic appearance of the primary tumor. The definition for lymphadenectomy was based on the Japanese Classification of Gastric Carcinoma, First English Edition (10).
Surgeons routinely removed lymph nodes from the excised specimens as soon as possible after operation. The specimens and retrieved lymph nodes were stained with hematoxylin and eosin and pathologically examined in the Gastric Laboratory of the Fourth Affiliated Hospital of the Harbin Medical University.
End Points and Follow-Up
The primary end points were 5-year overall survival. Overall survival was calculated from the day of surgery until death or the last follow-up contact. Data of patients were censored at last follow-up when they were alive. Follow-up assessments were done every 6 months for the first 5 years after surgery, and then every 12 months until the patient’s death.
Statistical Analyses
Data from all eligible patients were analyzed for overall survival. Survival curves were estimated by the Kaplan–Meier method, and treatment comparisons were made by use of the log-rank test.
Potential prognostic factors were entered into a Cox’s regression model. For univariate analyses, we put the prognostic factor of interest and treatment group as covariates in the Cox regression model.
Analyses of subgroups were also conducted to evaluate interactions between treatment and subgroup with the use of Cox regression; we report the results of all these analyses.
Two-sided p values were calculated for all tests and are reported here. A p value of less than 0.05 was considered to indicate statistical significance. Analyses were performed with the use of SPSS software, version 16.0.
Results
From 1990 to 2007, 1792 patients were enrolled: 1344 patients with median age of 60 years were allocated to D2 surgery, and 448 patients with median age of 63 were allocated to D2 plus PAND surgery (Table 1). All patients were followed up for at least 5 years (until 19 December 2012).
Characteristics of D2 and D2 plus PAND population (n = 1792).
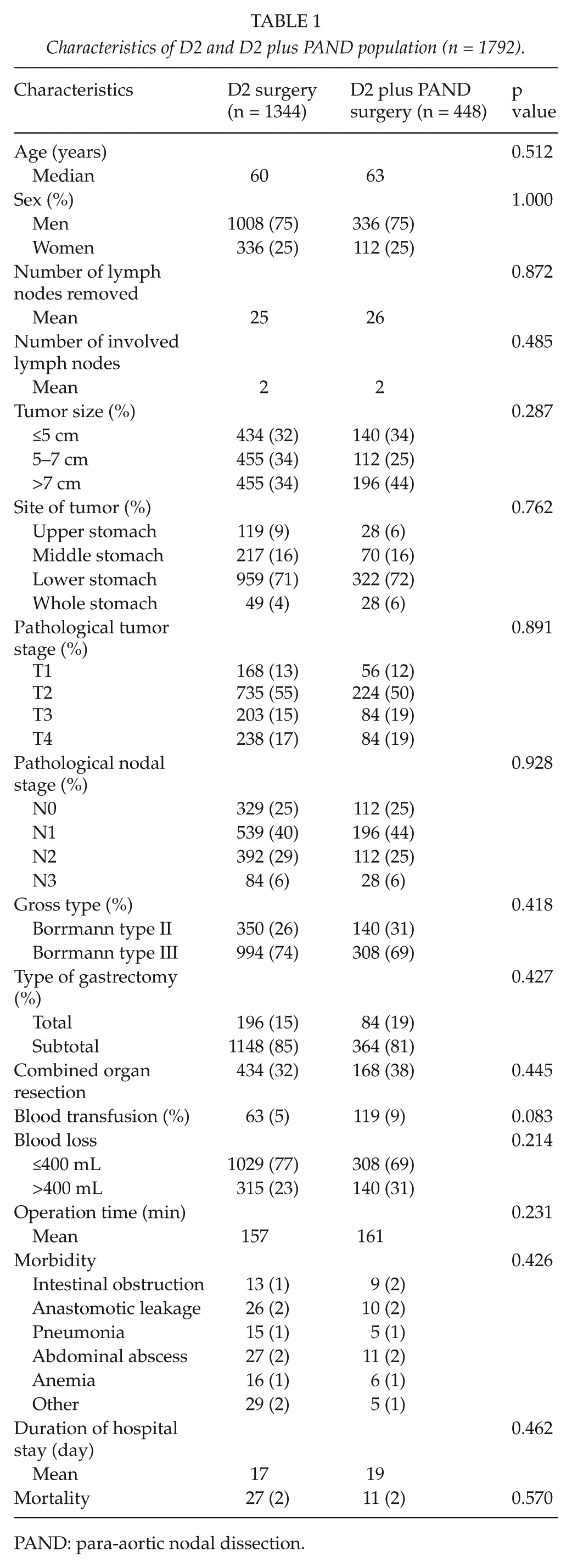
PAND: para-aortic nodal dissection.
The characteristics of the two groups, which are shown in Table 1, were well balanced. A total of 224 patients had early cancer (confined to submucosa or mucosa). All the patients had a curative resection. Total gastrectomy was performed in 196 patients assigned to D2 group (15%) and in 84 patients assigned to D2 plus PAND surgery (19%). Blood loss and blood transfusions were not more frequent (p = 0.241 and 0.083, respectively) in patients undergoing D2 plus PAND surgery than in those undergoing D2 surgery.
After median follow-up periods of 50 months for patients assigned to D2 group and 54 months for patients assigned to D2 plus PAND group, 1113 patients of D2 group and 364 patients of D2 plus PAND group had died. Neither the skill of the individual surgeon nor the period of diagnosis affected survival (p > 0.05, log-rank test). Fig. 1 shows the overall rates for all enrolled patients. There was no significant difference between D2 and D2 plus PAND group. Fig. 2 shows the 5-year survival rates for all enrolled patients. The 5-year survival was 26.6% (95% confidence interval (CI): 20.3%–32.9%) for the D2 group and 31.2% (95% CI: 19.8%–42.6%) for the D2 plus PAND group (log-rank p = 0.433).
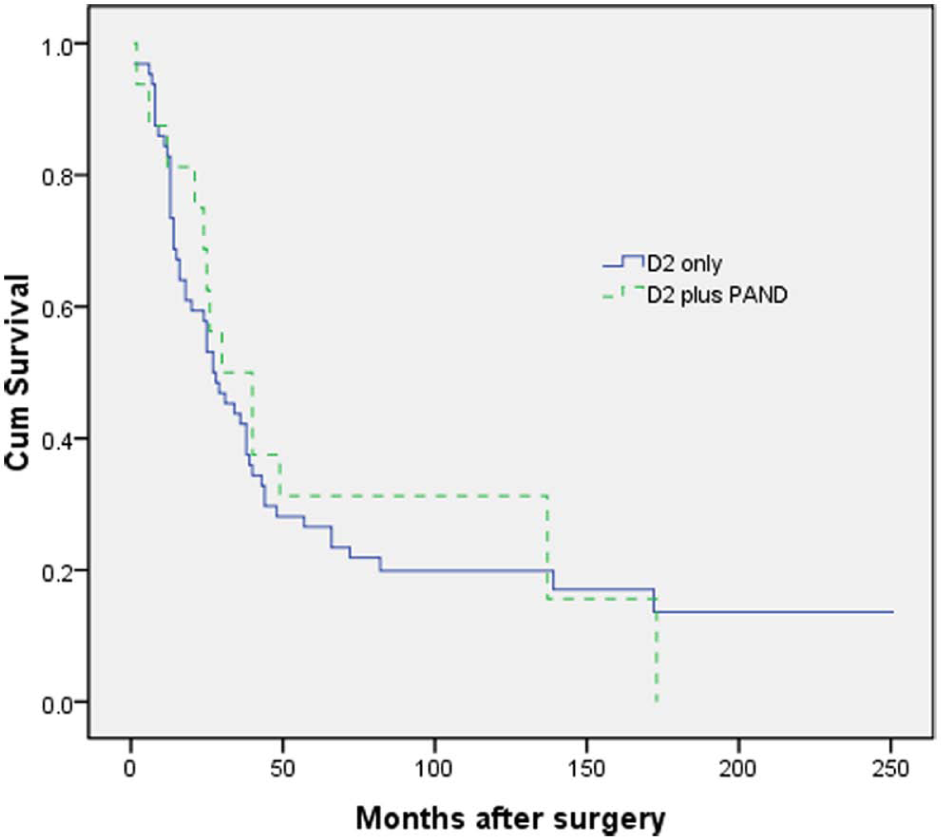
The overall survival rates for all enrolled patients.
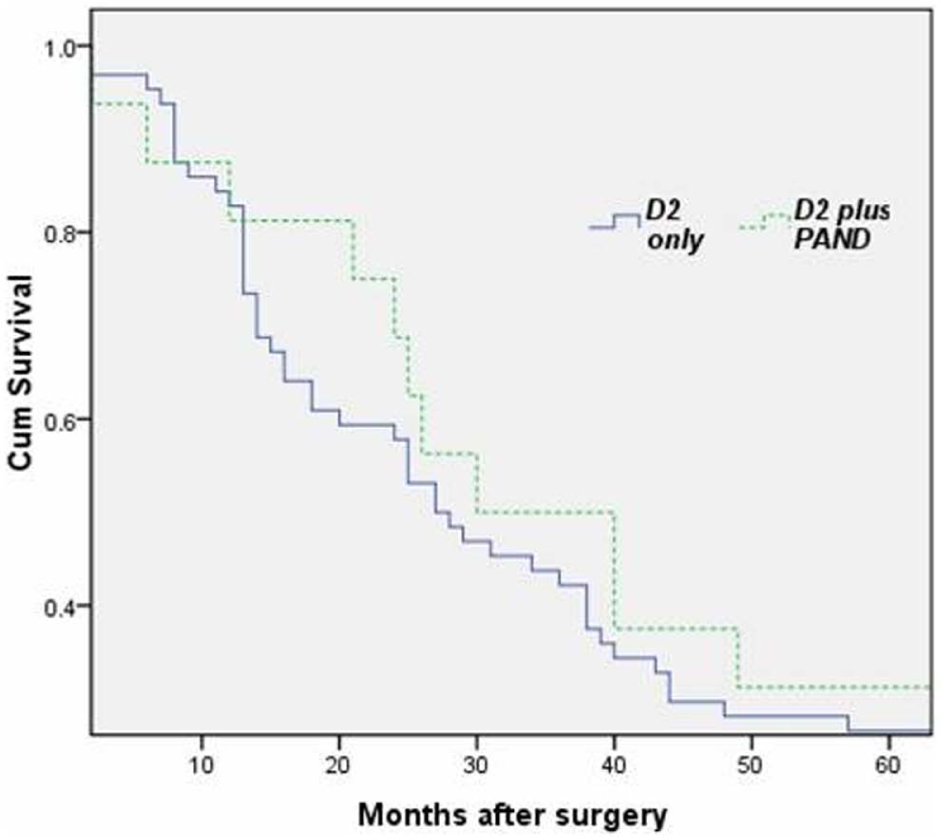
The 5-year survival rates for all enrolled patients.
The hazard ratio for death was 0.885 (95% CI: 0.647–1.211; p = 0.445) in the D2 plus PAND group (Table 2, univariable analyses). After adjustment for 11 baseline variables (age, sex, tumor size, tumor location, Borrmann type, T stage, lymph-node stage, joint organ removal, gastrectomy, blood transfusion, and blood loss) with the use of Cox regression analysis, the hazard ratio was hardly changed (hazard ratio: 0.719 (95% CI: 0.503–1.028); p = 0.070) (Table 2, multivariable analyses). As expected, the multivariate analyses showed that whole stomach tumor, T4 stage, and N1-3 disease was significantly associated with poor survival (Table 2).
HR for death in intention-to-treat population (n = 1792)—univariable and multivariable analyses.
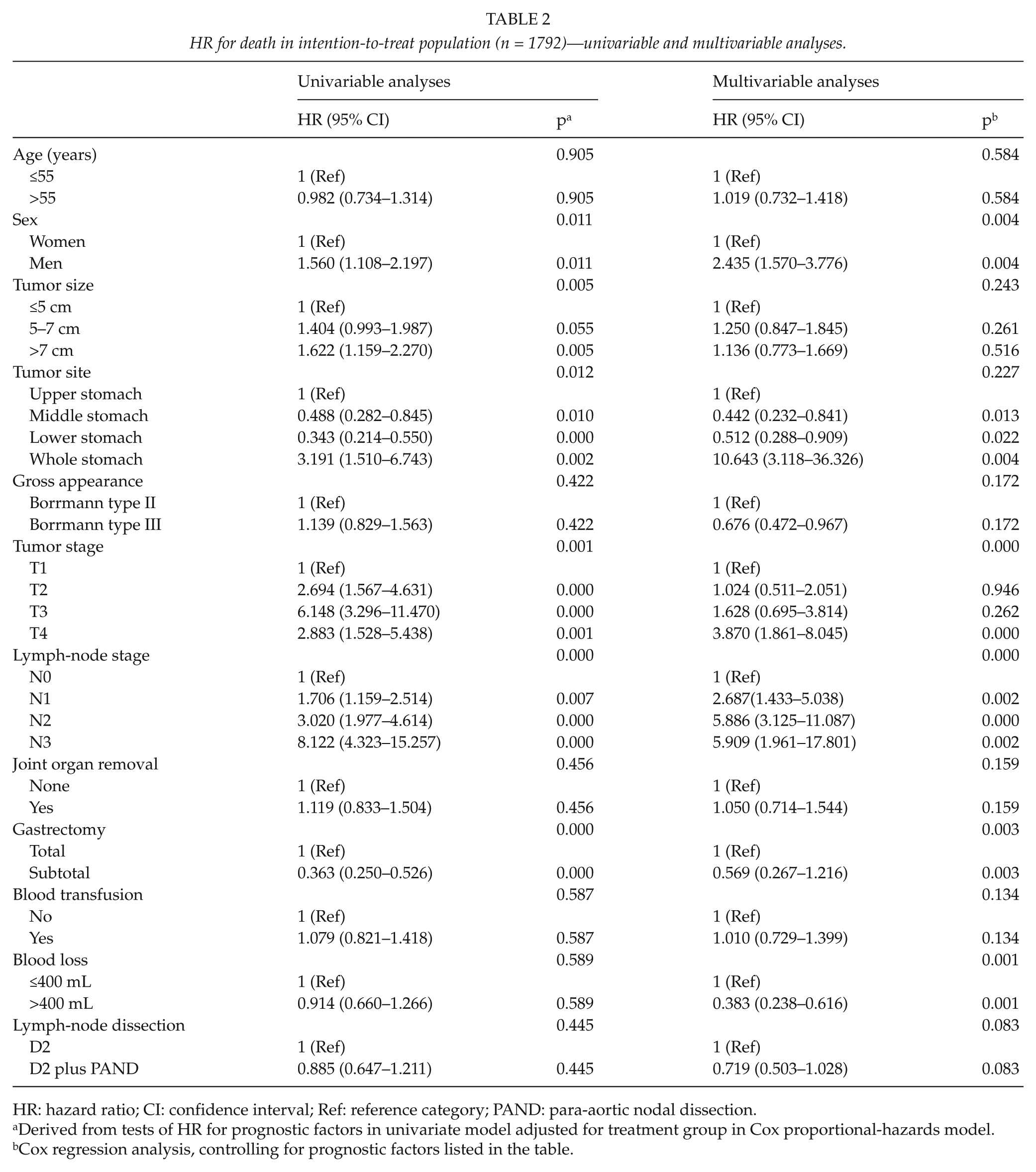
HR: hazard ratio; CI: confidence interval; Ref: reference category; PAND: para-aortic nodal dissection.
Derived from tests of HR for prognostic factors in univariate model adjusted for treatment group in Cox proportional-hazards model.
Cox regression analysis, controlling for prognostic factors listed in the table.
D2 surgery has significantly more benefit than D2 plus PAND surgery only for the total gastrectomy and T1 disease; the hazard ratios for death in the D2 group were 0.407 (95% CI: 0.195–0.850; p = 0.017) and 0.083 (95% CI: 0.022–0.315; p = 0.000), respectively. This result indicated that D2 plus PAND lymphadenectomy should not be used to treat curable gastric cancer in these two subgroups (Table 3).
Tests for heterogeneity of treatment effect according to the clinicopathological characteristics of the D2 and D2 plus PAND patients.
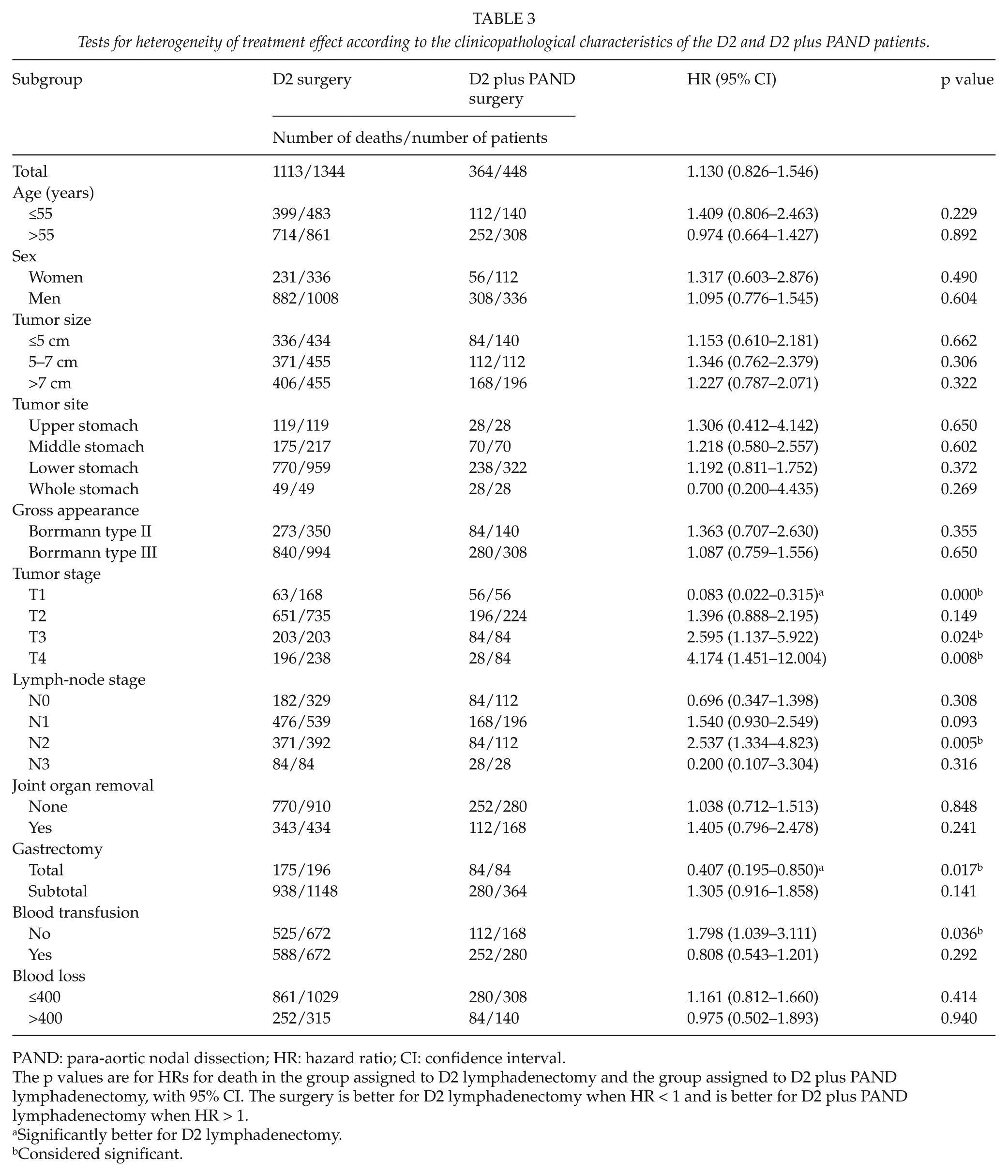
PAND: para-aortic nodal dissection; HR: hazard ratio; CI: confidence interval.
The p values are for HRs for death in the group assigned to D2 lymphadenectomy and the group assigned to D2 plus PAND lymphadenectomy, with 95% CI. The surgery is better for D2 lymphadenectomy when HR < 1 and is better for D2 plus PAND lymphadenectomy when HR > 1.
Significantly better for D2 lymphadenectomy.
Considered significant.
D2 plus PAND surgery has significantly more benefit than D2 surgery in the subgroups of T3-4 stage, N2 disease, and no blood transfusion (Table 3).
Discussion
In this trial, we found no significant improvement in overall survival with D2 plus PAND surgery as compared to D2 surgery with no significant differences between the two groups in blood loss, except for those patients with T3-4 and N2 disease, who are more suitable for D2 plus PAND surgery. This result is similar to the Japan Clinical Oncology Group (JCOG) study 9501 (11). In this randomized trial, the researchers found no improvement in overall or recurrence-free survival with D2 lymphadenectomy plus PAND gastrectomy as compared to D2 lymphadenectomy alone. The pattern of recurrence was similar in the two groups, and D2 lymphadenectomy plus PAND did not reduce the rate of the recurrence of cancer in the lymph nodes. There were no significant differences between the two groups in the rates of surgery-related complications. D2 lymphadenectomy plus PAND, however, was associated with a longer operation time, greater blood loss, and a significant increase in complications. For all these reasons, the authors did not recommend D2 lymphadenectomy plus PAND for curable gastric cancer patients. Here, we also reported the surgery-related complications, operation time, and blood loss; however, as we did not have the complete data on recurrence, we could not compare the recurrence-free survival between these two groups of patients. Because of the limitations of retrospective analysis, the possible survival benefit of D2 plus PAND surgery in T3-4 and N2 patients will need to be clarified in further studies.
In this trial, the 5-year overall survival rate was 26.6% for the D2 group and 31.2% for the D2 plus PAND group, and the period of diagnosis did not affect survival (log-rank p > 0.05), which was similar to a previous study (12). It can be seen that long-term survival is lower, when comparing our results with an historical report, in which the observed 5-year survival rates were 53.6% and 59.5%, respectively (8). This is likely to be due to the high proportions of T2-4 (88%) and N1-3 (75%) tumors, which have a relatively poor prognosis. This result indicated that the time of diagnosis of malignant tumors as well as gastric cancer is much later in China than in other countries, especially Western countries. At the same time, in our trial, adjuvant therapy was not used in both groups, and this may also explain the different survival rates between our trial and previous results.
In this study, we found that more than 70% of both the D2 and D2 plus PAND patients had lymph-node metastases. The most frequent lymph-node metastases were in the N1 level, followed by the N2 level, and then the N3 level. Our observation was consistent with the result that was previously reported by Maruyama et al. (13).
Extended surgery is thought to be closely related to the risk of operative morbidity and mortality, as has been reported with respect to nodal dissection increasing morbidity (1). The reported mortality for gastrectomy in Western countries is often 5% and even approaches 16% in some trials (2–4); similar rates were also reported in Hong Kong (47%) (14), Dutch (43%) (15), and UK trials (46%) (16); however, only Japanese studies consistently reported a mortality rate lower than 2% (5). However, whether the morbidity rate is higher or lower, it has been reported that lymph-node dissection does not adversely influence QOL (6), and operative morbidity does not influence survival (7, 8).
A previous study reported that the overall 5-year survival rate for 286 patients undergoing gastrectomy with additional organ resection was 32%, which was significantly less than the gastrectomy-alone group (17). In our trial, disregarding the type of surgery, the overall 5-year survival rate for the patients with additional organ resection was 29%; however, there was no significant difference whether or not an additional organ resection was performed.
In addition to the extent of surgery, the participating surgeons’ operative skill and experience and the workload cases are also important factors for survival rates (18, 19). There are many studies which have reported a relationship between the number of cases treated in a hospital and the outcomes of cancer treatment (19–24). Moreover, the uniformity of treatment is also important and is an advantage for single-center trials. Our study was carried out in a hospital that performs a high volume of nodal dissections for gastric cancer with low morbidity and mortality rates. The procedures in our study were performed by five experienced surgeons with a high volume of at least 300 cases. In our study, all participating surgeons were from the same department, which minimizes the variation in individual operating skill and management, and they did an equal number of D2 and D2 plus PAND resections during the trial, which balances the comparisons between the two groups avoiding the bias of the individual surgeon’s skill. Therefore, the factors of caseload, surgical skill, and case selection are very important (25–27).
As D2 gastrectomy is associated with low mortality and adequate survival times when performed in selected institutions that have had sufficient experience with the operation and with postoperative management, we recommend D2 lymphadenectomy for patients with curable gastric cancer except for those with stage T3-4 and N2 disease, who are more suitable for D2 plus PAND surgery.
Footnotes
Declaration of Conflicting Interests
The authors declare that there is no conflict of interest.
Authors’ Contributions
Yan Zhang had the original idea and designed the study. Yan Zhang and Suli Tian were participating surgeons, reviewed the operation reports, and contributed follow-up data. Yan Zhang performed the statistical analysis, designed the study, analyzed data, and reviewed the article. Suli Tian supervised the trial progress and modified the article.
