Abstract
Background and Aims:
While in the past, thoracotomy represented the traditional surgical approach for the treatment of epiphrenic diverticula, actually mini-invasive approach seems to be the preferred treatment as many series have been published in the recent years. This article describes the authors’ experience with the laparoscopic approach for performing diverticulectomy, myotomy, and Nissen–Rossetti fundoplication.
Material and Methods:
From 1994 to 2010, 21 patients (10 men and 11 women), mean age 58.5 years (range 45–74 years), with symptomatic epiphrenic diverticulum underwent laparoscopic diverticulectomy, myotomy and Nissen–Rossetti fundoplication.
Results:
The mean operative time was 135 min (range = 105–190 min). Mean hospital stay was 14.2 days (range = 7–25 days). In 5 patients (23.8%), a partial suture staple line leak was observed. Conservative treatment achieved leak resolution in all the cases. One patient (4.8%) died of a myocardial infarction in the postoperative period. After a mean clinical follow-up period of 78 months (range = 6–192 months), excellent or good outcome was referred with no dysphagia in 16 patients (80%) and only mild occasional dysphagia in 4 patients (20%).
Conclusions:
Surgical treatment of epiphrenic diverticula remains a challenging procedure also by mini-invasive approach, with major morbidity and mortality rates. For this reason, indications must be restricted only to selected and symptomatic patients in specialized centers.
Keywords
Introduction
Among the motility disorders of the esophagus, epiphrenic diverticula represent an uncommon disease characterized by outpouching of the esophageal lumen in the distal third of the esophagus close to the diaphragm (1–3). The true incidence is unknown as only 15%–20% of patients are symptomatic, while a great number of cases are diagnosed incidentally during radiographic or endoscopic examinations performed for other reasons. In 1833, Mondiere (4) hypothesized for the first time that increased intraluminal pressure could be a pathogenic factor. However, to date, there is no definitive conclusion on the physiopathology of this disease, while diffuse agreement exists about restriction of surgical indications only to symptomatic patients (3, 5–8). Different procedures have been described in literature (9–14). Thoracotomy is the traditional surgical approach, while, in the last 20 years, many authors have published their experience with mini-invasive approach (15–25), and also recently, with robotic-assisted approach (26, 27). Actually, the choice treatment appears to be laparoscopic diverticulectomy with myotomy and antireflux fundoplication.
Some years ago, we reported our experience on laparoscopic diverticulectomy, myotomy and Nissen–Rossetti fundoplication (20). Most authors do not agree with us considering that for this motility disorder, a partial fundoplication could be more appropriate. Aim of this work is to evaluate the long-term results of our choice treatment on a larger group of patients, together with a review of the literature.
Materials and Methods
In the period from January 1994 to March 2010, 21 patients (10 men and 11 women), mean age 58.5 years (range = 45–74 years), affected by symptomatic epiphrenic diverticulum came to our observation. Symptoms referred by the patients were classified, according to frequency, as mild (occasional occurrence), moderate (daily occurrence) and severe (symptoms with each meal). These symptoms were evaluated by a clinical investigation and a written questionnaire. Dysphagia for liquids and solid food was complained by all the patients: it was severe in 19 (90.5%) and moderate in 2 patients. Other symptoms were the following: regurgitation in 17 patients (81.0%; moderate 7, severe 10), chest pain in 16 patients (76.2%; mild 8, moderate 5, severe 3), chronic cough in 8 patients (38.1%; mild 2, moderate 3, severe 3), and pyrosis in 5 patients (23.8%; moderate 2, severe 3). In 8 patients, weight loss of more than 10 kg was observed, while aspiration pneumonia occurred in 4 patients (19.0%). Mean duration of symptoms was 118 months (range = 7–260 months). Preoperative instrumental evaluation included upper gastrointestinal barium meal, gastrointestinal endoscopy, stationary esophageal manometry and 24-h esophageal pH monitoring. Radiological examination showed a mean diameter of the diverticular sac of 47 mm (range 33–61 mm). Motility disorders evaluated by manometry were the following: achalasia in 12 patients (57.1%), hypertensive lower esophageal sphincter (LES) in 4 patients (19.0%), nonspecific esophageal motility disorders in 4 patients (19.0%), and nutcracker esophagus in 1 patient (4.8%).In 5 patients (23.8%), a pathologic gastroesophageal reflux was documented. The intervention was conducted by means of laparoscopic access. In the postoperative period, oral intake of liquids started on day 6 after a gastrografin esophageal and gastric transit. Follow-up was performed on the basis of an outpatient visit for all the patients. Each patient was provided with the same questionnaire given at the entry to evaluate symptoms and clinical outcome. The latter was classified as excellent (absence of symptoms), good (occasional occurrence), fair (daily occurrence), and poor (symptoms with each meal). Instrumental follow-up included endoscopy, upper gastrointestinal barium meal, stationary manometry, and esophageal pH monitoring.
Operative Technique
For laparoscopic management of epiphrenic diverticula, the patient is placed in the reverse Trendelenburg position with the operator standing between the patient’s legs. The first assistant is at the right of the surgeon, and the second is at his left. After pneumoperitoneum induction by a Veress needle introduced in the umbilicus, five trocars are positioned as already described (20). The left liver is lifted up, and the esophagogastric junction is identified. The abdominal esophagus is mobilized from the diaphragmatic pillars. Once a wide retroesophageal space has been created, gentle traction is applied to the esophagus by a retractor. Then, mediastinal dissection of the inferior thoracic esophagus is carried out until the diverticular pouch is reached. Intraoperative endoscopy ensures complete identification of the diverticulum. Ligasure (Valleylab, Boulder, CO, USA) is usually utilized to perform this part of the procedure. After preparation of diverticular pouch is completed, a linear stapler is introduced (Echelon 60 ENDOPATH® stapler, Ethicon Endo-Surgery Inc. or Endo-GIA™ USSC, Norwalk, CT, USA, blue cartridge). Once the stapler jaws are closed, endoscopy verifies the presence of an adequate esophageal lumen. The staple is fired (Fig. 1), and the integrity of the suture line is checked always under endoscopic assistance. Most of the diverticular pouches in the current series required a second staple firing to complete the diverticulectomy. After diverticulectomy, myotomy is performed below the inferior margin of the staple suture line and prolonged for 2 cm on the anterior gastric wall (Fig. 2). Endoscopy checks the absence of mucosal tears, whereas intraoperative esophageal manometry confirms the correct execution of myotomy (resting LES pressure < 4 mmHg) (20, 28, 29). For all patients, an antireflux procedure is performed at the end of the intervention to prevent pathologic acid gastroesophageal reflux (Fig. 3). We routinely perform a Nissen–Rossetti fundoplication without section of short gastric vessels or esophagus transfixing stitches (20, 28, 29). At the end of the intervention, endoscopy verifies the regular transit of the esophagogastric junction and the correct wrap realization. A further manometry measures the new high-pressure zone (HPZ), in which values should range between 20 and 40 mmHg. Whenever these values are out of range, the wrap is taken down and a new fundoplication is performed.
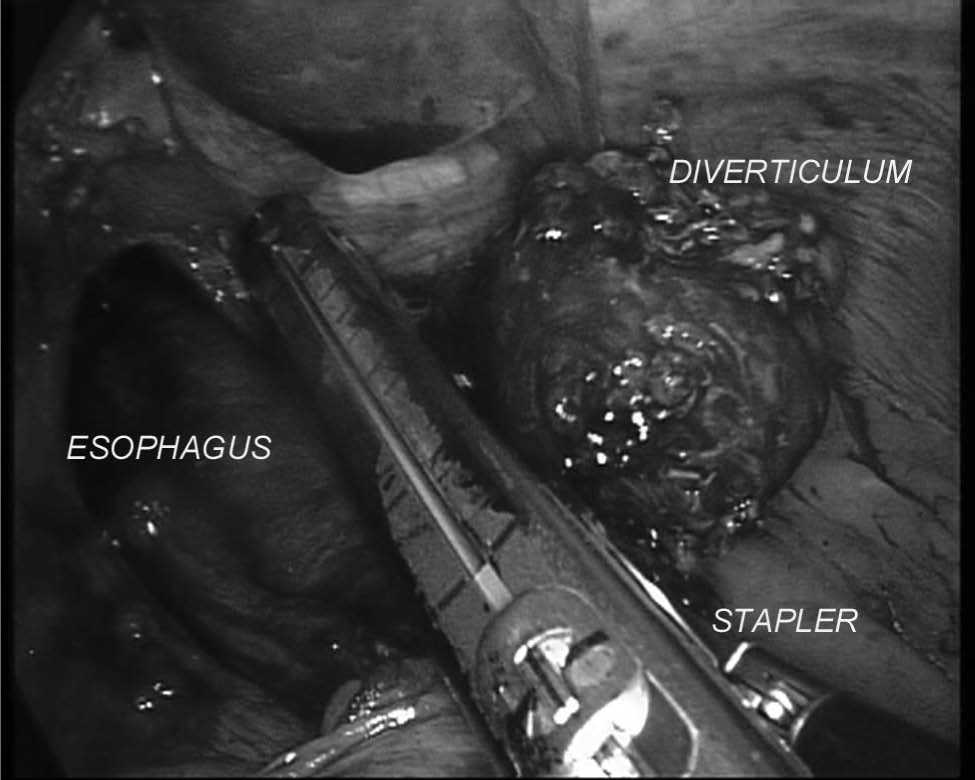
Stapled diverticulectomy.
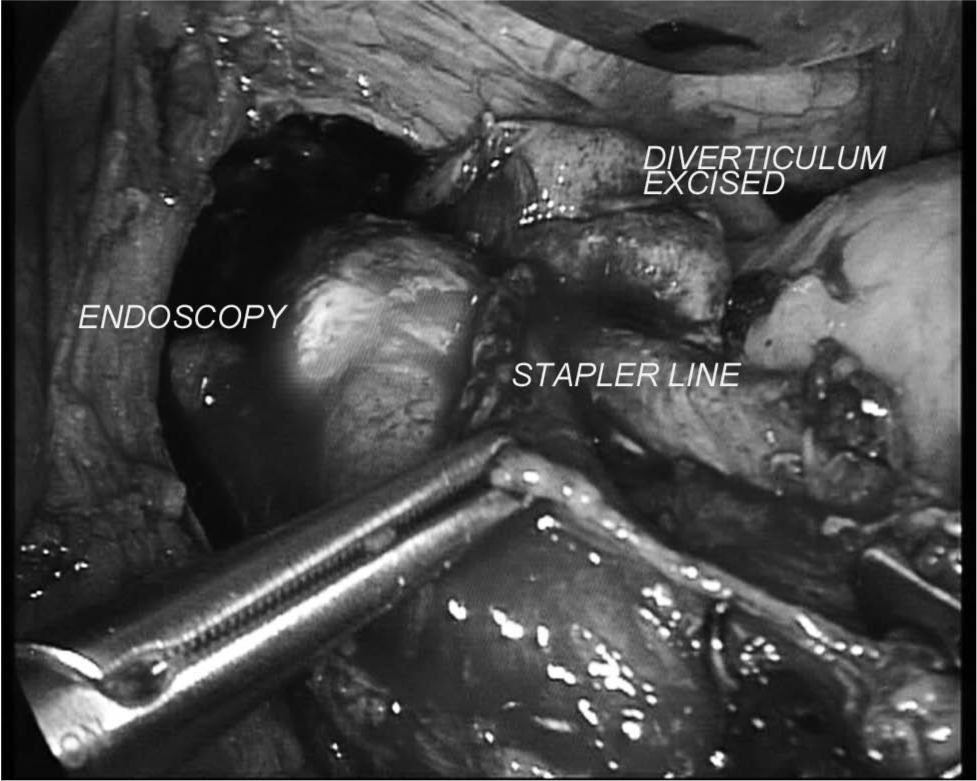
Esophagogastric myotomy.
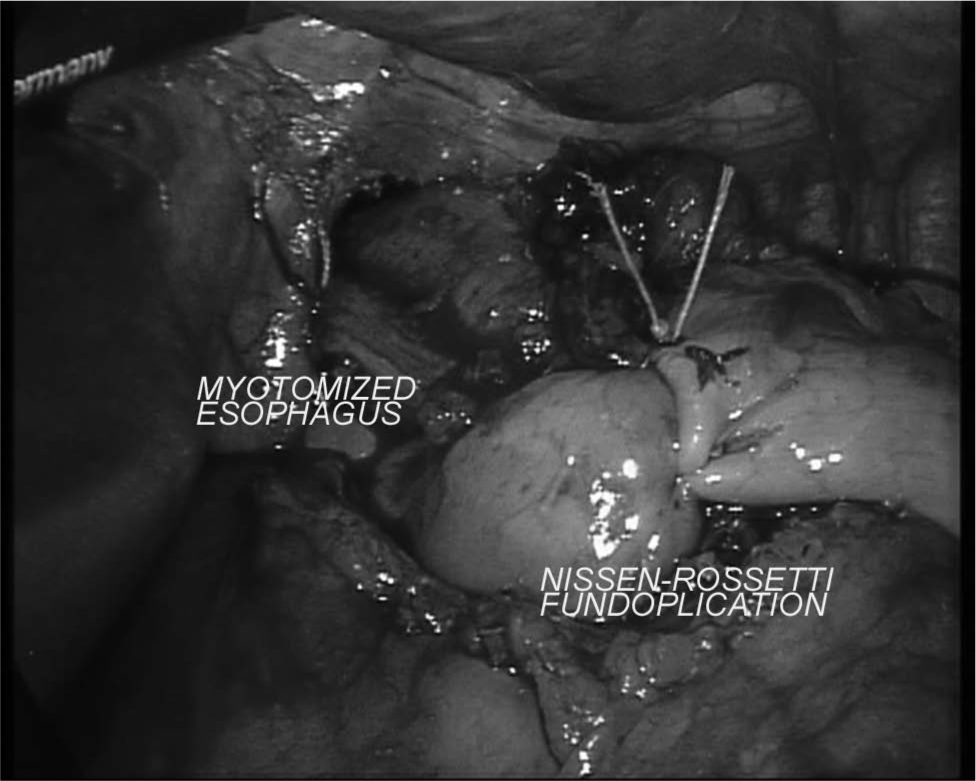
The confectioned Nissen–Rossetti fundoplication.
Results
All the operations were completed via laparoscopic approach. Mean blood loss was 120 ± 40 mL (range = 50–200 mL). The mean operative time was 135 min (range = 105–190 min). In 4 patients (19.0%), during myotomy, a mucosal tear occurred. It was repaired by direct suture. The postoperative complications included a partial disruption of the suture staple line in 5 patients (23.8%). Four of them were successfully treated with apposition of a computed tomography (CT)-guided drain and total parenteral nutrition (TPN), and one patient required only TPN. Another patient (4.8%) died of a myocardial infarction on postoperative day 4. No clinical sign of esophageal leak was observed in this patient. Mean postoperative hospital stay was 14.2 days (range = 7–25 days). The mean clinical follow-up time was 78 ± 53.7 months (range = 6–192 months; median 66 months); no patient was lost to follow-up. According to the classification of clinical outcome, excellent or good results were found, respectively, in 16 patients (80%) with total resolution of dysphagia and in 4 patients (20%) with mild residual dysphagia. Instrumental follow-up was performed on 15 patients (75%) because the remaining refused the investigations. They were evaluated with an upper gastrointestinal barium meal at 6 months from the intervention: it showed no diverticular recurrence and a regular esophagogastric transit. They all underwent a stationary manometry and esophageal pH monitoring at a mean follow-up of 61 ± 31.2 months (range = 4–92 months; median = 65 months). The mean value of the new HPZ was 19.3 mmHg (range = 14.8–23.8 mmHg), with 76.7% of postdeglutitive relaxations. Esophageal pH monitoring documented a total absence of pathologic gastroesophageal reflux.
Discussion
Alterations of the esophageal motility associated with epiphrenic diverticula are not simple to be detected despite great improvements in the study of physiopathology of esophageal functional diseases (30–32). Different disorders can be responsible for the occurrence of epiphrenic diverticula, such as achalasia, hypertensive LES, diffuse esophageal spasm, nutcracker esophagus, and nonspecific esophageal motor disorders (1, 33, 34). Furthermore, a recent study demonstrated histologic abnormalities of the esophageal myenteric plexus in almost 80% of these patients, although this was not strictly related to a specific motility disorder (35). As a result, there have been many controversies about the indications and the choice of an adequate treatment. Surgery is an effective treatment for epiphrenic diverticula but is associated with a significant morbidity and mortality (1, 11, 20, 24). Moreover, even in the long term, unoperated patients do not develop severe complications and potentially death for their diverticula, though a better subjective symptom outcome is reported by operated patients. The decision to operate or not has been a matter of debate for long time; to date, the following statement from Orringer (6) has to be considered the cornerstone: “A masterful inactivity in asymptomatic or mildly disturbing diverticula is a good practice even if, in this time of minimally-invasive surgery and stapling device, an esophageal diverticulectomy may represent a tempting trophy for an hyperactive surgeon.” The isolated presence of a diverticulum cannot be considered an indication for surgery: it has to balance the potential benefit and the surgery-related risks. In literature, the percentage of symptomatic diverticula with a surgical indication is between 0% and 40% (1, 3, 9). The diameter of the diverticular sac is not a determining factor for the surgical choice, as the size of the diverticulum does not relate strictly with the patients’ symptom score (3, 5–7). In our opinion, only the presence of severe dysphagia, regurgitation, and contrast retention on barium swallow, with a risk of aspiration pneumonia, suggest the surgical approach.
As far as the surgical technique is concerned, diverticulectomy with myotomy is now considered the choice treatment (30, 36). Diverticulectomy alone is associated with an incidence of diverticular recurrence of almost 10%–20% (1, 7, 11, 37). However, the myotomy length still remains a matter of debate: Streitz et al. (3) considered enough to perform a selective myotomy, preserving the gastric wall muscular fibers in patients with normal LES pressure; other authors, such as Nehra et al. (31), supported a long myotomy extended on the whole sphincter zone in order to prevent postoperative dysphagia and staple suture line leakage. With the diffusion of laparoscopic approach, most authors have preferred a complete myotomy extended to the gastric wall with an associated antireflux procedure (16–25). In agreement with this choice, we perform a myotomy prolonged for at least 2 cm on the anterior gastric wall and an antireflux wrap. In contrast to other authors (7), we believe it is easier to perform the myotomy on the same side of the diverticulectomy: the endoscopic properties of insufflation and transillumination can check any eventual mucosal tear, as occurred in four patients in our series, and allows its intraoperative repair. With regard to the choice of the wrap, most authors prefer performing a partial fundoplication to protect esophagus from acid reflux (5, 7, 17–19, 21–25). In our opinion, instead, a total wrap, the Nissen–Rossetti fundoplication, is preferable because it represents the best protection against reflux without increasing postoperative dysphagia, as we documented in previous studies (20, 28, 29, 33, 38–40). In fact, in our experience, even patients affected by achalasia or gastroesophageal reflux disorder (GERD) with defective peristalsis were operated with this technique achieving excellent long-term results in terms of postoperative dysphagia rates (29, 38).As we expected, the present study confirms these results with no persistent severe dysphagia, no reflux symptoms complained by the patients, and no pathologic gastroesophageal reflux detected at the 24-h pH monitoring.
The high leak rate (23.8%) reported in our experience can be a matter of discussion about the choice of a total wrap after diverticulectomy plus myotomy. First, we consider important to highlight that in our series published in 2004 (20), the only three leaks that occurred corresponded to the first three patients operated. Successively, out of the following 18 patients treated till date, only two leaks occurred, with an incidence of 11.1%. These data lead us to consider that the leak rate was, in some way, related to the learning curve. Another relevant consideration is the fact that in mini-invasive series published in literature, different authors reported a leak rate of almost 18%–20% even after performing a partial fundoplication (21, 23, 24). There is no agreement in literature about therapeutic choice for leak resolution, as some authors perform laparoscopic or thoracotomic reintervention (21, 25), while others (23, 24), and we agree with them, prefer a conservative treatment. No experience of esophageal stent placement has been reported for leaks that occurred after epiphrenic diverticulectomy.
Finally, regarding surgical approach, there is now wide preference for the laparoscopic treatment of epiphrenic diverticula (7, 16–25). Not only the well-known advantages in terms of morbidity rates and postoperative recovery led us to prefer this approach but also technical factors were determining in this choice, such as better visualization of the esophagogastric junction, easier myotomy, and realization of antireflux wrap and better alignment of the stapler cartridge with the longitudinal axis of the esophagus. Potential disadvantages could be a more difficult dissection of the upper part of the diverticular neck and the major risks of pleural lesions. In this article, we report the results of the largest published series via the traditional (Table 1) and the mini-invasive approach (Table 2). Morbidity and mortality are slightly higher in the mini-invasive series, whereas success rates are similar in the two different approaches. Our results are comparable with those reported in the other laparoscopic series. We believe that the disappointing morbidity rates detected in our and other authors’ series could be a consequence of the learning curve for this challenging surgery and are not dependent on the side of realization of myotomy or on the type of fundoplication. Maybe the increasing experience of surgeons together with the evolution of stapling and other devices for laparoscopic surgery could help to reduce, in the future, the morbidity and mortality rates for this approach.
Epiphrenic diverticula—results of surgical treatment (traditional approach).
Epiphrenic diverticula—results of surgical treatment (mini-invasive approach).
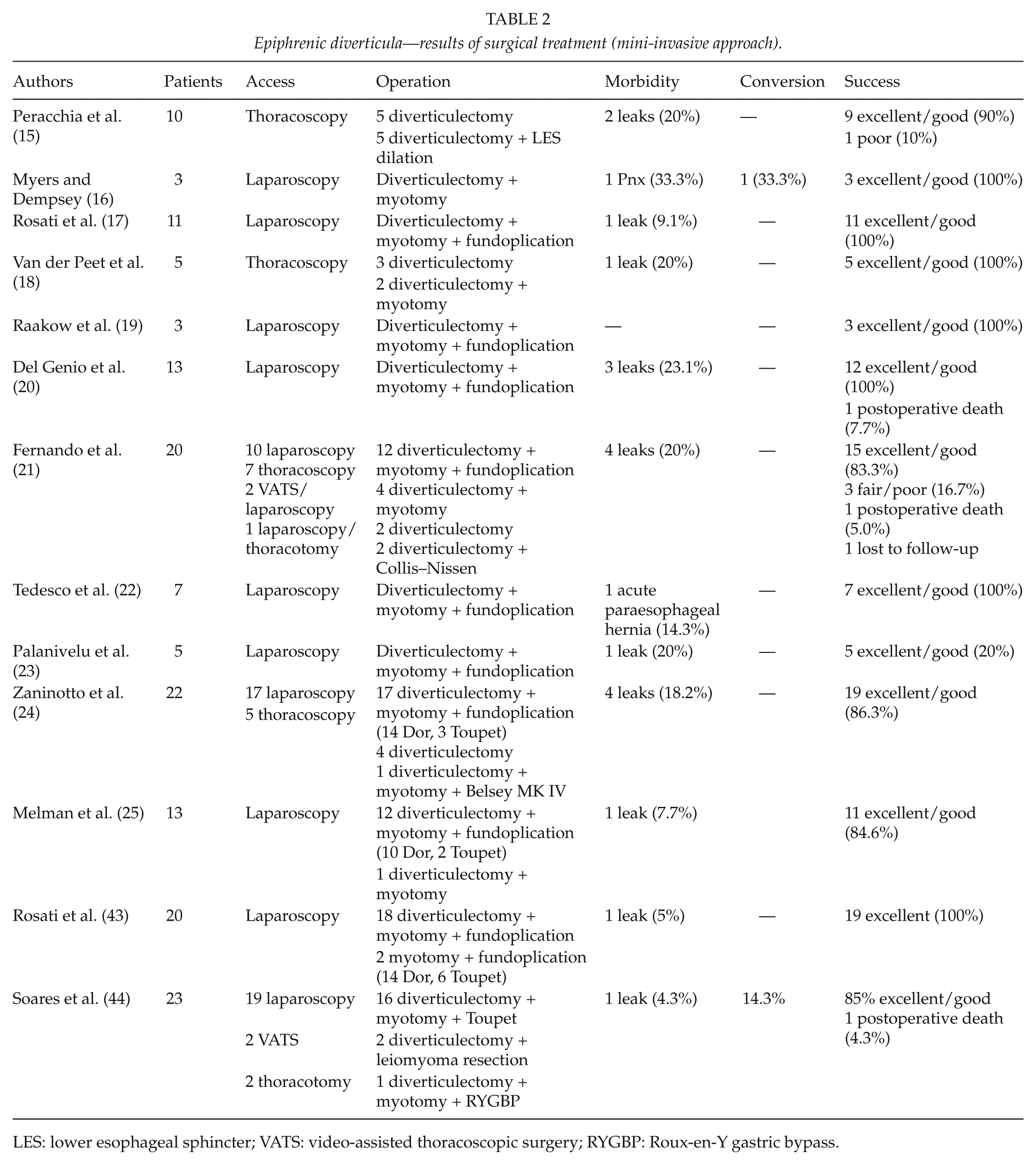
LES: lower esophageal sphincter; VATS: video-assisted thoracoscopic surgery; RYGBP: Roux-en-Y gastric bypass.
According to the literature data and our experience, laparoscopy seems a feasible approach in the treatment of epiphrenic diverticula. However, this kind of surgery remains a challenging procedure with higher morbidity and mortality rates in mini-invasive series compared to the traditional approach. For this reason, indications must be restricted only to selected and symptomatic patients treated in specialized centers. Moreover, in light of our findings, we conclude that Nissen–Rossetti fundoplication after diverticulectomy and myotomy can be considered an effective treatment in patients affected by epiphrenic diverticula.
