Abstract
Peripheral arterial disease (PAD) has often underlying risk factors, of which diabetes and cigarette smoking are the most common. Enhanced platelet activation and interaction with vessel wall associate with atherothrombotic disease, but also increased fibrinogen levels, thrombin generation and fibrin turnover are typical for PAD. The pathogenic role of fibrinogen, thrombin formation and fibrin degradation is suggested not only in acute thrombotic complications, but also in the stable form of PAD, where these markers associate with the functional severity (ankle-brachial blood pressure index). The coagulation-specific etiologies of PAD should be suspected if the atherothrombotic disease has severe manifestations, especially while the traditional risk factors are absent, or if the patient has also a history of venous thromboembolism. Malignant disease may be present in form of peripheral arterial thrombosis as well. Thrombophilia may expose patients to idiopathic thrombosis – both spontaneously and after vascular interventions. The management of these patients includes often combination therapies with antiplatelet agents and anticoagulants. Obviously, the strict policy to avoid risk factors and to treat them well in avoidance of progression of arterial disease is highly important. In the absence of published follow-up data the evidence to support the management strategies is weak and individual tailoring of efficacious and safe antithrombotic drug therapy remains our challenge. These patients benefit from continuous medical attention by the experts in the field of angiology. Management of PAD is an excellent example of the multidisciplinary approach where the hematologist meets the vascular surgeon or interventional radiologist to secure the best available patient care.
Keywords
COAGULATION DISORDERS
Coagulation disorders associate with thrombosis and are typically familial, occur at young age, even in childhood, without significant usual risk factors and predisposing conditions. A typical thrombophilia patient is less than 40 years of age, has had a history of venous thromboembolism or females a history of miscarriage or still birth. When typical risk factors, such as diabetes, hyperlipidemia, obesity and heavy cigarette smoking coincide with coagulation disorders, cardiovascular disease – including peripheral arterial disease (PAD) – appears with severe and extreme presentation. “When sick blood interacts with sick vessel wall, the disease becomes sick” (1 –5).
Classically thrombophilia predisposes to thrombosis in veins more often than in arteries. However, if arteries are injured the risk of thrombosis is increased by thrombophilia due to increased thrombin generation in comparison with normal blood. Obviously, if the classical risk factors are present the risk increases many-fold (3, 4). Similarly, thrombophilia predisposes to thrombotic complications after vascular interventions (6, 7). These clinical findings: thrombosis occurring at an early age, with limited other risk factors, but an inherited pattern, indicate the need for laboratory screening of thrombophilias (Table 1). In all, thrombophilias are present in 10–15% at the population level. Thrombophilias are both inherited or acquired. The inherited thrombophilias include coagulation factor V (FV) Leiden mutation. It is characterized by enhanced thrombin generation due to limited regulation via protein C, which does not bind to FV to regulate its activity. Prothrombin mutation FII G20210A leads to enhanced rate and extent to thrombin generation. The two gene defects can be heterozygous or homozygous, the latter of which carries again a several fold enhanced risk of thrombosis. High levels of FVIII, fibrinogen or dysfibrinogenemia associate with the risk of early atherothrombosis, and are related to impaired fibrinolytic capacity. These etiologies have both inherited and acquired nature. Merely fibrinogen being above normal limits (2–4 g/l) is linked with earlier appearance of PAD than when fibrinogen is within the normal range (8).
Acquired and inherited thrombophilias

Rare defects (presence together around 1%) of antithrombin, protein C and protein S are inherited and associated with the strongest history of early multiple thrombotic episodes in the family. When acquired they relate to inflammation, sepsis and consumption by uncontrolled thrombin formation. Acquired coagulation disorders include antiphospholipid antibodies, myeloproliferative diseases, and hyperhomocysteinemia. Also, some platelet and endothelial activation syndromes, such as heparin-induced thrombocytopenia, immunological conditions of vasculitis and involvement of the complement system as in paroxysmal nocturnal hemoglobinuria (PNH) expose to severe thrombotic complications. These can be diagnosed based on blood cell counts (paradoxical thrombocytopenia), and specific markers (antinuclear antibodies and lack of CD51/59 from red cells and leukocytes). Additionally, high numbers of receptor copies of platelet glycoprotein Ia/IIa, VI, II/IIIa and Ib associate with increased platelet activation and may sustain thrombogenicity, but have not been systemically studied in PAD.
In all, clinicians should be able to recognize the signs of thrombophilia because of the abnormal presentation of PAD (2, 5). Coagulation disorders promote vascular complications due to thrombin and enhanced platelet activation and interaction with arterial wall (Fig. 1). Also, the possibility of cancer should be kept in mind upon an atypical presentation of PAD. These patients may be prone to microvascular thrombosis, causing various organ failures and distal ischemia. This thrombin formation and platelet interaction support local fibrin formation and resistance to fibrinolysis, ultimately leading to symptomatic occlusion of even large arteries. Finally, also in patients with end stage renal disease demanding dialysis vascular interventions have been shown to occlude more if thrombophilia is present (Salmela et al. submitted). Individual design of antithrombotic treatment and other specific remedies is important to avoid the reoccurrence of vascular complications (2, 5) (Fig. 2).
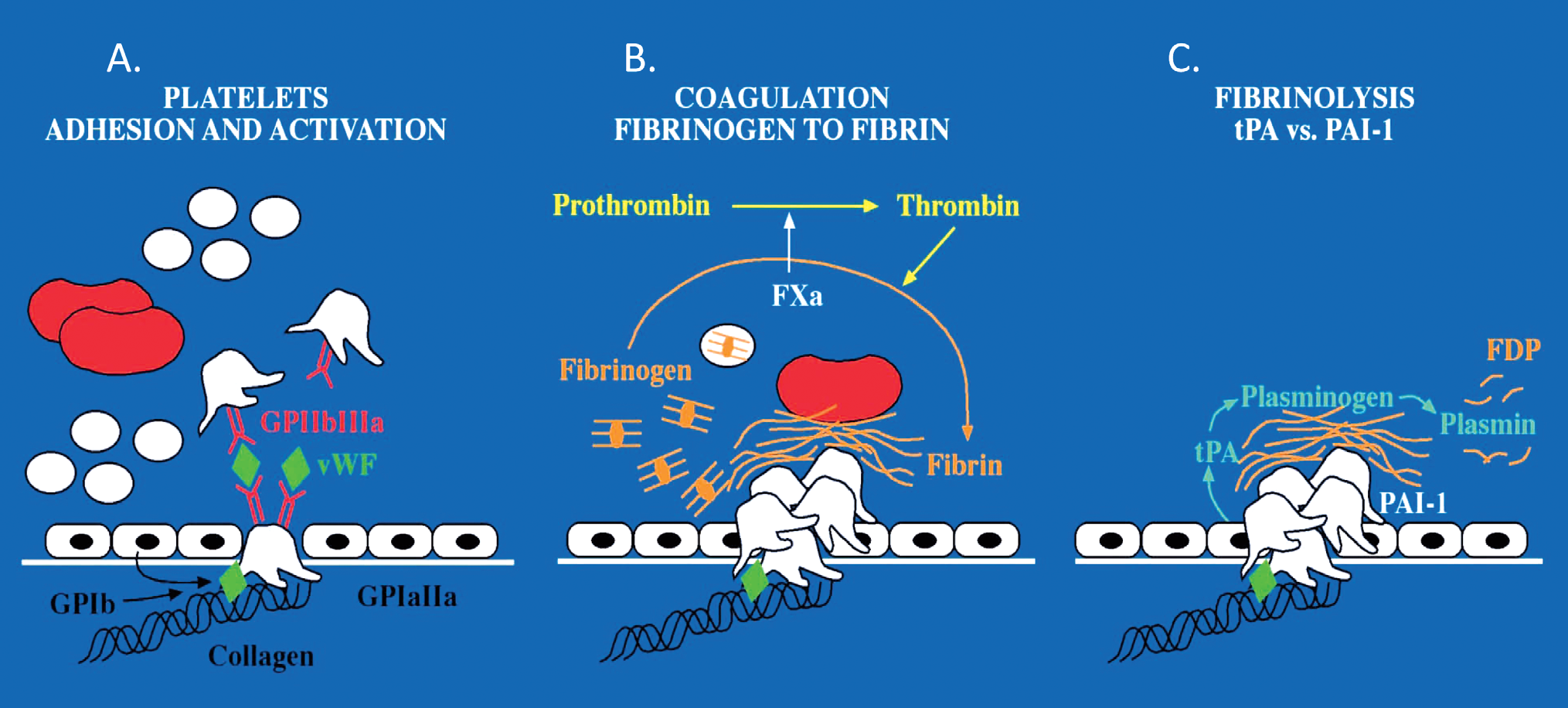
Three phases of blood interacting with vessel wall to induce hemostasis, implications for peripheral arterial occlusive disease. If the reactions are enhanced and uncontrolled thrombosis ensues. All three mechanisms of platelet adhesion and activation (A), coagulation (B) and fibrinolysis (C) are involved. In the first step platelets may accumulate too extensively and engage with the coagulation system to stabilize the forming thrombus too strongly for the fibrinolysis to clear it. Fibrinolysis may also be impaired resulting in the occlusion of the vessel.
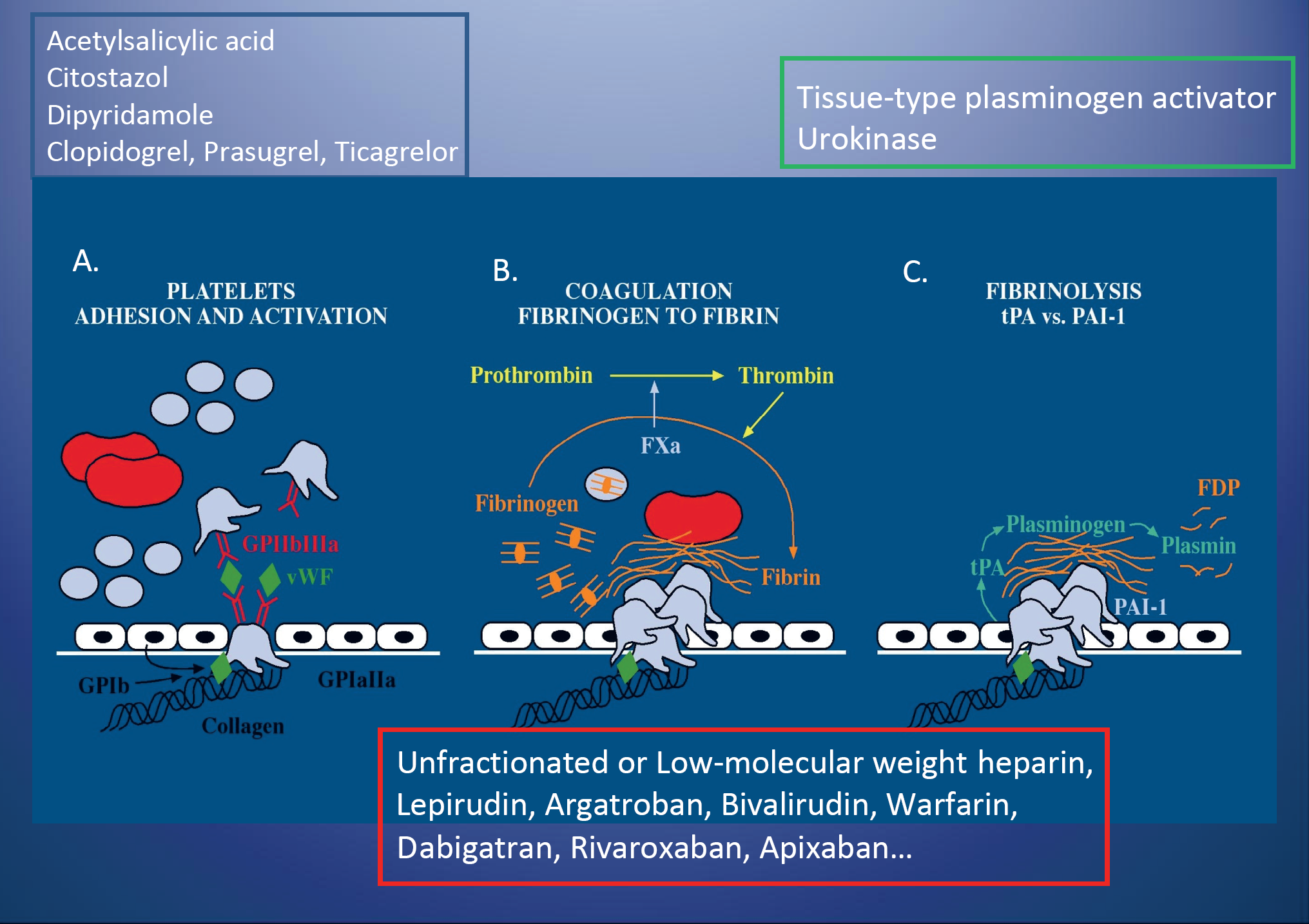
Antithrombotic and fibrinolytic therapies are introduced in association with their target in the vessel wall and blood interphase. The armamentarium of antiplatelet agents is broadening, especially in the field of platelet ADP P2Y12 receptor inhibitors. Also, novel anticoagulants are entering the market. Their role in inhibiting systemic embolisation in association with atrial fibrillation is clear, however, specifically in PAD more studies are needed.
COAGULATION ACTIVITY AND PAD
We have observed that fibrinogen and D-dimer measuring fibrin turnover associated with the functional severity of stable PAD, a finding which has been later confirmed (9 –11). Ankle-brachial index correlated with fibrinogen and with D-dimer with the exactly same correlation coefficient, 0.57, suggesting that common mechanisms are involved (9). Synthesis of fibrinogen is regulated by degraded fibrin products, such as D-dimer, compatible with our finding. In the same study thrombin generation (thrombin-antithrombin complexes, TAT) in the absence of overt thrombosis associated with radiological severity of atherosclerosis. Also, platelet glycoprotein Ib status associated with PAD compatible with thrombin activity (12).
Physical exertion further induces sudden thrombin generation, but also fibrin degradation in patients with peripheral arterial disease when compared with healthy controls (13). The marker levels assessing coagulation and fibrinolysis were compared among patients with acute and subacute thrombosis and with stable disease PAD, overt thrombosis elevated the marker levels by 5–7 fold (14). These markers of thrombin generation and fibrin turnover related to indicators of skeletal muscle damage. Increased TAT, plasminogen activator-1 antigen and myoglobin concentrations associated with poor outcome among patients with acute arterial thrombosis.
In our further studies on vascular reconstructions we found that vein grafts engaged with the upregulation of PAI-1 mRNA expression and markedly increased amounts of PAI-1 antigen after the development of neointima (15). Furthermore, urokinase plasminogen activity was augmented in the graft wall, but tissue plasminogen activator, responsible for fibrinolysis in blood, was depleted. Altogether, these findings imply decreased fibrinolytic potential in the stenosed graft, which may ultimately contribute to the graft occlusion.
CONSERVATIVE MANAGEMENT AS AN IMPORTANT ADJUNCT FOR VASCULAR SURGERY
When the diagnosis of PAD is obtained, the classical cardiovascular risk factors: hypertension, hyperlipidemia, diabetes and cigarette smoking should be actively managed. Overweight is not usually the most common risk factor, but diabetes is (16). Cigarette smoking is, as about 90% of the patients with PAD used to be cigarette smokers 25 years ago, but fortunately this share is decreasing (17). Smoking increases adrenergic activation and fibrinogen levels in addition to its several toxic components injuring vascular wall.
The most important evidence-based recommendations for the conservative management of PAOD, are presented by the American College of Chest Physicians Evidence-Based Clinical Practice Guidelines (8th Edition) (18) and the guidelines by the European Society of Vascular Surgery (16). Lifelong antiplatelet therapy in comparison with no antiplatelet therapy in PAD patients with or without clinically manifest coronary or cerebrovascular disease is strongly recommended (Grade 1A). Anticoagulants are not recommended upfront, unless there are other indications, such as atrial fibrillation with or without an acute embolisation. In patients with short-term (< 14 days) arterial thrombosis or thromboembolism intraarterial thrombolytic therapy is the method of choice provided the low risk of myonecrosis and ischemic nerve damage during the time to achieve revascularization. For patients undergoing major vascular reconstructive procedures, IV unfractionated heparin (UFH) prior to the application of vascular cross clamps should be provided (Grade 1A). For all patients undergoing infrainguinal arterial reconstruction and vein or prosthetic bypass, aspirin (75–100 mg, begun preoperatively) is needed (Grade 1A). According to one trial (CASPAR) dual antiplatelet therapy with aspirin and clopidogrel in the below-the-knee bypass prosthetic grafting appeared beneficial in reducing composite events of graft occlusion and revascularization, above knee amputation and even death (16, 17). However, considerable number, 30% of the patients with atherothrombosis are likely to be nonresponders to clopidogrel, which needs urgent re-evaluation (18).
Some patients with a severe distal form of the disease are beyond the reach of vascular surgery. We have described eight such relatively young patients who had thrombophilia and a severe history of coagulation problems prior to entering our study (2). We chose to manage them with direct thrombin inhibitor, lepirudin, because it is highly efficacious in heparin-induced thrombocytopenia, which has extremely strong preponderance to thrombin generation and spread large vessel thrombosis. Lepirudin has been proven to be effective in acute coronary syndrome which is a relevant mimic to acute atherothrombosis in other vessels, too. Strong inhibition of thrombin fosters fibrinolysis, as thrombin activates an inhibitor to fibrinolysis (TAFI) (19), a mechanism suggested to be involved in diabetic cardiovascular risk. In our small patient group lepirudin therapy succeeded well in providing relief of distal ischemia (2). We have further explored laboratory monitoring tools for lepirudin treatment (20). Also, we obtained similar outcome in five patients with mesenterial thrombosis and in one patient with threatening liver transplantation surgery due to extensive thrombosis in the portal system (21, 22).
SOME KEY RULES TO AVOID BLEEDING COMPLICATIONS
Platelet antagonists are the mainstay of the antithrombotic management in PAD. When platelet antagonists are used acutely or even chronically on top of anticoagulants the risk of defective platelet function may predispose to bleeding complications, which most often occur early at the surgical site or later in form of mucocutaneous bleeds, such as gastrointestinal bleeds. To avoid bleeding complications some basic rules need to be filled.
Anemia and thrombocytopenia lead to impaired primary hemostasis. This is because red cells create rheological forces which help platelets to adhere to injured vessel wall. Analogically in polycythemia vera too high hematocrit leads to enhanced platelet adhesion and aggregation and thereby reinforce thrombus formation. When hematocrit is below 30% and platelet count is below 100 × 109/l, the adhesive potential of platelets is significantly impaired. When using antiplatelet therapy, efforts to diagnose and manage anemia (deficiency of iron and/or folate and vitamin B12) and thrombocytopenia are important, and therefore blood cell counts should be followed up on regular basis. Other platelet inhibitors, such as non-steroidal anti-inflammatory drugs and selective serotonin reuptake inhibitors should be avoided, and other alternatives should be offered. Liver function should be monitored in terms of synthesis of coagulation factors, as such screening of vitamin K dependent coagulation factors with prothrombin time (PT) and other extrinsic factors measured with activated partial thromboplastin time (APTT). If there is deficiency of vitamin K due to temporary problems with diet or absorption, vitamin K can be given intravenously a couple of times (dose 1–5 mg iv). Usually next day the synthesis is improved if liver functions properly and PT normalizes. When using low molecular weight heparins for a longer time periods, PT and APTT should be monitored and be in normal range. The indications of warfarin should be clarified and thrombophilia adds to them in severe cases. We do not yet have any information of the new oral anticoagulants in the thrombophilia cases.
All the above topics together underline the important role of platelets and coagulation activity, fibrin formation and regulation of fibrinolysis in PAD and its surgical and conservative management (Fig. 3), the target of which is thrombosis. The interactive cowork between vascular surgeon or interventional radiologist and hematologists or internists, such as angiologists, is beneficial and supports the further development in this field. It has been a personal honor and priviledge to enjoy of such a collaboration with professor Mauri Lepäntalo in both unraveling these mechanisms in PAD and treating patients with extreme vascular problems. In the coming years the growing options for antithrombotic medication continue to open new avenues in this field.
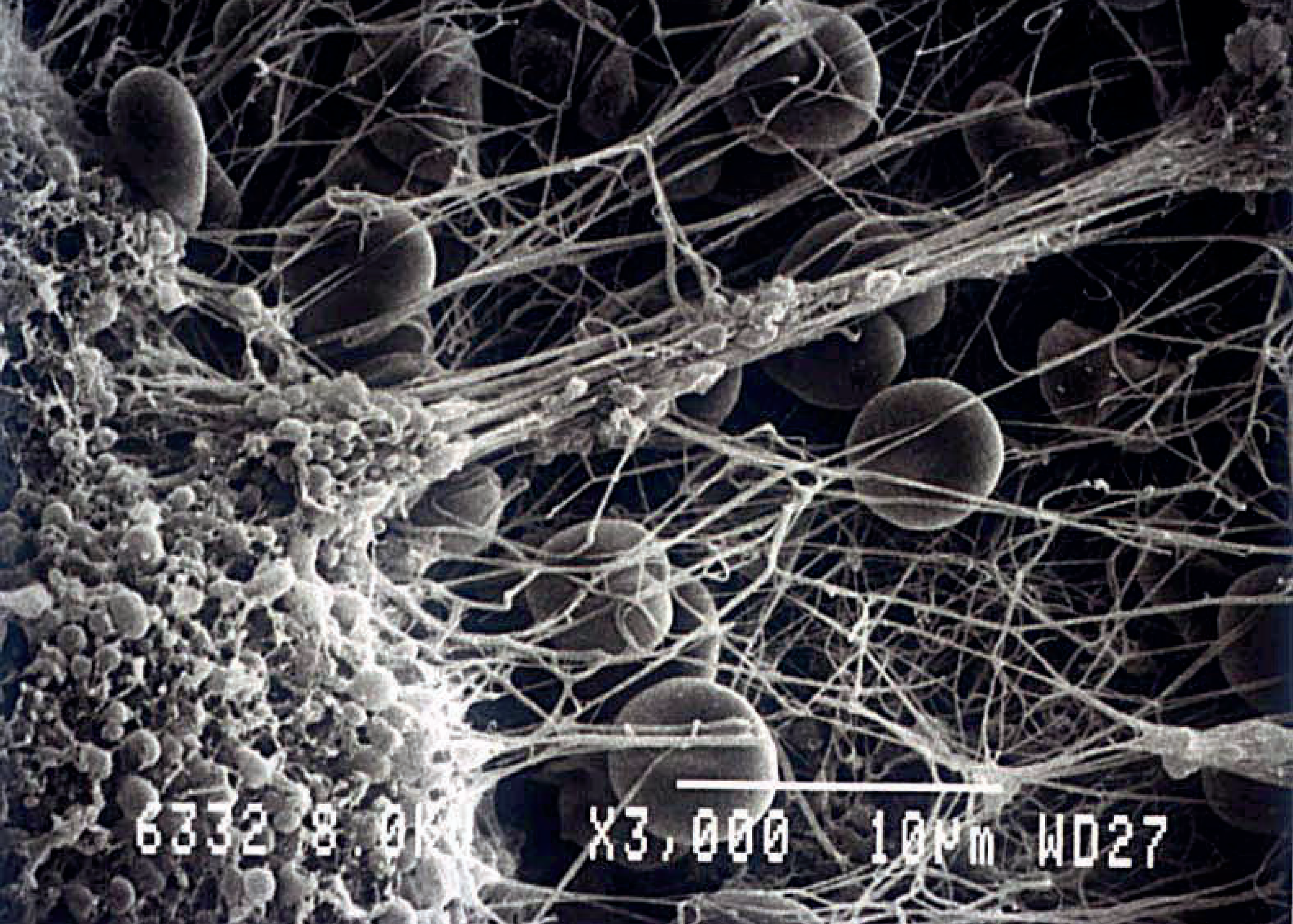
Scanning electron micrograph of platelet-rich thrombus interacting with fibrin strands formed under arterial blood flow conditions.
Footnotes
ACKNOWLEDGEMENT
This manuscript is devoted to Professor Mauri Lepäntalo. His commitment, enthusiasm and interactive skills have provided the background and requisite not only for this review, but for the initial steps of Coagulation Disorders Unit in Helsinki University Central Hospital, Helsinki, Finland.
