Abstract
Introduction:
Implementing new treatment regimens entails a learning phase with a concomitant risk of suboptimal results. High caseload has generally been shown to improve results in complex surgical procedures. This study aimed to evaluate results of Cytoreductive Surgery (CRS) and Intraperitoneal Chemotherapy (IP), given as Early Postoperative Intraperitoneal Chemotherapy (EPIC) or Hyperthermic Intraperitoneal Chemotherapy (HIPEC) in a mid-size volume center, stratified by time periods.
Method:
This retrospective single-center cohort study included all CRS-IP procedures in Malmö, Sweden. All data were retrieved from a prospective quality control registry. Primary outcomes were: 5-year overall survival (5-Yr OS) and 30-day severe and total postoperative complication rates. Secondary outcomes were: recurrence rate, 3-year disease-free survival (DFS), and 90-day mortality rate. Outcomes were analyzed by time periods divided into initiating phase (n = 56) and established phase (n = 208).
Results:
In total 264 CRS-IP in 240 patients were performed from 2004 to 2021 (35 EPIC, 229 HIPEC). The 5-Yr OS after primary CRS-IP was 43% in colorectal cancer (n = 169) and 87% in pseudomyxoma (n = 53). In pmCRC, the 5-year OS was significantly lower in the initiation phase (29%) compared to the established phase (48%), verified in an adjusted multivariable analysis showing a hazard ratio of 0.51 for mortality in the established phase. The risk of recurrence did however not differ. Total 30-day complication rate was 67%, severe complication rate 15% and 90-day mortality was 1%. None of which differed depending on time period in multivariable analysis.
Conclusion:
Our results for both pseudomyxoma peritonei (PMP) and pmCRC are in line with previously published results, implying that high volume is not a prerequisite for high-quality CRS-IP. Although long-term survival in pmCRC was significantly better in the established phase, recurrence rates remained unchanged. Further, postoperative mortality and risk of complications did not differ between time periods, suggesting that the impact of the learning curve was limited.
Keywords
Context and Relevance
Peritoneal metastasis of colorectal origin (pmCRC) presents a significant clinical challenge, historically associated with poor prognosis. The introduction of cytoreductive surgery (CRS) combined with intraperitoneal chemotherapy (IP), either as Early Postoperative Intraperitoneal Chemotherapy (EPIC) or Hyperthermic Intraperitoneal Chemotherapy (HIPEC), has significantly improved survival outcomes. While high-volume centers have been associated with better results, establishing new centers, especially low or mid-size centers, remains critical for increasing patient accessibility to these advanced treatments. However, concerns regarding the learning curve and the potential for suboptimal oncological outcomes during the early phases of implementation persist. This study evaluates the long-term oncologic and postoperative outcomes of CRS and HIPEC in a Swedish mid-volume center over time, addressing the impact of the learning curve and providing insights into the feasibility of achieving favorable results in mid-size centers.
Introduction
Peritoneal metastases originating from colorectal cancer (pmCRC) account for 25% of all colorectal cancer metastases, 1 affecting 4%–19% of colorectal cancer patients, 2 either as synchronous or metachronous metastases. pmCRC has very poor survival with best supportive care or systemic chemotherapy alone. Following the introduction of CRS combined with IP either as Early Postoperative Intraperitoneal Chemotherapy (EPIC) or Hyperthermic Intraperitoneal Chemotherapy (HIPEC) in the 1990s, 3 significantly improved survival rates have been reported in selected patients. Comparative studies have shown an improvement of median survival from 13 to 24 months with systemic chemotherapy alone to 22 to 63 months following CRS-IP. 4 – 7 For patients with pseudomyxoma peritonei (PMP), CRS-HIPEC is considered the standard of care, offering the best chance for long-term survival and, in selected cases, potential cure.
Nevertheless, the procedure carries a high risk for complications8,9 and recurrences. 10 Due to the intricacy of CRS-HIPEC procedures, only a few specialized centers performed the procedure initially. 11 However, about 15 years ago, CRS-HIPEC gained recognition as an efficient therapeutic option, especially for patients with PMP and patients with pmCRC,12,13 leading to a need for new centers. Starting up these centers raised the issue of learning curves and which caseload was necessary to achieve good short-term and long-term results.
Although several studies indicate that a high caseload and high center volume are associated with superior outcomes in complex oncologic procedures,14,15 there is no consensus on the optimal caseload for CRS/HIPEC centers. Setting up an HIPEC center without jeopardizing patient safety and ensuring acceptable outcomes is a delicate task, especially in low- and medium-volume centers. The objective of this study was to evaluate the results in a medium-volume center from start-up, including the impact of the learning curve, with a focus on postoperative complications and long-term oncological results.
Methods
This retrospective single-center cohort study was based on prospectively collected data since the start of CRS in 2004 at Skåne University Hospital, Malmö, Sweden. Initially, CRS was combined with EPIC plus sequential postoperative intraperitoneal chemotherapy (SPIC) and later with HIPEC. The registry started for quality assurance purposes by prospectively collecting data on patient and tumor characteristics, perioperative data, and follow-up. Registry data was completed with medical chart reviews if needed.
Patient selection
All patients, irrespective of histology, who underwent CRS combined with EPIC plus SPIC or HIPEC at Skåne University Hospital, Malmö, from the start in April 2004 until 31 December 2021, were included. All prophylactic HIPECs and “open-and-close” procedures were excluded. Throughout the study period, CRS-IP was offered to fit patients with peritoneal metastases considered curatively resectable with preservation of at least 1.5 m of small intestine (2 meters if no remaining colon). In pmCRC, a peritoneal carcinomatosis index (PCI) score > 20 was regarded as a relative contraindication, particularly if major resections were required. Since 2013, patients have been routinely assessed using the Karnofsky Performance Status, with a score below 70 regarded as a relative contraindication. The presence of synchronous non-peritoneal metastases was considered a contraindication until 2010. Prophylactic HIPEC procedures were discontinued in 2018. A CRS-IP team was established from the start. At least two of the four surgeons involved in the team over the whole period performed all operations together.
Patients were followed up with computed tomography (CT) scans of the abdomen and thorax together with serum tumor markers at 6, 12, 18, 24, 36, 48, and 60 months and with clinical examinations on demand. The PMP cases were further followed every 24 months up to 10 years altogether. Medical files of all cases have been scrutinized until May 2025, resulting in a median follow-up time of 19 months for recurrence and 52 months for survival.
Data retrieved were: baseline characteristics including age, sex, synchronous or metachronous presentation, histology, type of intraperitoneal chemotherapy regimen (i.e. EPIC + SPIC or HIPEC); type of chemotherapy and dose; PCI score; complete cytoreduction (CC) score; blood loss; duration of surgery; and postoperative complications. Starting April 2014, all complications were prospectively graded according to the Clavien-Dindo (CD) classification, 16 whereas this was done retrospectively for all cases before 2014. Furthermore, data on survival and recurrences were retrieved from the registry and updated from medical files at the time of data collection.
The primary endpoints were: Five-year overall survival (5-Yr OS); 30 day total and severe (CD-grade ⩾ 3b) postoperative complication rate. Secondary endpoints were: recurrence rate, 3-year disease-free survival (DFS), and postoperative mortality within 90 days. Cases lost to follow-up before 5 years from operation were censored at the last follow-up. Risk factors for complications, mortality, and recurrence were estimated by multivariable analyses.
To assess the impact of the learning curve, the material was divided into two time periods, the initiating phase (2004–2013) and the established phase (2014–2021). During the initiating phase, only patients from the region of Skåne were operated on. In 2013, an evaluation of the outcomes resulted in a general acceptance of the procedure, leading to referrals from the whole southern part of Sweden during the established phase. With around 30 cases performed annually during the established phase, the center has medium-volume. To describe the influence of time, irrespective of the two time periods, we also present scatter smoothing graphs. PMP cases were originally classified according to Ronnet 17 but later as low grade or high grade according to the PSOGI guidelines, 18 following the international shift.
Statistical analysis
Categorical variables are presented as numbers and proportions, and group comparisons were analyzed using the Chi-2 test. Continuous variables are presented as median with interquartile range (IQR) and group comparisons were made using the Mann–Whitney U-test or the Kruskal–Wallis’ test. No data imputation was used. Cases with missing data were excluded from the actual analysis. Multiple logistic regression was used to calculate odds ratio for severe postoperative complications, whereas the Cox’s proportional hazard model was used to calculate relative risks (i.e. Hazard Ratios, HR) for recurrence and long-term mortality. Besides the time period, previously described risk factors and variables with a p-value ⩽ 0.2 in the univariable analysis were included in the multivariable analysis, except for associated variables. Analyses including all variables were also performed as a two-step sensitivity analyses, presented in Supplementary Tables 1–3. No formal correction for multiple testing was applied.
Kaplan-Meier (KM) curves were used to describe OS and recurrence rates, starting at the date of the CRS-IP operation. The 5-year survival estimates and median OS for each time period were obtained from the Kaplan–Meier survival curves using the survival probability at the corresponding time point. Log-rank test was used for comparisons based on KM curves. In cases who received a re-HIPEC, only the index operation was included in the survival and recurrence analysis. Hence, OS was calculated starting at the first CRS-IP procedure. For learning curve analyses, local polynomial smooth graphs were used (Stata version 19, StataCorp LLC, Texas, USA). All other statistical analyses were performed using SPSS version 28 (IBM Corp., Armonk, NY, USA).
This study was approved by the Swedish Ethical Review Authority, dnr 2020-03504, which waived the need for patient consent. It was conducted in accordance with the Declaration of Helsinki. Results are reported according to the STROBE guidelines. 19
Results
Demographic and operative data
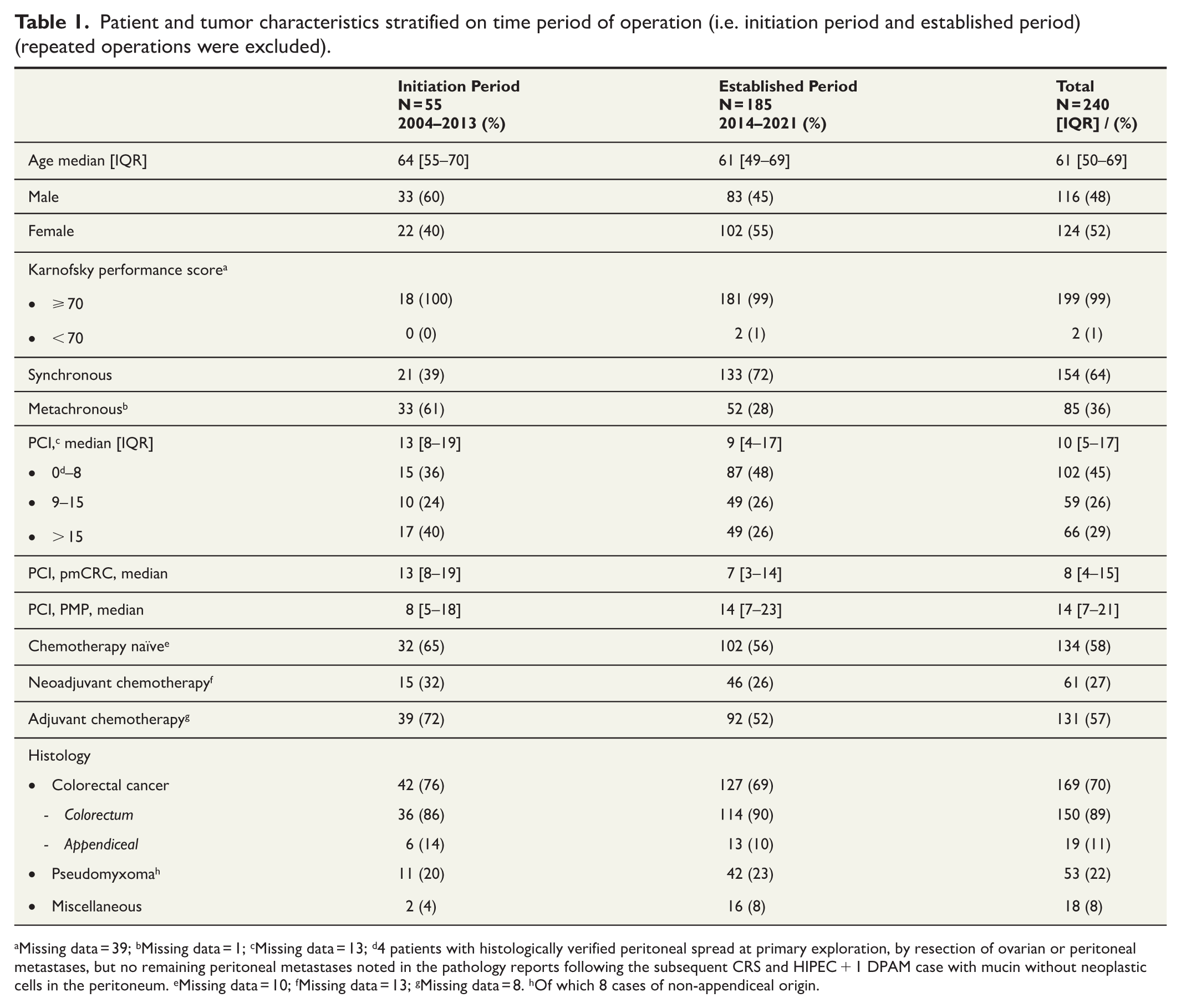
A total of 315 procedures were identified. Of these, 51 interventions were excluded, being either prophylactic HIPEC or open and close procedures, leaving 264 surgical interventions in 240 patients included in the study (Fig. 1). The median age at surgery was 61 years (initiation period 64 years, established period 61 years). Overall, 116 patients (48%) were male and 124 (52%) were female. In the initiation, 9% of patients, and in the established phase, 91% were classified according to the Karnofsky performance status. It was ⩾ 70 in 99% of patients. A score of 100 (maximum score) was observed in 71% of all patients. The median PCI score was 10 (8 in pmCRC and 14 in PMP). The median PCI score in the initiation phase was 13 (13 in pmCRC and 8 in PMP) and did not differ statistically significantly from the median PCI of 9 in the established phase (7 in pmCRC and 14 in PMP). Otherwise, no major differences were noted between the groups (Table 1). Baseline characteristics stratified by pmCRC and PMP are presented in Supplementary Table 1A and B.
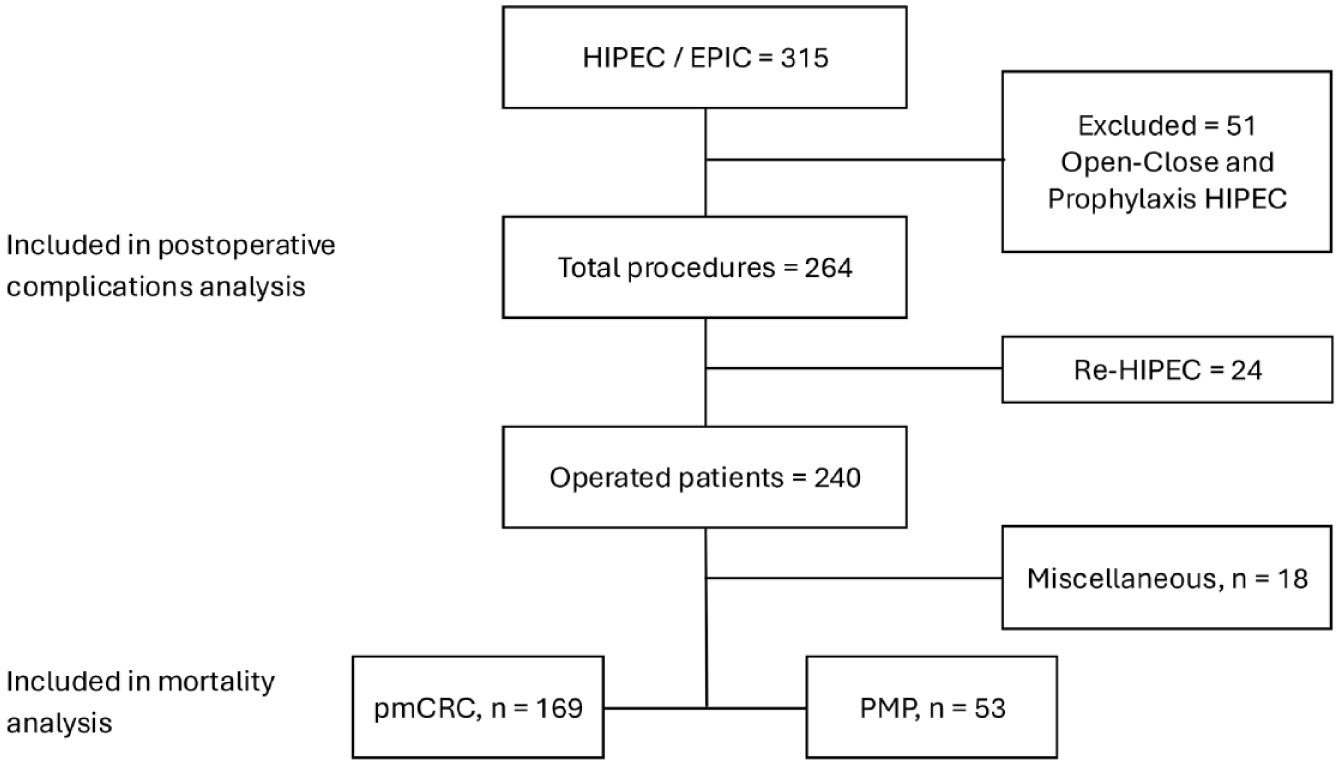
Flowchart of patient selection, exclusions, and tumor type distribution.
Patient and tumor characteristics stratified on time period of operation (i.e. initiation period and established period) (repeated operations were excluded).
Missing data = 39; bMissing data = 1; cMissing data = 13; d4 patients with histologically verified peritoneal spread at primary exploration, by resection of ovarian or peritoneal metastases, but no remaining peritoneal metastases noted in the pathology reports following the subsequent CRS and HIPEC + 1 DPAM case with mucin without neoplastic cells in the peritoneum. eMissing data = 10; fMissing data = 13; gMissing data = 8. hOf which 8 cases of non-appendiceal origin.
During the initiation phase (2004 to 2013), 56 CRS-IP operations were performed (one case of re-HIPEC), with a mean of 5.6 operations per year (range = 1–15). During the established phase (2014 to 2021), 208 CRS-HIPEC operations were performed (23 cases of re-HIPEC), with a mean of 27 operations (range = 21–34) performed annually.
Of the 240 included patients, 169 had pmCRC (including 19 with appendiceal origin); 53 PMP (of which 37 were DPAM/low-grade PMP and 16 PMCA/PMCA-i/high-grade PMP) and 18 miscellaneous (mesothelioma, small intestine, urachus, fallopian tube, unknown origin) (Table 1). In total, 64% had synchronous peritoneal metastasis and 36% had metachronous peritoneal metastasis.
Neoadjuvant chemotherapy was administered to 61 patients (27%), 50 (31%) of those in pmCRC and 5 (9%) in PMP. In total, 131 (57%) received adjuvant treatment after surgery, 118 (72%) in pmCRC, and 5 (10%) in PMP. A total of 134 patients (58%) were chemo-naive before CRS, and 42% were never treated with systemic chemotherapy at any time during the disease.
Operative data are shown in Table 2. The first 35 patients were operated with CRS combined with EPIC plus SPIC. In 2013, the routine was changed to HIPEC. Oxaliplatin-based HIPEC has predominantly been used for pmCRC, and Mitomycin C for PMP, with some alterations. A total of 24 re-HIPEC procedures (9%) were performed for intraperitoneal recurrence (15 pmCRC, 7 PMP, 2 miscellaneous). One of which was in the initiation phase, whereas 23 cases (96%) were in the established phase. All re-operations involved HIPEC and no EPIC, Supplementary Table 2. Besides peritonealectomies, bowel resections were performed in 202 (84%) of the cases, and major organ resections (liver, stomach, diaphragm, pancreas, or spleen) in 67 (32%) of the cases. Complete cytoreduction (CC0) was achieved in 95% of the cases, without any differences between the two time periods. The median length of operation was 611 min (495–686) in the initiation phase and 612 min (524–725) in the established phase. Median intraoperative blood loss was 800 mL, which decreased from 900 mL to 700 mL between the two time periods (p < 0.01). Temporal development of length of operation and intraoperative bleeding is depicted in Supplementary Figure 1A and B, showing a trend toward less bleeding but otherwise minor fluctuations.
Operative characteristics (primary operation), stratified by time period of operation (i.e. initiation phase and established phase).
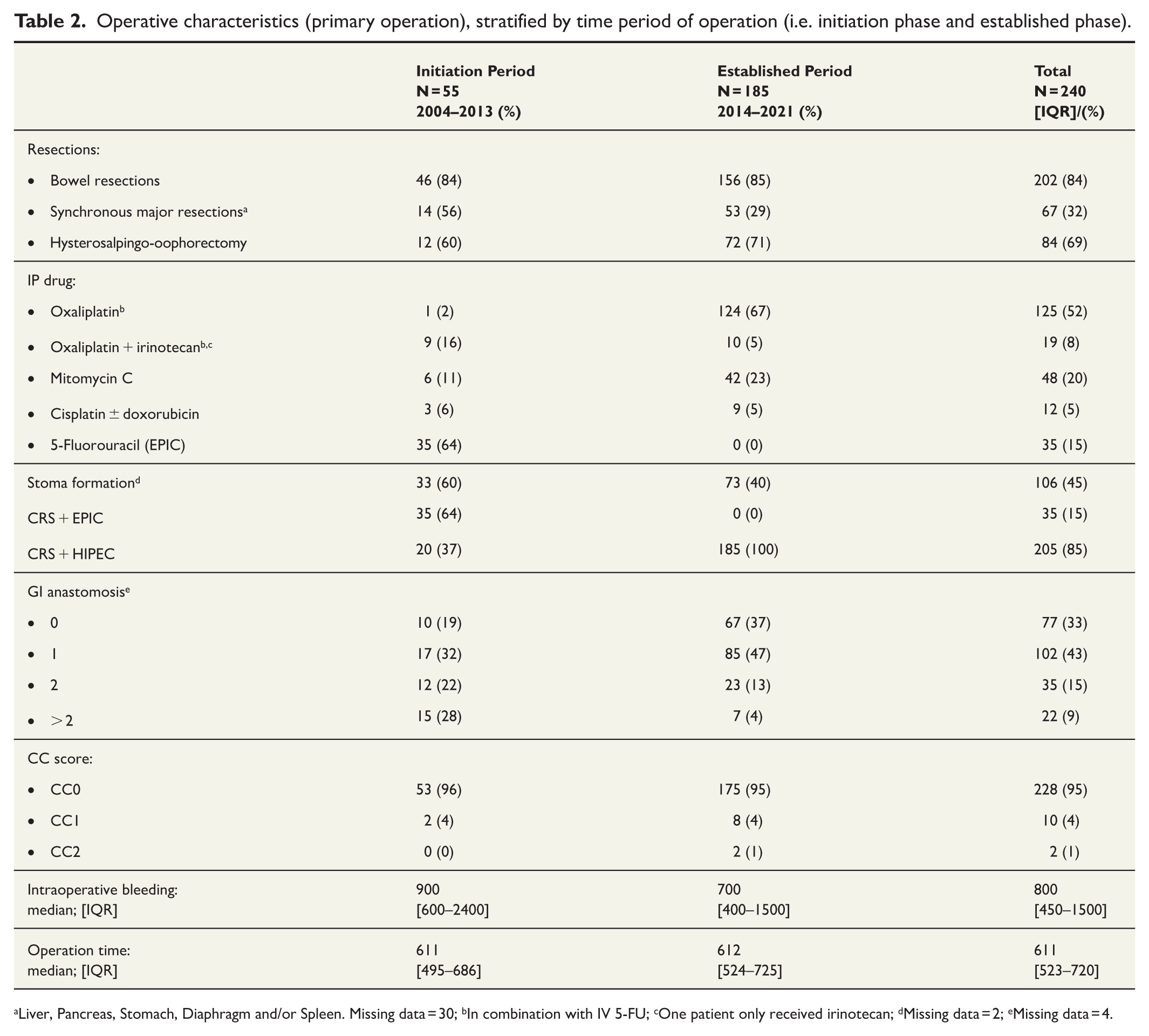
Liver, Pancreas, Stomach, Diaphragm and/or Spleen. Missing data = 30; bIn combination with IV 5-FU; cOne patient only received irinotecan; dMissing data = 2; eMissing data = 4.
Postoperative morbidity and mortality
Postoperative complications within 30 days, including re-HIPEC cases, occurred in 178 cases (67%), whereas serious complications (Clavien-Dindo ⩾ 3b) were noted in 39 cases (15%) (Table 3). Total overall complication rates were significantly higher in the established phase (p = 0.005), whereas severe postoperative complications were more frequent in the initiation phase (p = 0.002). Re-operation due to complications was performed in 34 patients (13%), due to bleeding (n = 9), anastomotic leaks (n = 7), bowel perforation (n = 5), and other reasons (n = 13), such as wound dehiscence or intestinal obstruction. One patient died due to cerebral infarction on postoperative day 5, and two more patients died within 90 days due to multiple complications originating from kidney failure with subsequent sepsis and late bleeding with subsequent sepsis and bone marrow depression, respectively. This corresponds to a 90-day mortality of 1.1% (Table 3).
Postoperative complications within 30 days and 90-day mortality stratified by time period of operation (i.e. initiation phase and established phase), including risk analysis (re-HIPEC procedures included).
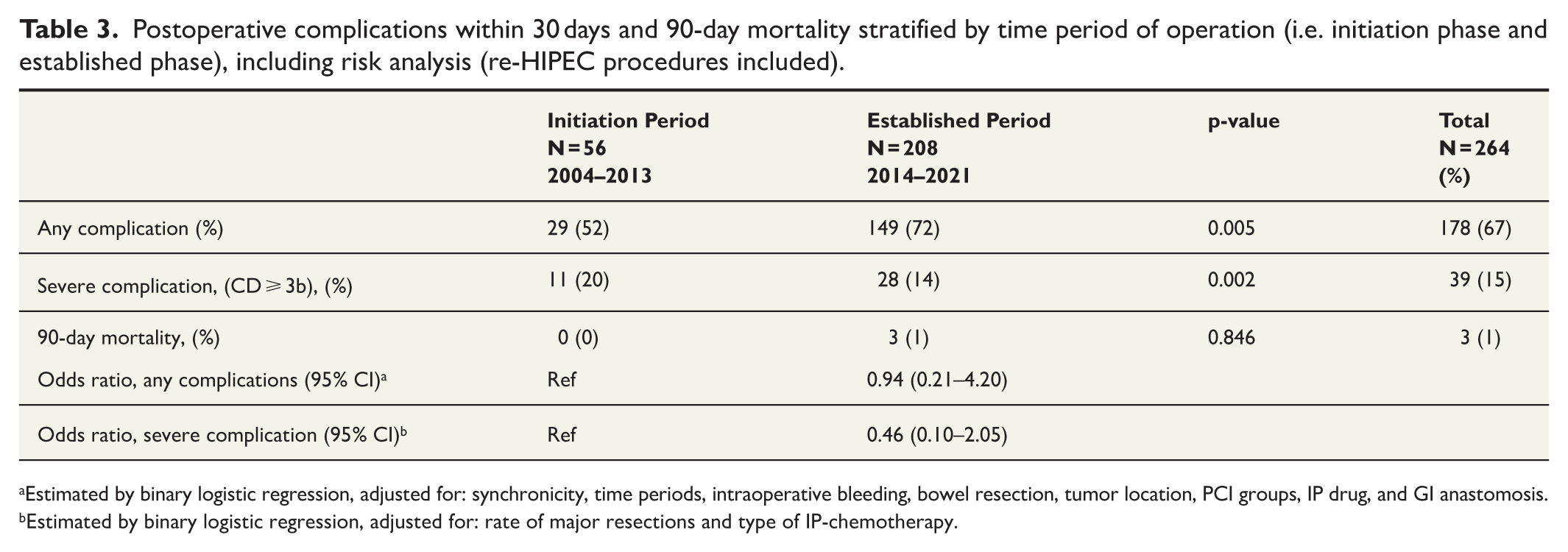
Estimated by binary logistic regression, adjusted for: synchronicity, time periods, intraoperative bleeding, bowel resection, tumor location, PCI groups, IP drug, and GI anastomosis.
Estimated by binary logistic regression, adjusted for: rate of major resections and type of IP-chemotherapy.
Univariable and multivariable logistic regression analyses showed that major resections and use of Cisplatin at HIPEC were risk factors for severe complications. Time period of operation was not a risk factor for complications or severe postoperative complications. The temporal development of complication rate in relation to CD classification is depicted in Fig. 2, showing some fluctuations but no obvious trend.
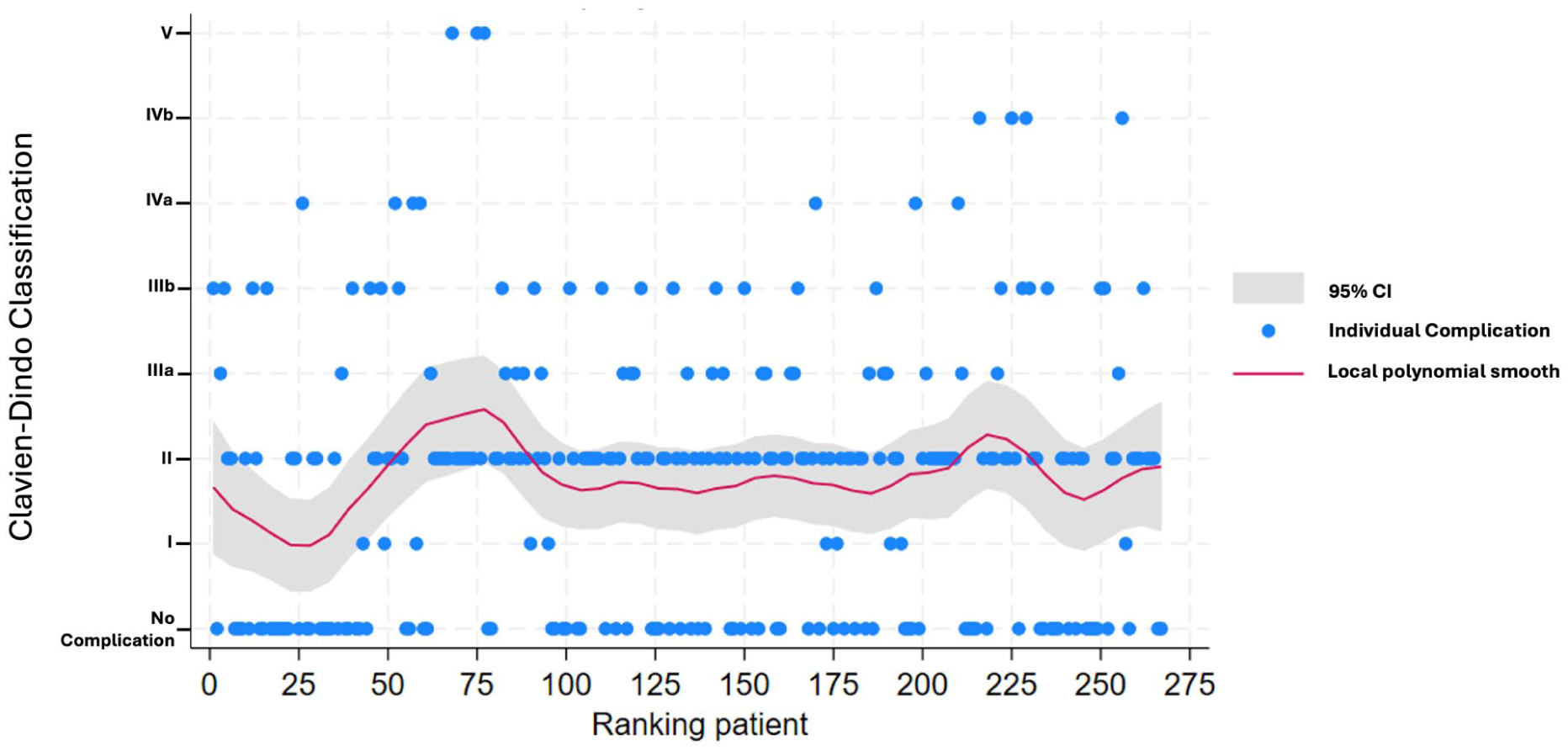
Development over time presented as scatter smoothing graphs in the rate of complications in relation to CD classification.
Survival and recurrence
Median estimated 5-year OS (median follow-up = 52 months) was 43% in colorectal cancer (median OS = 49 months) and 87% in PMP (Fig. 3A and B). For pmCRC, there was a statistically significant increase in 5-year OS from 28.6% in the initiation phase to an estimated 47.5% in the established phase (p = 0.008). This finding was confirmed in multivariable analysis showing a decreased risk of mortality in pmCRC in the established phase (HR = 0.51 (0.31–0.84)). Age, PCI > 8, postoperative complications, and nodal positivity of the primary tumor were noted as risk factors for mortality (Table 4). Sensitivity analysis revealed no significant differences compared to these findings (Supplementary Table 3). The Kaplan-Meier curves on pmCRC stratified by colorectal and appendiceal origin are presented as Supplementary Figure 2.
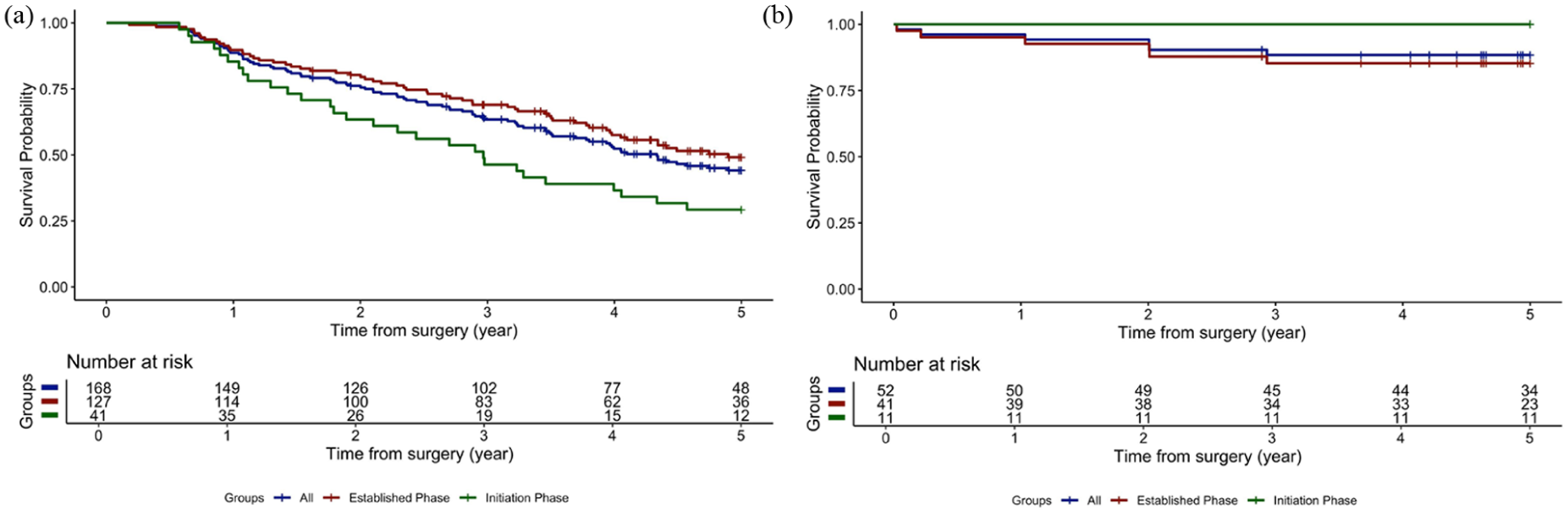
5-Year overall survival, presented as Kaplan-Meier curves, stratified on time period of operation (re-HIPEC excluded): (A): Colorectal cancer: 5-Year OS: 42.8%; in the initiation phase 28.6%, and 47.5% in the established phase. Median survival 49 months. (B): Pseudomyxoma: 5-Year OS: 87.2%; median survival not reached.
Risk of mortality within 5 years in pmCRC following CRS and EPIC or HIPEC (n = 169), using multivariable Cox proportional hazard model.
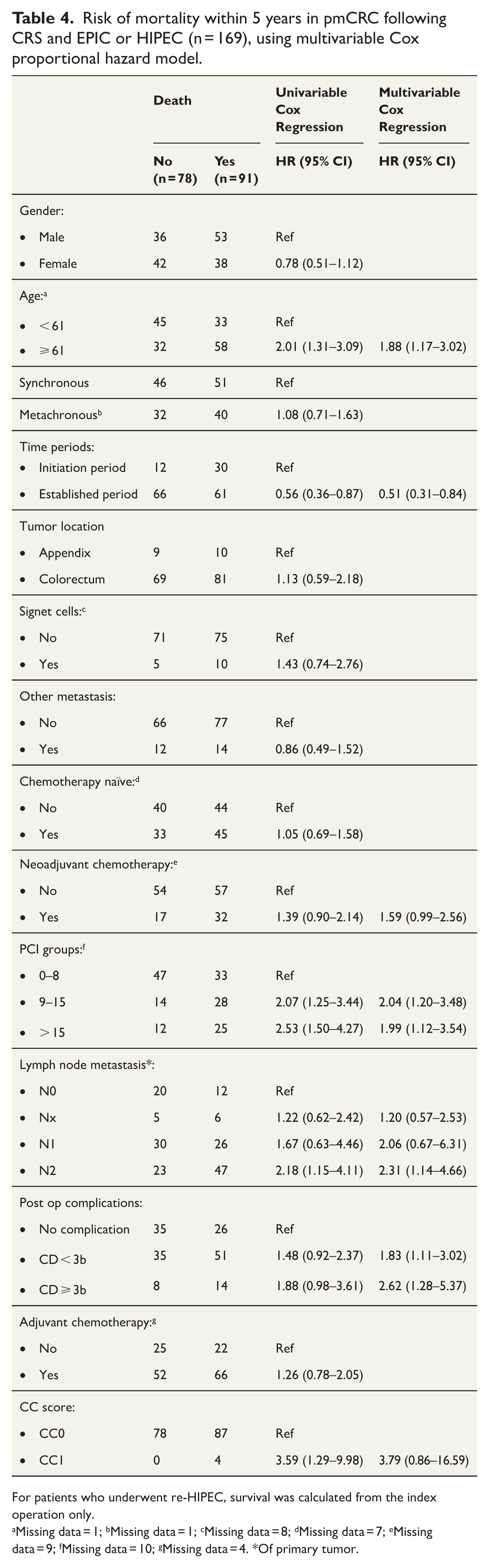
For patients who underwent re-HIPEC, survival was calculated from the index operation only.
Missing data = 1; bMissing data = 1; cMissing data = 8; dMissing data = 7; eMissing data = 9; fMissing data = 10; gMissing data = 4. *Of primary tumor.
The recurrence rate (CC0 only, no re-HIPECs) in colorectal cancer was 70%, with a median follow-up time of 19 months (18 months in the initiation phase and 19 months in the established phase). In multivariable analysis, nodal positivity of the primary tumor was identified as a risk factor of recurrence, whereas PCI > 15, other concomitant metastases, and colorectal origin (as opposed to appendiceal) were borderline significant risk factors (Table 5). Operation in the initiation phase showed no increase in risk of recurrence. The sensitivity analysis revealed no major differences compared to these findings (Supplementary Table 4).
Risk of recurrence within three years for pmCRC following CC0 CRS with EPIC or HIPEC (n = 159), estimated using Cox proportional multivariable hazards model.
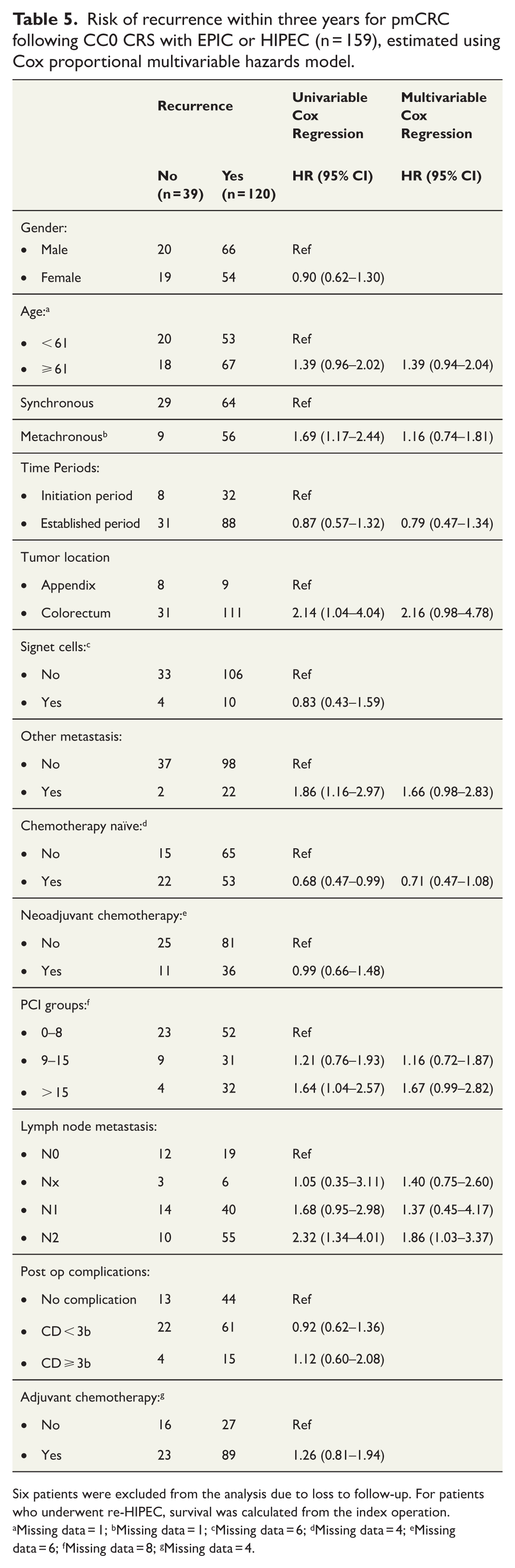
Six patients were excluded from the analysis due to loss to follow-up. For patients who underwent re-HIPEC, survival was calculated from the index operation.
Missing data = 1; bMissing data = 1; cMissing data = 6; dMissing data = 4; eMissing data = 6; fMissing data = 8; gMissing data = 4.
The 3-year DFS for pmCRC was 25.5% with a median DFS of 14.3 months, whereas it was 86% in the PMP group. There was no statistically significant difference between time periods in 3-year DFS (Log Rank, p = 0.51). Temporal development of recurrences is presented in Fig. 4, confirming this result.
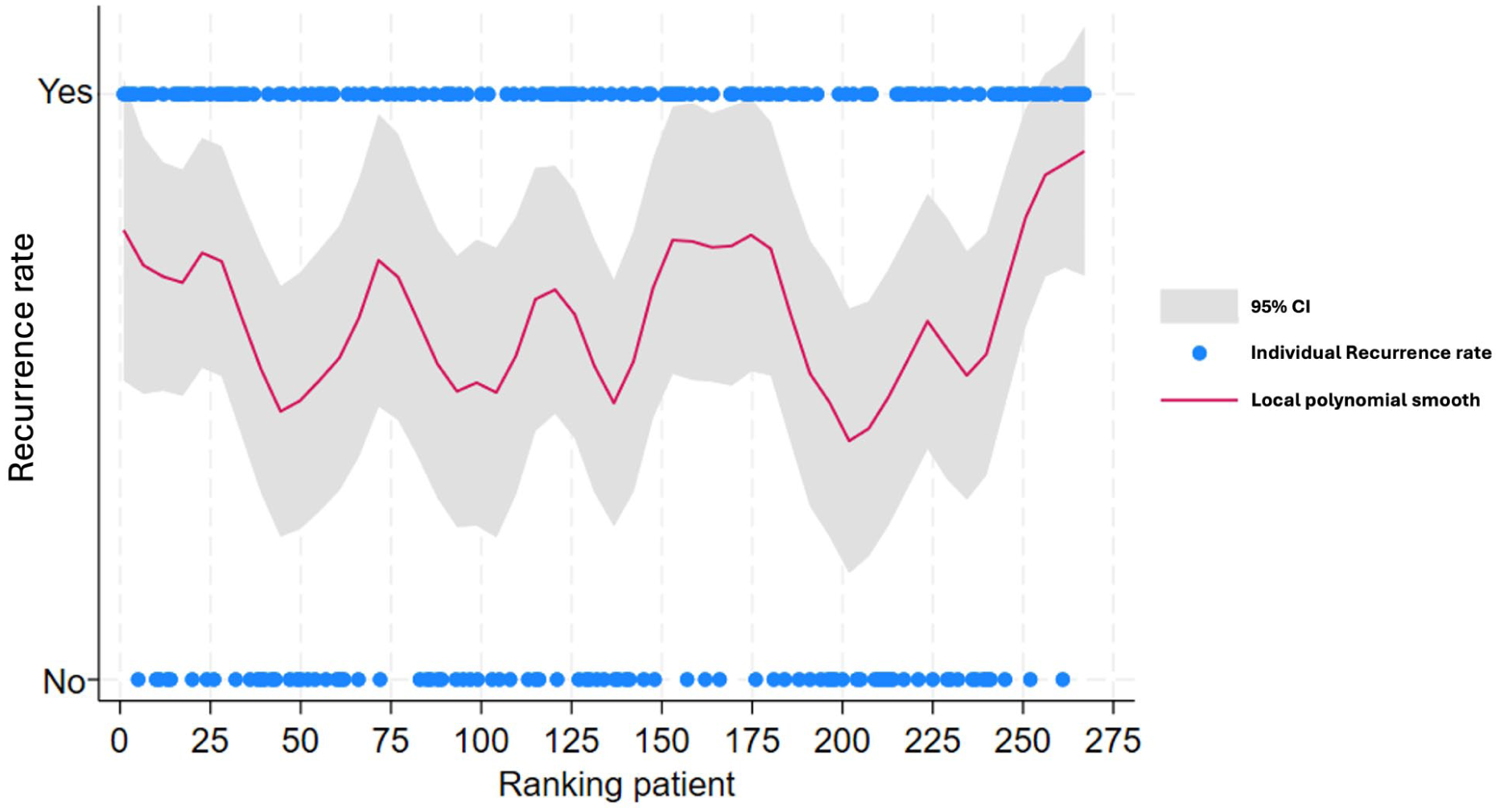
Development over time presented as scatter smoothing graphs in recurrence rate in colorectal cancer.
Discussion
This single-center cohort study of patients treated with CRS-IP showed a 5-year OS of 43% in pmCRC and 87% in PMP. In comparison, a systematic review published in 2022, summarizing overall survival in 16 studies, showed that 5-year OS for pmCRC ranged from 0% to 48.5%. 20 Furthermore, a multicentric study of 523 CRS-IP procedures for pmCRC showed a 5-year OS of 27%, whereas a Dutch study of 433 pmCRC patients reported a 5-year OS of approximately 25%. 21 A more recent national register study from Spain on 1716 HIPEC procedures reported a 5-year OS of 35.6% in pmCRC and 74% in PMP. 22
In the present study, age, PCI > 8, nodal positivity of the primary tumor, and postoperative complications were identified as risk factors for long-term mortality, which is in line with previous work.9,23,24 The pmCRC group during the established phase had a lower mortality compared to the initiation phase (OS: 48% versus 29%, p = 0.008), verified in multivariable analysis, revealing an HR for mortality of 0.51. However, no difference in recurrence rates or risk of recurrence depending on time period of operation was noted. The reason for this discrepancy is elusive, although improved palliative treatment possibly contributed. Sex and the observed decrease in PCI score and lower age of participants in the established phase as compared to the initiating phase also possibly contribute but were adjusted for in the multivariable analysis; however, still showing some differences. The difference in PCI score reflects that patients were referred in a later phase of the disease in the initiation period, also noted by an over twice as high rate of metachronous disease in this time period. EPIC was predominantly used in the initiation phase, whereas HIPEC was used in all cases in the established phase. HIPEC regimens have changed somewhat during the years according to international trends. These facts hamper somewhat the assessment of which impact the learning curve had on the results. However, most previous studies report quite similar results of EPIC and HIPEC, with possibly just slightly worse outcomes after EPIC.25,26 Our interpretation is that the worse survival of pmCRC patients during the initiation phase most likely is a learning curve effect. However, the initiation phase was quite limited in numbers (n = 55), and survival was comparable to other publications from this time period. 27
Nodal positivity of the primary tumor was the only noted risk factor for recurrence of pmCRC, whereas PCI score above 15, other concomitant metastases, and colorectal origin (as opposed to appendiceal) were borderline significant. The follow-up time does not enable a reliable estimation of the total risk of recurrence of PMP due to the slow evolution of this disease.
The present study showed a severe postoperative complication rate of 15%, whereas the 30-day postoperative mortality rate was 0.4% which is lower than previously reported major postoperative complication rates between 19% and 34% and 30-day mortality between 1.0% and 7.7%.8,28 In a systematic review, postoperative mortality rates of 0.9% to 5.8% and morbidity rates between 12% and 60% were reported. 29 In the multivariable analysis, time period of operation was not a risk factor for complication, whereas major organ resection and the use of Cisplatin at HIPEC were independent risk factors for the development of severe postoperative complications, which is in line with previous publications.30–33
Center volume
Several studies have shown that high center and surgeon volume are associated with superior outcomes in complex oncologic procedures.14,15 This includes reduced postoperative complication rates 34 and lower postoperative and long-term mortality. 35 A systematic review showed that the postoperative complication rates were significantly lower among high-volume surgeons and specialists, whereas hospital volume had less impact. 36 Several retrospective studies have investigated oncological outcomes and postoperative morbidity depending on the size of HIPEC centers.37,38 A large French multi-institutional study found the level of institutional experiences to be a strong prognostic factor for morbidity and mortality. 24 Furthermore, Rajeev et al. reported better outcomes in high-volume centers compared to low-volume centers 39 and speculated that this was likely due to increased surgical volume and experience, exposure to diverse cases, strong mentorship and a collaborative environment. 39 However, Chatani et al. 40 noted no association between hospital volume and postoperative outcomes. The result of the present study supports the finding that a high caseload is not a prerequisite for achieving good results in CRS-HIPEC.
Learning curve
The impact of a learning curve in CRS-HIPEC procedures has been reported in several papers, with somewhat diverging results.41,42 Most studies have noted a learning curve effect, but the numbers needed to reach a plateau have differed. For example, in a 15-year material from Singapore, based on prospectively collected data from 188 patients, a decrease in the rate of severe postoperative complications from 34% in the earliest phase compared to 14% in the latest phase was noted. 43 Kusamura et al. 42 reported a caseload of 140 cases to ensure surgical proficiency, whereas Polanco and colleagues showed that high-volume centers reached gradual improvement in completeness of cytoreduction and OS after 180 and 90 cases, respectively. 44 Whether these figures apply to low- and medium-volume centers is unknown. In the present study, an acceptable but lower long-term survival of pmCRC was noted in the initiation phase consisting of 42 patients. As discussed above, several factors, such as improved palliative treatment of recurrences, including a higher rate of re-HIPEC and refined patient selection, probably contribute to this finding. This also reflects a learning phase, although not so much of surgical technique.
Although there was a continuous improvement during the study period, only a minor difference in intraoperative bleeding (200 mL) was noted comparing the initiation and the established phases and no significant differences in length of operation or postoperative mortality were noted. Furthermore, in the multivariable analysis, time period was not associated with an increased risk of complications.
Besides the individual learning curve, there is a collective collection of experience, making the learning curve shorter and flatter by time due to shared data from the pioneer centers and the scientific community. That is a worldwide learning curve of which we are all part. This is exemplified by Kuijpers et al., who showed that after receiving hands-on training, the performance of new institutions on four outcomes (complete cytoreduction rate, blood loss, length of operation, and morbidity) was better in the first 10 procedures compared to the first 100 procedures in the pioneer hospital. 45 The impact of an external mentor was studied upon establishing a new peritoneal surface malignancy unit, showing high completeness of cytoreduction along with favorable morbidity and mortality rates in the first 50 cases. 46 These results indicate that new HIPEC centers can achieve good oncological outcomes with acceptable postoperative complications despite the low number of cases, utilizing mentorship and collaboration, which is supported by the results in the present study.
A strength of this study is that all consecutive patients undergoing CRS-IP at our center during the study period were included, thereby minimizing selection bias and ensuring that outcomes reflect a complete, real-world cohort. Furthermore, patient selection criteria were stable over time. A limitation of this study is that it is retrospective; however, it is mainly based on prospectively collected data. The retrospective design hampers control of biases and confounding factors, although it is in part compensated for in multivariable analyses. Other limitations are the long study period, which inevitably implies that some routines have been changed during the study, and the relatively small sample size which restricts the ability to perform subgroup analyses and makes it difficult to draw firm conclusions about infrequent outcomes. Finally, the shorter than 5-year follow-up in the last year limits the evaluation of long-term survival for this group.
Taken together, the figures for both short-term and long-term outcomes in this study stand up well with published results in the early and late phases. We speculate that factors contributing to these results were the collective collection of experience and that standardized treatment protocols and a dedicated team were established early on with a total of four consultants performing all surgery and the responsible surgeon (IS) attending virtually all procedures.
Conclusion
The results of the present study show that high caseload is not a prerequisite for satisfactory results of CRS-IP. It is possible to achieve good outcomes in medium-volume peritoneal malignancy centers, and the impact of the learning curve on oncologic and perioperative outcomes can be limited.
Supplemental Material
sj-doc-3-sjs-10.1177_14574969251397312 – Supplemental material for Cytoreductive surgery and intraperitoneal chemotherapy for peritoneal malignancy—outcomes and learning curves in medium volume center
Supplemental material, sj-doc-3-sjs-10.1177_14574969251397312 for Cytoreductive surgery and intraperitoneal chemotherapy for peritoneal malignancy—outcomes and learning curves in medium volume center by N Algethami, V Valdimarsson, H Thorlacius, V Verwaal and I Syk in Scandinavian Journal of Surgery
Supplemental Material
sj-docx-1-sjs-10.1177_14574969251397312 – Supplemental material for Cytoreductive surgery and intraperitoneal chemotherapy for peritoneal malignancy—outcomes and learning curves in medium volume center
Supplemental material, sj-docx-1-sjs-10.1177_14574969251397312 for Cytoreductive surgery and intraperitoneal chemotherapy for peritoneal malignancy—outcomes and learning curves in medium volume center by N Algethami, V Valdimarsson, H Thorlacius, V Verwaal and I Syk in Scandinavian Journal of Surgery
Supplemental Material
sj-docx-2-sjs-10.1177_14574969251397312 – Supplemental material for Cytoreductive surgery and intraperitoneal chemotherapy for peritoneal malignancy—outcomes and learning curves in medium volume center
Supplemental material, sj-docx-2-sjs-10.1177_14574969251397312 for Cytoreductive surgery and intraperitoneal chemotherapy for peritoneal malignancy—outcomes and learning curves in medium volume center by N Algethami, V Valdimarsson, H Thorlacius, V Verwaal and I Syk in Scandinavian Journal of Surgery
Footnotes
Author contributions
N.A. and V.V. wrote the draft and performed statistical analyses. H.T., V.V., and I.S. supervised the study and verified the final version of the manuscript. All authors reviewed and validated the final version of the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the ALF agreement between the Swedish government and the county councils (2018-Project 0264) by an unconditional grant.
The consent statement and clinical trial registration
N/A.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
