Abstract
Despite numerous attempts, chronic critical limb ischaemia (CLI) has not been unequivocally defined as yet. Its epidemiology is poorly investigated and its prevalence probably higher than anticipated. It is accompanied by high mortality and morbidity irrespective of the way it is treated. Its management is very expensive. Additionally, the prevailing diabetes epidemic is increasing the need for revascularizations although there is a clear lack of evidence as to when to revascularize an ulcerated diabetic foot. The fast development of endovascular techniques blurs the vision as the window of opportunity for gathering proper evidence keeps narrowing. The notion of endovascular artistry prevails, but attempts to conduct proper studies with clear definitions, strict criteria and appropriate outcome measures in a standardised manner should continue – preferably using propensity scoring if randomised controlled trials are not possible.
This review highlights some of the steps leading from art to evidence and illustrates the difficulties encountered along the path. In parallel with this overview, the progress of the treatment for CLI in Finland is described from the perspective of the work concluded at Helsinki University Central Hospital.
Keywords
THE ART
Early surgery aimed to relieve the symptoms of severe leg ischaemia. After the grim periods of mere amputations and sympathectomies the art of vascular surgery developed from innovations that opened, almost literally speaking, new avenues for healing. Charles Rob has reviewed the early experiences quite incisively (1): “Probably the first surgeon to realize the importance of a direct attack on the occluded segment of artery was Leriche, who excised the thrombosed portion of the artery (arteriectomy) in 1937. Dos Santos (1947) took this step further with his operation of disobliterative endarterectomy, the first procedure designed to restore the blood flow through the blocked artery. Although better than the previous indirect procedures the success rate was not very high. A better method is to replace the thrombosed segment of artery by an autologous vein graft or a homologous artery graft. The first successful grafting of peripheral artery was done by Lexer in 1912, when he excised a popliteal aneurysm and replaced it by a vein graft.” The first breakthrough in the treatment of lower leg ischaemia came from an attempt in 1948 by Jean Kunlin of France who used saphenous vein as a bypass conduit (2). In the beginning, mostly human arterial grafts were used after rapid freezing or other measures, but these grafts did not survive; they died and were replaced by the tissue of the host, merely acting as scaffolding while the process of creeping substitution occurred (1). Eventually, the introduction of arterial reconstructions to restore circulation to a gangrenous limb was welcomed as the new treatment of choice, although only 14% of patients were found suitable in the early series reported by Rob (3).
By the mid-1950s, femoral bypass grafts were confined to the popliteal artery, and the use of venous grafts increased (4). The application of femoral arteriography allowed surgeons to define the operability of the bypass graft in relation to the arteriographic pattern (4). Sven-Ivar Seldinger of Sweden developed the co-axial puncture technique of arteriography (5). It enabled better visualisation of the distal arterial tree and facilitated the assessment of run-off, i.e. the adequacy of outflow. Björn Lindström was the first in Finland to perform femoral angioplasty with a vein graft in 1957 (Fig. 1a). In the chronology of the development of infrainguinal vascular reconstructions, Yao and Pearce emphasise the of role of Charles Rob (1959) and Karl Viktor Hall (1961) in introducing in-situ vein grafts, whereas the extension of bypass grafts down to the infrapopliteal and pedal arteries is attributed to a number of surgeons, such as JJ McCaughan in 1958, EC Palma in 1960, WA Dale in 1962 and JJ Ochsner in 1966 (4). In Finland, PT Harjola (Fig. 1b) was the first to report on a long bypass down to the ankle level (6). Whether these revascularizations were performed due to intermittent claudication or more advanced leg ischaemia is unclear. The concept of chronic critical limb ischaemia (CLI) emerged late in the history of peripheral arterial occlusive disease (7). In Finland, Michael Luther introduced in-situ bypass surgery for CLI (Fig. 1c). Team work by vascular and plastic surgeons, with Erkki Tukiainen (Fig. 1d) in an essential role, made the correction of large ischaemic tissue defects possible by combining bypass surgery and free tissue transfer (8).

Key persons contributing to the development of surgery for CLI in Helsinki and in Finland.
The shift towards less invasive revascularization techniques symbolizes the evolution of vascular practices. Charles Dotter was the first to introduce image-guided percutaneous catheter dilatation of the femoral artery (9). After the introduction of balloon catheters, percutaneous transluminal angioplasty became increasingly adopted (10). Currently, endovascular techniques and equipment are developing rapidly. Subintimal angioplasties, drug-eluting balloons and stents, stent grafts, cryoplasties and other latest endovascular innovations may offer fascinating outcomes. Their use in the hands of the best experts is a form of art. As yet, however, the long-term outcome of endovascular revascularizations remains ill defined. Since new techniques are introduced all the time, the target is shifting so rapidly that it is difficult to collect proper scientific data.
SEARCH FOR EVIDENCE
TERMINOLOGY
It is often unclear what we are talking about when we are talking about critical limb ischaemia. The term critical implies a level of disease beyond the stable condition. Critical ischaemia describes the most extreme degree of lower extremity signs and symptoms, suggesting a limb-threatening condition. The history of modern classification of ischaemia starts with the purely clinical classification by Fontaine (11). The objective criteria for CLI were first included much later (7). Recently, the history of the definitions was clearly presented by Becker (12). Yet the use of objective criteria is not properly highlighted in the Transatlantic Inter-Society Consensus (TASC) Documents, which accept rest pain, ulcer or gangrene attributable to objectively proven peripheral arterial disease as presenting CLI (13–14). While the documents are easy to use in clinical practice, this has led to problems in the development of the clear concept of critical limb ischaemia in the search for evidence (12). The diabetic with an ulcerated foot represents another problem as the tissue perfusion needed to heel the ulcer or keep the tissue intact is not the same as in non-diabetics (15). This is also apt to cause confusion.
NON-INVASIVE VASCULAR STUDIES AND HAEMODYNAMIC THINKING
The aim of non-invasive testing of patients with critical leg ischaemia is, first of all, to provide reproducible physiological data concerning disease severity (Lepäntalo M, unpublished review, 1979). Arterial hypotension of the foot caused by peripheral arterial disease was first measured by a fall in ankle pressure by Winsor in 1950 (16). However, vascular surgeons did not really become interested until the 1970s after the papers by Carter in 1968 (17) and Yao et al. in 1969 (18). In Finland, objective assessment of peripheral arterial disease first became systematic in the Surgical Hospital in Helsinki after the visit in 1977 by the author of this review to Växjö, Sweden (Figs 1e, f). Further knowledge was obtained from Seattle and Chicago (Figs 1g, h). Thereafter, a number of different methods were tested (19) and, relatively often, abandoned (Table 1). At present, the ankle/brachial pressure index (ABI) is the initial method, but toe pressure, not biased by incompressible arteries, should be measured for all patients with suspected CLI, preferably supplemented with TcPO2 measurement (12). Unfortunately, none of these methods always have the desired reproducibility (20 –22). Different criteria for diabetic and non-diabetic lesions have also been proposed (23, 24). The armamentarium has changed during the 35-year vascular laboratory practice, while more than 135 000 studies have been conducted or recorded at the HUCH vascular laboratory in Helsinki.
Vascular laboratory workup for critical leg ischaemia in HUCH, Helsinki in 1977–2011

A total of 88397 patient visits in the vascular laboratory of the vascular surgical unit/Department of Vascular Surgery, HUCH functioning in Surgical Hospital 1977–1998, Meilahti Hospital 1998–2011 and Jorvi Hospital 2002–2011.
Since the pioneering work by Strandness et al. (25), duplex ultrasound imaging has emerged as a noninvasive means of scanning the arterial tree, allowing the identification of the anatomical location and degree of occlusive lesions. Unfortunately, it is strongly operator-dependent and its diagnostic accuracy is poorer in the infrapopliteal region. However, it is gaining acceptance as a substitute for more invasive imaging, and the method is being increasingly used by vascular technicians/nurses as well as vascular surgeons.
THE PROBLEM
Patients with critical limb ischaemia (CLI) represent less than 5% of the occurrence of symptomatic peripheral arterial disease. It mostly occurs late in life, and the natural outcome is poorly studied. Chronic kidney disease, tissue loss and coronary heart disease as well as impaired ambulatory status are predictors of poor outcome, especially after revascularization (26 –28). Despite revascularization, ischaemic lesions have a tendency to heal slowly, especially in diabetics (29).
The fast progress in medical management, imaging and revascularization techniques, as well as the differences in their implementation and the unstable criteria for CLI, in addition to the varying inclusion criteria and a lack of clear and standardised reporting criteria, make it impossible to find proper data to provide clear evidence-based guidelines. This problem was well illustrated in a systemic review of the effectiveness of revascularization of the ulcerated foot in patients with diabetes and peripheral arterial disease (30). Out of the 15,227 original papers searched, 49 could be included in qualitative and none in quantitative analysis (30).
EVIDENCE FROM OUTCOME MEASURES AVAILABLE
Patency is a direct measure of revascularization success when reopening or bypassing occlusions. Patency data are available far less frequently than leg salvage rates, which is the most popular outcome parameter for the treatment effect of revascularizations. However, amputation-free survival has been emphasised as a main composite end point (31, 32). Other measures of outcome analysis include wound healing, quality of life and sustained ambulation.
LEG SURVIVAL OR LEG SALVAGE
Leg salvage or foot preservation – a favoured and easy to retrieve end point of CLI studies – is problematic as a number of factors besides revascularization affect the outcome. Leg salvage is an indirect measure of the success of revascularization. The key question is what the leg outcome would be if left untreated or treated only conservatively. Indeed, in studies reporting on the outcome of patients with CLI unsuitable for revascularization, one-year leg survival rates of 54% if ankle pressure was < 50 mmHg or toe pressure < 30 mmHg (33), 58% among controls for spinal cord stimulation (34), 66% if ABI was < 0.5 (35), and 79% in patients with minor tissue lesion and ankle pressure < 70 mmHg, toe pressure < 50 mmHg or TcPO2 < 30 mmHg (36) have been reported.
The results of invasive therapeutic measures, any type of revascularization in particular, should be considered in the face of these data. Four large recent series of bypass surgery for CLI legs reported leg salvage rates of 88%–92% at one year (37 –40). The 5-year leg salvage rate of 78% underlines the durability of bypass surgery (37). Endovascular treatment has been found to be connected with an 82%–86% leg salvage rate at one year (41, 42) (Fig. 2). Accordingly, no difference was found in limb salvage between infrapopliteal surgery and infrapopliteal endovascular interventions in a recent meta-analysis (38, 42). However, primary and secondary mid-term patency rates were better after bypass (42). Therefore, there is a wider so-called patency/leg-salvage gap in endovascular than in surgical series – i.e. occlusion of the revascularized segment more seldom leads to amputation after endovascular procedure than after surgical bypass (42). This wide gap may be explained in several ways. One hypothesis is that leg salvage exceeding the patency of the revascularization procedure is attributable to the early patency which provides adequate perfusion until the ischaemic lesions are healed. Thereafter, the leg stays vital if infection is cleared and proper foot care sustained, especially in diabetic lesions. This “tide-over” concept may, of course, be partly true, but a more obvious explanation is that legs treated with endovascular methods have milder lesions to begin with, as indicated by a large Italian study (43). It is clear that milder lesions have better healing potential with or without revascularization (33 –36, 43). Leg salvage is actually the improvement achieved by therapeutic measures beyond natural leg survival (Fig. 2). In other words, leg salvage is a composite end point and should thus be used cautiously as an outcome measure for revascularizations.
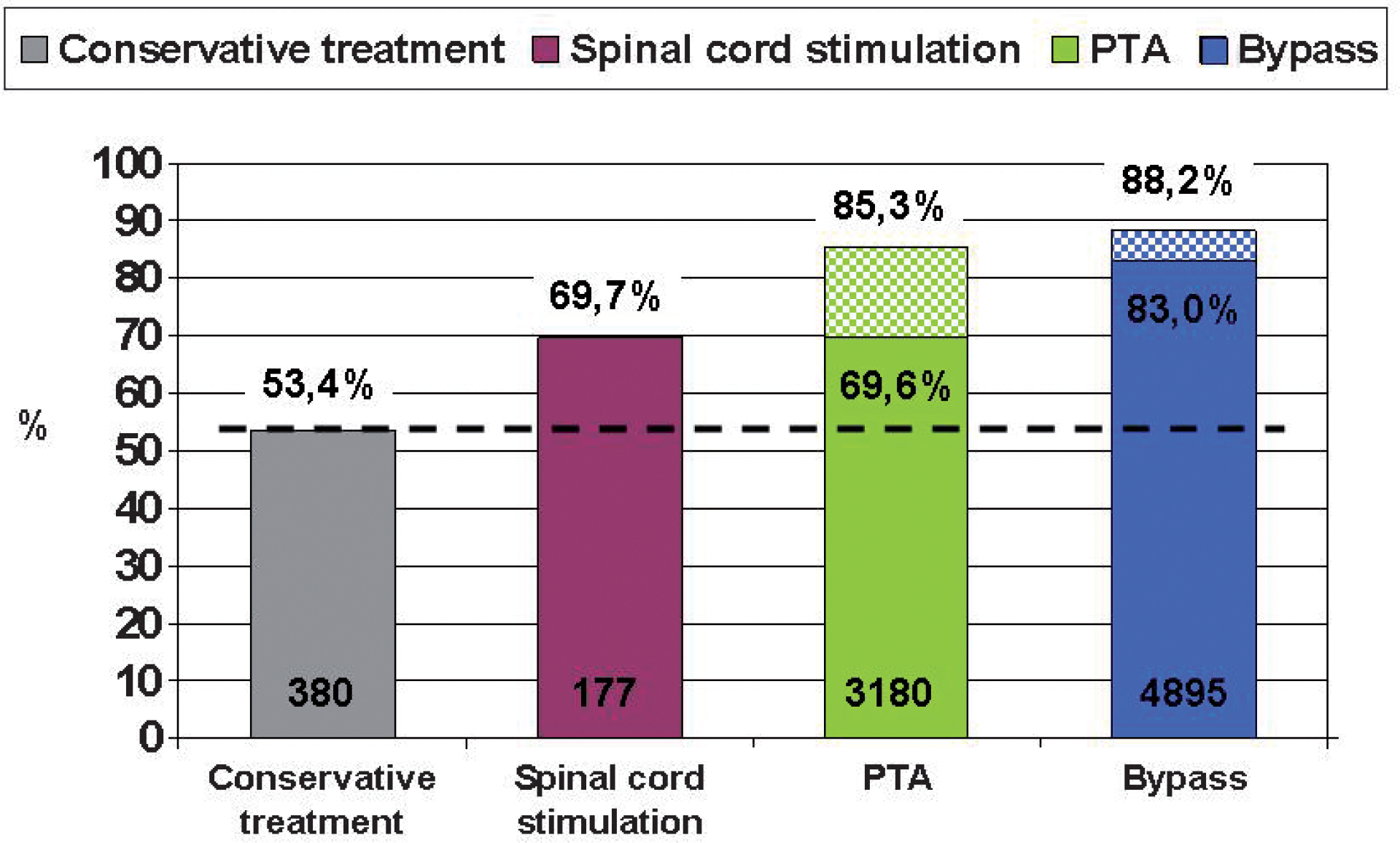
One-year leg survival after different modes of treatment for CLI; the improvement achievable by conservative means (largely natural outcome) is shown above the horizontal line, whereas leg salvage despite occlusion of the revascularizations is indicated by a hatched area of the column (data from references 33–36 for conservative treatment, 41–42 for PTA and 37–40 for bypass).
Despite inadequate evidence, endovascular therapy for infrainguinal arterial disease is gaining acceptance as a first-line revascularization method to improve ulcer healing and limb salvage. Accordingly, a clear paradigm shift has also occurred at the Helsinki University Central Hospital with an increase of endovascularly treated patients from 34% to 70% during the last ten years (Fig. 3). Comparisons of case series are biased by differences in indications, risk factors and treated arterial segments as regards their number, lesion type (occlusion vs. stenosis), lesion length, anatomy, wall characteristics and outflow. It is most likely that a bypass group includes patients with more severe disease and thus poorer outcome to be expected. Only a rather small share of infrainguinal lesions are equally well treatable with either method as shown by the only large trial that compares endovascular revascularization and bypass surgery in a randomised fashion in the setting of severe limb ischaemia (31). Both approaches yielded similar results in terms of amputation-free survival up to two years. Surgery was associated with higher early morbidity, predominantly due to infections, as well as more hospital days and higher costs. Angioplasty was associated with a higher need for further revascularization procedures, which abolished the difference in cost over time (44). Yet, the long-term results suggested that surgical repair was more durable (44). No patency data were available. The findings of the BASIL trial suggest that the decision as to whether to perform bypass surgery or PTA first for CLI due to infrainguinal disease appears to depend on life expectancy (44). Of note, in patients with ischaemic tissue loss, short life expectancy may shadow the outcomes despite successful revascularization (45).
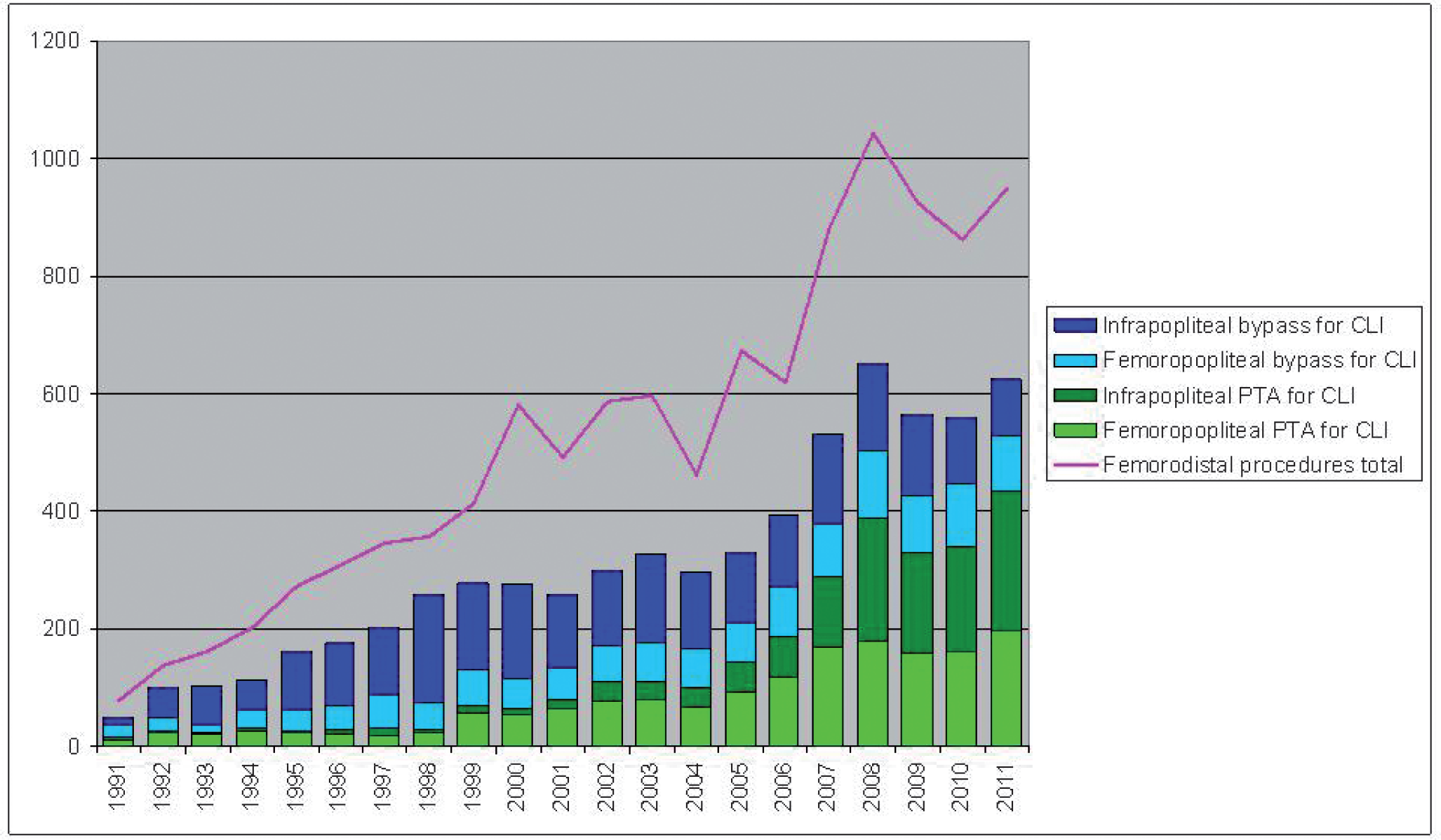
Infrainguinal bypass surgery and endovascular revascularization for CLI in 1991–2011 as well as the total annual number of infrainguinal revascularizations at Helsinki University Central Hospital (incomplete regional coverage in the 1990's).
Loss of ambulation is an important cause of increase in costs. Goodney et al. reported an 81% sustained ambulation rate at one year for patients treated with bypass for CLI (40). The costs outside the health care system are not known. Similarly, it is unclear what the costs of unnecessary interventions are. The approaches to maximize early detection and optimize therapy are widely emphasised as CLI patients are understood to suffer from the worst form of peripheral arterial disease (46). This is absolutely true in regard to conservative treatment, but there is no data to show whether the indications for revascularization should be extended. However, neuroischaemic diabetic feet probably necessitate wider indications (12, 15, 24). Although there is an increasing amount of RCTs comparing different endovascular techniques, the scientific evidence is still inadequate for assessing the treatment effect of endovascular therapy on critical ischaemia (47). Even the data on open surgery is scarce (47). All in all, there seem to be more questions than answers despite numerous attempts, also in Helsinki (Table 2). There is an urgent need to apply a standardised definition of CLI, with clinical and objective criteria, to demonstrate and compare potential advances in the management of these patients (12). As RCTs tend to have low external value, alternative approaches, such as propensity score analyses, may provide useful information (48, 49).
Diagnostics, outcome and treatment of CLI: DMSc/PhD theses from the Department of Vascular Surgery, Helsinki University Central Hospital, and the Institute of Clinical Medicine, Faculty of Medicine, University of Helsinki, Finland

EVIDENCE BY GUIDELINES AND RECOMMENDATIONS
There is an increasing number of recommendations and guidelines available, the latest by the ESVS (12) and partly also by the International Working Group on the Diabetic Foot – Peripheral Arterial Disease (24). As a new approach, both emphasise the detrimental role of a combination of ischaemia and neuropathy in an ulcerated diabetic foot. The eternal problem of guidelines is how to gather all available evidence and interpret it without bias. All valid data and different specialities should be involved. The data should be analysed collaboratively in a stepwise and systematic fashion, followed by a discussion and collaborative writing and rewriting of the guidelines. Only scientific views should be accepted. Disagreements on the interpretation of the data may cause problems, even to the extent that members of the expert panel may withdraw during the work. Therefore, any guidelines are only as good as the efforts of the members and may become skewed by an unbalanced participation of different specialities. Furthermore, commercial interests, even if indirect, are difficult to avoid.
MANAGEMENT ISSUES
As to quality control, Scandinavian vascular registries have been a success story, especially in Sweden and Denmark, with a well-established and independently sponsored organisation. The Swedvasc and Karbase provide outcome data on the internet (http://www.ucr.uu.se/swedvasc/ and http://www.karbase.dk/). They allow surgeons, patients and policymakers to assess the activity of all Swedish and Danish vascular centres as regards the numbers and outcomes. Unfortunately, Finnvasc, the Finnish Vascular Registry (50), faded away after a good start due to national bureaucratic restrictions and a lack of financial support. Currently, only one third of the Finnish CLI data is included in the international Vascunet cooperation, HUSVasc being a part of it. Registries show remarkable differences between centres and countries.
As the first in Finland, Michael Luther conducted a profound treatment outcome study on critical leg ischaemia, including a cost-effectiveness analysis (51). Its conclusions are still valid: clear reduction in amputations and costs as well as better ambulation can be achieved with an aggressive reconstruction, surveillance and re-operation policy.
The effectiveness of treatment for CLI is not only affected by surgical decision-making, but cooperation with interventional radiologists and other medical specialities is needed for the care of this multi-morbid patient group. Managerial decisions affect both effectiveness and productivity. This is a typical problem in all Scandinavian capitals where expertise is diluted to two or more centres. According to the Helsinki experience, understanding the economy of scale and scope could increase productivity by more than 30% (52). Simultaneously, the centralisation of all expertise needed to treat multi-morbid CLI patients would improve the results.
CONCLUSIONS
The art is to revascularize the critically ischaemic limb properly with a resulting warm, well-perfused foot to allow ulcer healing. Bypass surgery and endovascular interventions are complementary techniques for revascularization. If both endovascular and bypass procedures are available with an equal outcome to be expected, endovascular treatments should be preferred. Unfortunately, the evidence is scarce and there are numerous issues to be resolved.
Footnotes
ACKNOWLEDGEMENTS
I thank Olof Lindfors, Björn Lindström, T.M. Scheinin, Pekka-T. Harjola, Johan Edgren, Michael Luther, Erkki Tukiainen, Riitta Lassila, Juha-Pekka Salenius, Mikael Railo, Ilkka Kantonen, Anders Albäck, Fausto Biancari, Maarit Venermo, Pekka-S. Aho, Pirkka Vikatmaa, Wolf-Dieter Roth, Kimmo Lappalainen, Sorjo Mätzke, Maritta von Bell, Päivikki Määttälä, Anna Mannerkoski, Anita Mäkelä and Leena Multanen, in addition to all vascular surgeons or interventional radiologists, trained or in training, who have visited, passed through or stayed with me in Helsinki or whom I have met anywhere in the world, for the shared burning interest, the friendship and the opportunities I have been granted.
