Abstract
In recent years, several epidemics by transmissible respiratory viruses have emerged, especially pandemics caused by coronaviruses. The most significant public health emergency may be the COVID-19 pandemic. Identifying the transmission of infectious disease plays an important role in healthcare for protecting and implementing effective measures for public health. However, studies on SARS-CoV-2 transmission dynamics are lacking. Infection with the airborne virus is very important and airborne transmission is likely to cause major problems. However, research on the aerosol route of the virus is very limited. Here, we aimed to present airborne coronavirus detection methods in previous studies and address the importance of methodology for the future. In previous studies on airborne coronavirus, detection methods were different in each study. Therefore, comparison between the airborne virus detected in each study is impossible, and the risk assessment could not be properly analysed due to limitations in applying it as basic data. There is currently a risk assessment for coronavirus, but the risk assessment due to airborne transmission is insufficient. Therefore, recommending accurate guidelines for airborne transmission is difficult. Future research should be conducted to standardize airborne virus detection methods to prevent transmission through rapid risk assessment and monitoring.
Introduction
Pandemics by coronavirus
The leading cause of global disease and mortality is respiratory viral infections. 1 In recent years, several epidemics by transmissible respiratory viruses have emerged, especially the pandemics caused by coronaviruses. In November 2002, the first patient with severe acute respiratory syndrome (SARS) was identified in Foshan, China and by February 2003, there were more than 300 cases of SARS, a novel coronavirus (SARS-CoV). 2 By July 2003, 8096 cases and 774 deaths in 29 countries were reported. 3 In June 2012, a person in Saudi Arabia died by acute pneumonia and renal failure because of a Middle East respiratory syndrome coronavirus (MERS-CoV) infection. 4 MERS-CoV continued to emerge and spread to countries because of the travel of infected persons; often, these MERS cases resulted in nosocomial transmissions. 5 By December 2019, there were 2494 confirmed cases of MERS and 858 deaths. 6 In late 2019, a novel human coronavirus, SARS coronavirus 2 (SARS-CoV-2), firstly reported from Wuhan, China; and coronavirus disease 2019 (COVID-19) spread globally at a rapid rate.7,8 In March of 2020, there were more than 180,000 confirmed COVD-19 cases and more than 7000 death in 96 countries. A global SARS-CoV-2 pandemic was declared by the World Health Organization (WHO) on 11 March 2020. 9 This pandemic has more than 1,237,000 confirmed COVID-19 cases in at least 183 countries, including more than 67,000 deaths and more than 253,000 recoveries as of 5 April 2020. 10 The most significant public health emergency in a century may be the COVID-19 pandemic. 1
Potential of coronavirus transmission through aerosols
Determining the route of transmission of viral infections is hard in an environment with undefined parameters. 11 Viral infection can occur via direct contact when infected hosts are close. However, infected hosts can transmit viral infections without direct contact since infectious viruses can exist outside their hosts for a prolonged time. 11 Identifying the transmission of infectious disease plays an important role in healthcare for protecting and implementing effective measures safeguarding public health. 1 Three sources are suggested for the transmission of SARS-CoV-2 in humans: (i) inhalation of liquid droplets, (ii) close contact with infected persons, and (iii) touching surfaces contaminated with SARS-CoV-2. 12 As COVID-19 has spread rapidly and globally, experts have conducted investigations on the potential of airborne transmission of the virus and the characteristics of the disease. 13 Furthermore, previous studies have proven that pathogens are transmitted via airborne routes in specific spaces, and many respiratory diseases are caused by airborne routes, such as tuberculosis, measles and chickenpox. 12 Generally, bioaerosols ranging from 1 to 5 µm float in the air, whereas larger particles are deposited on surfaces. 14 Thus, an aerosol can float for a long time and move far away, especially nano-sized aerosols. 15
Since studies on SARS-CoV-2 transmission dynamics are lacking, guidelines for airborne and droplet isolation precautions were published by the WHO, the Centres for Disease Control and Prevention (CDC) in the USA, and other public health organizations. 1 The transmission of SARS-CoV-2 in hospitals has been reported, but the role of aerosols in the transmission is unclear and controversial.13,16 The guidelines for COVID-19 published by the National Health Commission of the People’s Republic of China stated that the transmission could potentially be caused by long-term exposure to high aerosol concentrations. 14 The WHO also believes that further evidence is needed to assess the possibility of aerosol transmission. Previous studies have suggested the airborne transmission potential of other coronaviruses, such as SARS-CoV-1 and MERS-CoV.17,18 In addition, a role for airborne transmission was reported in epidemiology studies of SARS-CoV-1 and MERS-CoV.19,20 However, complete and conclusive evidence of airborne transmission has not been reported. 13 Several studies have identified that aerosols were involved in the spread of viral diseases, such as SARS, MERS and influenza A virus subtype H1N1.21–23 Due to a lack of information on airborne SARS-CoV-2 and the difficulties in sampling, investigating concentration patterns and behaviour during airborne transmission is difficult. This limits the risk assessment, prevention and control of COVID-19. To determine the probability of infectious disease transmission through airborne routes, epidemiological and bioaerosol studies should be conducted.11,21 Here, we aimed to present detection methods for airborne coronavirus used in previous studies and thus emphasizing the importance of methodology for any future studies.
Air sampling and detection methods for airborne coronavirus
Many sampling and detection methods have been used to detect airborne viruses, such as bacteriophage, influenza virus, foot-and-mouth disease virus, coronavirus and porcine reproductive and respiratory syndrome virus. 11 Specifically, many studies have targeted Enterobacteria phage MS2 but only a few studies on airborne coronavirus have been conducted. Presently, there is neither a single sampling method suitable for the collection of different types of airborne microorganisms nor a standard testing protocol available. 24 Several studies on coronavirus-targeted human coronavirus 229E and other studies were conducted during the SARS-CoV-1, MERS-Cov and SARS-CoV-2 outbreaks. Table 1 shows the studies on the measurement of airborne coronavirus, SARS-CoV-1, MERS-CoV and SARS-CoV-2.
Summary of sampling method studies for coronavirus.

Filters for coronavirus sampling
Since most samplers are not efficient in capturing particles with an aerodynamic diameter under 500 nm, various filters, differing in composition, pore size and thickness, have been used to capture airborne viruses. 11 Generally, polytetrafluoroethylene (PTFE) filters and gelatine filters are used. The physical collection efficiencies of gelatine filters, as well as PTFE filters, exceed 96%. 25 However, since filters might damage the structure of viruses, filters have not been commonly used for airborne viruses. 11 Recently, gelatine filters have been used to analyse viral infectivity. For example, an MD8 air scan sampling device with 80 mm gelatine membrane filters with a pore size of 3 µm (Sartorius, Goettingen, Germany) has been successfully used for airborne MERS-CoV26,27 and SARS-CoV-2. 12 The indicated pore size of the gelatine filter does not accurately represent the collection particle size of the filter. This is because capturing of the aerosol is performed by physical factors, e.g. Brownian motion, inertial impact, diffusion, interception and electrostatic attraction, etc. Therefore, it can be concluded that a 3 µm pore size filter can successfully capture sub-micron aerosols. Although this can be very useful for sampling virus, gelatine filters are affected by environmental conditions, such as humidity. 11 PTFE filters with a pore size of 0.3 µm are the best method for long-term sampling of virus-laden particles ranging in size from 10 to 900 nm in diameter. 25 Polyvinylidene fluoride nanofibre filter has been suggested for filtering particles under 100 nm, such as airborne SARS-CoV-2. 28 This filter can be used for airborne virus sampling.
When a coronavirus pandemic occurs, the monitoring of airborne viruses is very important to prevent transmission. For the real-time monitoring of airborne virus, the standardization of filters is mandatory, especially to analyse infectivity, necessary for monitoring coronavirus. The standardization of filters for virus sampling has not been pursued because of the damage filters can cause to the viruses. 11
Detection method
Many previous studies detected viruses using reverse transcription-polymerase chain reaction (RT-PCR) or cultures. Culture-based measurements are mainly used to determine airborne virus concentrations. However, air sampling using the culture medium is inadequate for calculating infectious viruses, since most sampling methods have the potential to influence viral infectivity. In contrast, PCR detects viruses regardless of infectiousness in air samples. 11 Recently, to accurately and speedily detect virus, droplet digital PCR (ddPCR) has been used in many studies for pathogen diagnosis, mutation detection and transgenic studies.29–31 An airborne SARS-CoV-2 detection study was also performed using ddPCR. 12 ddPCR can measure the endpoint of the nucleic acids’ quantity without standard samples. 32 Also, RT-ddPCR was developed and used for detecting virus.33,34 Furthermore, in a recent study, a real-time on-site bioaerosol monitoring system was used to measure bioaerosols of various sizes 35 and might have the potential to measure airborne coronavirus.
Risk of COVID-19 by aerosol
Airborne SARS-CoV-2 concentrations
Table 2 shows the field measurements of airborne SARS-CoV-2 concentrations. All research was conducted in a hospital. In the patient rooms, the maximum airborne SARS-CoV-2 concentrations were 9 copies m−3 in Fangcang Hospital, China, 12 and 8.224 ± 8.266 copies per dm 3 in the University of Nebraska Medical Center, USA. 1 In addition, the total airborne SARS-CoV-2 levels were ND (not detected), 3384 RNA copies m−3 and 1843 RNA copies m−3 in the three airborne infection isolation rooms (AIIRs) at the National Centre for Infectious Disease, Singapore, respectively. 36 The airborne SARS-CoV-2 was detected in the patient rooms and also in the toilet, staff rooms and hallways. The airborne SARS-CoV-2 concentrations in the patient toilet were higher than in the patient rooms. 12 However, these airborne SARS-CoV-2 concentrations do not represent concentrations in the entire room. For example, the airborne SARS-CoV-2 concentrations near the patients were higher than those more than 1.8 m from the patients 1 and airborne SARS-CoV-2 was not detected in patients’ rooms over 2 m from patients’ beds.13,35 The possibility of infection could become especially high if a COVID-19 patient nearby has a high level of dispersion viral copies. The SARS-CoV-2 concentration after coughing was 2.56 log copies mL−1 without a mask, 2.42 log copies mL−1 with a surgical mask and 1.85 log copies mL−1 with a cotton mask, respectively. All swab samples of outer mask surfaces were positive for SARS–CoV-2, whereas most swab samples of inner mask surfaces were negative in the patient room. 37 This result was not airborne. However, coughs have the potential to distribute airborne viruses, although the size and levels of airborne SARS-CoV-2 generated by coughing are unknown. Thus, the distance from the patient is important when measuring airborne SARS-CoV-2 concentrations, and airborne virus studies should consider the sampling site in relation to the patients. Furthermore, airborne SARS-CoV-2 was detected in staff rooms and hallways. Most medical staff areas were positive. Specifically, 42 copies m−3 was found in the protective apparel removal room, 12 and the airborne SARS-CoV-2 concentration in hallways was similar to that in patient rooms. 1 Airborne SARS-CoV-2 was also detected in outdoors. 12 Positive airborne SARS-CoV-2 findings outside patient rooms, including medical staff rooms, hallways and outdoors, suggested that infectious airborne SARS-CoV-2 could remain outside for a long time and cause infections.
Summary of studies of airborne SARS-CoV-2 concentrations.
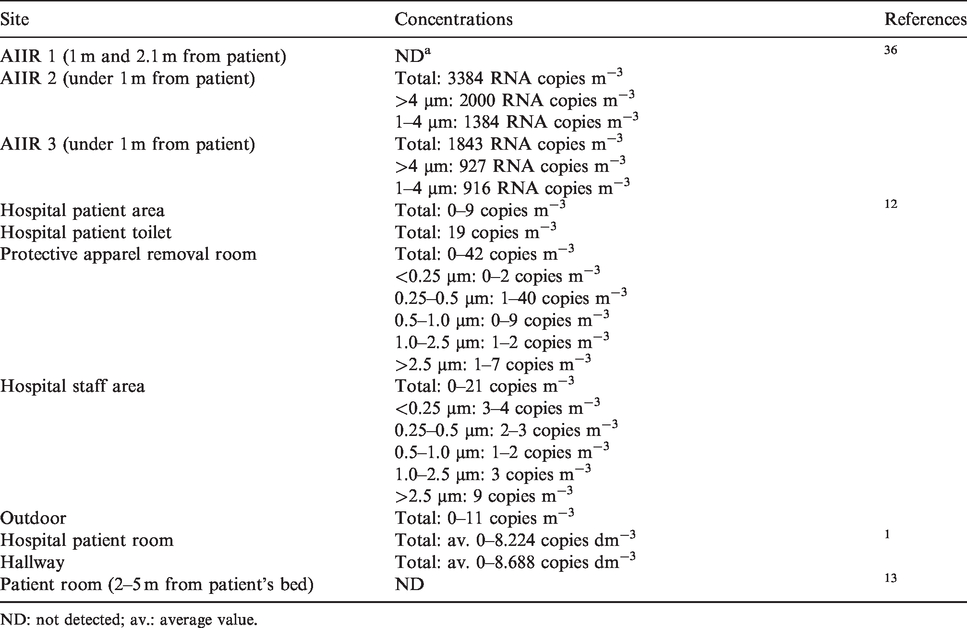
ND: not detected; av.: average value.
Studies evaluating airborne virus concentrations showed different results. Several studies showed positive results, whereas other studies reported negative results, probably because the standardization of measurement methods, indoor environments and patient conditions, such as coughing and patient location, were different. Although the studies were able to determine the concentration of airborne SARS-CoV-2, they tended to conclude that the detection itself would represent a danger.
Size distribution of SARS-CoV-2 aerosol
According to a recent study, the minimum size of SARS-CoV-2 is about 60 nm, and it can be attached to a larger carrier aerosol with a combined size of about 100 nm or larger.8,28 SARS-CoV-2 aerosols measured by the Fangcang Hospital in China were divided into two distribution groups according to the aerodynamic diameter, a submicron group (0.25 to 1.0 µm) and a super-micron group (over 2.5 µm). 12 The reason for the various particle sizes is that the aerosol size generated varies depending on human activity. The particle size emitted from normal mouth breathing was less than 1 µm in diameter, but the average droplet size generated through coughing was 12 to 14 µm. 38 In other previous studies, the diameter of the aerosols emitted by human activity, such as breathing, coughing and speech, was under 100 µm particle sizes that human can inhale.10,39 Coughing, sneezing, breathing or speaking of a person infected with virus results in the exhalation of virus particles ranging in size from 0.3 to 100 µm in diameter, which may become bioaerosols. For this reason, the SARS-CoV-2 size distribution ranges from nano to micron sizes. Small-sized particles can penetrate the lungs easily if inhaled, causing respiratory diseases and infections. 24 The viruses that manage to enter through airways or ingestion, especially through the air, are particles sized 1 to 4 µm. Furthermore, they can penetrate even through the immunological barrier. For example, in the case of virus type influenza, influenza RNA was detected in aerosol particles generated by human coughs. Of these particles, 35% contained particles over 4 µm in diameter, while 23% contained 1–4 µm particles and 42% contained particles under 1 µm. These results suggest that the aerosols generated by coughing contained viral RNA of many particle sizes. 40
Risk assessment of airborne SARS-CoV-2
SARS-CoV and MERS-CoV infections are known to be caused by aerosol transmission.22,41 Accordingly, researchers have tried to analyse the risk of exposure through a risk assessment of aerosols containing viruses. Although the scientific evidence for the role of airborne transmission can be applied in an epidemic, airborne transmission as a route of contagion has been insufficiently investigated. 42 Therefore, the risk assessment study was not conducted due to the lack of information on aerosol exposure paths, the virus concentration in aerosols, the amount of exposure and dose-response evaluations. Only infection risk assessments were conducted for researchers and medical staff. As a case study for MERS-CoV, a comparative evaluation was conducted for nurses, healthcare workers, family visitors and patients using the Quantitative Microbial Risk Assessment (QMRA) approach. 22 However, the dose-response model for MERS-CoV has not been completed, and some items were evaluated based on existing research literature and had limitations. In the case of SARS-CoV, there are no risk assessment case studies. Only basic dose-response model development studies have been reported. 43 In the case of SARS-CoV-2, the European Centre for Disease Prevention and Control (ECDC) COVID-19 rapid risk assessment report has stated many unknowns, including the virulence/pathogenicity, the mode of transmission, the reservoir and the source. There have been reports of significant uncertainties. 44 The US Centres for Disease Control and Prevention (CDC) and World Health Organization (WHO) also do not provide information on risk assessment due to the lack of information regarding SARS-CoV-2. Only private and healthcare providers have developed and provided guidance via a questionnaire survey method to assess the risk of potential exposure and manage themselves.45,46
Conclusion
Reports of COVID-19 have emphasized the transmission and lethality by SARS-CoV-2. 47 However, the airborne SARS-CoV-2 has not been clearly determined. The sources, transport, clearance, transmission and inactivation of airborne SARS-CoV-2 RNA should be identified and then interdisciplinary collaborations are required. 10
Airborne virus sampling methods have been improved for promoting human health, and previous studies on coronavirus were conducted during world pandemics. However, since the standardization of sampling and detection methods is lacking, the recommendations for monitoring and management are limited. Given the wide aerodynamic diameter of airborne viruses, the issue of standardization of filters, airflow and time is the most important issue. 11 To date, studies on coronavirus have rarely included quantitative analyses for risk determinations and most sampling methods are inadequate for detecting the exact concentrations of infectious airborne coronaviruses. In addition, there were problems of technologies, for example, RT-PCR has limitations because it may quantitate virus that is no longer infectious. To prevent transmission through aerosols, the infectivity of coronavirus is important to determine and an appropriate method for detecting infectious airborne coronavirus has to be suggested. Whatever methods are proposed, nanoparticles should be included together with larger aerosol particles. 11 Standardization of the suggested method is important to monitoring and assessing the risk of airborne coronaviruses, including SARS-CoV-2. In addition, when a new coronavirus pandemic emerges in the future, an airborne coronavirus monitoring system should be applied immediately and risk assessment for the transmission through aerosols should be performed. Future studies should be conducted to standardize the detection method and the potential risk of infectious airborne coronavirus.
Footnotes
Authors' contribution
Yun H and Yang J contributed equally in the preparation of this manuscript. This manuscript was written and analysed by Yun H and Yang J. Seo JH investigated data and Sohn JR supervised the project.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Korea University and the BK21 Plus program.
