Abstract
Background and aims:
The combination of cytoreductive surgery (CRS) and hyperthermic intraperitoneal chemotherapy (HIPEC) is widely used to treat pseudomyxoma peritonei (PMP), peritoneally metastasized colorectal or appendiceal cancer (pmCRC or pmAppCa) and peritoneal mesothelioma, although the specific efficacy of HIPEC remains inadequately proven. The aim of this retrospective study was to evaluate the outcomes of our CRS-HIPEC procedures and compare them with prior reports.
Materials and Methods:
All patients operated with the intent of CRS-HIPEC at Oulu University Hospital (OUH) between September 2013 and August 2023 were prospectively registered in a CRS-specific quality control registry that included preoperative, intraoperative, and pathological data, as well as 30-day postoperative complications. The follow-up included thoracoabdominal computed tomography (CT) every 6 months for 3 years and after this yearly until 6 years postoperatively. Survival data were retrieved both from hospital records and from a national registry to analyze both short- and long-term outcomes of CRS-HIPEC.
Results:
Since its introduction at OUH in 2013, CRS-HIPEC was performed with curative intent in 272 patients. Peritoneal disease was confirmed in 223 (82%) cases. Among 171 resectable patients operated with CRS, 162 (95%) received HIPEC. Indications included pmCRC in 67 patients (41%), PMP in 61 (37%), pmAppCa in 23 (14%), and peritoneal mesothelioma in 7 (4%, other histology 4). Severe complications within 30 days (Clavien-Dindo class III–V) occurred in 68 patients (40%) of the CRS patients with a postoperative mortality of 0.6% (n = 1). The mean follow-up was 39 months (range 1–138). The estimated 5-year overall survival after CRS-HIPEC was 87% for PMP, 41% for pmCRC and 58% for pmAppCa. Estimated 5-year disease-free survival was 70%, 24%, and 36%, respectively.
Conclusion:
CRS-HIPEC is associated with good OS and DFS in PMP, whereas survival after both pmCRC and pmAppCa is considerably lower. The procedure is associated with a considerable risk of severe complications and should therefore be performed in specialized centers only.
Keywords
Social media summary: This single center experience demonstrates good survival after cytoreductive surgery (CRS) and hyperthermic intraperitoneal chemotherapy (HIPEC) for pseudomyxoma peritonei. The survival after CRS-HIPEC for peritoneally metastasized colorectal or appendix malignancies was poorer, but in line with that in high-volume centers.
Context and Relevance
Cytoreductive surgery (CRS) and hyperthermic intraperitoneal chemotherapy (HIPEC) are used with curative intent for the treatment of pseudomyxoma peritonei (PMP), peritoneal metastases of colorectal cancer (pmCRC), peritoneal metastases of appendiceal cancer (pmAppCa) and peritoneal mesothelioma. This curative approach has significantly improved survival outcomes for patients with peritoneal malignancies. This retrospective study presents the short- and long-term outcomes from the first 10 years of our CRS and HIPEC center.
Introduction
Cytoreductive surgery (CRS) and hyperthermic intraperitoneal chemotherapy (HIPEC) were first described in the 1980s for the treatment of pseudomyxoma peritonei (PMP).1,2 Since then, CRS and HIPEC have also been used to treat peritoneal mesothelioma and peritoneal metastasis of colorectal cancer (pmCRC), as well as ovarian and gastric cancers. 3 While CRS recommendations are quite homogeneous, 4 HIPEC recommendations and HIPEC protocols vary significantly. 5 Different chemotherapeutic agents, such as mitomycin C, oxaliplatin, and doxorubicin, have been used with various carrier solutions. Furthermore, treatment durations and target intraperitoneal temperatures differ widely, with no consistent evidence of improved survival outcomes.5,6
PMP refers to a peritoneal dissemination of mostly appendiceal mucinous neoplasms accompanied by mucinous ascites. 6 The primary appendiceal neoplasms are histologically classified in low- or high-grade appendiceal mucinous neoplasm (LAMN or HAMN). 6 The peritoneal disease can further be categorized in acellular mucin, low-grade peritoneal neoplasia, or high-grade peritoneal neoplasia with some variations in terminology across classifications.7,8 The current widely accepted combination of CRS-HIPEC as mainstay treatment of PMP, is based only on observational studies suggesting prolonged overall survival (OS) compared to debulking surgery alone.9,10 With CRS-HIPEC, the 5-year OS for low-grade PMP is reported to be over 85%, 11 whereas high-grade PMP has a significantly lower 5-year OS of around 48%. 12 The use of CRS-HIPEC in other peritoneal malignancies is more controversial and its use varies between centers. Although CRS-HIPEC has been shown to improve OS in pmCRC compared to systemic chemotherapy alone, 13 the additional effect of HIPEC compared to CRS alone and the optimal HIPEC protocol remains a matter of debate.14,15
In 2013, CRS-HIPEC was introduced at Oulu University Hospital (OUH), which is one of two centers in Finland performing these procedures. From introduction, all procedures have been recorded in a prospective database for scientific and quality assessments. Given the ongoing debate surrounding the role of HIPEC with CRS, all available data should be reported to support further analyses. The aim of this study was to compare our patient demographics, operative findings, postoperative complications, and survival rates with previously published CRS and HIPEC data from other centers.
Methods
Patients and data collection
This single center observational cohort study included all patients operated with the intention of CRS-HIPEC at OUH from the introduction in September 2013 to August 2023. Patients with confirmed or suspected peritoneally metastasized tumors considered amendable to CRS-HIPEC and selected patients with high risk of developing peritoneal metastases after resection of a primary tumor were referred to the CRS multidisciplinary team (MDT) consisting of CRS surgeons, abdominal radiology specialists, and clinical oncologists. Patients with suspected peritoneal tumors deemed amendable to CRS-HIPEC were evaluated by a CRS surgeon and a CRS team anesthesiologist in the outpatient clinic, to determine the patient’s fitness for CRS prior to proceeding with the surgical treatment plan. Patients with ASA score IV were considered unfit for CRS-HIPEC and were referred for oncological treatment.
Over the first 10 years, the indications for second-look operations have been modified. Previously second-look operations were performed in patients with high risk of peritoneal metastases including after tumor perforation, limited peritoneal surface dissemination, or ovarian metastases detected during primary cancer surgery. However, routine second-look operations are no longer performed, as randomized trials have demonstrated no benefit of CRS-HIPEC in these patients.16,17 No other significant changes have been made to the selection criteria.
Since the beginning of CRS-HIPEC at OUH, all patients operated with the intent of CRS-HIPEC have been prospectively registered in a quality control registry. Data collected included demographics, ASA score, histology and origin the primary tumor, details on preoperative diagnostics, perioperative data (presence and resectability of peritoneal disease, peritoneal cancer index (PCI), performance of CRS and or HIPEC), postoperative complications, length of hospital stay, readmissions, as well as postoperative surveillance data. Patient survival and cause of death data were retrieved both from patient records and from the national registry (Statistics Finland).
Surgical procedure
The standard access was a long midline incision. In patients deemed borderline operable, laparoscopy was typically performed before proceeding to laparotomy only if no clear unrespectability was found. In PMP patients, the peritoneum was preserved following the incision along the linea alba to facilitate lateral dissection before entering the abdominal cavity. Any ascites was removed and all adhesions released to allow a thorough examination of the abdominal cavity, conducted to define the PCI, assess operability, and plan the surgical approach. No strict PCI cutoff for operability was adhered to, but for pmCRC and pmAppCa, patients with a PCI higher than 20 were considered borderline operable. In these cases, the operation proceeded only after thorough assessment of the patient’s overall circumstances. For pmCRC and pmAppCa, the resection aim was CC-0 (no visible residual tumor) and only CC-0 was considered a radical resection. For PMP patients, CC-1 (all remaining tumor nodules less than 2.5 mm) was accepted and considered a radical resection. CC-0 is regularly not achievable in this context and CC-1 is therefore commonly considered a complete CRS in these patients. 18
For extensive disease, the procedure generally commenced with the removal of the parietal peritoneum over the diaphragm, proceeding through the right upper abdomen, left upper abdomen, pelvic area, and finally, the small bowel. Only disease-affected segments of the peritoneum were excised, and previous scars were removed. Alongside parietal peritonectomies, necessary (multi)visceral resections were performed to remove all visible metastases and any primary tumor if present. In addition, the greater omentum and ovaries (particularly in patients of post-childbearing age or with widely disseminated disease) were commonly removed to reduce further peritoneal invasion, as supported by previous and recent studies as well as surgical tradition.19–21 Bowel anastomoses and stomas were created following the HIPEC treatment.
HIPEC
Following CRS, the skin edges were elevated with sutures to facilitate open HIPEC. The abdomen was rinsed with 10 L of heated Ringer’s acetate to remove any loose tissue that could obstruct fluid circulation during HIPEC. Intraperitoneal chemotherapy, with a drug solution temperature of 41°C–43°C in Ringer’s acetate, was administered. Mitomycin C was used for PMP, pmCRC, and pmAppCa at a dose of 35 mg/m², while a combination of doxorubicin and cisplatin was used for peritoneal mesothelioma. A 90-min incubation period was applied for both regimens. After the treatment, the abdomen was rinsed with heated Ringer’s acetate in an amount matching the circulating drug volume.
Postoperative procedures
After HIPEC, hand-sewn bowel anastomoses were created using single layer seromuscular continuous 4-0 absorbable sutures. Any necessary stomas were positioned through the abdominal wall before its closure, with stomas everted and opened following the closure of the abdominal wall and wound.
Postoperative care
Patients undergoing CRS-HIPEC were routinely monitored at an intensive care unit (ICU) at least until the day after surgery. A nasogastric tube was retained for at least 1 day postoperatively and removed once nausea subsided. Antibiotic prophylaxis (cefuroxime and metronidazole) was started right before surgery and continued until 24 h after surgery. Further prolongation was at the surgeon’s discretion. Patients were encouraged to resume eating and mobilization as soon as their condition allowed.
Medical and surgical postoperative complications were evaluated within the first 30 days after surgery and were graded according to the Clavien-Dindo classification. 22 The failure-to-rescue rate (FTR) was defined as the death of a patient who experienced at least one severe complication (Clavien-Dindo ⩾ 3) within 30 days after surgery.
Postoperative oncological treatment
All patients with pmCRC, pmAppCa, or HAMN-PMP were referred to clinical oncologists for evaluation of adjuvant chemotherapy. Due to different electronic patient record systems and national policy, we were unable to maintain a detailed surveillance of possible chemotherapy regimens received by these patients apart from local patients. However, adjuvant chemotherapy is our recommended treatment of choice unless otherwise contraindicated.
Postoperative follow-up
Patients were followed up in outpatient clinics at 1 month post-surgery, and then every 3 months for the first 3 years, and subsequently every 6 months from years 3 to 6 post-surgery. At each follow-up, clinical status, Carcinoembryonic antigen (CEA), CA19-9, and hemoglobin levels were assessed, and a thoracoabdominal computed tomography (CT) scan was performed at every other visit. Additional follow-ups are planned at 8, 10, 15, and 20 years. Survival data, available up to the end of 2022, were obtained from Statistics Finland, and other relevant clinical data were collected prospectively.
Statistical analysis
Statistical analyses were conducted using IBM SPSS Statistics 24.0 (IBM Corp., Armonk, NY, USA). All values are presented as means with 95% confidence intervals (95% CI) or medians with interquartile ranges (IQR). Survival rates were estimated using Kaplan–Meier life tables. Patients were censored at the last confirmed follow-up if lost to follow-up, or at the end of follow-up if event free at the end of the observation period. The log-rank test was used to compare differences in survival. Median follow-up was calculated as median time to censoring. Patients with missing values were excluded from analyses of that particular variable.
Ethics
The study was conducted in accordance with the Declaration of Helsinki. The study was approved by the Scientific Committee of the Northern Finland Hospital District (84/2017) and individual consent for this retrospective study was waived by the Scientific Committee.
Results
Patient demographics
Between August 2013 and July 2023, we operated on 272 patients with the intent of performing CRS and HIPEC (Figure 1). Patient demographics are presented in Table 1. Yearly number of CRS ± HIPEC operations and complications of inoperable patients are presented in the supplementary material. During 2013–2019 (early period), we operated 85 CRS ± HIPEC operations, and during 2020–2023, 86 CRS ± HIPEC operations (data shown in supplementary material). Patients in the later period were older (median age 62 vs 58 years, p = 0.029) and had higher ASA class (p = 0.017). Mean operative duration decreased (p = 0.015), more patients received protective stomas instead of end ostomies (p = 0.013), and hospital stay was shorter (p = 0.033). No significant differences were observed in PCI, operative blood loss, or postoperative complications. The distribution of PMP, pmCRC, and pmAppCa cases remained similar.
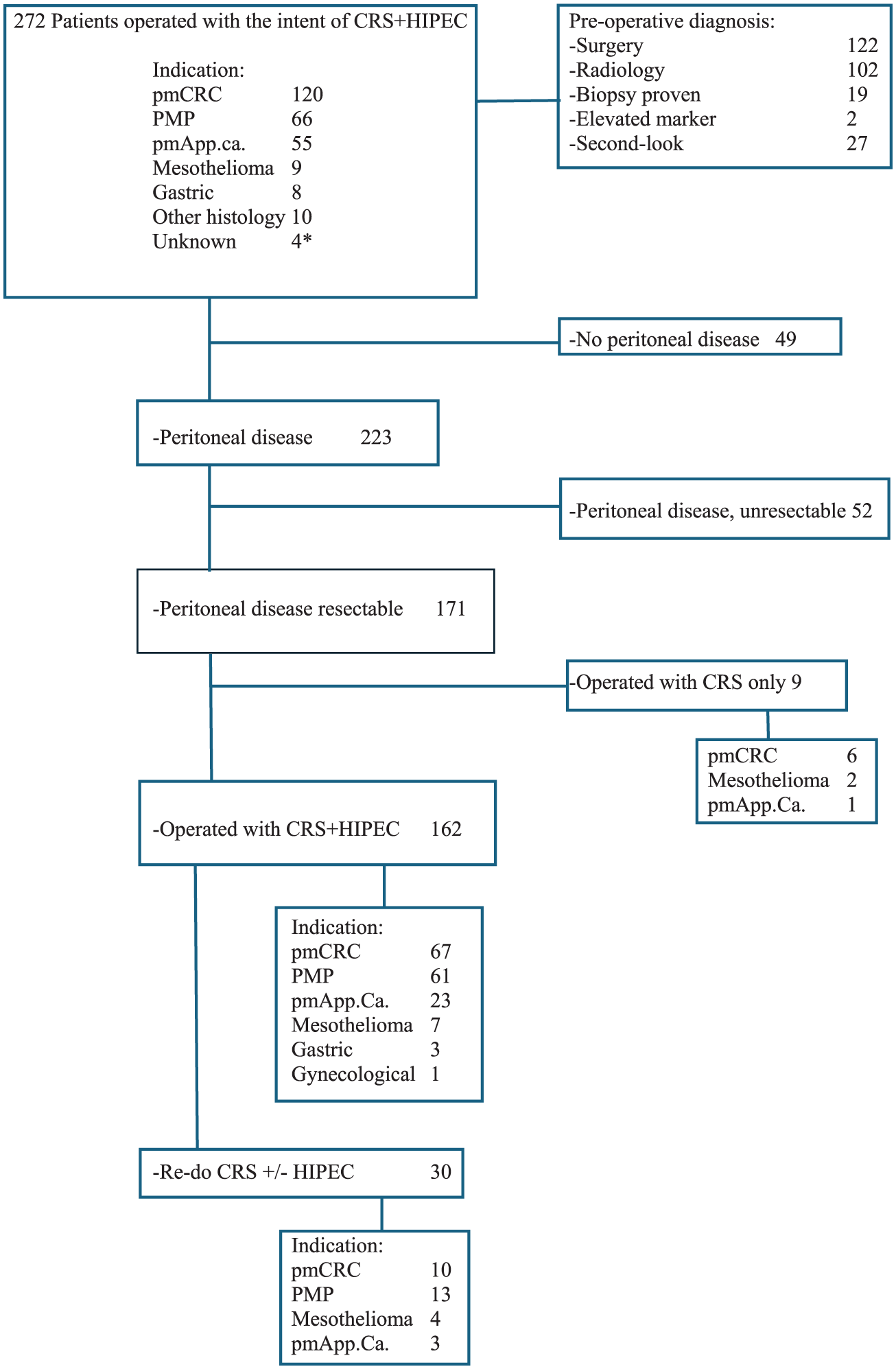
Flow chart of patients operated with the intent of CRS-HIPEC. *Unresectable peritoneal adenocarcinoma with unknown primary tumor location.
Patient demographics.
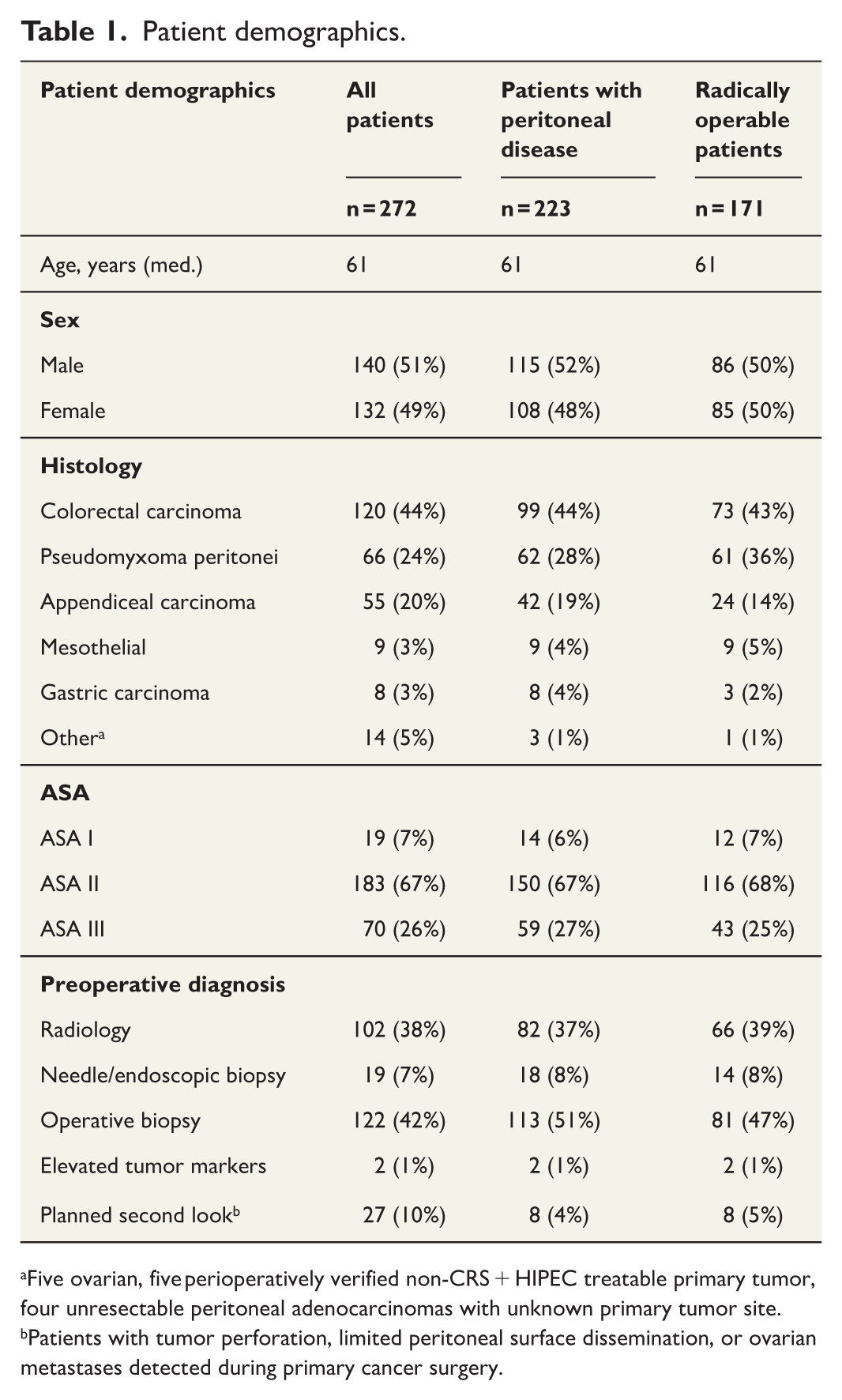
Five ovarian, five perioperatively verified non-CRS + HIPEC treatable primary tumor, four unresectable peritoneal adenocarcinomas with unknown primary tumor site.
Patients with tumor perforation, limited peritoneal surface dissemination, or ovarian metastases detected during primary cancer surgery.
Preoperative findings
In 122 cases (45%), peritoneal dissemination was discovered during elective surgery, leading to discontinuation of the procedure and referral to OUH for CRS-HIPEC. In 102 patients (38%), peritoneal metastases were identified through preoperative imaging. Needle biopsies and/or endoscopic biopsies were taken of 19 (7%) patients. Two patients were suspected of having peritoneal disease based on elevated cancer markers during follow-up. In addition, 27 patients underwent a second-look operation due to an elevated risk for peritoneal disease after primary cancer operation.
Operative findings
In 223 of the 272 operated patients (82%), peritoneal disease was confirmed. Fifty-two of whom were unresectable. Among 171 (77%) radically resectable cases, 162 (95%), had no contraindications for HIPEC and received intraperitoneal chemotherapy. Nine patients (six with pmCRC, two with mesothelioma and one with pmAppCa) were treated with CRS without HIPEC. The main contraindications for HIPEC were age and/or comorbidities (seven patients). In 27 second-look operations, 8 patients (30%) had peritoneal disease and met the criteria for CRS-HIPEC (Tables 1 and 2).
Patient Demographies and operative findings of main entities of CRS-HIPEC.
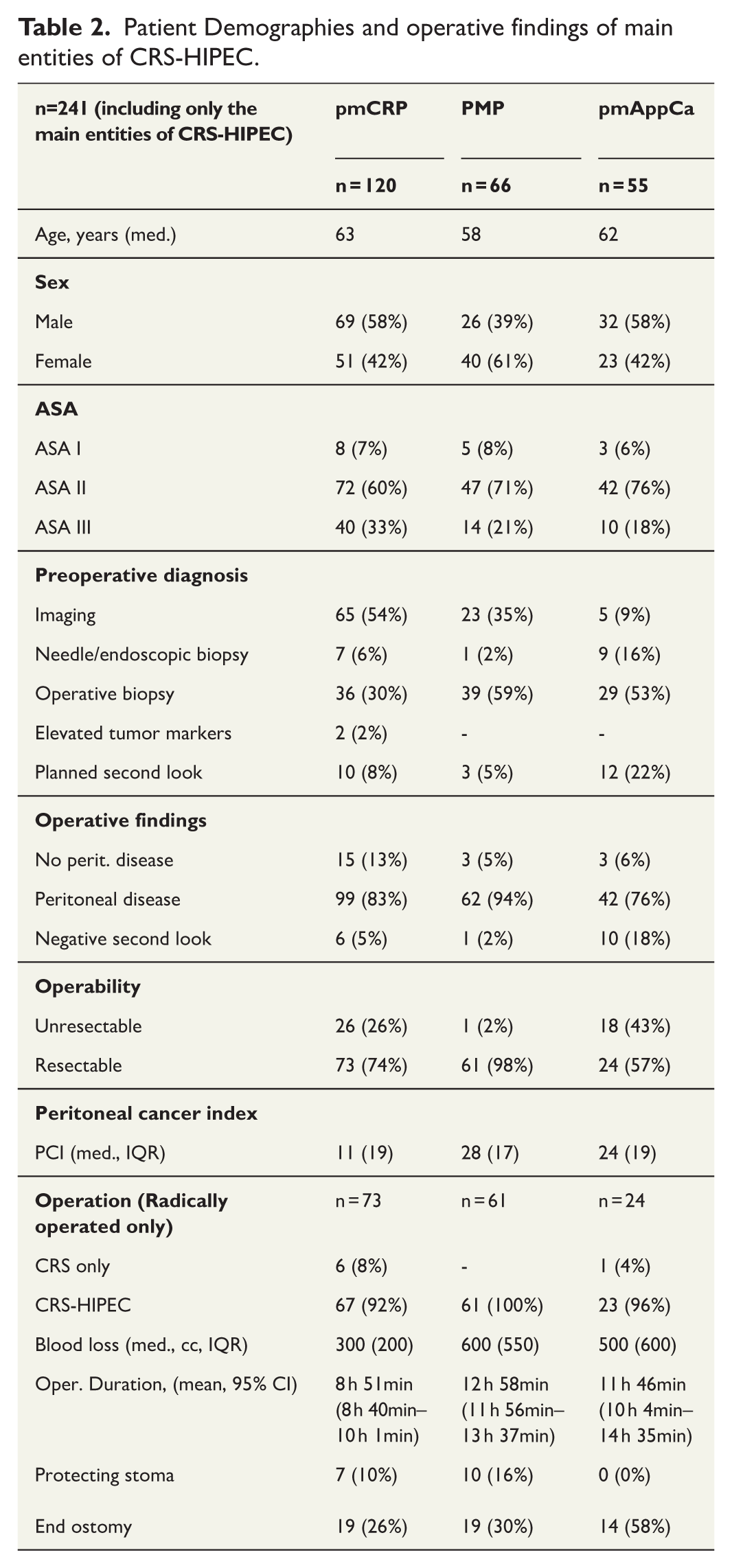
The median PCI was 28 for PMP, 24 for appendiceal cancer, and 11 for pmCRC (Table 2). Median operative time was 10 h and 58 min for CRS-HIPEC procedures. Median blood loss during CRS-HIPEC procedures was 400 mL.
Pathological findings
A total of 66 patients were operated for suspected PMP (Figure 1, Table 2). In four of these (6%), no peritoneal disease was detected, while one patient (2%) was inoperable. Thus, 61 operations (92%) achieved radical cytoreduction (CC-0 or CC-1). Among the 66 patients with suspected PMP, 57 (86%) of the primary tumors were classified as low-grade appendiceal neoplasia (LAMN), and 9 (14%) as high-grade appendiceal neoplasia (HAMN). Peritoneal histology of confirmed PMP cases revealed low-grade features in 34 cases (55%), high-grade features in 4 (6%), and acellular mucin in 24 (39%).
Among 120 patients operated for pmCRC, peritoneal disease was verified in 99 (83%). Of these, 73 (74%) were operated with radical cytoreduction (CC-0), 27 (37%) for synchronous, and 46 (63%) for metachronous disease. The histological type of the primary CRC was non-mucinous adenocarcinoma in 44 (60%), mucinous adenocarcinoma in 21 (29%), and signet ring cell carcinoma in 6 (8%). The primary CRC tumor was located in the right colon in 53%, in the left colon in 39%, and in the rectum in 8%.
In 55 patients operated for suspected pmAppCa, peritoneal disease was confirmed in 42 (76%). Of these, 24 (57%) were radically operable. The histological subtypes included 13 (54%) mucinous adenocarcinomas, 5 (21%) signet ring cell adenocarcinomas, 3 (13%) goblet-cell adenocarcinomas, 2 (8%) non-mucinous adenocarcinomas, and 1 (4%) adenoneuroendocrine carcinoma.
Short-term outcomes
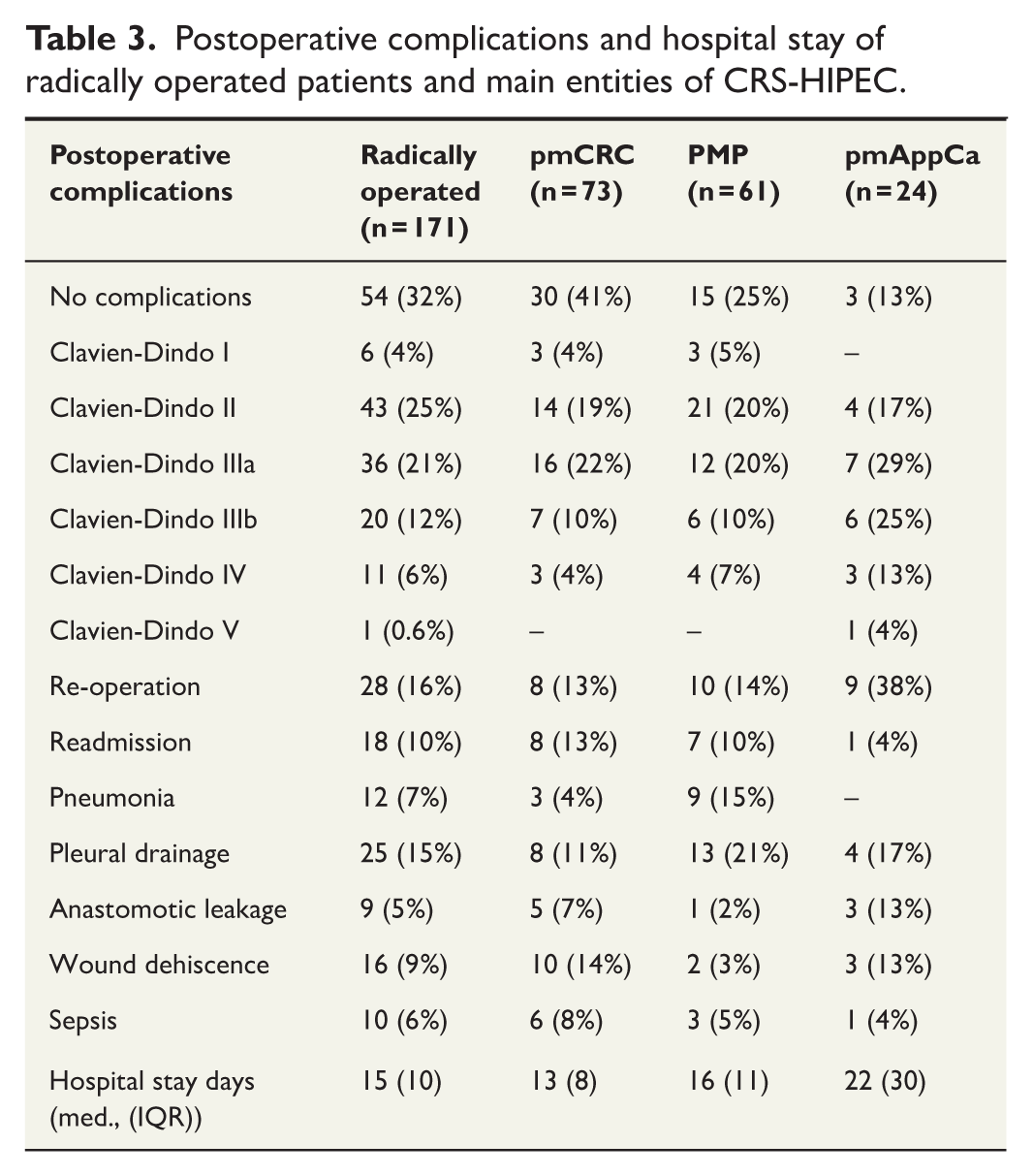
Postoperative complications and hospital stay for operations with peritoneal metastases and radically operated patients are detailed in Table 3. Of 171 radically operated patients, 68 (40%) experienced severe complications, 1 of whom died, corresponding to a mortality rate of 0.6% and an FTR of 1.5%. The mean ICU stay was 1.9 days. Among patients who received HIPEC, the postoperative neutropenia rate was 22 patients (14%).
Postoperative complications and hospital stay of radically operated patients and main entities of CRS-HIPEC.
Long-term outcomes
The median follow-up time of censoring for OS of radically operated patient was 39 months (range 1–138). After CRS-HIPEC for PMP, the estimated 5-year cancer-specific survival (CSS) was 94.9%, OS was 87.1%, and disease-free survival (DFS) was 70.0% (Figure 2). For pmCRC patients treated with CRS-HIPEC, the estimated 5-year OS and CSS were both 41.4%, with a median survival of 51 months (Figure 3a). The median DFS for pmCRC was 18 months, with an estimated 5-year DFS of 23.7%. The PCI level had a significant effect on OS for pmCRC patients after CRS-HIPEC. Patients with a PCI <11 had a better estimated 5-year OS of 54.6%, while OS was 21.9% for PCI 11–15 and 23.9% for PCI > 15 (p = 0.03, Figure 3b). Among patients with pmAppCa, the estimated 5-year OS was 57.7%, with a median survival of 63 months (Figure 4). The estimated 5-year DFS was 36%.
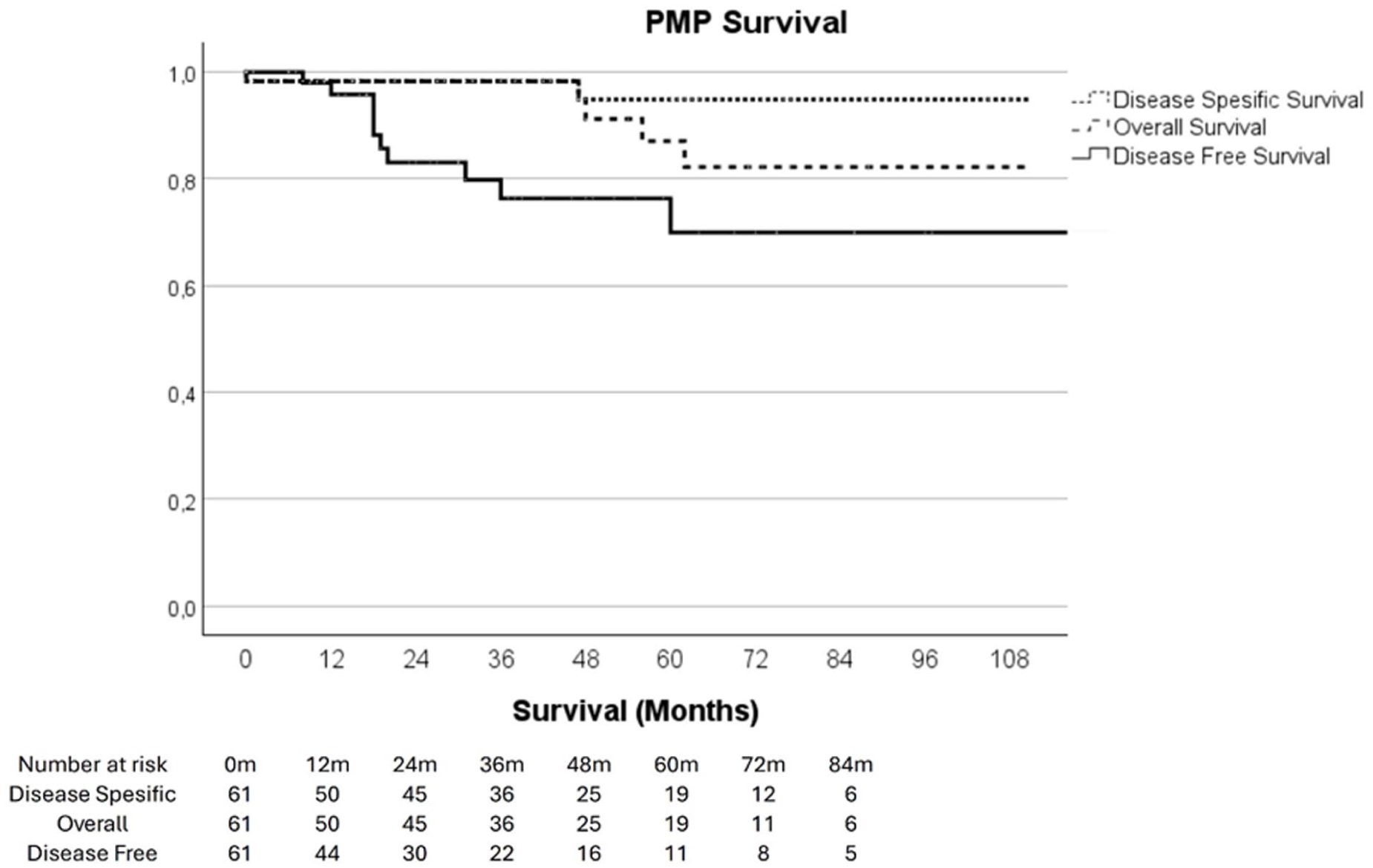
Disease-specific, overall, and disease-free survival of PMP patients after primary CRS-HIPEC (n = 61).
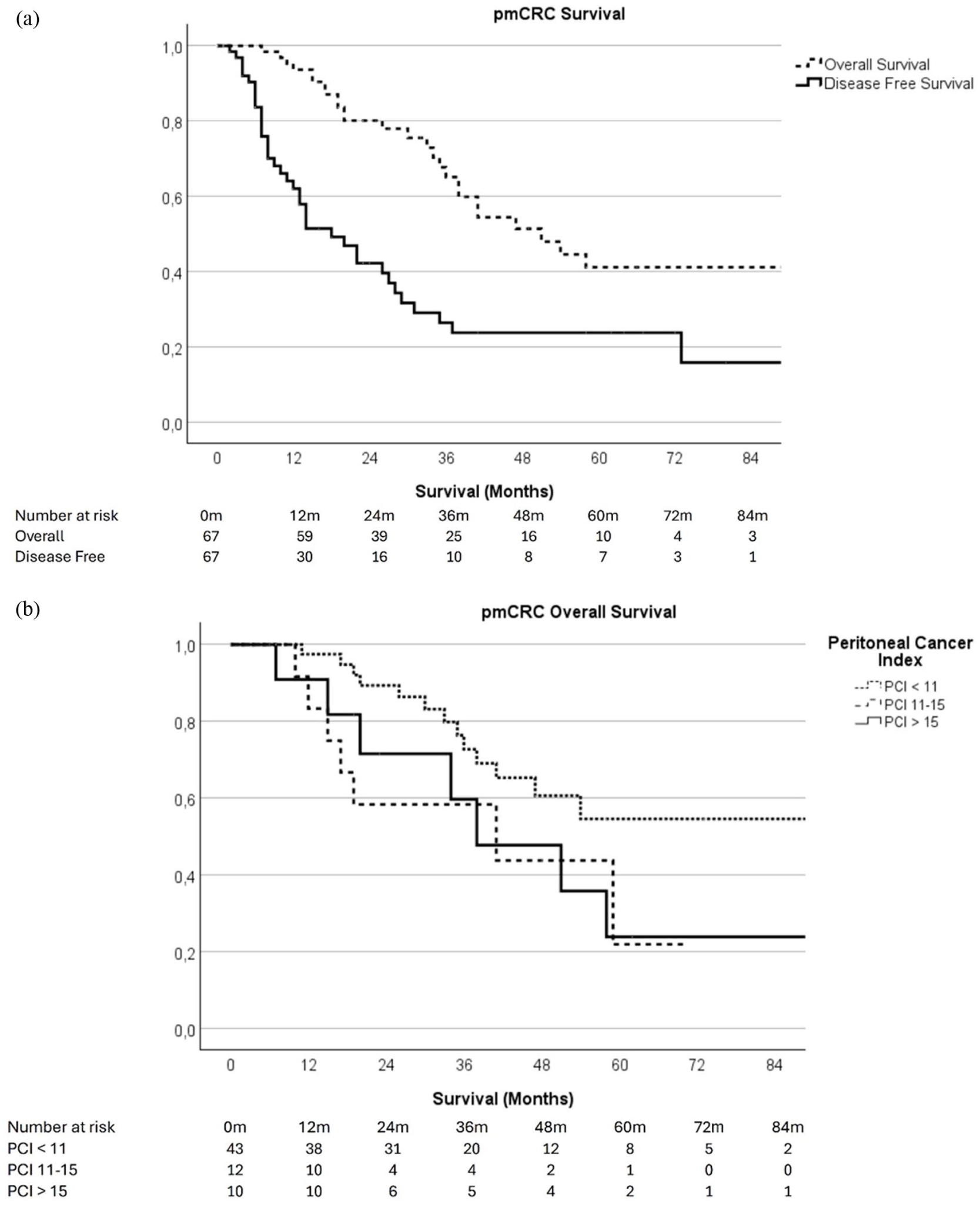
(a) Overall and disease-free survival of pmCRC patients after CRS-HIPEC (n = 67). (b) Overall Survival of pmCRC patients according to PCI.
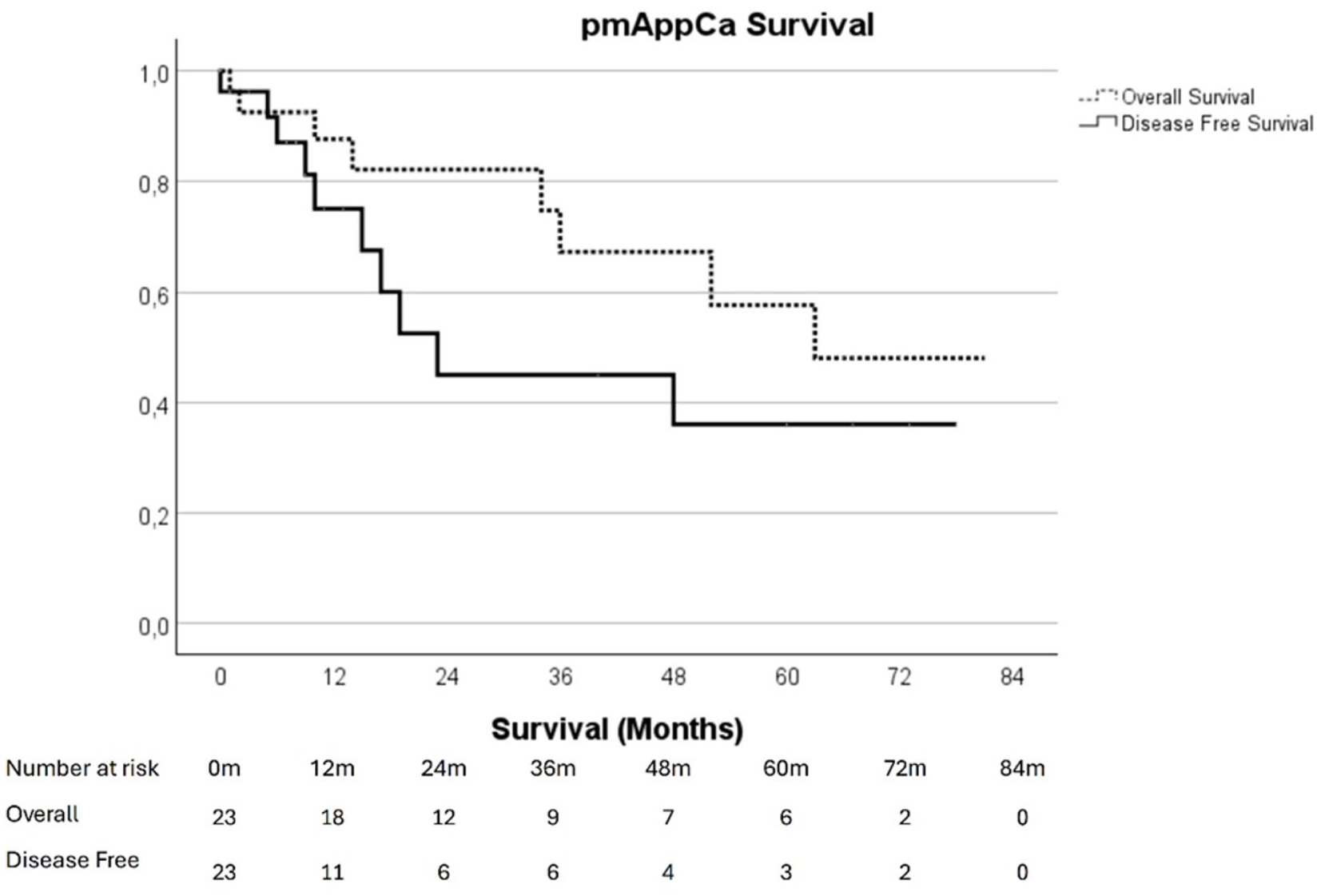
Overall and disease-free survival of pmAppCa after CRS-HIPEC (n = 23).
Redo operations
Redo CRS-HIPEC was primarily considered for pmCRC (10 patients, all radically operated) and PMP (13 patients, all radically operated). The 5-year OS following the first redo cytoreduction was 100% among PMP patients (median follow-up time 45 months) and 16.7% among pmCRC patients (median follow-up time 21 months).
Discussion
This retrospective study prospectively collected data of the establishment and development of a CRS-HIPEC center, to compare short- and long-term outcomes at this center with results from other centers. Previous studies suggest that CRS-HIPEC is an effective treatment for pmCRC and PMP and long-term survival surpasses that of systemic chemotherapy alone.9,23 In addition, there is evidence that centralization in high-volume centers could improve postoperative morbidity and mortality of CRS-HIPEC. 24 The main finding of this study is that CRS-HIPEC led to an estimated 5-year OS of 87% after PMP, 41.4% after pmCRC, and 57.7% after pmAppCa. This study included all patients from the inception of our CRS-HIPEC program, also those operated during the initial learning curve of our center. Despite this, we demonstrate here that the 5-year survival rates for PMP and pmCRC patients at OUH are comparable to previously published data.14,25
Although both CRS and HIPEC are major surgical procedures, hospital stay and postoperative complications may not differ substantially from other major abdominal oncological surgeries. 26 In our cohort, 40% of patients experienced major postoperative complications (Clavien-Dindo class III and IV), a rate consistent with some prior studies, 14 although also lower rates have been reported. 27 Postoperative mortality was low (0.6%), which is similar to rates reported in the majority of other studies. HIPEC is known to contribute to specific postoperative complications, such as cytopenia. 28 In addition, lung complications are not uncommon although their incidence varies in different publications.24,29,30 In our study, 37 patients (21%) experienced pulmonary complications, with 25 requiring pleural drainage. Similar rates of pulmonary complications have also been shown in larger cohort studies. 24 Twenty-two patients (14%) experienced neutropenia, a somewhat lower rate than the previously reported 20%–40%. 31 The average hospital stay in our center was 15 days, a prolonged duration that may reflect the considerable travel distances within our region; however, this length of stay aligns with findings in recent studies.32–34
Frøysnes and colleagues 27 identified PCI as an important prognostic factor in pmCRC, with PCI > 12 as a strong indicator of disease relapse. Our results support these findings; the estimated 5-year OS for patients with PCI < 11 was 54.6%, whereas OS estimate decreased significantly to <25% in patients with PCI > 11. However, the survival of pmCRC in this study is still remarkable, when considering that 5-year OS for pmCRC patients treated with systemic chemotherapy is close to zero. 16 Unfortunately, we are unable to report postoperative chemotherapy treatments for all pmCRC patients and are therefore unable to evaluate a possible effect on survival. However, as we routinely refer our pmCRC patients to clinical oncologists for chemotherapy, one has to assume that the majority was treated according to the recommendation.
pmCRC has a high risk for recurrence, with a median DFS of 18 months observed in our study. Therefore, it is essential to keep in mind that despite acceptable OS after CRS-HIPEC, most patients still require oncological treatment for their advanced disease at a later stage. In cases of local recurrent pmCRC after CRS-HIPEC, redo CRS-HIPEC may be an option, but the scientific evidence of any benefits is limited. 35 We have performed redo CRS-HIPEC after thorough individual assessment for selected pmCRC patients with limited local residual tumor and good overall health status. The OS and DFS in PMP are longer than those in pmCRC but some patients still experience recurrent disease requiring treatment. In selected PMP patients, redo CRS-HIPEC appears to provide favorable survival, 36 consistent with our findings. While the number of redo patients is certainly limited, we believe that these results indicate that selected patients may benefit from secondary CRS-HIPEC. These findings also align with previous meta-analyses, indicating that selected patients may benefit from reCRS-HIPEC when feasible. 37
The efficacy of HIPEC in improving the survival of pmCRC patients remains uncertain. The PRODIGE 7 trial did not show a significant survival benefit of oxaliplatin-based HIPEC compared to CRS alone. 14 However, the wide variability in HIPEC regimens and patient selection complicates the generalizability of the trial results to the overall effectiveness of HIPEC. After PRODIGE 7, concerns have been raised that its results could lead to an increase of CRS performed in non-HIPEC centers, decreasing their case load. This in turn could impair the outcomes, since complete tumor removal with CRS has an extensive learning curve with outcomes improved in centers of experience above 100 procedures.24,38 Also, the annual caseload (>31 procedures per year) has been associated with reduced postoperative mortality. 24 In our center, we have gradually operated on older patients and on patients with higher ASA class since the establishment phase, while postoperative complication rates have remained stable. Furthermore, operative duration has decreased, more patients have received protective stomas, and hospital stay has shortened. After PRODIGE 7, there has been a decrease in the rate of CRS-HIPEC compared to CRS alone. 39 At the same time, it has been shown that high-volume HIPEC centers perform CRS alone more radical with lower mortality and lower FTRs than low-volume centers or non-HIPEC centers. 39 Therefore, it is likely that also the centralization of CRS without HIPEC could improve patient outcomes in pmCRC. 39 However, in the lack of randomized controlled trials, it is impossible to draw definitive conclusions. Our results are consistent with others and demonstrate that the 5-year OS of resectable pmCRC after CRS-HIPEC approximates 40%. 14 This underscores the importance of thorough assessments for surgical treatment possibilities in these patients. Taken together, we argue that the continued treatment of pmCRC patients in specialized centers is essential.
One strength of our study is the use of the prospective quality registry combined with a comprehensive access to patient records and a national registry offers more complete data coverage compared to conventional registry-based studies. In addition, our patient registry contains consecutive patients of our center without selection bias. Our approach allowed for the detection of all minor hospital complications. Survival information obtained from Statistics Finland ensured that there was no missing data in the OS analysis.
This study has several limitations. The majority of patients were referred to our center from other hospitals. Thus, part of the follow-up visits has been performed at the referring units, partly based on patient preferences. This may introduce bias in disease-free and disease-specific survival outcomes, as not all recurrences may have been reported to us. Furthermore, due to the lack of a control group, we can only compare our results to those of other centers and to historic cohorts. In addition, due the relatively small sample size, the generalizability of our findings is limited, especially for the smaller subgroups. Unfortunately, we have not prospectively registered those patients who were considered for CRS-HIPEC but disregarded after consultation or MDT, so we cannot present the ratio of patients who underwent surgery after being referred to OUH. It is therefore difficult to tell how patient selection may have influenced the outcomes.
Conclusion
Our results demonstrate that patients undergoing CRS-HIPEC represent a heterogeneous group. CRS-HIPEC is associated with good OS and DFS in PMP, whereas survival after both pmCRC and pmAppCa is considerably lower. The procedure is associated with a considerable risk of severe complications and should therefore be performed in specialized centers only.
Supplemental Material
sj-docx-1-sjs-10.1177_14574969251397325 – Supplemental material for Cytoreductive surgery and HIPEC: A 10-year single-center retrospective cohort study
Supplemental material, sj-docx-1-sjs-10.1177_14574969251397325 for Cytoreductive surgery and HIPEC: A 10-year single-center retrospective cohort study by Jukka M. Rintala, Marjo Koskela, Pekka Peroja, Heikki Huhta, Juha Saarnio, Vesa-Matti Pohjanen and Heikki Takala in Scandinavian Journal of Surgery
Footnotes
Authors contributions
Jukka M. Rintala: designing the work, analysis and interpretation of data, drafting of the article, revisiting the article critically for important intellectual content, approving the version to be published.
Marjo Koskela: designing the work, analysis and interpretation of data, revisiting the article critically for important intellectual content, approving the version to be published.
Pekka Peroja: designing the work, analysis and interpretation of data, revisiting the article critically for important intellectual content, approving the version to be published.
Heikki Huhta: designing the work, analysis and interpretation of data, revisiting the article critically for important intellectual content, approving the version to be published.
Juha Saarnio: designing the work, revisiting the article critically for important intellectual content, approving the version to be published.
Vesa-Matti Pohjanen: designing the work, revisiting the article critically for important intellectual content, approving the version to be published.
Heikki Takala: designing the work, analysis and interpretation of data, revisiting the article critically for important intellectual content, approving the version to be published.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: HH: (grant no. 00210395) Finnish Culture Foundation, https://skr.fi/en (grant no. 202210072); Mary and George C. Ehrnroot Foundation, https://marygeorg.fi/en/home/ (grant no. 5785); Finnish Medical Foundation, https://laaketieteensaatio.fi/en/home/; Sigrid Jusélius Foundation, ![]() . None of the funding sources had involvement in study design; in the collection, analysis, and interpretation of data; in the writing of the report; and in the decision to submit the article for publication.
. None of the funding sources had involvement in study design; in the collection, analysis, and interpretation of data; in the writing of the report; and in the decision to submit the article for publication.
Ethical approval
The study was conducted in accordance with the Declaration of Helsinki. The study was approved by the Scientific Committee of the Northern Finland Hospital District (84/2017) and individual consent for this observational study was waived.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
