Abstract
Despite the health outcomes associated with exposure to indoor microbial aerosols, the environmental factors contributing to indoor microbial proliferation in residential environments have not been extensively investigated. This study assessed factors associated with indoor bacterial and fungal aerosols in residential apartments within Ibadan, Nigeria. A 12-month survey on housing characteristics and estimation of indoor bacterial and fungal aerosols was carried out in a sample of 356 residential apartments in Ibadan using a standardized home walkthrough checklist and an indoor sampling method. Indoor levels of microbial concentration, total bacterial count, total fungal count, richness (R) and Simpson diversity (D) were assessed and categorized as either high (>median) or low (≤median) exposures. The results showed that occupant density (adjusted odds ratio (aOR) = 1.03, 95% confidence interval (CI) = 1.01–1.05; aOR = 1.04, 95%CI = 1.02–1.06; aOR = 1.04, 95%CI = 1.02–1.06), leaking roofs (aOR = 2.51, 95%CI = 1.52–4.27; aOR = 1.79, 95%CI = 1.13–2.88; aOR = 1.85, 95%CI = 1.17–2.97) and visible dampness (aOR = 4.67, 95%CI = 2.21–10.34; aOR = 4.37, 95%CI = 2.22–8.81; aOR = 4.28, 95%CI = 2.17–8.59) were independently associated with higher levels of bacterial concentrations, richness and diversity respectively. Culturally relevant interventions targeted at these factors would be promising in managing microbial proliferation in residential environments.
Introduction
Residential exposures to indoor microbial aerosols have been linked to diverse health outcomes, especially respiratory tract infections, including asthma,1–3 rhinitis,4,5 lower respiratory tract infections (LRTIs)6–8 and respiratory symptoms (such as recurrent wheezing, coughing, difficulty breathing etc.),9,10 especially amongst children aged 1–59 months. 7 Over the past two decades, the United States Environmental Protection Agency conducted numerous studies revealing that indoor air quality in residential areas can be significantly more polluted than outdoor air. These studies indicated that indoor contamination levels can be 70–100 times higher than those outdoors. 11
Indoor exposure to microbial aerosols has been reported to be of significant public health importance because most human activities occur in such environments. 12 The majority of people, especially children, spend more than 90% of their time in indoor environments, mostly in homes, kindergartens, schools and others,12,13 which are, by implication, residential environments with the potential of nurturing relatively diverse microorganisms that may turn pathogenic and detrimental once the immune system weakens. The built environments are expected to be free from any harmful air pollutants; 14 however, certain indoor pollutants and at times, microbial agents can proliferate leading to higher concentrations indoors than outdoors. 15 Additionally, vulnerable populations, including children, older adults and those with underlying complications who are at higher risk of being negatively impacted by indoor microbial aerosols, tend to stay indoors longer. 16
The levels and distribution of microbial aerosols in residential settings exhibit significant variation across different geographical locations, seasons and weather conditions. 17 These variations are affected by several factors, including the microbial species present, the environmental conditions both indoor and outdoor, and the impact of human activities and urban development. 18
Nevertheless, the influence of environmental factors on microbial growth in residential environments has not been thoroughly studied, esspecially in Africa. However, most current studies assessing the environmental determinants of microbial proliferation in residential environments were conducted in developed countries such as Europe and the United States7,9 with limited information from low-middle-income countries. It is likely that the environmental factors promoting microbial proliferation in indoor residential environments differ by regions of the world, including but not limited to differences in the culture, housing pattern, climatic conditions and other environmental parameters.19,20 It is essential to assess the environmental factors influencing microbial proliferation in residential environments to design population-level interventions such as public health education and advisories to guide the public on factors promoting microbial proliferation in indoor environment to reduce the burden of microbial proliferation-related health outcomes.
In a previous report, we detailed the levels of bacterial and fungal aerosols in homes of children under-five, both with and without LRTIs. 6 In this paper, we focused on identifying the environmental factors that contribute to high and low levels of bacterial and fungal richness and diversity, an area which has not been previously explored.
This study, therefore, assessed the association of environmental factors with indoor microbial counts (IMCs), including bacterial and fungal richness and diversities in residential apartments within Ibadan, Nigeria.
Materials and methods
Data source
The current study used data from the home assessment for indoor microbiome study (HAIMS) conducted in Ibadan, Nigeria, to discern the environmental factors associated with microbial proliferation in residential environments in Ibadan, Nigeria. Details of the HAIMS study have been published elsewhere. 7 Briefly, the HAIMS study sought to assess the levels expressed as microbial counts, and diversity of microbial aerosols expressed as richness and diversity in 356 residence of children under the age of 5 years with and without LRTI in Ibadan, Nigeria's third largest urban centre. 21
Assessment of indoor environmental factors and conditions
Trained environmental inspectors were recruited to assess selected homes and document real-time observations using a validated walkthrough checklist. 22 Housing conditions inspected included type of house, age of building categorized as <10 or ≥10 years, presence/absence of air-conditioning system/ceiling fan, floor covers, roofing material, leaking roof categorized as extensive (if more than two spots), one or two spots and none, visible mould growth described as identifying algal growth on walls or roof, and dampness on surfaces within the house. Also, a survey was undertaken to assess various household characteristics, including house ownership, occupant density (calculated by dividing the total number of occupants by the total number of rooms), 6 types of cooking fuel and facilities used and the presence of pets. The cooking fuel used was categorized as either clean (electricity and liquid petroleum gas) or dirty (kerosene, charcoal and wood). 22 To monitor indoor meteorological conditions such as relative humidity (RH) and temperature (°C) in specific residences over a 24-h period, a real-time datalogger model 42,270 was employed.
Assessment of bacterial and fungal concentrations and diversity
To assess the presence of indoor airborne bacteria and fungi, an air sampler known as BioStage Anderson was utilized. This sampler was furnished with a 90 mm petri dish that held agar media, including Nutrient, Blood and McConkey Agar for bacteria isolates as well as Saboraud Dextrose Agar for fungi isolates. The sampler collected aerosol samples during the day (between 8:30 am and 11:00 am, assuming that most of the occupants had spent the whole night and early hours of the day in their apartment, during which activities such as cleaning, cooking, bathing and washing took place before leaving the building) at a height of approximately 1.5 m in the centre of a room (which represents the occupants’ breathing zone) and at an airflow rate of 1.698 ± 0.12 m3/h for 15 min. 23 After collection, the samples were transported to the laboratory in an ice pack and incubated according to the manufacturer's specifications. After incubation, a Quebec darkfield colony counter (Cambridge Instruments, Inc., Buffalo, NY) was used to count the number of colonies on each agar medium as colony-forming units per cubic metre (cfu/m3) using a standard formula. 24 The identification of bacterial and fungal isolates was done following standard procedures.23,24 The assessment of IMC, total bacterial count (TBC), total fungal count (TFC), microbial richness (R) and Simpson diversity Index (D) was conducted based on the individual species of bacteria and fungi identified. R was computed based on the number of individual species of bacteria and fungi per sample. D was calculated as the square of the ratio of bacterial and fungal species to the total bacteria and fungi genera. The sum of the squared proportions for all bacterial and fungal species was calculated, and the reciprocal sum was recorded.
Statistical analysis
All walkthrough checklists and aerosol data were entered into an Excel spreadsheet, validated and uploaded to IBM version 25 and R 4.1.1 software for further analyses.
Bacterial and fungal estimations, including microbial counts and microbial diversity measured by richness and diversity, were categorized using the median values. The categories were defined as high (> median) and low (≤ median) exposures. To compare environmental factors associated with IMC, TBC, TFC, R and D, bivariate analysis was conducted using a t-test (for continuous data) and Chi-square test (for categorical data). Furthermore, multivariable-adjusted regression models were used to determine the odds ratio (OR) and 95% confidence interval (CI) of residential environmental factors associated with microbial proliferation in indoor environments. The OR and 95% CIs were estimated in our models, and all statistical tests of hypotheses were two sided with statistical significance considered for p-value < 0.05.
Results
A large proportion, 179 (50.3%) of homes visited were mini-flat. The mean occupant density was 4.46 ± 1.89, and more than half of homes, 180 (50.6%) had a low occupant density. A higher proportion, 237 (66.6%) of homes were rented by occupants, while less than half (27.2%) had one or two spots with leaking roofs. Visible dampness and mould growth anywhere in the house were observed in 30.1% and 17.1% of homes, respectively, while 29.5% were observed to own a pet. The mean indoor temperature and RH were 31.26 ± 2.17°C and 66.15 ± 11.98%, respectively (as shown in Table 1).
Description [n (%)] of home characteristics.
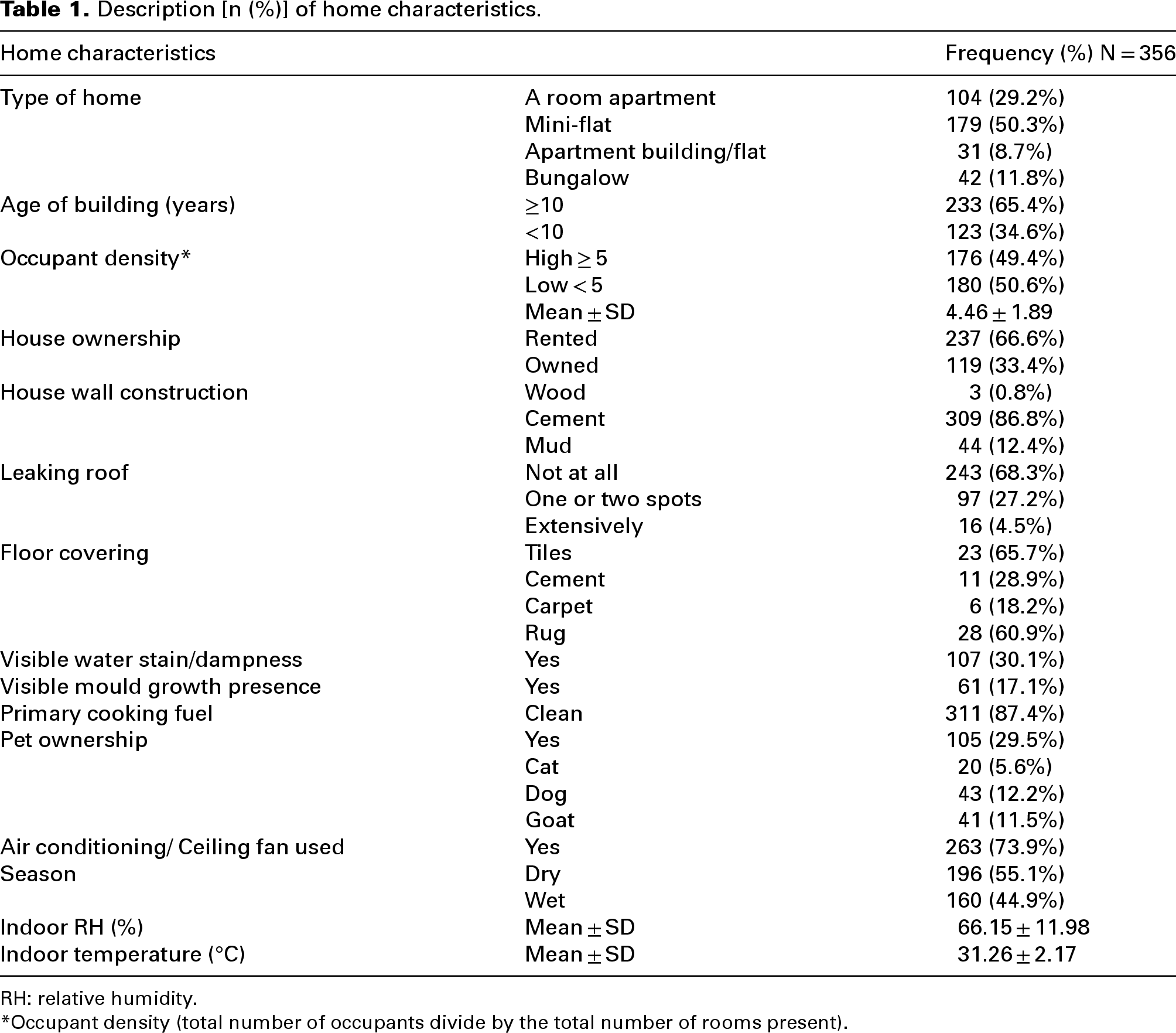
RH: relative humidity.
*Occupant density (total number of occupants divide by the total number of rooms present).
The majority of homes visited had higher levels of TBC (50.6% vs 49.4%) and TFC (51.7% vs 48.3%). Also, a higher proportion of homes had high levels of bacterial (R = 61.8%; D = 61.5%) and fungal (R = 66.9%; D = 67.1%) richness and diversity, respectively (Figure 1).
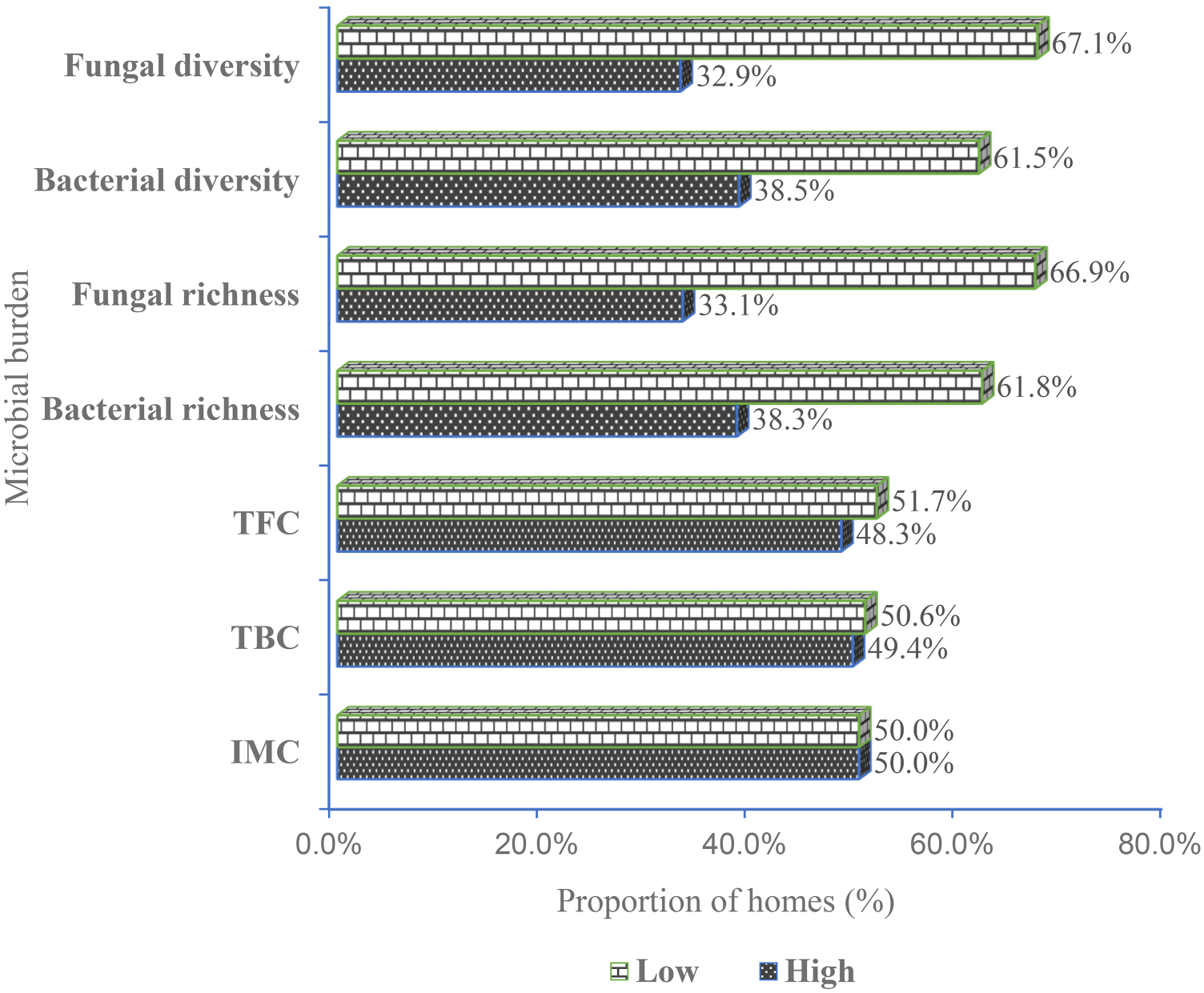
Proportion of homes with low vs high bacterial and fungal levels. IMC (low: ≤ 515 cfu/m3 vs high: > 515 cfu/m3), TBC (low: ≤ 456 cfu/m3 vs high: > 456 cfu/m3), TFC (low: ≤ 54 fu/m3 vs high: > 54 cfu/m3), bacterial richness (low: ≤ 2 vs high: > 2), bacterial diversity (low: ≤ 2.0 vs high: > 2.0), fungal richness (low: ≤ 2 vs high: > 2) and fungal diversity (low: ≤ 2.0 vs high: > 2.0). IMC: indoor microbial count; TBC: total bacterial count; TFC: total fungal count.
Buildings ≥10 years were found to have significantly higher levels of IMC (75.3% vs 55.6%), TBC (75.0% vs 56.1%), TFC (72.1% vs 59.2%), bacterial proliferation (R = 80.8% vs 55.9%; D = 80.3% vs 56.2%) and fungal proliferation (R = 72.0% vs 62.2%; D = 72.6% vs 69.1%). Also, high occupant density was significantly associated with higher levels of IMC (59.0% vs 46.6%), TBC (59.0% vs 46.7%) and bacterial proliferation (R = 59.6% vs 45.0%; D = 59.1% vs 45.2%). In addition, homes with extensively leaking roofs were associated with high levels of IMC (7.9% vs 1.1%), TBC (8.0% vs 1.1%), TFC (7.0% vs 2.2%), bacterial proliferation (R = 8.1% vs 2.3%; D = 8.1% vs 2.3%) and fungal proliferation (R = 6.8% vs 3.4%; D = 6.8% vs 3.3%). Similarly, a higher level of IMC (11.2% vs 2.2%), TBC (11.2% vs 2.2%), TFC (8.1% vs 5.4%), bacterial proliferation (R = 24.3% vs 5.9%; D = 24.1% vs 5.9%) and fungal proliferation (R = 21.2% vs 8.8%; D = 21.4% vs 8.8%) were found in homes with visible dampness or moisture on surfaces. Also, using an air-conditioning system or fan in the house was associated with a lower level of IMC (67.4% vs 80.3%), TBC (68.2% vs 79.4%) and TFC (68.0% vs 79.3%), but not with the level of diversities (Tables 2 and 3).
Description [n (%)] of home characteristics and exposure levels by microbial concentrations.
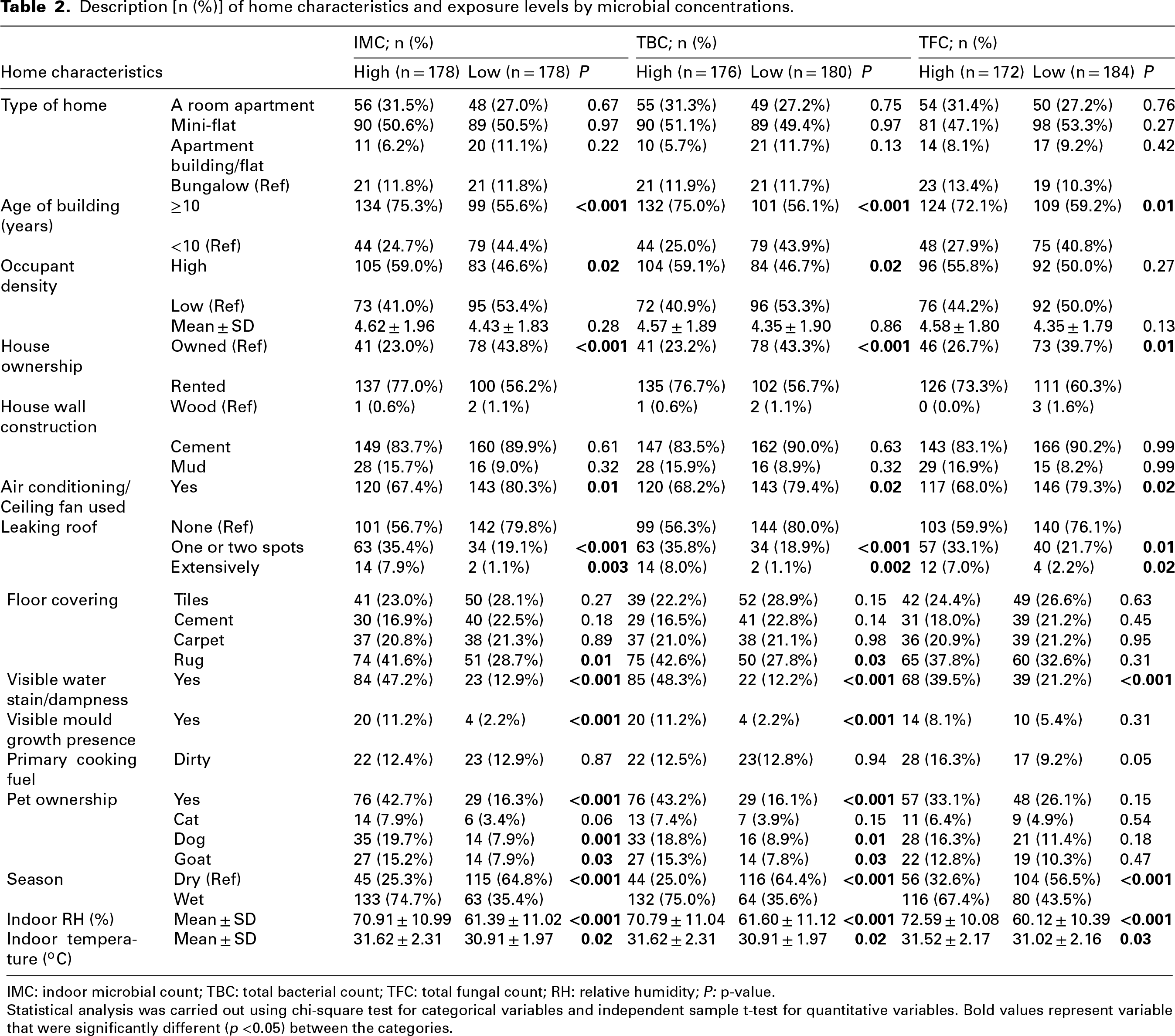
IMC: indoor microbial count; TBC: total bacterial count; TFC: total fungal count; RH: relative humidity; P: p-value.
Statistical analysis was carried out using chi-square test for categorical variables and independent sample t-test for quantitative variables. Bold values represent variable that were significantly different (p <0.05) between the categories.
Description [n (%)] of home characteristics and exposure levels by microbial richness and diversity.
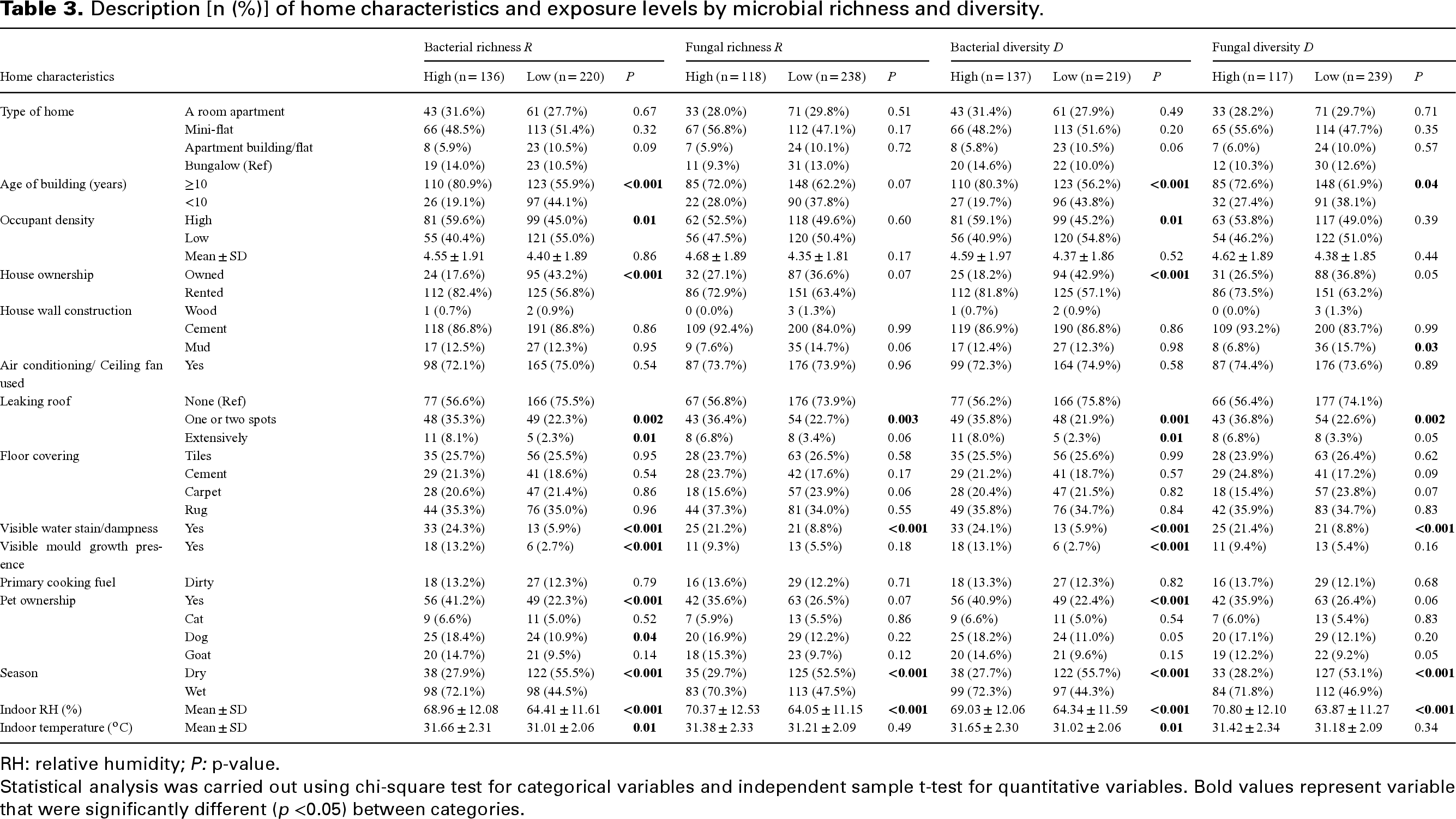
RH: relative humidity; P: p-value.
Statistical analysis was carried out using chi-square test for categorical variables and independent sample t-test for quantitative variables. Bold values represent variable that were significantly different (p <0.05) between categories.
In the unadjusted regression model (Table 4), high occupant density was associated with higher levels of IMC (OR; 1.43, 95%CI: 1.01, 2.18), TBC (OR; 1.44, 95%CI: 1.02, 2.48), bacterial richness (OR; 1.43, 95%CI: 1.01, 2.18) and bacterial diversity (OR; 1.75, 95%CI: 1.14, 2.70), but not with TFC (OR; 0.99, 95%CI: 0.72, 1.66) and fungal richness (OR; 1.13, 95%CI: 0.72, 1.75). Visible dampness and leaking roofs were also associated with both bacterial and fungal counts, richness and diversities.
Unadjusted regression model for indoor environmental factors associated with levels of microbial count, richness and diversity in residential environments.
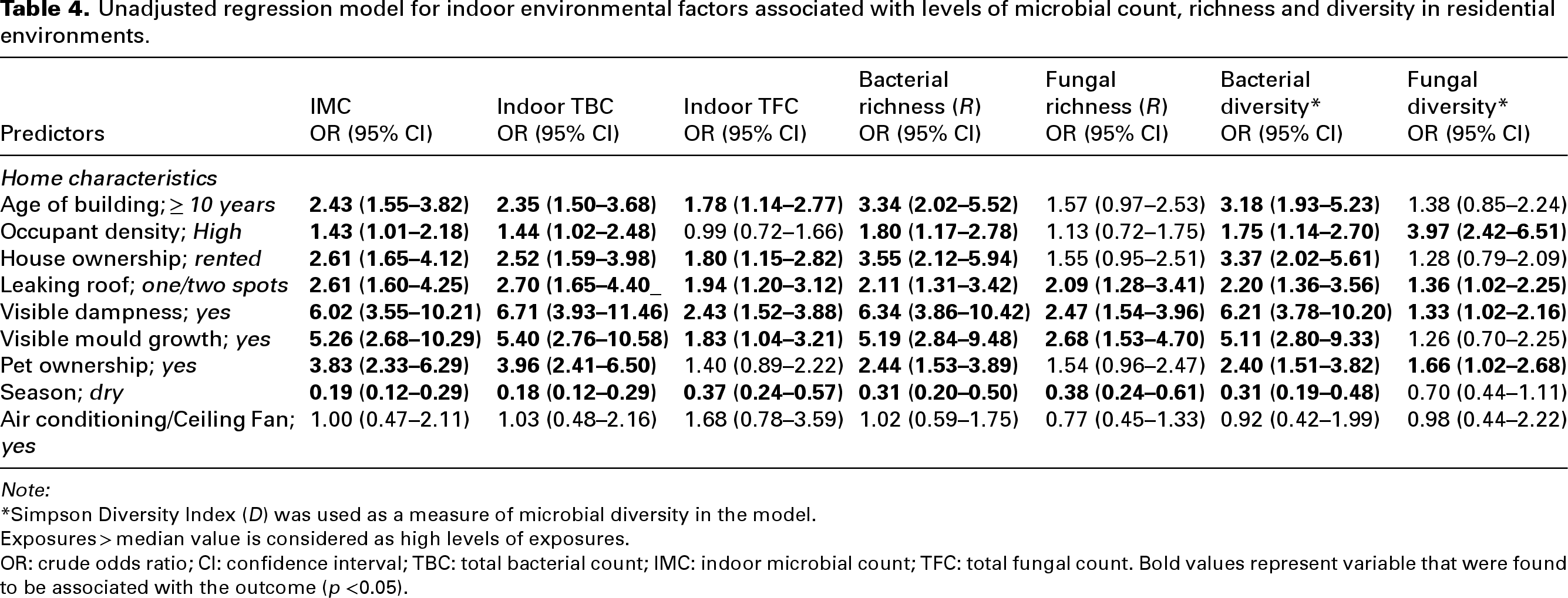
Note:
*Simpson Diversity Index (D) was used as a measure of microbial diversity in the model.
Exposures > median value is considered as high levels of exposures.
OR: crude odds ratio; CI: confidence interval; TBC: total bacterial count; IMC: indoor microbial count; TFC: total fungal count. Bold values represent variable that were found to be associated with the outcome (p <0.05).
In the multivariable-adjusted regression model (Table 5), high occupant density was independently associated with higher levels of IMC (adjusted odds ratio (aOR); 1.03, 95% CI: 1.01, 1.05) and TBC (aOR; 1.03, 95% CI: 1.01, 1.05), but not with indoor TFC (aOR; 1.00, 95% CI: 0.98, 1.01). Similarly, high occupant density was associated with bacterial richness (aOR; 1.04, 95% CI: 1.02; 1.06) and bacterial diversity (aOR: 1.04, 95% CI: 1.02, 1.06) but not with fungal richness (aOR; 1.00, 95% CI: 0.98, 1.02) and fungal diversity (aOR; 1.01, 95% CI: 0.99. 1.02). Also, compared to homes with no leaking roof, homes with leaking roofs at one or two spots were more likely to have higher levels of IMC (aOR; 2.42, 95% CI: 1.47, 4.12), TBC (aOR; 2.51, 95% CI: 1.52, 4.27), TFC (aOR; 1.74, 95% CI: 1.16, 2.66), bacterial proliferation (R: aOR; 1.79, 95% CI: 1.13, 2.88 and D: aOR: 1.85, 95% CI: 1.17, 2.97) and fungal proliferation (R: aOR: 1.57, 95% CI: 1.05, 2.36 and D: aOR: 1.59, 95% CI: 1.06, 2.39). Visible dampness or moisture in homes was observed to significantly independently contribute to higher levels of IMC (aOR: 3.82, 95% CI: 1.82, 8.32), TBC (aOR: 4.67, 95% CI: 2.21, 10.34), TFC (aOR: 1.93, 95% CI: 1.02, 3.75) and bacterial proliferation (R: aOR: 4.37, 95% CI: 2.22, 8.81 and D: aOR: 4.28, 95% CI: 2.17, 8.59). Notably, using an air-conditioning system or ceiling fan in the house was negatively associated with higher levels of IMC (aOR; 0.43, 95% CI: 0.23, 0.78) and TBC (aOR; 0.50, 95% CI: 0.23, 0.90).
Multivariate regression models for indoor environmental factors associated with levels of microbial count, richness and diversity in residential environments.
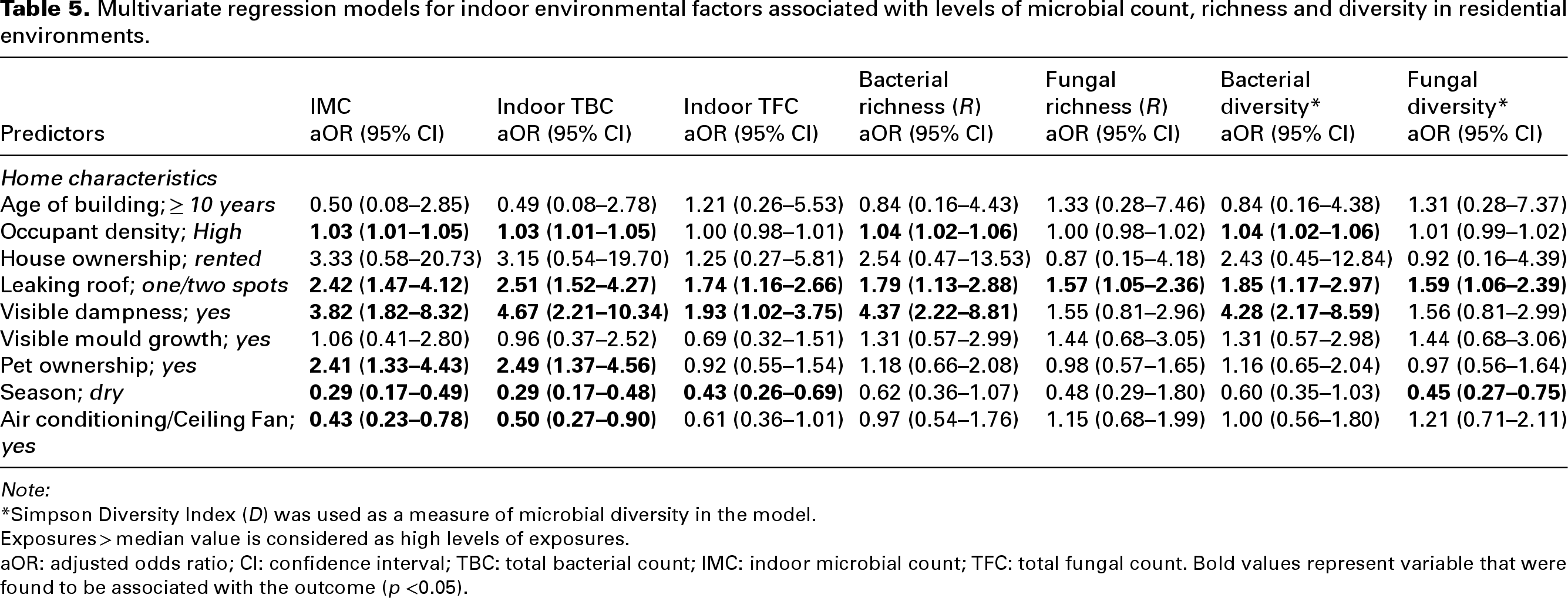
Note:
*Simpson Diversity Index (D) was used as a measure of microbial diversity in the model.
Exposures > median value is considered as high levels of exposures.
aOR: adjusted odds ratio; CI: confidence interval; TBC: total bacterial count; IMC: indoor microbial count; TFC: total fungal count. Bold values represent variable that were found to be associated with the outcome (p <0.05).
Discussion
In this study, we assessed environmental factors associated with microbial proliferations, including (IMC, TBC, TFC, bacterial richness and diversity, fungal richness and diversity) in residential environments, and found that high occupant density, leaking roofs and the presence of visible dampness were three unique environmental factors associated with microbial proliferation in residential environments. Of the three factors, visible dampness presented the highest odds (fourfold odds for high IMC, fivefold odds for high TBC, twofold odds for high TFC, fourfold odds for high bacterial richness and diversity and twofold of being at odds of high fungal richness and diversity).
The human population is surrounded by a diverse bacterial community from various places like human skin, the oral cavity and even clothes.25,26 The concentration of bioaerosols has been found to increase in indoor environments such as University classrooms, offices, health care facilities and other indoor settings when they are in use and decrease when they are empty. 27 This study revealed that occupancy contributes significantly to the concentration and diversity of aerosols in residential environments, corroborating a meta-analysis. 28 Findings from our study suggest that humans are likely the primary source of airborne bacteria in residential environments as high occupancy level was directly associated with increased bacterial concentration and diversity.
A study by Degois et al. 29 found a correlation between occupancy and skin-related bacteria or yeasts irrespective of the ventilation system in place. However, another study by Fujiyoshi et al. 30 reported that different levels of occupancy in residential or indoor environments have no impact on microbial composition. According to Nevalainen et al., 31 fungal particles from outdoor environment can be carried into indoor space by building occupants and pets through their clothes, skin, shoes and so on therefore elevating the fungal levels indoors.
As evident in our multivariate regression model, water leaks may also facilitate bacterial and fungal growth in buildings. Dales et al., 32 in their study, examined over 400 bioaerosol samples collected from selected homes and found that water damage was associated with a 50% increase in fungal concentrations. According to our study, fungal concentration and diversity were significantly associated with moisture and dampness in the home. Several epidemiological studies considered water activity as a supporting factor for fungal development. 33 Several studies have indicated a direct relationship between dampness and indoor airborne fungi concentrations.34,35 For instance, a case–control study conducted in Australia reported that residential settings with a musty odour and water intrusion exhibited notably higher levels of airborne fungi. 34 Additionally, a cross-sectional study demonstrated that homes with water sources and visible mould displayed two or three times greater concentrations of airborne fungi. 35
In our study, visible dampness in homes correlated significantly with bacterial and fungal proliferations. This is in keeping with the findings of Cox et al., 36 in their study to explore quantitative metrics of observed dampness and mould on fungal and bacterial microbiomes. One of the major indicators of poor ventilation in residential environments is dampness, which may lead to increased levels of other potentially harmful indoor pollutants affecting vulnerable populations, especially children, resulting in health implications such as childhood helicobacter pylori infections, 37 childhood asthma 38 and the development of childhood otitis media. 39 A high moisture level in homes will likely promote the proliferation of several microbial species.40,41 The concentrations and diversity of microorganisms are relatively higher in buildings with visible dampness than those without. 41 Therefore, the monitoring and control of residential humidity levels through the use of dehumidifiers, chillers and humidifiers and the regulation of ventilation rates can reduce aerosol formation. 12
We found a negative association between using a functional air-conditioning system or ceiling fan and a high level of bacteria in the home. As reported by Pandey et al., 42 increased circulation speed of the ceiling fan in residential environments reduces the parabolic path of bacterial and fungal particles. This study reveals that with an increase in the rotation speed of the ceiling fan, the larger particles settle down quickly towards the floor area and disperse along the floor, while the smaller particles are transported to various locations within the residential environment under the influence of airflow produced by the ceiling fan.43,44 While studies on the impact of ceiling fans have attracted little or no attention with respect to infectious diseases, a relatively high proportion of studies have investigated the impact of ceiling fans on airflow patterns.45–47 Although the ceiling fan is not an effective device as an Air Conditioner (AC) for cooling purposes and maintaining the temperature, it is an inexpensive and practical air circulation device that can improve occupants’ thermal sensation by circulating the air through an indoor area.48,49 Therefore, the airflow dynamics provided by the ceiling fans or ACs increases the ventilation rate, thereby subduing microbial growth and proliferation within the residential environments. 50 In addition, AC naturally dehumidifies the indoor air by lowering the air temperature, causing moisture to condense and be removed from the air. 51 The removal of moisture from such environment directly interferes with microbial growth and proliferation. 52
One of the key strengths of the study was the employment of a culture-dependent technique to assess microbial abundance, offering in-depth insights into microbial concentrations and diversity in low-resource settings like Nigeria. Evaluating microbial diversity through species richness and diversity provides a straightforward method to understand the structure of the microbial community. The study's limitation was the sample size, which may not have been sufficiently powered for this study. Therefore, future studies should expand the size to accommodate diverse geographical regions to enhance external validity. We acknowledge the limitations of culture-dependent approaches and the possibilities of missing some non-culturable species. However, we strongly believe that our study has provided a foundation for future studies to consider supplementing with molecular approach for a more comprehensive understanding of the microbial diversity. The assessment of the temporal relationship between dampness and microbial proliferation was difficult due to the design employed. Therefore, future longitudinal studies are required to strengthen our understanding of the cause-and-effect dynamics between dampness and microbial growth.
Conclusion
This study found high occupant density, visible dampness and leaking roof as major environmental factors independently associated with higher burden of bacterial and fungal proliferation in residential environments. Therefore, culturally relevant interventions such as improving roof integrity, encouraging the use of ceiling fans and air-conditioning systems, would be promising in managing microbial proliferation in living environments, thereby mitigating the burden of communicable and non-communicable disease transmission in Ibadan, Nigeria.
Footnotes
Acknowledgements
We are also grateful to the families whose residential apartments were made available and accessible for indoor microbial assessment and analysis.
Author contributions
AGF did conceptualization, writing – original drafted preparation and statistical analysis. RNN and NJ participated in study design and conceptualization, reviewed the study instruments, guided the statistical analysis and provided extensive review of the drafts; APO and JOO contributed to the statistical analysis and writing – review. All authors have read and agreed to the final version of the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval and consent to participate
The study was approved by the University of Ibadan/University College Hospital Ethics Committee (Ref No.: UI/EC/17/0077) and the Oyo State Research Ethical Review Committee (Ref. No.: AD13/479/462).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We acknowledge the support received from the College of Health (CHS) PhD Scholarship of the University of KwaZulu-Natal to facilitate field activities.
