Abstract
Background and Aims:
We analyzed the feasibility of cytoreductive surgery and hyperthermic intraperitoneal chemotherapy in patients with pseudomyxoma peritonei.
Material and Methods:
A prospective database comprised 90 consecutive patients with demonstrable pseudomyxoma peritonei collected during 48 months. These patients, referred to our unit for consideration for cytoreductive surgery and hyperthermic intraperitoneal chemotherapy, received both cytoreductive surgery and hyperthermic intraperitoneal chemotherapy if possible. We evaluated the factors associated with a successful procedure.
Results:
Hyperthermic intraperitoneal chemotherapy was successfully delivered to 56 of 90 patients (62%) with demonstrable pseudomyxoma peritonei. Tumor morphology of low grade (p = 0.013), age under 65 years (p = 0.004), and serum carcinoembryonic antigen level under 5.0 µg/L (p = 0.003) were associated with successful administration of cytoreductive surgery and hyperthermic intraperitoneal chemotherapy. Mean peritoneal cancer index was lower (18.9 vs 32.6, p < 0.001) and age was younger (54.3 vs 61.6, p = 0.003) in patients who underwent hyperthermic intraperitoneal chemotherapy than in patients who did not. Four patients had complete cytoreductive surgery alone, and 20 patients underwent palliative debulking, but 10 were ineligible for this operation.
Conclusions:
Although the combination of cytoreductive surgery and hyperthermic intraperitoneal chemotherapy is currently suggested the standard practice for pseudomyxoma peritonei, not all patients are eligible for this protocol. In this study, hyperthermic intraperitoneal chemotherapy was suitable for 62% of patients with pseudomyxoma peritonei of appendiceal origin.
Keywords
Introduction
Pseudomyxoma peritonei (PMP) is a rare syndrome with an estimated incidence of 1 to 2 persons per million annually (1). Its diagnosis is clinicopathological, and it is characterized by accumulation of extracellular mucin and epithelial mucin-secreting cells within the abdominal cavity. Movement and absorption of peritoneal fluid plus gravity result in accumulation of epithelial cells at specific sites in the abdominal cavity; this is called a redistribution phenomenon (2). An omental mass, ovarian masses, and a mucinous burden on the right hemidiaphragm are characteristically present in PMP and can be explained by this effect. Eventually, the space-taking disease compromises organ functioning and causes intestinal obstruction, dyspnea, and malnutrition. Without surgical intervention, mortality will be the outcome.
The primary origin of PMP has been controversial, ever since the early description of the syndrome as being associated with an ovarian tumor. Nowadays, in the vast majority of cases, the appendix can be identified as the origin (3, 4). Synchronous ovarian metastases are not uncommon, and PMP of appendiceal origin can be confused with primary ovarian neoplasia (5). Moreover, molecular evidence suggests that PMP is usually derived from an appendiceal neoplasm (6). However, PMP is not synonymous with appendiceal neoplasm with peritoneal spread because other tumor types may also induce this clinical syndrome (6). In particular, the classification of PMP has been challenging, with various classification schemes proposed (7–10). In this study, we used the World Health Organization (WHO) 2010 classification, where the tumor is classified as “high-grade” or “low-grade” tumor (11).
In the past, PMP was treated by iterative debulking operations. However, relapses occurred in most cases, and repeated surgeries were more challenging when the disease progressed and scarring, adhesions, and distortion of the anatomy developed. A serial debulking protocol resulted in overall survival figures of 21%–32% after 10 years’ follow-up (12–14). Currently, the treatment considered optimal for PMP is aggressive cytoreductive surgery (CRS), to be followed by hyperthermic intraperitoneal chemotherapy (HIPEC) (15). In case of optimally cytoreduced series of PMP patients, combination of CRS and HIPEC has been reported to yield 10-year overall survival of up to 85% (16).
As CRS and HIPEC represent complex surgery, morbidity and mortality rates are also considerable (17). The complication rate does, however, decrease with increasing surgical experience, particularly in a specialized center. As the Basingstoke center experience points out, not only does surgical technique influence the learning curve, but so does the emphasis on case selection (18). Undoubtedly, not all cases of PMP are amenable to aggressive surgery and HIPEC. Some patients are ineligible due to the extent of their disease. Other patients would, technically, be able to undergo the surgical procedure, but their general condition would prohibit any attempt at surgery. This study analyzed the feasibility of CRS and HIPEC in patients with PMP and then evaluated factors associated with a successful procedure—using an analysis of prospectively and consecutively recruited patients numbering 90.
Material and Methods
This study included 90 patients with histologically proven and demonstrable PMP of appendiceal origin. The database of 121 patients with suspected or diagnosed PMP, who were referred to the Department of Colorectal Surgery in Helsinki University Central Hospital since the establishment of our HIPEC center in November 2007 up to October 2011, 48 months later, was collected prospectively. The inclusion criterion was demonstrable mucinous neoplasm of the appendix and of the peritoneum. The exclusion criteria were as follows: no extra-appendiceal spread (2 patients), mixed adenoneuroendocrine carcinomas of appendiceal origin (MANEC) (8 patients), the peritoneal carcinomatoses of nonappendiceal origin such as ovarian (3 patients), colorectal (5 patients), and ileal (1 patient) and mucinous tumors of unknown origin (2 patients) as well as peritoneal mesothelioma (two patients). Seven patients referred to our unit were only monitored as they showed no demonstrable disease after preceding operation(s) and were not included. One patient with low-grade PMP who refused any further surgical treatment was excluded. Thus, a total of 90 patients fulfilled the inclusion criterion for analysis.
The patients referred underwent consecutive evaluation regarding possible surgical intervention. Only their initial evaluation or initial HIPEC operation in our unit was included here. One patient with high-grade disease showed signs of early relapse after the combined-modality treatment and was offered re-HIPEC during the timeline, but only the initial HIPEC operation was included to avoid demographic design bias. Otherwise, both primary and recurrent PMP were included in the study, regardless of the preceding treatment strategy. There were 65 patients with primary disease and 25 patients with recurrent disease.
Diagnosis of PMP was based on intraoperative findings confirmed by histopathological sampling. The diagnosis was established either in our unit (13 patients) or in referring units (77 patients). Every patient was further evaluated and had her diagnosis confirmed. A total of 83 patients (92%) were invited to a clinical meeting and one (1%) was assessed ineligible for HIPEC during an emergency operation. Six (7%) were excluded from the complex procedures of CRS and HIPEC after our consulting the patient records. Of 90 patients, 80 (89%) had undergone at least one major operation before the clinical meeting. Of those 80 patients who had surgery before the meeting, 73 were operated on elsewhere, 4 had surgery in our unit, and 3 had undergone operations both in our unit and elsewhere. The mean number of preceding operations per patient was 1.4 ± 0.1 (median = 1, range: 0–8). Three of the patients evaluated had undergone HIPEC operations elsewhere before receiving a referral to our unit, with two of them each having had two HIPEC surgeries.
Each patient’s carcinoembryonic antigen (CEA) was measured in 89 of 90 cases (99%) prior to consideration for surgery. The patient with a missing CEA measurement had poor overall status and severe comorbidities and underwent surgery. The nature of this emergency operation was palliative debulking surgery. Values of CEA below 5 µg/L were considered normal. The preoperative tumor-load evaluation included both computed tomography (CT) and the surgical result of the preceding operation(s) (81 patients) or included CT only (9 patients). Histological classification of PMP was based on the WHO classification of tumors: low grade or high grade. Histological samples retrieved underwent review by our pathologists (A.R. and J.K.).
The clinical procedures were as follows: patients were offered combined treatment with CRS and HIPEC, conventional surgery, or active follow-up (Fig. 1). Every amenable patient who would seem to benefit from combined treatment was offered an attempt at the regimen. Conventional surgery was offered to patients ineligible for HIPEC, and follow-up was initiated in patients ineligible for any surgery. Conventional surgery and follow-up were arranged either in our unit or in the referring hospital. Contraindications for intraperitoneal chemotherapy were poor overall status, high age (1 patient, 77 years), and severe comorbidities. Those patients with disseminated disease without any considerable likelihood for complete CRS were also excluded from any attempt at combined-modality therapy. Active follow-up was the choice for patients of very high age (1 patient, 86 years) with very poor general status, or with extensively disseminated disease. Active follow-up included iterative clinical evaluation, serum CEA measurement, and CT every 6 or 12 months.
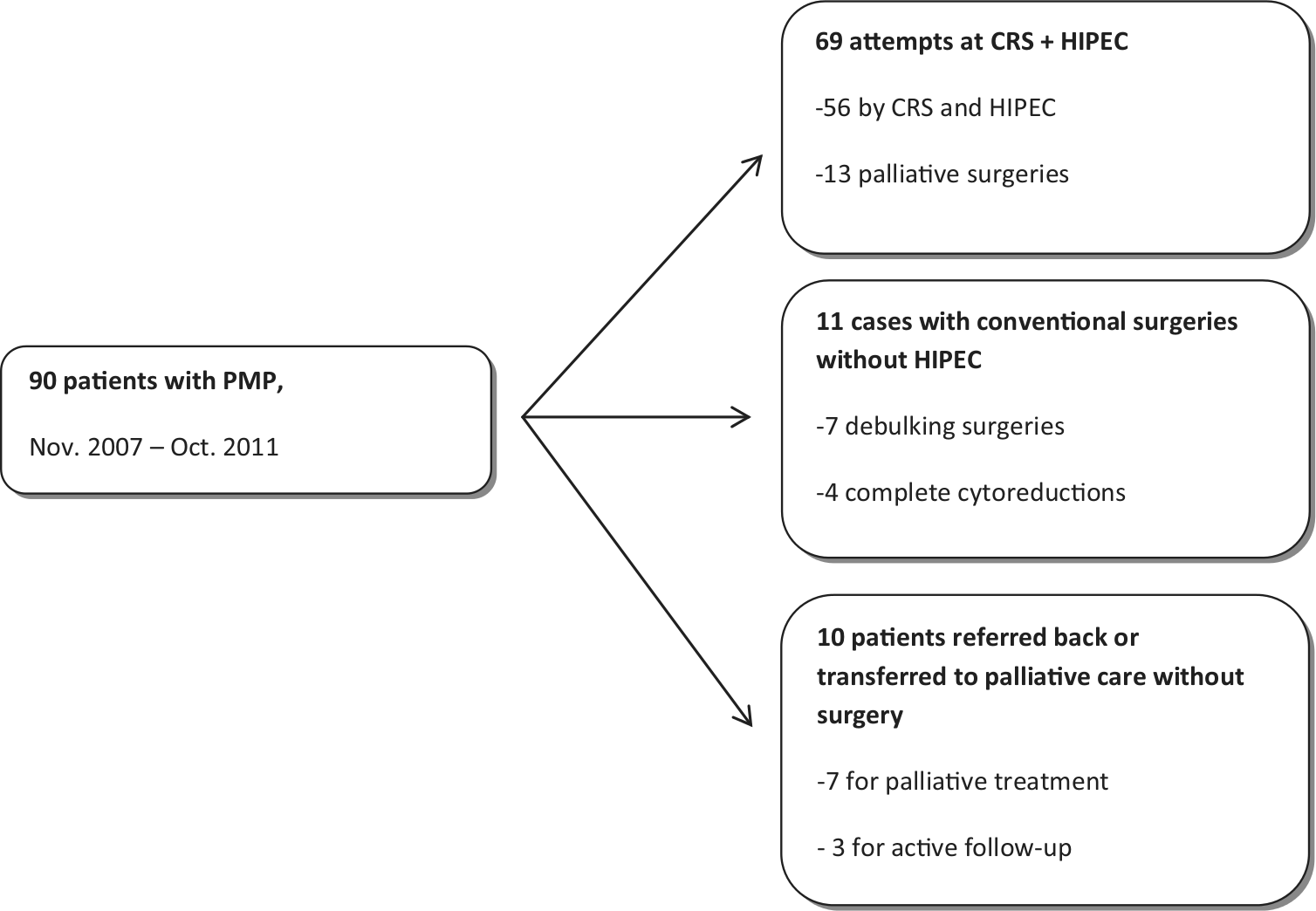
Three clinical pathways of treatment. A total of 90 patients with demonstrable PMP evaluated and considered for surgery in Helsinki during 4 years.
If none of the contraindications existed, an attempt at CRS and HIPEC was scheduled. Aggressive surgery with peritonectomy procedures combined with necessary organ resections was attempted, as described by Sugarbaker (19). After complete exploration of the abdomen and pelvis, every patient who underwent an attempt of HIPEC (69 patients) received a peritoneal cancer index (PCI) score determined intraoperatively (20). No PCI score was determined routinely for the group who underwent debulking surgery. The final decision whether or not to administer intraperitoneal chemotherapy was based on intraoperative assessment of cytoreduction. The radicality of surgery was assessed based on the completeness of the cytoreduction scoring system (21). A surgical result of complete cytoreduction-0 (CC-0) signified no macroscopical residual tumor, CC-1 signified residual nodules under 0.25 cm, CC-2 signified residual nodules between 0.25 cm and 2.5 cm, and CC-3 signified residual tumor over 2.5 cm in diameter. The scores CC-0 and CC-1 were considered successful cytoreduction, and CC-2 and CC-3 as palliative surgery. Intraperitoneal chemotherapy with mitomycin C was administered routinely after successful complete cytoreduction by the modified Coliseum technique (22). The dosage of chemotherapeutic agent was calculated by each patient’s clinical oncologist. The standard dosage was 30 mg/m2.
All analyses were performed with SPSS® Statistics software (IBM® Version 20.0.0). The data were in proportions (percentages) or number of patients for categorical variables and as means ± standard errors (medians, ranges) for continuous variables. Associations between patient characteristics and successfully administered HIPEC were measured by Pearson’s χ2-analysis. The characteristics tested were morphology of the tumor (low-grade or high-grade tumor), age with a cutoff above 64 years, whether or not the serum CEA value was elevated, and gender. Age and PCI, when available, were compared by Student’s t-test for differences in means between patients who underwent or did not undergo a successful HIPEC operation. The delay, determined in months, between diagnosis and assessment for feasibility of HIPEC was compared by Mann–Whitney U-test. Ages and PCI scores were normally distributed, but the delay was not (the Kolmogorov–Smirnov test 0.20, 0.20, and <0.01, respectively). The p-value < 0.05 was considered statistically significant (two-sided). This study was approved by the local ethics committee (permission number 265/13/03/02/2011).
Results
Of the 90 cases, 58 were women (64%) and 32 men (36%); they were assessed for feasibility of CRS and HIPEC. Mean age at assessment was 57.1 ± 1.19 years (median = 58, range: 30–87 years). The tumor was a low-grade carcinoma in 57 patients and high grade in 33. Of 89 cases, serum level of CEA was elevated in 41 (46%). Mean of CEA was 46.7 ± 16.8 (median = 4.4, range: 1–1325.2).
Of the 90 cases, 69 (77%) underwent an attempt at delivering combined treatment with CRS and HIPEC. In 56 (81%) of those 69 surgeries (62% of all patients), HIPEC was successfully administered after a surgical score of CC-0 or CC-1 was achieved. Tumor burden preventing radical surgery was located in the hepatic hilum (7 patients), in the lesser omentum (4 patients), and extensively in the abdominal cavity (2 patients, PCI scores 37 and 39). Conventional surgery with major laparotomy was offered to 11 (12%) patients in our unit, as those patients were considered unamenable to intraperitoneal chemotherapy. A radical end result (CC-0 or CC-1) was achieved in four of those 11 cases, of whom 10 were electively operated but preoperatively excluded from combined treatment with CRS and HIPEC. One of those 11 patients had Alzheimer’s disease as well as atrial fibrillation and was operated on as an emergency case; this debulking surgery was considered definitive treatment. Ten patients (11%) were not scheduled for surgery and were referred back to their referral hospitals or were transferred to palliative care in our unit: 6 patients had widely disseminated disease, and 4 were ineligible for surgery because of severe comorbidity or old age (Table 1). The choices of treatments for the 3 patients who had undergone HIPEC operation elsewhere before obtaining a referral to our unit were the following: one HIPEC, one conventional surgery, and one active follow-up.
Reasons for exclusion from attempt at HIPEC in 21 patients with PMP.
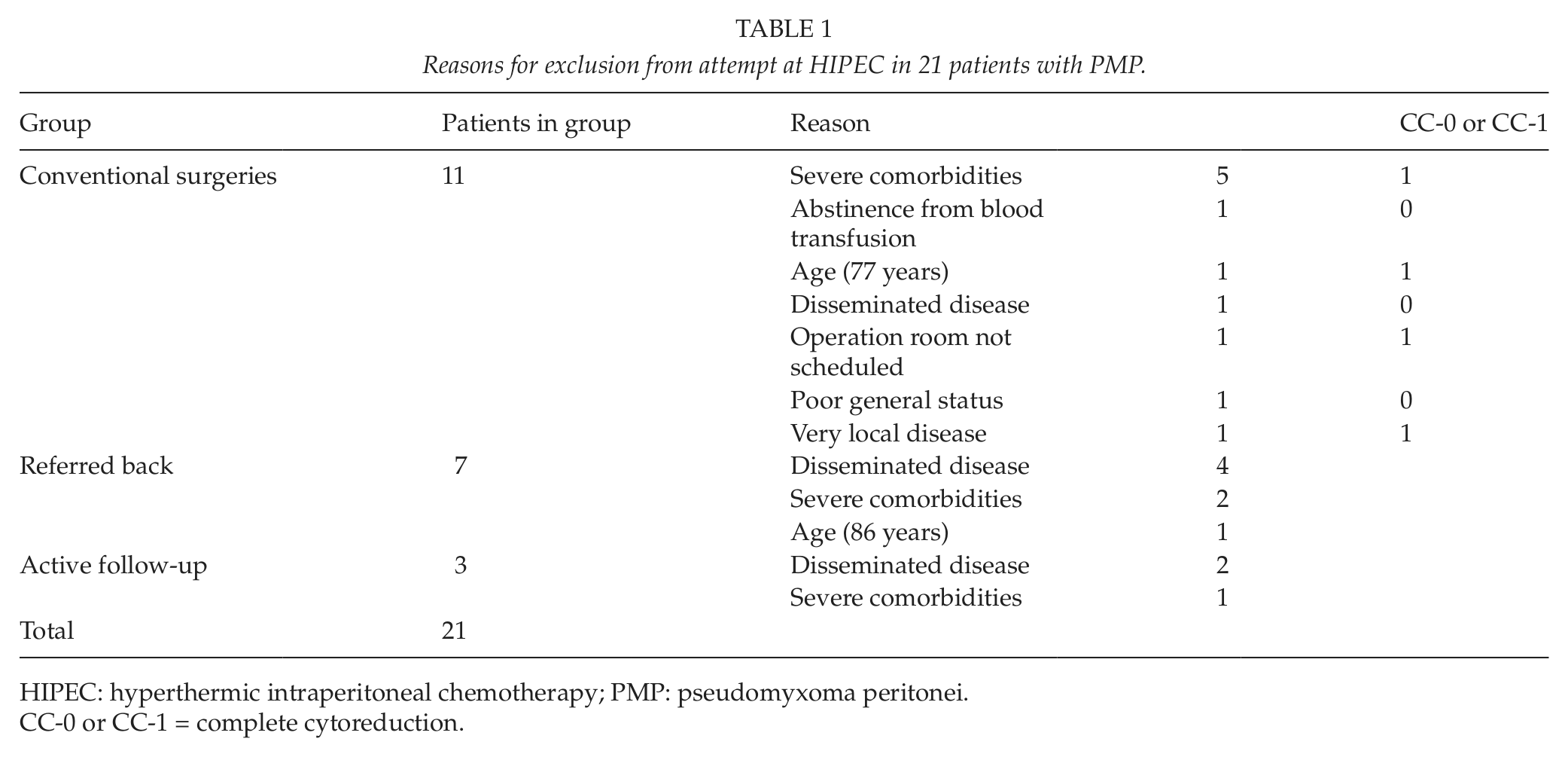
HIPEC: hyperthermic intraperitoneal chemotherapy; PMP: pseudomyxoma peritonei.
CC-0 or CC-1 = complete cytoreduction.
Analysis of variables associated with a successful cytoreduction (CC-0 or CC-1) and HIPEC included 90 patients with demonstrable PMP at the time of their evaluation. Successful combined treatment was associated with low-grade morphology (p = 0.013), serum CEA level under 5.0 µg/L (p = 0.003), and age < 65 years (p = 0.004). Gender showed no effect on surgical result (p = 0.385). There was, however, significant overlap in some variables, such as morphology and CEA (Table 2). Patients undergoing successful HIPEC also had significantly lower PCI values (18.9 vs 32.6, p < 0.001) and were younger (54.3 years vs 61.6 years, p = 0.003) than those with no HIPEC treatment. The highest PCI value in the HIPEC group was 29 years, and the oldest patient was aged 73 years (Table 3). We found no differences in statistical significance in the means for delay between diagnosis and assessment for feasibility of HIPEC, although the mean delay was longer in the non-HIPEC group (24.1 vs 18.3, p = 0.124).
Characteristics associated with HIPEC administration.
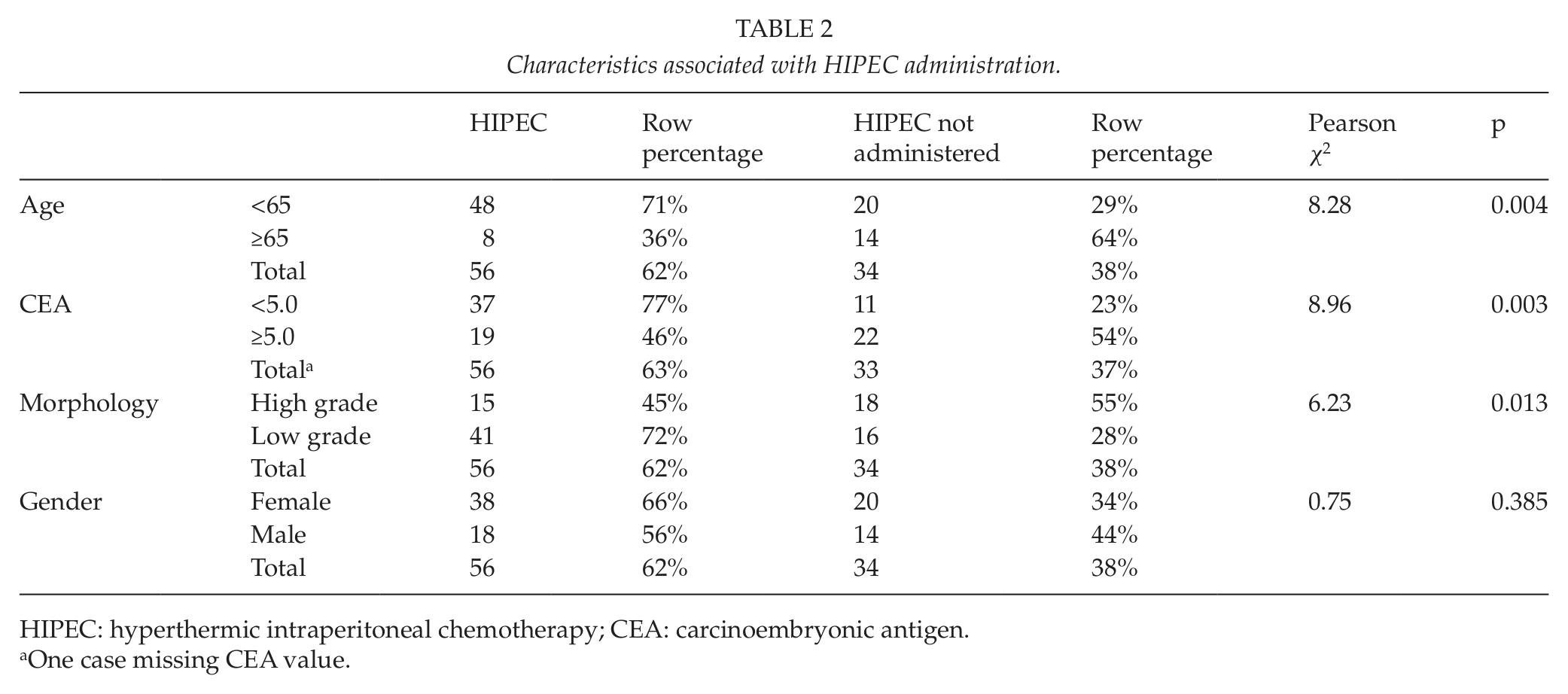
HIPEC: hyperthermic intraperitoneal chemotherapy; CEA: carcinoembryonic antigen.
One case missing CEA value.
Comparison of means in patients undergoing successful HIPEC and other treatment modalities.
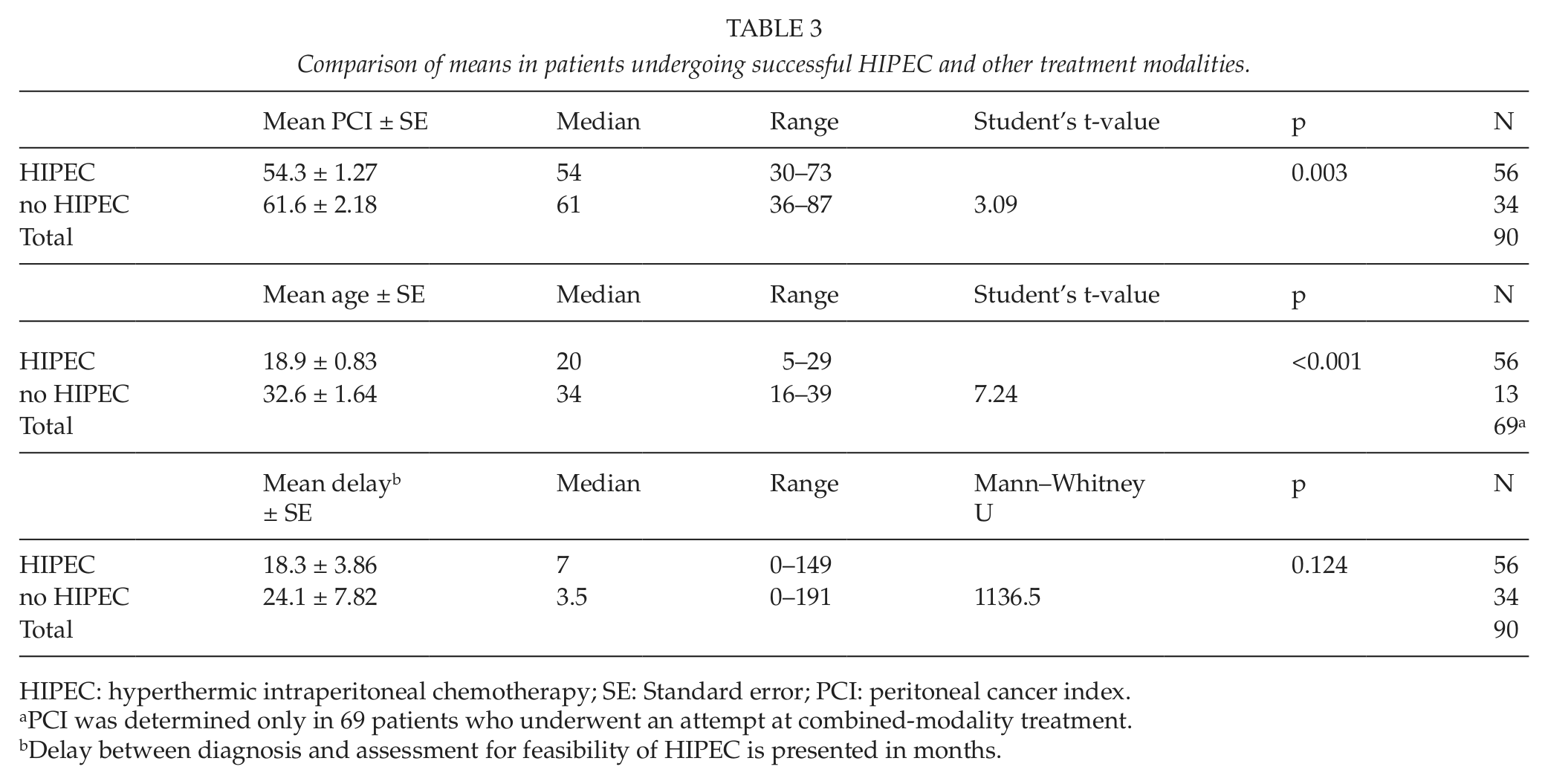
HIPEC: hyperthermic intraperitoneal chemotherapy; SE: Standard error; PCI: peritoneal cancer index.
PCI was determined only in 69 patients who underwent an attempt at combined-modality treatment.
Delay between diagnosis and assessment for feasibility of HIPEC is presented in months.
Discussion
Of the 90 PMP patients with demonstrable disease, 69 (77%) were considered suitable for an attempt at CRS and HIPEC. Of those 69 patients, eventually, 56 (81%) also received HIPEC treatment, 62% of the total. In a series of 203 patients with PMP from Manchester, UK, 69 (34%) underwent CRS, and 32 (16%) had debulking surgery (23). Their proportion of complete cytoreduction was 68% of those undergoing laparotomy, similar to our figure of 75%, even though the number of cases with no operation was higher. In a series of 38 patients referred from Milan (24), 31 (82%) received CRS and HIPEC, but that study included only patients with favorable histology, that is, disseminated peritoneal adenomucinosis (DPAM) and peritoneal mucinous carcinomatosis with intermediate or discordant features (PMCA-I/D). In our own series, however, 37% (33 of 90) belonged to the high-grade group. Moreover, in our series more patients had undergone previous debulking surgery than in theirs (90% vs 53%), which is known to diminish probability for complete CRS.
For the 13 patients in whom complete CRS failed, tumor load was located in the liver hilus, or in the lesser omentum or extensively in the abdominal cavity, which is in line with previous findings. Lesser omentum involvement is especially difficult to evaluate in preoperative CT with adequate accuracy, indicating that even as the best modality for PMP evaluation (25), CT sensitivity is far from optimal. The finding of a heavy general tumor burden in preoperative CT cannot remain the sole exclusion criterion for an attempt at CRS and HIPEC because for some of the patients with heavy tumor load but low-grade histology, radical surgery is possible. Neither has the Verwaal score (26) proven to adequately exclude patients from an attempt at CRS and HIPEC. The lack of a sensitive method for patient exclusion is nearly entirely the problem of waste of surgical resources—not the problem of the patient. Nearly every patient benefits from debulking surgery or “near to complete CRS without HIPEC” because no better alternative treatment exists. The only disadvantage for patients is the longer wait, when operative resources reserved on HIPEC are wasted on palliative treatment.
We noted that patients who underwent nonradical surgery and thus were more prone to undergo debulking surgery only with no HIPEC had higher PCI scores than patients with successful HIPEC. A similar effect has also occurred (23, 27). This finding is reasonable and points out the importance of early diagnosis and consideration for surgery before the tumor bulk accumulates. Although we found no difference in statistical significance in delay between diagnosis and (intraoperative or preoperative) assessment for feasibility of HIPEC, the mean delay was shorter in the HIPEC group. It is not impossible that this effect would become more apparent in larger patient populations.
Age under 65 years was associated with successful HIPEC, and the mean age of patients who underwent HIPEC was, for obvious reasons, lower. As high age itself was a contraindication for an attempt at HIPEC in only 2 cases, the increase in comorbidities by age explains the figures. However, of 56 patients who received HIPEC, 19 (34%) were aged 60 years or older and 4 (7%) were 70 years or older. This suggests that healthy elderly patients in good overall condition and with a moderate tumor burden deserve an attempt at combined-modality treatment, as suggested by Klaver et al. (28) in cases of peritoneal carcinosis of colorectal cancer.
Serum markers are predictors of adequate cytoreduction (29), and this study also showed an association between elevated CEA and inadequate cytoreduction and thus unsuccessful HIPEC. This finding shows that PMP patients with elevated CEA are not only more likely to develop recurrent disease and have a significantly shorter recurrence-free interval, as elsewhere noted (30), but are more likely to undergo nonradical surgery in the first place. Considering that elevated CEA levels are correlated with tumor burden and malignant histological morphologies, the surgical result is understandable.
The predictive value of these findings on PCI and morphology as associated with successfully delivered HIPEC is limited. Not all our patients had undergone a previous operation, and determination of PCI is possible only intraoperatively. Biopsy specimens for determining morphology often were lacking before an attempt at HIPEC. Even with knowledge of factors associated with successful HIPEC, to determine feasibility of HIPEC before an attempt in a specialized center is still difficult. Nevertheless, when there is a diagnosed PMP case or a strong suspicion of one, we suggest that the initial operation, when possible, should be undertaken in a center with capability of performing HIPEC.
As combined-modality treatment of CRS and HIPEC is widely considered standard practice for patients with PMP, what must be clear is that not all patients are eligible for these complex procedures. In this study, HIPEC was successfully delivered to 62% of patients with demonstrable PMP who were referred to our unit for consideration for surgery. This study suggests that nearly two-thirds of patients with PMP are amenable to these complex procedures. Indeed, we have a possible referral bias in this study design, as patients in very poor condition or with extensive disease are unlikely to be referred to our unit. This bias is hard to eliminate. As long as there exists controversy in management and even in classification of PMP, it is obvious that not all patients with PMP are gaining referrals to a specialized center. However, regardless of location of treatment, a considerable proportion of the patient population has still to be treated with conventional methods: iterative debulking surgery, active follow-up, or sometimes even palliation without surgical intervention. When evaluating the use of CRS and HIPEC in the long term and when comparing it to the results of previous debulking surgery, comparison is necessary between entire PMP populations because comparing the results of HIPEC with the whole group of patients treated with debulking surgery will lead to bias in favor of HIPEC.
Footnotes
Funding
This study was supported by a grant from the Mary and Georg C. Ehrnrooth foundation.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
