Abstract
Background and Aims:
Distal pancreatectomy is the only effective treatment for cancers of the pancreatic body and tail. The recurrence rate after DPhas remained high. In an effort to overcome this problem, we developed a no-touch surgical technique for DP. This is a pilot study to see if distal pancreatectomy can be technically done using a no-touch surgical technique without deteriorating the post-operative prognosis.
Patients and Methods:
From November 2000 through May 2011, 16 pancreatic ductal adenocarcinoma patients have been operated on using a no-touch technique by a single operator. We described the surgical technique, and we reported our preliminary experience. During the procedure, the pancreatic body and tail is neither grasped nor squeezed by the surgeon. And all drainage vessels from the pancreatic body and tail are ligated and divided during the early phase of the operation. Furthermore, for improved dissection of the retroperitoneal tissue (rightward and posterior margins), we use a hanging and clamping maneuver and dissection behind Gerota's fascia.
Results:
In the current series, the posterior and rightward resection margins were free in all patients, although seven were positive for anterior serosal invasion. The post-operative prognosis was not deteriorated with this technique.
Conclusion:
No-touch distal pancreatectomy technique may have some theoretical advantages, which merit future investigation in randomized controlled trials.
Keywords
INTRODUCTION
Whereas operable cancers of the pancreatic body and tail are less common than those of the pancreatic head, distal pancreatectomy (DP) is the only effective treatment for cancers of the pancreatic body and tail. DP has been the standard procedure for these cancers for decades. However, the recurrence rate after DP has remained high (1 –8). One of the possible factors for poor prognosis after resection of pancreatic cancer may be a squeezing out of cancer cells during resection. In an effort to overcome this problem, we developed a no-touch surgical technique for DP. We ligate all drainage vessels during the early phase of the operation. The purpose of this manuscript is not the analysis of effects of our operative method, but is to present the technical possibility of no-touch distal pancreatectomy without deteriorating the post-operative prognosis.
The no-touch isolation technique was originally adopted as a strategy to protect cancer cells from spreading as a result of handling malignant tumors during colon and eye cancer surgery (9, 10). Because a pancreatic tumor is generally often grasped by the surgeon prior to the ligation of surrounding vessels during pancreatectomy, squeezing and handling the tumor could increase the risk of shedding cancer cells into the portal vein, retroperitoneum, and/or peritoneal cavity (11, 12). Hence, there is a potential benefit for using a no-touch approach in distal pancreatectomy as well.
MATERIALS AND METHODS
PATIENTS TOKA
From November 2000 through May 2011, 16 pancreatic ductal adenocarcinoma patients have been operated on using this technique by a single operator with the assistance of other authors. All patients were Japanese. If there were distant metastases, infiltration to the colon and/or stomach, peritoneal dissemination, the patient was excluded from this study.
SURGICAL TECHNIQUE
Transection of the pancreatic neck and division of splenic vessels
Following the division of the gastrocolic ligament, the posterior surface of the pancreatic neck is tunneled by blunt dissection. The pancreas is transected after ligating the left side of the pancreas. The splenic artery and vein are ligated and divided at the origin and at the confluence with the superior mesenteric vein, respectively. As mentioned by Fagniez and Munoz-Bongrand, early division of the pancreatic neck provides superior access to control the splenic vessels (13).
Division of gastrosplenic, splenorenal, and splenocolic ligaments
Retrosplenic Gerota's fascia is transected on the surface of the left kidney. The dissection plane is continued along the surface of the kidney. After this blunt dissection of the retrosplenic space, the splenorenal ligament is divided. Then, splenocolic and gastrosplenic ligaments including short gastric and left gastroepiploic vessels are divided. At this point, all drainage vessels from the pancreatic body and tail have been ligated and divided.
Hanging up, clamping and transection of the peripancreatic retroperitoneal margin
The anterior surface of the abdominal aorta, which corresponds to plane posterior to the Gerota's fascia, is exposed around the ligament of Treiz. From this point, the anterolateral surface of the aorta is bluntly dissected towards a cranial direction up to the crus of diaphragm, and a tape was passed (Fig. 1). Caudal end of the tape is repositioned relative to the pancreatic side of the left renal vein and mesocolon. Then the tape lifts up the peripancreatic retroperitoneal tissue, tissue adjacent to the superior mesenteric artery (SMA) and celiac trunk including nerves and lymphatic vessels (hanging maneuver). The peripancreatic retroperitoneal tissue is clamped by a long-nosed, right-angled DeBakey type aortic clamp. Using leftward traction of the clamp, the peripancreatic retroperitoneal tissue (rightward resection margin) is transected along left surface of the SMA and celiac trunk and anterolateral surface of the abdominal aorta. The margin is sliced along the clamp and is submitted to frozen microscopy.
The dissection is behind the Gerota's fascia. The rightward margin is usually set along the left surface of superior mesenteric artery (SMA) and the celiac trunk.
Medial-to-lateral dissection of peripancreatic retroperitoneal tissue
After transection of the rightward retroperitoneal margin, the posterior plane of Gerota's fascia is dissected from medial to lateral direction, allowing exposure of the left adrenal gland and vein, and left kidney and renal vessels (Fig. 1). As mentioned by Strasberg, early identification of the left renal and adrenal veins sets up the proper dissection plane (14, 15). After the procedure, extensive peritoneal lavage was performed with 10 L of warm saline to remove any potential dissemination of cancer cells (16).
ETHICS
The operation was performed after obtaining the patients' written informed consent. The study protocol conformed to the ethical guidelines of the 1975 Declaration of Helsinki.
RESULTS
As for the tumor stages in the 16 patients, final pathological stage (I/II/III/IVa/IVb) according to the Japan Pancreas Society (JPS) classification system was 1/1/8/6/0. The International Union against Cancer (UICC) stage according to pTNM pathological classification (IA/IB/IIA/IIB/III/IV) was 1/1/5/9/0/0. The mean number of lymph nodes examined was 16 and seven patients (44%) had at least one positive node. Fifteen patients were with tubular adenocarcinoma, and one was with adenosquamous carcinoma. Among these cases, there was no hospital mortality. Two patients (13%) developed a postoperative complication (Grade B pancreatic fistula), giving an overall morbidity rate of 13%. The posterior and rightward resection margins were free in all patients, although seven were positive for anterior serosal invasion. In addition, eight tumors demonstrated invasion to the splenic vein, and three to the splenic artery.
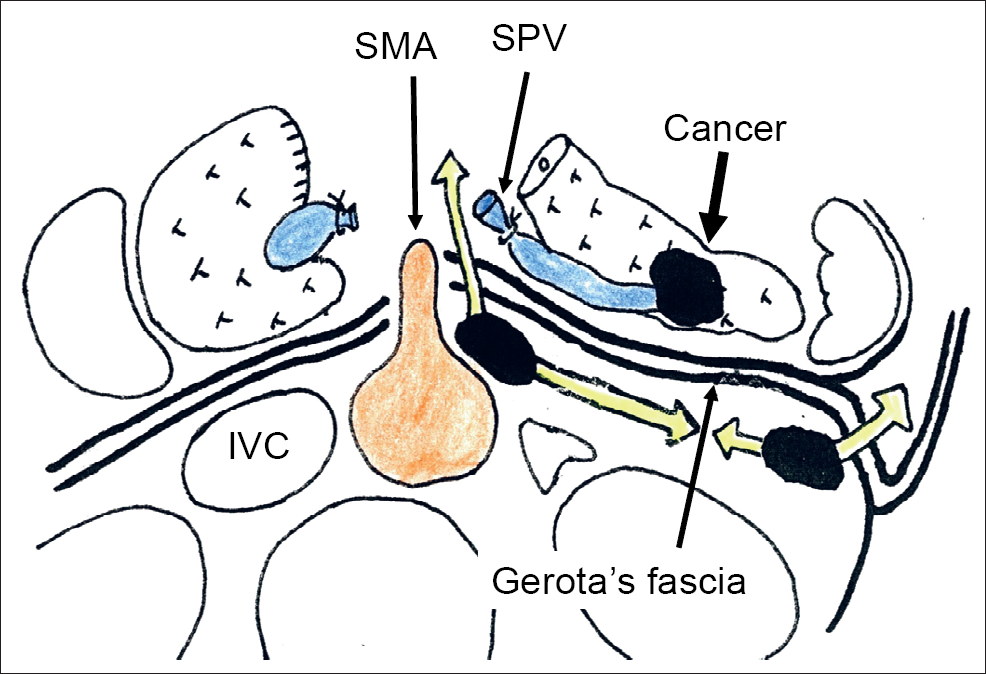
Dissection line in no-touch distal pancreatectomy
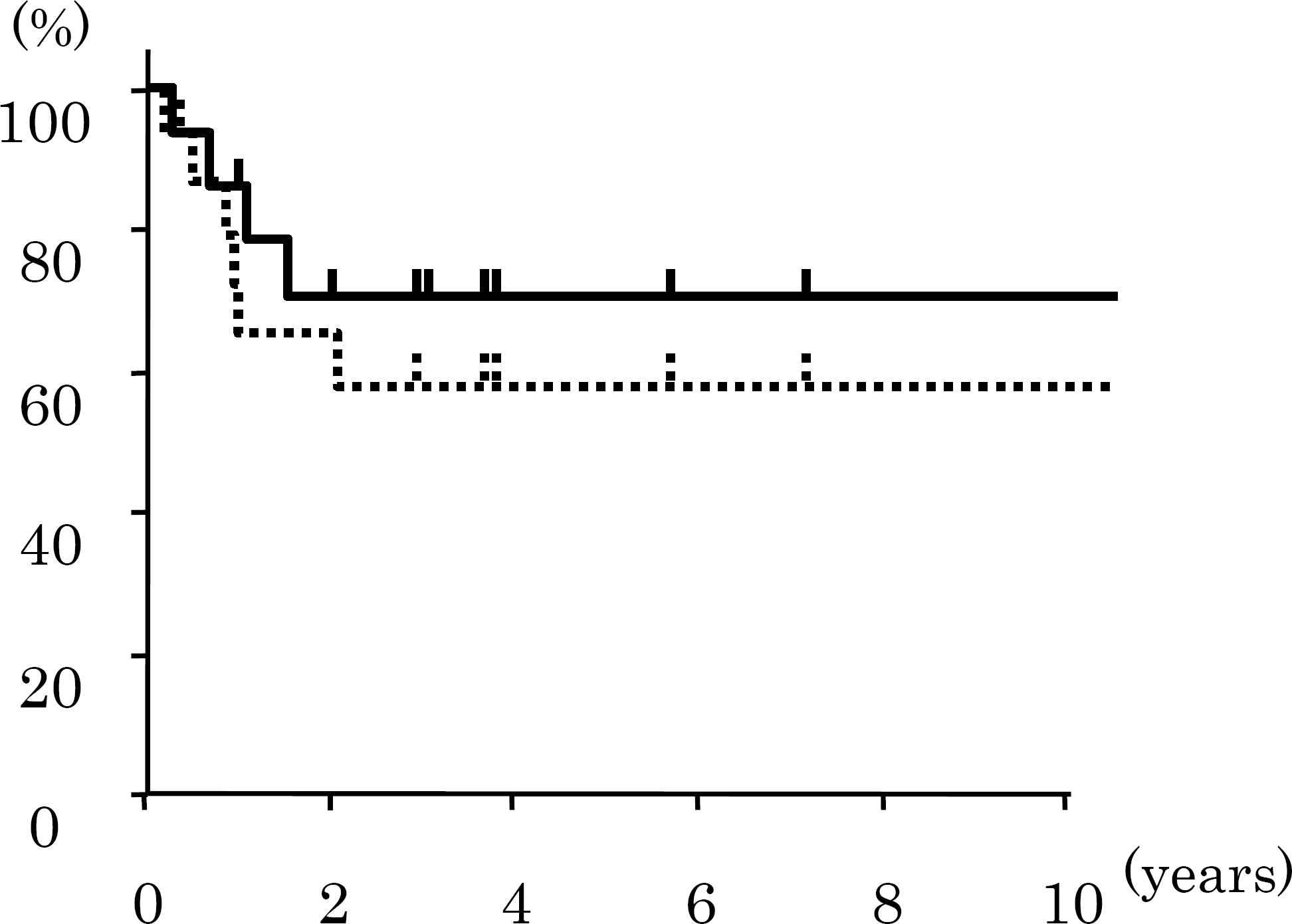
Survival curves (Kaplan-Meier) after no-touch distal pancreatectomy. Solid and dotted lines represent overall survival and recurrence free survival curves, respectively.
Overall one- and five-year survival rates were 86 and 71% with mean follow-up periods of 48.4 months (range: 2–125 months). Recurrence free one- and five-year survival rates were 72 and 57% (Fig. 2). The numbers of observations for one- and five-years were 14 and six. Median overall survival was 30 months. Three patients with tubular adenocarcinoma were actual five-year survivors. One patient is surviving over 10 years, where stage, histology, and lymph node status were IVa in JPS stage, IIB in UICC stage, well differentiated tubular adenocarcinoma, with one positive peripancreatic node, respectively. Six patients were developed with any recurrence. The recurrence sites were local in four, lung in two, and skin in one. There was no hepatic metastasis observed so far.
DISCUSSION
We have described a distal pancreatectomy involving a no-touch isolation technique. The aim of our method is to decrease the shedding of cancer cells, and to achieve negative transection margins. All drainage vessels from the pancreatic body and tail have been ligated and divided during the early phase of the operation. Squeezing and handling the tumor prior to ligation of the surrounding vessels during pancreatectomy may increase the risk of shedding cancer cells into the portal vein, retroperitoneum and/or peritoneal cavity. Although the no-touch isolation technique has not been shown to increase cancer survival or decrease recurrence, it is theoretically promising (11, 12).
Our another aim is to resect cancers by wrapping them within Gerota's fascia. Perirenal tissue beyond Gerota's fascia is often protected from the autodigestion in severe acute pancreatitis. Because cancer cell invasion is dependent on protease activity, Gerota's fascia may function as a barrier against protease-mediated invasion of cancer cells.
A hanging and clamping maneuver of the peripancreatic retroperitoneal tissues allows not only proper clearance of tissue along SMA, celiac artery and abdominal aorta but also easier dissection of the plane posterior to the Gerota's fascia. Since the Gerota's fascia may potentially function as a barrier against protease-mediated cancer invasion, one of our aims was to resect cancers wrapped within Gerota's fascia. Strasberg et al. selected two dissection planes according to the extent of the retroperitoneal invasion of the cancers (14, 15). They dissected along the anterior surface of the adrenal gland if the cancer did not invade to the gland, and the posterior plane of the adrenal gland if the cancer penetrates to the adrenal gland or more deeply. Their method is similar to ours, although they remove only the medial part of Gerota's fascia judging from the attached photo (Fig. 3 in ref. 5). However, their method is not associated with no-touch manner, which is the main characteristic of our operation. Our another aim was to facilitate negative rightward and posterior margins. A majority of studies confirm the importance of R0 resection for pancreatic head cancers (17,–19), which may only be accomplished by techniques moving the dissecting line further away from the tumor. Even for pancreatic body and tail cancers, the importance of R0 resection is applicable (15, 20). By leftward traction of the clamp on the peripancreatic retroperitoneal tissue may improve the clearance of the nervous and lymphatic tissues along the SMA and celiac artery. Because pancreatic body and tail cancers often invade to the splenic vessels and/or the anterior pancreatic serosa, the recurrence rate would become high even though we achieved negative retroperitoneal transection margins. Nevertheless we aim to endeavor to accomplish R0 resection. The traction procedure and dissection of the posterior plane of Gerota's fascia increase the possibility to perform R0 resection of pancreatic adenocarcinoma.
The reported overall one- and five year survival rates after distal pancreatectomy for pancreatic cancer are 33–69, and 19–29%, respectively (1 –8). Our results were not inferior to these data, although patients' number is very small. Hence, no-touch pancreatectomy is technically possible without deteriorating the post-operative prognosis. There was no hepatic metastasis observed so far. Because the liver is the most frequent recurrence site usually, no-touch distal pancreatectomy may have some effects to reduce the hepatic metastases after surgery. Because all patients were associated with ductal carcinoma, the results can be evaluated without any revision. Although all patients were Japanese, this technique should also be applicable in non-Japanese patients because of no big difference among races. This technique may have some theoretical advantages, which merit future investigation in randomized controlled trials.
