Abstract
Background
Preclinical risk for Alzheimer's disease (AD), including amyloid-β (Aβ) deposition, begins 10–15 years prior to diagnosis. In addition to genetics, hypertension, type 2 diabetes, and depression in midlife are major risk factors for AD.
Objective
Here, we assessed sex differences in associations of AD risk status with memory circuitry function and AD pathology in midlife.
Methods
High- (HR) and low-risk (LR) participants (N = 99; ages 52–71 years) were recruited from the Mass General Brigham Biobank. HR participants have genetic risk (APOE4) plus hypertension, type 2 diabetes, and/or depression; LR participants have no genetic or clinical risk. Participants underwent neuropsychological assessments of verbal, associative, and working memory, functional MRI (fMRI) scans while completing a working memory and verbal encoding task, and PET imaging scans. Aβ deposition was detected using PET C-11PiB and calculated as distribution volume ratio.
Results
HR status was significantly associated with lower scores in associative memory, altered fMRI BOLD activity in memory circuitry regions, and higher Aβ deposition, primarily in women. Further, altered task-based fMRI activity was related to worse memory performance and higher Aβ accumulation in women. While some effects were observed in men, effect sizes were smaller and did not survive correction for multiple comparisons.
Conclusions
Results demonstrated that genetic and clinical risk factors can help identify in a sex-dependent manner those in midlife who are at increased risk of developing AD to target for early intervention.
Introduction
Addressing the increasing rate of Alzheimer's disease (AD) in the population, the majority of whom will be women, is a critical public health challenge. 1 While it is known that AD risk can vary considerably by age and preclinical disease state, 2 many proposed preclinical AD diagnostic algorithms do not take into consideration sex differences.3,4 Preclinical disease state, involving major chronic diseases such as major depressive disorder (MDD) and cardiovascular disease (CVD) are critical risk factors for AD, for which women are at increased risk of multimorbidity. 5 The prevalence of MDD, and its co-occurrence with CVD, is twice as high in women compared to men. 5 Consequently, although greater life expectancy rates partially explain the higher prevalence of AD in women, there are other contributing sex-specific and sex-dependent genetic and physiologic factors. 6 Thus, effectively addressing healthy brain aging and therapeutic strategies for AD, and the sex differences therein, are important public health priorities that can significantly reduce lifetime risk of AD. 2
Most studies on memory aging and AD enroll study participants over age 65. However, asymptomatic accumulation of AD pathology has been found in much younger individuals, as early as 15–20 years prior to the onset of clinical symptoms, particularly in those at increased risk for AD. 7 Early symptoms of dementia risk include amyloid pathology and related functional alteration in memory circuitry.8–11 Increased task-evoked functional activity has been observed in a wide-spread network of regions in relation to AD risk, potentially reflecting compensatory strategies or neural inefficiency. 12 These regions were primarily part of the default mode network and sensorimotor network (i.e., frontal and parietal regions). In contrast, the hippocampus and other medial temporal lobe regions generally show decreased task-related activity with increasing AD risk. However, several studies have shown the opposite effect (i.e., increased hippocampal activity) depending on task type and memory stages assessed. 12 Both the presence of at least one APOE ε4 allele (APOE4; the genetic variant of the APOE gene most highly associated with late-onset AD) and the clinical diagnoses of MDD,13,14 type 2 diabetes (T2D), and hypertension (HYP) in midlife can increase the risk of AD.15,16 Further, having diabetes and carrying one or more APOE4 allele is associated with lower cognitive performance, 17 suggesting that the comorbidity of these factors exacerbates AD pathology. 18 Despite higher prevalence of CVD in men, CVD has a stronger impact on cognitive decline in women in midlife, 19 yet most studies simply adjust for sex and do not examine sex differences in CVD risk factors for AD. 20 HYP and APOE4 genotype also interact to increase amyloid-β (Aβ) deposition. 21 Longitudinal data extracted from the Alzheimer's Disease Neuroimaging Initiative database suggest that the APOE4 allele exacerbates the impact of HYP on neuronal damage and synaptic loss. 22 Furthermore, APOE4 carriers are at increased risk for white matter lesions if they suffer from HYP as well. 23 As such, midlife chronic health conditions may have a critical impact on late-life cognitive health, especially when they co-occur with APOE4.
The consideration of sex differences is critical when examining AD risk, given that AD disproportionately affects women and the disease manifests differently between men and women. Women with AD have more severe tau pathology 24 and the association between pathology and clinical symptoms is stronger in women compared to men. 25 In previous studies among early midlife males and females, we demonstrated significant sex differences in memory circuitry structure and function. These regions include the hippocampus (HIPP), dorsolateral prefrontal cortex (DLPFC), ventrolateral prefrontal cortex (VLPFC), anterior cingulate cortex (ACC), and inferior parietal cortex (iPAR),26–29 consistent with previous work.30–32 There is also evidence for sex differences in susceptibility to the effects of APOE4 and to CVD risk factors as well as sex–genotype interactions in response to cholinesterase inhibitors. 33 APOE4 risk has been shown to have sex-dependent effects, whereby the ε3/ε4 genotype is associated with a significantly higher risk for AD in women compared to men. 6 In contrast, the ε3/ε4 genotype is associated with increased risk in both men and women. Ovariectomization in rodents also leads to increased Aβ deposition, an effect that is reversed with estradiol treatment.34–36 In clinical studies, reduced grey and white matter volumes in AD-vulnerable regions 37 and Aβ accumulation are observed across the menopausal transition. 37 These studies demonstrate significant sex differences in AD and suggest that estradiol decline may increase vulnerability to AD pathology in women, making menopausal women particularly vulnerable. The Society for Women's Health Research Interdisciplinary Network on AD comprised an expert panel of scientists and clinicians (including JMG) who reviewed ongoing and published research related to sex and gender differences in AD. They note that studies rarely explicitly examine sex differences and exclusion of sex and gender has impeded faster advancement in the detection, treatment, and care of AD across the clinical spectrum. 20
Here, we investigate the shared impact of genetic and clinical risk factors on AD pathology, and the sex differences therein. We hypothesize that in the absence of overt clinical symptoms, high-risk individuals, characterized as those with genetic and clinical risk for AD, will demonstrate poorer memory performance, increased Aβ deposition, and altered functional activity in the memory circuitry, compared with low-risk individuals in midlife. Further, we expect a greater impact in women compared to age-matched men.
Methods
Participants
Participants were recruited from the Mass General Brigham (MGB) Biobank. The MGB Biobank is a large research data and sample repository sponsored and funded by Mass General Brigham with >150,000 individuals. It provides access to high-quality, consented samples and data to help foster research. It also provides banked samples (plasma, serum, and DNA), genomic data, and other health information, including data from the Electronic Medical Record (EMR), to investigators who are affiliated with an MGB institution. Participants have been broadly consented for linkage of biosamples to health information in their electronic health records, a survey on lifestyle, environment and family history, and genetic analyses. Here, we leveraged the MGB Biobank to identify individuals at high and low risk for AD based on genetic and clinical information queried from EMRs. Clinical risk factors included HYP, T2D, and depression. Clinical risk factor diagnoses were determined using curated variables created by the MGB Biobank, using a validated phenotypic algorithm based on the frequency of coded diagnoses and medications in the patient's EMR. Clinical chart review was used to establish a gold standard training set. Clinician-reviewed classifications were used to train models on the basis of a logistic regression classifier using the adaptive least absolute shrinkage and selection operator (LASSO) procedure (Biobank Portal Wiki). The biobank positive predictive value was set at 0.90 and negative predictive value at 0.99 for all three clinical risk factors. Depression diagnosis included major depressive disorder (MDD) and Depressive disorder not otherwise specified (NOS). Genetic risk was based on the presence of either the APOE4 allele (based on either whole exome sequencing or HRC-imputed genotype data, released in 2020) or an APOE4 proxy: exm-rs4420638 (based on genotype data available in 2019). The proxy SNP is in strong linkage disequilibrium with rs429358 (one of the two SNPs defining ε4 allele) and is coinherited with the ε4 allele. 38 Five participants met genetic HR status with only the proxy. We initially recruited 50 high- (HR) and 50 low-risk (LR) participants, with a target age range between 50–70 years old (final age range 52–71 years old), equally divided by sex. HR participants have one or two APOE4 alleles (or proxy) plus one or more clinical risk factors (HYP, T2D, or depression). LR participants do not have the APOE4 allele (or proxy) and have no clinical risk factors. A composite score reflecting cumulative risk of AD, calculated as the sum of genetic and clinical risk factors, was also generated. Exclusionary criteria included any history of neurological disease including AD, central nervous system (CNS) damage, hormone replacement therapy (HRT), endocrine disorders, current moderate to severe alcohol or substance use disorder, current or history of psychosis, heart failure, chronic kidney disease, other medical illnesses that may significantly alter CNS function, severe autoimmune disorders, and any MRI contraindication. Participants were excluded if they sought medical attention for any memory problems or reported any cognitive impairment. Participants were defined as cognitively healthy as they have never been diagnosed by a medical professional with cognitive impairment or AD. The Partners (now Mass General Brigham (MGB)) Human Research Committee Institutional Review Board granted Human Studies participants’ approval. All volunteers gave written informed consent and were paid for their participation.
Study design
Participants were seen over two virtual visits and two in-person visits at Massachusetts General Hospital. The first in-person visit consisted of an MRI scan at the Athinoula A. Martinos Center for Biomedical Imaging and the second in-person visit consisted of a blood draw at the Translational and Clinical Research Center and PET scan at the Gordon Center for Medical Imaging. Participants were asked to refrain from taking pain medication, recreational drugs, and drinking alcohol 24 h prior to their visit. They were also asked to refrain from drinking caffeine the morning of their in-person visits. Prior to a morning baseline blood draw, participants fasted for ≥8 h and after were offered a light standardized breakfast. All women attested to being postmenopausal with no menstrual period in the last 12 months. Postmenopause was validated serologically (FSH, progesterone, estradiol). Neuropsychological testing, a health survey, and structured clinical interviews administered by an experienced clinical interviewer/clinician were conducted virtually. Demographic, self and family medical history, reproductive history, and additional clinical information, including body mass index (BMI), marital status, race/ethnicity, psychiatric history, and history of alcohol or substance abuse or dependence, were collected as part of the clinical interview via self-report using the Structured Clinical Interview for DSM-V (SCID) and a health survey. Clinical information, in part, was used to determine eligibility.
Neuropsychological assessments
Participants completed a neuropsychological battery to measure executive function (attention and working memory), verbal memory, associative memory, and estimated verbal intelligence (IQ). Digit Span Forward (DSF) and Backward (DSB) 39 were used to assess attention and working memory (WM), respectively. Participants were asked to repeat sequences of numbers that became progressively longer in the same order (DSF) or reverse order (DSB). The total number of correct trials (maximum 14) was used as the outcome measure for each task. The American National Adult Reading Test (AMNART) 40 was used to assess estimated verbal intelligence (IQ).
Measures of episodic memory included the 12-item Face-Name Associative Memory Exam (FNAME)41,42 and the Buschke 6-trial Selective Reminding Test (SRT).43,44 These tests were chosen because they are particularly challenging and sensitive to memory and learning deficits associated with early aging. The FNAME is a paired-associative face-name task that is sensitive to temporolimbic integrity. Using associative learning paradigms, in contrast to list learning procedures, has the benefit of controlling attention within the learning process by pairing items together, thus requiring the participant to make associations between them. Memory impairments on associative learning paradigms are often consistent with temporolimbic amnestic dysfunction.45,46 As reported by Papp et al., 41 the FNAME asks the participant to study 12 unfamiliar face-name-occupation groupings. The test consists of two learning exposures, followed by the presentation of the face and the request to recall the name and occupation associated with that face. After a 10-min delay during which participants are shown 12 generationally-specific celebrity faces and asked to provide their name and occupation as an interference trial, participants are shown the face and asked to recall the name and occupation. A multiple-choice format was presented that asked the participants to choose the name and occupation from three choices of names and three choices of occupations. While forming face-name associations is particularly difficult, pairing the face with an occupation is an inherently easier task because of its association with previously stored semantic knowledge. 42 Performance on the FNAME was previously found to decline with age 45 and other factors that impact memory performance, such as depression. 47 Furthermore, FNAME is thought to isolate medial temporal lobe memory function capacities apart from frontal executive mechanisms involved in memory processing.
The SRT, on the other hand, is a selective reminding procedure that relies on feedback tailored to the individual, i.e., individuals are only reminded of words they failed to recall in the previous trial. This procedure taps into executive processing because it requires the individual to hold information online, within working memory, as only a portion of the list that was not immediately recalled is presented for learning. More specifically, once the individual is exposed to all the words in trial 1, they are reminded only of the words that were not immediately recalled in the previous trial. While more challenging than a traditional list learning test, the SRT was chosen because it taps into executive compromise, 48 an important component to learning and memory. Executive compromise refers to executive dysfunction and the inability to plan and organize to complete a task. Thus, our rationale for utilizing these two specific episodic memory tests was to determine whether memory changes were related to frontal executive retrieval deficits common in aging and hippocampal dysfunction (i.e., as detected on the SRT and FNAME).
Z-score composites were created for FNAME including: 1) Initial learning of names and occupations (Initial recall); 2) Cued recall of names and occupations (Cued recall) and; 3) FNAME summary score combining both initial learning and cued recall of names and occupations (Summary score). SRT performance was assessed using individual scores from list learning over 6 trials (Total Recall) and delayed recall at 10 min (DR) and 30 min (30DR) 43 as well as an SRT summary z-score combining both list learning and 30 min delayed recall (Summary score).
fMRI data acquisition
MRI data were acquired with a Siemens 3 T Skyra scanner (Siemens, Erlangen, Germany), equipped with a 20-channel head coil. Functional data were obtained using a T2*-weighted echo-planar imaging sequence sensitive to blood oxygenation level-dependent (BOLD) contrast (repetition time, 2000 ms; echo time, 30 ms; field of view, 210 mm; flip angle, 90°; voxel size, 2.5 × 2.5 × 2.2). Each functional volume consisted of 56 (2.2 mm) oblique axial slices. A T1-weighted image was collected using a high-resolution 3D Multi-Echo (ME) MPRAGE sagittal sequence with an isotropic resolution of 1 mm3. Following acquisition, MRI data were converted to Nifti format and preprocessed in SPM12 (Wellcome Department of Cognitive Neurology, London, UK). Preprocessing included realignment and geometric unwarping of echo-planar imaging images using magnetic field maps, correction for head motion, nonlinear volume-based spatial normalization (Montreal Neurological Institute template MNI-152), and spatial smoothing with a Gaussian filter (6 mm [full width at half maximum]). Additional software (http://web.mit.edu/swg/software.htm) was used to identify and exclude outliers in the global mean image time series (threshold 3.0 standard deviation (SD) from the mean) and movement (threshold 1.0 mm; measured as scan-to-scan movement, separately for translation and rotation) parameters. Statistical parametric maps of BOLD activation were calculated in SPM12 using the general linear model approach. 49
Working memory fMRI paradigm
Participants performed a verbal working memory N-back task during fMRI scanning. 27 The task consisted of two conditions, 0-back and 2-back. In each condition, participants were presented with a sequence of white upper-case letters on a black background presented centrally (200 ms duration, 1800 ms interstimulus interval) in a pseudo-random order. Participants performed two experimental runs of the task, with each run lasting 5 min 44 s. Each run contained six 32-s blocks. Each block was preceded by a 200-s fixation period and a 4-s instruction screen. During 0-back blocks, participants responded to every letter using one of two buttons to indicate whether or not the target letter (X) appeared. During 2-back blocks, participants responded to every letter using one of two buttons to indicate whether it matched or did not match the letter seen two previously. Contrasts were generated for each participant as a function of WM load (2-back > 0-back). Response times and accuracy (d’ 2back) were recorded. Response time (RT) values <100 ms were considered null and not included in the computation of participants’ average RT. The sensitivity index d’ 2back was calculated 50 as d’ 2back = z[probability(hits)] - z[probability(false alarms)]. In accordance with signal detection theory, a higher d’ represents a greater distinction between signal and noise (i.e., better signal detection).
Verbal encoding fMRI paradigm and subsequent memory retrieval task
Participants also performed a verbal encoding task during fMRI scanning.28,32,51 The task consisted of two conditions, “Novel” and “Repeat.” In each condition, participants were presented with a pair of common nouns on a black background presented centrally (4000 ms duration) with a variable interstimulus interval (600–1500 ms). Participants were asked to silently generate a sentence using both words and were instructed to remember the stimuli for a later test. In the Repeat condition, participants viewed the same noun pair repeated throughout each block of a run and were instructed to generate the same sentence each time they saw the word pair. In the Novel condition, participants viewed novel word pairs and generated a new sentence in response to each pair. Participants were instructed to respond to every word pair with a single button press (pointer finger) to indicate that they had successfully formed a sentence in their mind. Participants performed two experimental runs of the task. Each run contained three Repeat blocks and three Novel blocks, for a total of six blocks per condition. Contrasts were generated for each participant as a function of encoding load (Novel > Repeat).
A subsequent recognition memory task was administered immediately following the encoding task using the same response box and while the participant remained in the scanner. Participants viewed single nouns, including 24 previously presented nouns and 24 foils. Each stimulus was presented for 4000 ms with a variable interstimulus interval. Participants were instructed to indicate, using one of two buttons, whether they had seen the item on the screen in the previous task (yes) or not (no). Response times (RT) and accuracy (d’ VR) were recorded. RT values <100 ms were considered null and not included in the computation of participants’ average RT. Same as above, the sensitivity index d’ VR was calculated 50 as d’ VR = z[probability(hits)] - z[probability(false alarms)].
fMRI data analyses
Hemodynamic responses were modeled using a gamma function and convolved with onset times of 2-back and 0-back blocks to form the general linear model (GLM) at the single participant level. Outlier time points and the 6 rigid-body movement parameters were included in the GLM as covariates of no interest. We used a small volume correction (SVC) approach in SPM12, which limits voxel-wise analyses to voxels within a priori hypothesized ROIs. Target ROIs were defined as anatomical masks and implemented as overlays on the SPM12 canonical brain. Working memory Nback task ROIs included bilateral anterior cingulate cortex (ACC: amCGa/BA24/32, pmCGa/BA24, pregenual-CGa/BA24/32, subgenual-CGa/BA24/32), inferior parietal cortex (iPAR: AG/BA39, PO/BA40/S-II, SGa/BA40, SGp/BA40), dorsolateral prefrontal cortex (DLPFC: F2/BA6, F2/BA8, F2/BA9, F2/BA46), and hippocampus (HIPP). For the verbal encoding task, ROIs included bilateral ACC, iPAR, ventrolateral prefrontal cortex (VLPFC: F3o/BA44, F3orb/BA47, F3t/BA45, FO/BA44-45, FOC/BA47-12-VL), and HIPP. ROIs were anatomically defined using a manually segmented MNI-152 brain (based on methods previously published by the Center for Morphometric Analysis at Massachusetts General Hospital and Harvard Medical School; see Makris et al.52,53). False positives were controlled using conservative FWE-correction to control for multiple comparisons. Within an anatomical ROI, results identified using SVC were reported as significant if they met the peak-level threshold of p < 0.05, FWE corrected, with a cluster threshold >10. ANOVA was applied to examine interactions between sex and risk status on functional activity in ROIs with age, education, and race/ethnicity as covariates. Peak values for each participant from significant ROIs were obtained using the VOI extraction tool in SPM12. Box dimensions were set to the voxel size of 2.5 × 2.5 × 2.2.
PET imaging
We analyzed 11C-Pittsburgh Compound-B (PiB) HATCH PET data (N = 87) using the methods we previously published. 54 Whole-brain Aβ deposition was measured as the mean standardized uptake value ratio (SUVR) from 40 to 60 min after radiotracer injection, with cerebellar gray matter as the reference region. Group-specific whole-brain SUVR maps were generated by averaging all individual subject maps within each group. Aβ deposition was measured as the distribution volume ratio (DVR) within the frontal, lateral temporoparietal, and retrosplenial regions of interest.
Statistical analysis
Descriptive statistics of study sample characteristics— age, BMI, education (above versus below a 4-year college degree), race/ethnicity (% White), HYP status, T2D status, and depression status—were reported by sex (men versus women) and risk status (HR and LR). Comparisons by group status were performed using nonparametric Wilcoxon rank-sum or chi-square tests, for continuous and categorical data, respectively.
The associations of risk status with memory performance measures (FNAME, SRT), fMRI BOLD activity peak values, and Aβ levels were assessed using general linear models adjusted for age, education, and race/ethnicity. Models were run for the overall sample, as well as stratified by sex. Sex differences in the association between risk status and outcomes measures were assessed by including an interaction term between sex and risk status in the model. Similarly, the composite score reflecting cumulative risk of AD was also examined in relation to outcome measures. We considered age, education, income, marital status, race/ethnicity, BMI, and smoking status (history of or current, yes versus no) as potential confounding variables. Covariates associated with both the outcome and exposure with a p < 0.2 were included in the models. Although income met our criteria for confounding, it was highly related to education, marital status, race/ethnicity, and age, and therefore was removed to avoid over-correcting the models. Further, models that include risk status as an exposure variable were conducted with and without covariates to assess the level of variance removed by including these potential confounders (Supplemental Tables). Normality was formally assessed using the Shapiro-Wilk test. All outcome variables were normally distributed except Aβ levels, as expected. Given the skewness of Aβ, Aβ was examined both as a continuous and categorical measure, the latter defined as Aβ+ versus Aβ- using a clinical DVR cutoff of ≥1.20. 54 Sex differences in the association of risk status with memory performance and fMRI BOLD peak values were assessed with the inclusion of an interaction term between sex and risk status. We used general linear models in SAS to also examine the relationship between peak values and behavioral outcomes and Aβ pathology. Tests of our hypotheses were two-sided with an α = 0.05. To address multiple comparisons, we used false discovery rate (FDR)-corrected p-values (pFDR) to account for the number of tests within each assessment. All analyses were performed using SAS 9.4 software (SAS institute, Cary, NC).
Results
Demographics and clinical characteristics
The sample included 100 participants recruited from the MGB Biobank (25 HR women; 25 HR men; 25 LR women; 25 LR men). Cognitive data from one LR male was excluded from analyses due to unreliable data, resulting in a final sample of N = 99. Working memory fMRI data were available for 90 participants, verbal encoding fMRI data were available for 92 participants, and PET imaging data were available for 85 participants. All participants were ages 52–71 (mean age = 62.35 ± 5.42 years), with an average verbal IQ of 121.62 ± 6.81, as assessed with the AMNART, and 83.4% had a college degree or higher. All women were postmenopausal with permanent amenorrhea, low estradiol levels, and signs of follicular failure (elevated FSH and oligomenorrhea).
Table 1 reports demographic and clinical characteristics of the sample and group comparisons. HR participants were slightly older than LR (63.34 versus 61.10 years old). LR men were the youngest group and differed significantly in age from HR men and HR women. HR individuals also had significantly higher BMI compared to LR. LR women had the lowest BMI and differed significantly from HR men and HR women. There was a higher percentage of individuals that identified as White in the LR group compared to the HR group, particularly in the LR men. Education also differed between groups, with a higher percentage of college graduates in the LR group compared to the HR group. The HR women were the least educated and differed significantly on education from LR women and LR men. There was no significant difference in verbal IQ between groups.
Demographic and clinical characteristics of a sample of N = 99 men and women assessed at ages 52–71.
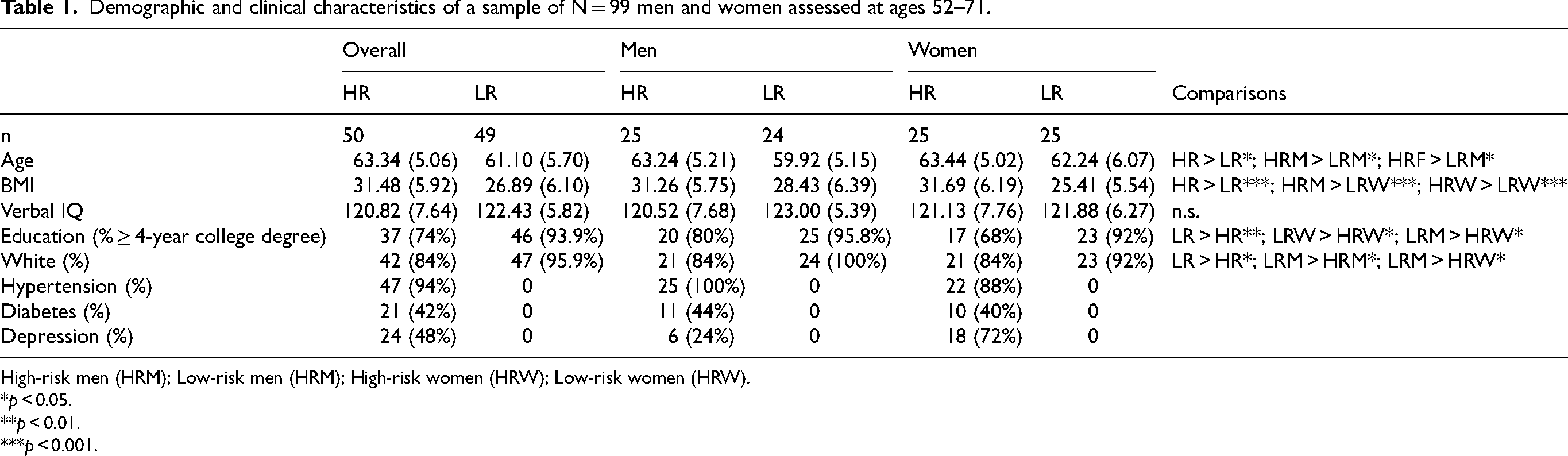
High-risk men (HRM); Low-risk men (HRM); High-risk women (HRW); Low-risk women (HRW).
p < 0.05.
p < 0.01.
p < 0.001.
Overall sex differences in memory outcomes and AD pathology
Women performed better than men on the SRT verbal memory task (Summary score: β=0.43, pFDR = 0.05) and tended to perform better on the FNAME associative memory task as well (Summary score: β=0.35, pFDR = 0.09). There was no significant sex difference in task performance on the working memory Nback, verbal encoding, and DSF and DSB tasks (all p's > 0.1). Aβ deposition also did not differ between men and women (β=0.02, pFDR = 0.73).
During the working memory Nback task, men had significantly higher activity in the left DLPFC (F(1,89) = 3.82, pFWE = 0.047; cluster of 104 voxels with peak at x = −39, y = 5, z = 34) and tended to have higher activity in the left ACC (F(1,89) = 3.65, pFWE = 0.086; cluster of 61 voxels with peak at x = −9, y = 41, z = −11) and right DLPFC (F(1,89) = 3.55, pFWE = 0.098; cluster of 197 voxels with peak at x = 42, y = 17, z = 34) compared to women. There were no regions with greater fMRI BOLD activity in women compared to men on the working memory Nback task.
During the verbal encoding task, women had significantly higher task-related BOLD activity in the right and left ACC (Right: F(1,91) = 5.8, pFWE < 0.001; cluster of 198 voxels with peak at x = 6, y = 32, z = 34; Left: F(1,91) = 3.96, pFWE = 0.036; cluster of 64 voxels with peak at x = 0, y = 41, z = 31), iPAR (Right: F(1,91) = 6.09, pFWE < 0.001; cluster of 554 voxels with peak at x = 45, y = −52, z = 46; Left: F(1,91) = 5.36, pFWE < 0.001; cluster of 351 voxels with peak at x = −39, y = −43, z = 43) and right VLPFC (F(1,91) = 3.88, pFWE = 0.035; cluster of 68 voxels with peak at x = 48, y = 8, z = 31). A separate region in the left ACC (F(1,91) = 3.93, pFWE = 0.04; cluster of 62 voxels with peak at x = −6, y = 41, z = −11) showed increased activity in men compared to women.
Impact of risk status on memory performance, memory circuitry, and AD pathology
Memory performance. Overall, HR individuals performed significantly worse on the FNAME associative memory task compared to LR (Initial recall: β=−0.67, pFDR = 0.002; Cued recall: β=−0.60, pFDR = 0.003; Summary score: β=−0.63, pFDR = 0.002; Figure 1(a) and Table 2). They also tended to perform worse on the verbal encoding task (β=−0.10, pFDR = 0.06; Table 2). The composite score was also negatively correlated with associative memory, such that a greater number of risk factors was associated with worse FNAME performance (Initial recall: β=−0.24, pFDR = 0.001; Cued recall: β=−0.24, pFDR = 0.001; Summary score: β=−0.24, pFDR = 0.001; Figure 1(b) and Table 3), and with verbal encoding (β=−0.04, pFDR = 0.02; Table 3).
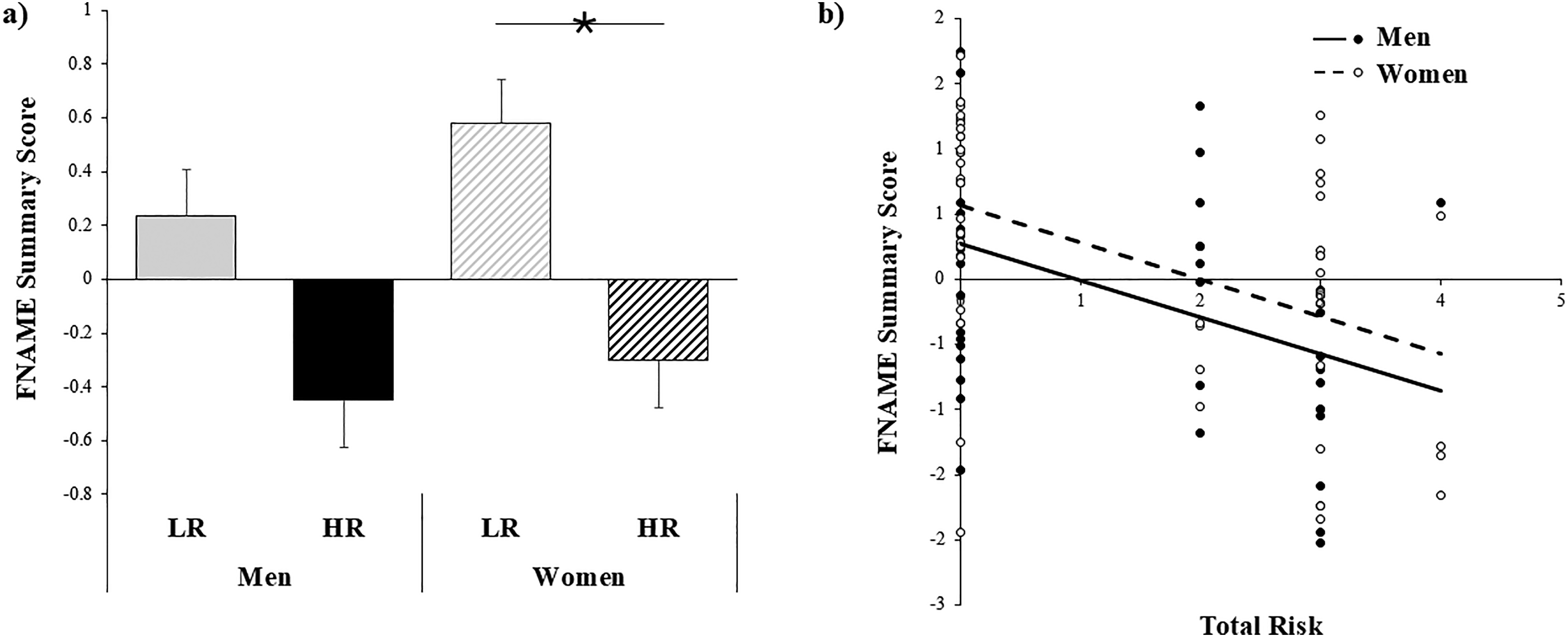
Relationship between AD risk and associative memory performance. (a) HR women had significantly lower summary scores on the FNAME task compared to LR women (β=−0.75, pFDR = 0.009). There was only a trend level difference between HR and LR men (β=−0.47, pFDR = 0.12). (b) Overall, greater number of risk factors (i.e., total risk) was related to worse associative memory performance on the FNAME task.
Associations of risk status with adult memory performance and amyloid pathology by sex.
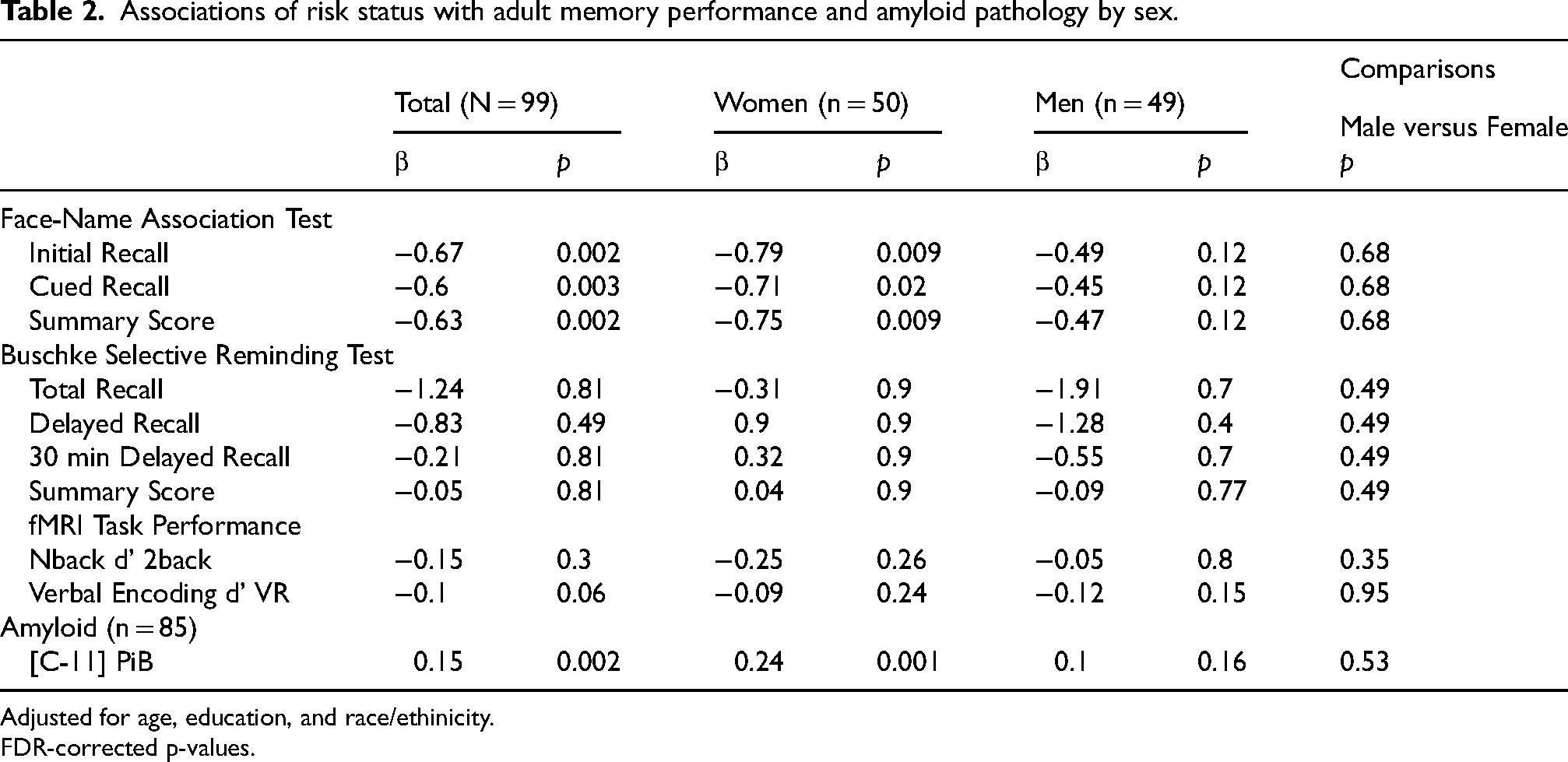
Adjusted for age, education, and race/ethinicity.
FDR-corrected p-values.
Associations of composite score of cumulative risk with adult memory performance and amyloid pathology by sex.
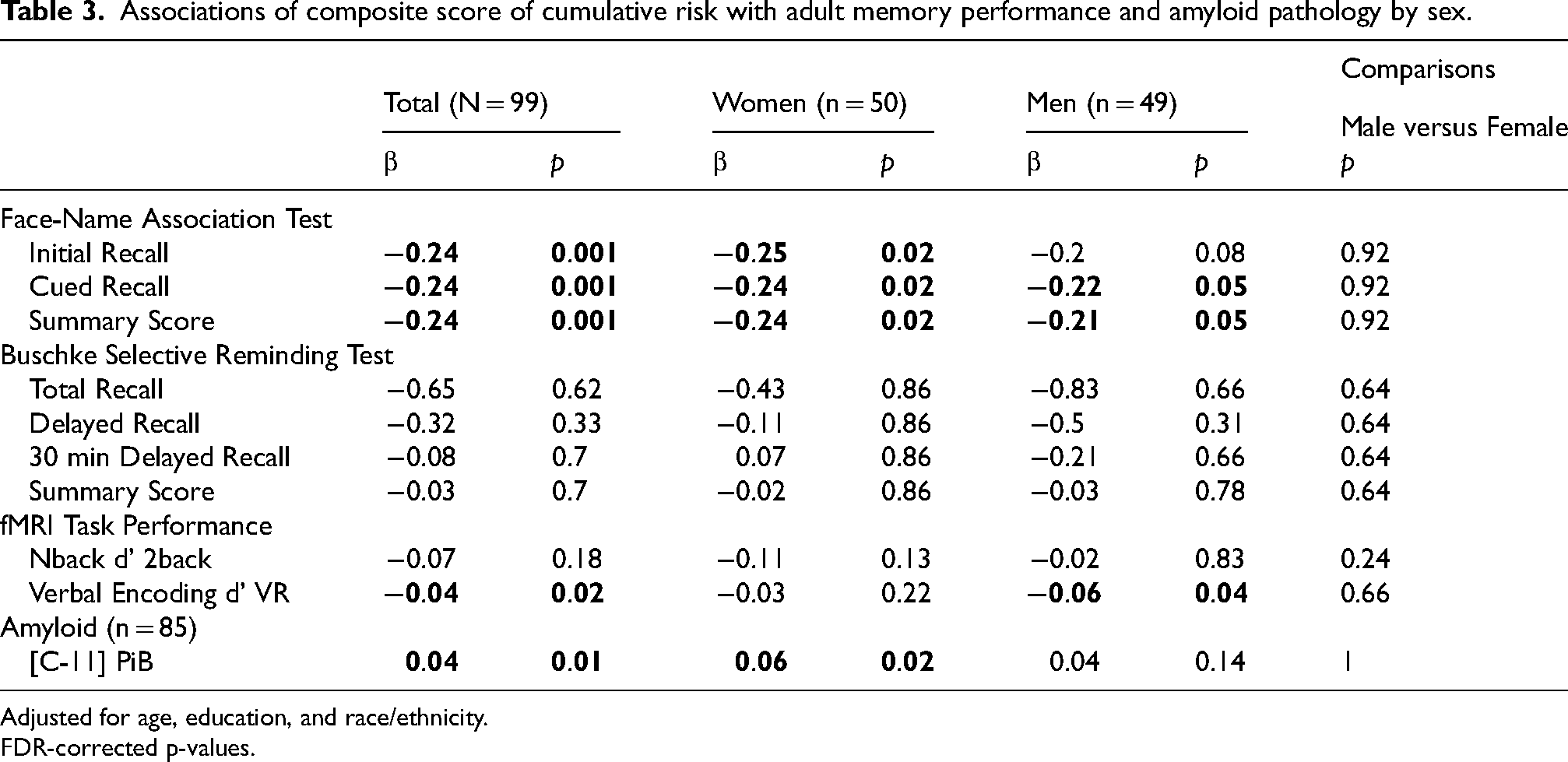
Adjusted for age, education, and race/ethnicity.
FDR-corrected p-values.
Stratified by sex, HR women performed significantly worse on all measures of associative memory compared to LR women (Initial recall: β=−0.79, pFDR = 0.009; Cued recall: β=−0.71, pFDR = 0.02; Summary score: β=−0.75, pFDR = 0.009; Table 2). HR men also exhibited impairments in associative memory compared to LR men; however, effects were only trend levels after controlling for age, education, and race/ethnicity and correcting for multiple comparisons (Initial recall: β=−0.49, pFDR = 0.12; Cued recall: β=−0.45, pFDR = 0.12; Summary score: β=−0.47, pFDR = 0.12). Unadjusted results are reported in Supplemental Tables 1 and 2. A higher composite score was significantly associated with worse associative memory performance in both men (Initial recall: β=−0.20, pFDR = 0.08; Cued recall: β=−0.22, pFDR = 0.05; Summary score: β=−0.21, pFDR = 0.05) and women (Initial recall: β=−0.25, pFDR = 0.02; Cued recall: β=−0.24, pFDR = 0.02; Summary score: β=−0.24, pFDR = 0.02; Table 3). The composite score was also inversely associated with verbal encoding in men (β=−0.06, pFDR = 0.04; Table 3), while there was no association in women. There were no significant sex by risk status interactions.
Aβ accumulation. Overall, HR individuals had significantly higher levels of Aβ deposition compared to LR individuals (β=0.15, p = 0.002; Figure 2(a)). When participants were further dichotomized into Aβ+ and Aβ- groups using a clinical DVR cutoff of 1.20, 32.6% of HR participants were classified as Aβ+, while only one LR participant (2.4%) was classified as Aβ+ (χ 2 (1) = 13.31, p < 0.001; Figure 2(b)). The composite score of cumulative risk of AD was also associated with greater Aβ deposition (β=0.04, p = 0.01; Table 3). Higher Aβ levels in turn were related to lower associative memory performance (FNAME Summary score: β=−1.27, pFDR =0.02; Initial recall: β=−1.34, pFDR =0.02; Cued recall: β=−1.20, pFDR =0.02).
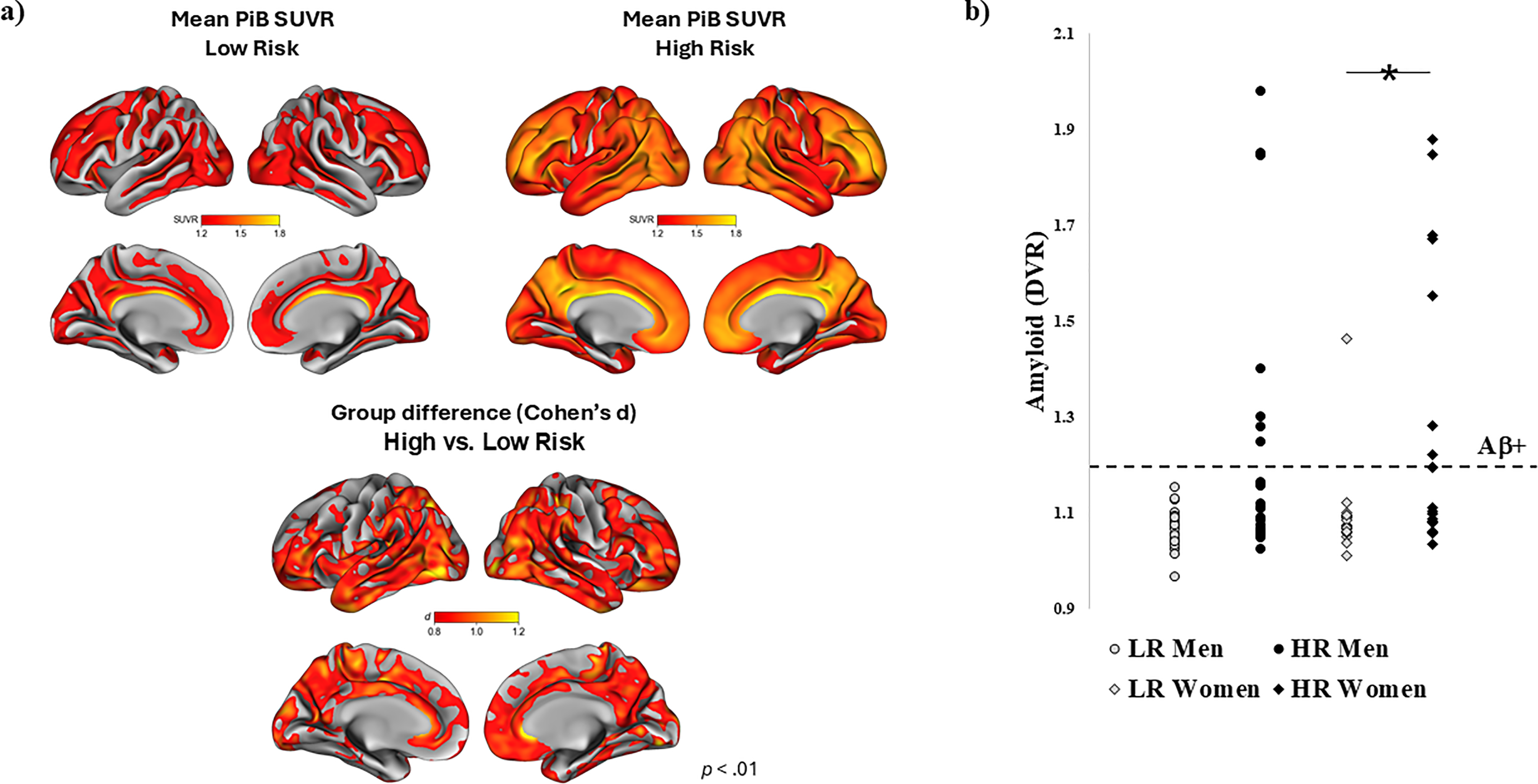
C-11 PiB PET by risk status. (a) HR status was related to significantly higher levels of Aβ deposition (β=0.15, p = 0.002). Aβ deposition was measured as the distribution volume ratio (DVR) within the frontal, lateral temporoparietal, and retrosplenial regions of interest. (b) Stratified by sex, Aβ deposition differed significantly between HR and LR women (β=0.24, p = 0.001). The relationship between risk status and Aβ was also present in men (β=0.16, p = 0.009) but did not remain significant after controlling for age, education, and race/ethnicity (β=0.10, p = 0.16).
By sex, HR women had significantly higher levels of Aβ deposition compared to LR women (β=0.24, p = 0.001). The relationship between risk status and Aβ was also present in men (β=0.16, p = 0.009) but did not remain significant after controlling for age, education, and race/ethnicity (β=0.10, p = 0.16). In men, higher Aβ levels were related to worse associative memory performance (FNAME Summary score: β=−1.63, pFDR =0.03; Initial recall: β=−1.70, pFDR =0.03; Cued recall: β=−1.57, pFDR =0.03). In women, this relationship was not present in the whole group and was only found in those with the lowest memory performance scores (bottom tertile: β=−4.88, pFDR =0.04). There were no significant sex by risk status interactions in relation to amyloid pathology.
fMRI memory circuitry activity: Working memory task. There were no significant differences in fMRI BOLD activity overall between HR and LR participants on the working memory Nback task (Unadjusted results reported in Supplemental Table 3). Overall, higher activity in the right HIPP during the working memory Nback task was related to worse task performance (β=−0.25, pFDR = 0.03; Figure 3(b)), poor performance on the DSB (β=−0.06, p = 0.03), and greater Aβ pathology at trend levels (β=0.36, p = 0.05; Figure 3(c)).
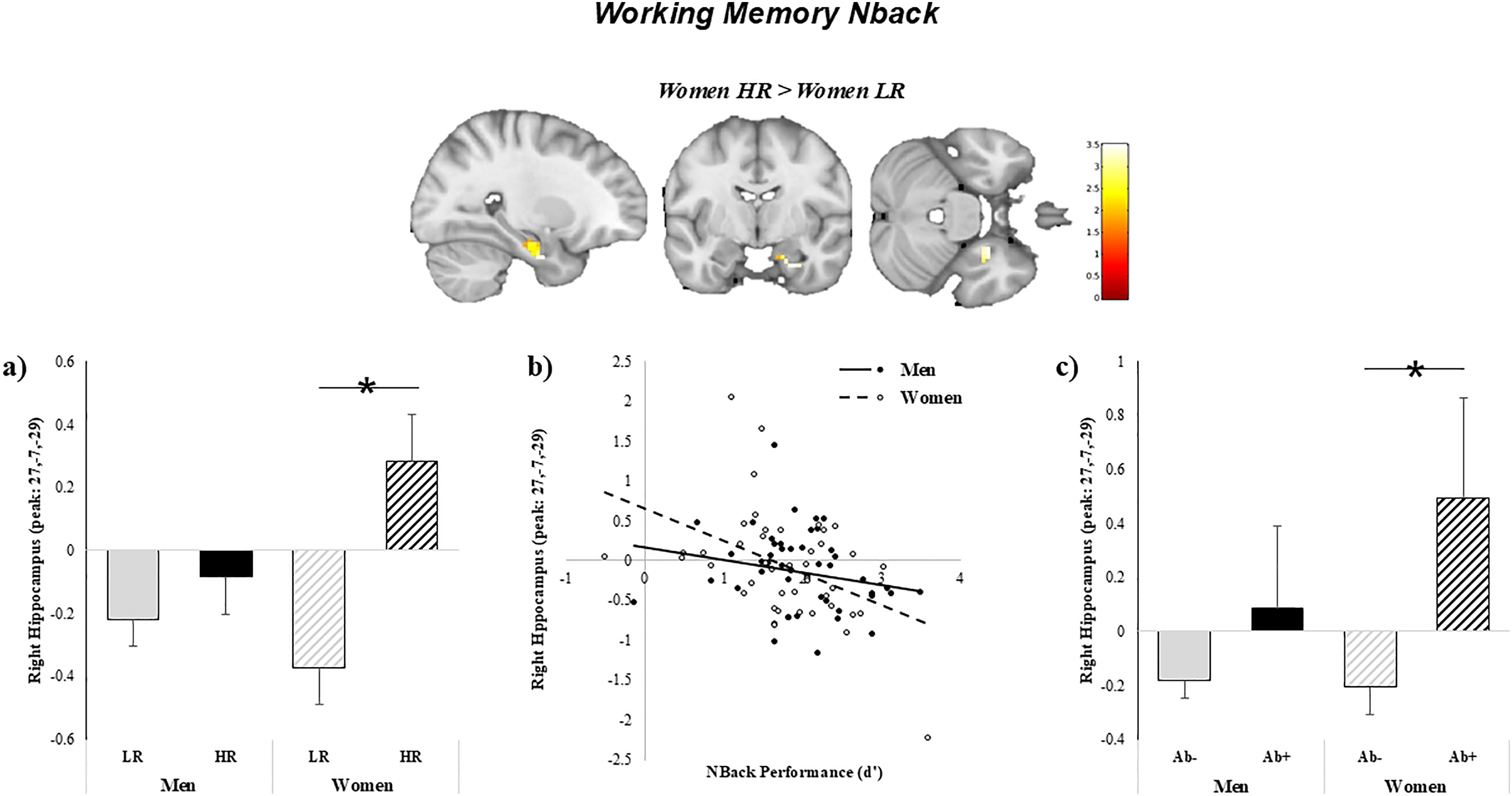
Relationship between task-based functional BOLD activity, working memory, and Aβ deposition by sex. (a) HR women had significantly higher fMRI BOLD activity in the right hippocampus during the Nback working memory task compared to LR women. Activity in the right hippocampus was related to (b) worse task performance and c) greater Aβ deposition in women.
Stratified by sex, HR women had significantly higher task-related BOLD activity in the right HIPP compared to LR women (F(1,42) = 3.51, pFWE = 0.034; cluster of 55 voxels with peak at x = 27, y = −7, z = 29; Figure 3(a)). In turn, higher activity in the right HIPP in women was related to worse task performance (β=−0.38, p = 0.01; Figure 3(b)) and greater Aβ pathology (β=0.64, p = 0.04; Figure 3(c)). In men, LR status was associated with significantly higher fMRI BOLD activity in the right iPAR (F(1,46) = 4.1, pFWE = 0.031; cluster of 319 voxels with peak at x = 60, y = −43, z = 31) and tended to be associated with higher DLPFC activity (F(1,46) = 3.75, pFWE = 0.058; cluster of 211 voxels with peak at x = 36, y = 11, z = 61) compared to HR. No significant relationships were found for task-based fMRI activity with cognitive performance and Aβ pathology (all p's > 0.1) in men. There were no significant sex by risk status interactions on the Nback task.
fMRI memory circuitry activity: Verbal encoding task. Overall, LR participants had significantly higher fMRI BOLD activity in the left VLPFC compared to HR participants (Unadjusted results reported in Supplemental Table 4). HR participants did not have higher activity in any ROIs compared to LR participants. Functional activity in the left VLPFC during the verbal encoding task was related to better associative (FNAME: Initial recall: β=0.39, pFDR = 0.0002; Cued recall: β=0.28, pFDR = 0.005; Summary score: β=0.39, pFDR = 0.0002) and verbal (SRT trend-level effects: Total recall: β=0.03, pFDR = 0.05; Delayed recall: β=0.09, pFDR = 0.05) memory performance as well as lower amyloid levels (β=−0.57, p = 0.02).
Stratified by sex, HR women had significantly higher verbal encoding-related BOLD activity in the left HIPP compared to LR women (F(1,42) = 3.65, pFWE = 0.023; cluster of 17 voxels with peak at x = −12, y = −10, z = 23; Figure 4(a)). In turn, higher activity in the left HIPP during verbal encoding in women was related to worse task performance (β=−0.71, p < 0.05; Figure 4(b)) and greater Aβ pathology (β=0.67, p = 0.02; Figure 4(c)). However, the relationship with task performance did not remain significant after controlling for covariates (β=−0.56, p = 0.18). In men, there was a trend level effect whereby LR status was associated with higher fMRI BOLD activity in the left iPAR (F(1,46) = 3.7, pFWE = 0.094; cluster of 319 voxels with peak at x = −45, y = −61, z = 34) compared to HR. There were no significant sex by risk status interactions on the verbal encoding task.
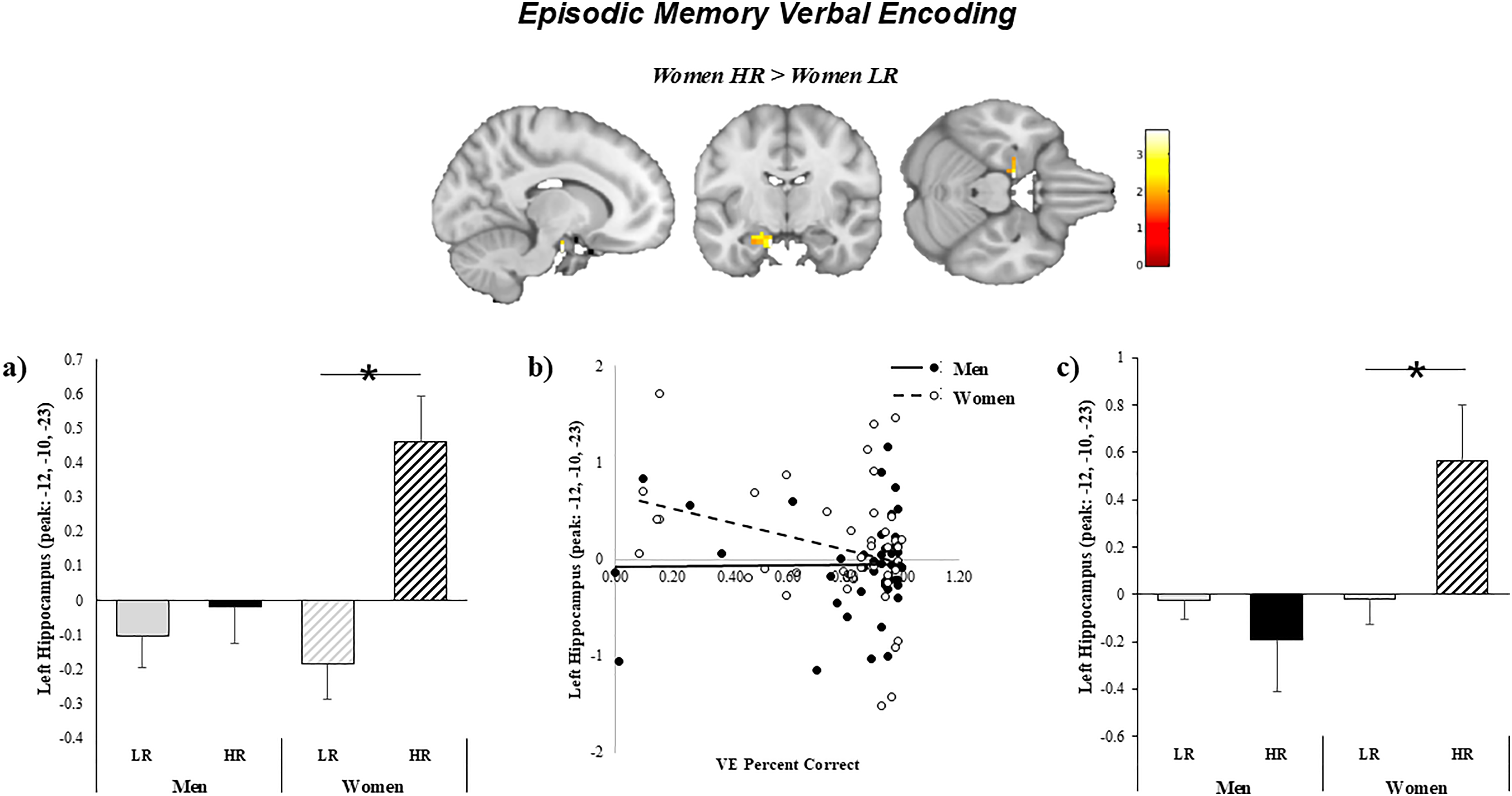
Relationship between verbal encoding task-based functional BOLD activity, verbal encoding accuracy, and Aβ deposition by sex. a) HR women had significantly higher fMRI BOLD activity in the left hippocampus during the verbal encoding task compared to LR women. Activity in the left hippocampus was related to b) worse task performance and c) greater Aβ deposition in women.
Discussion
This study investigated the shared impact of genetic and clinical risk factors in midlife on memory circuitry function and Aβ accumulation, and the sex differences therein. Overall, genetic and clinical risk factors in midlife were associated with poorer memory performance, altered task-based functional activity in the brain, and higher AD pathology in otherwise cognitively healthy midlife adults. Significant differences by risk status were primarily observed among women. HR women had worse memory, greater Aβ accumulation, and altered fMRI BOLD activity in memory circuitry regions compared to LR women. Altered task-based activity in women was related to overall worse memory performance and higher Aβ accumulation. While some effects were observed in men, effect sizes were smaller and did not survive adjustment for covariates and correction for multiple comparisons. Total cumulative risk, an alternative method of examining the additive impact of clinical risk factors in addition to genetic risk, was associated with worse memory performance in both men and women, and in women, with Aβ pathology. We did not find significant sex differences between risk status and AD pathology. However, effect sizes were larger in women and may be significant with larger sample sizes. Taken together, these results suggest cardiometabolic diseases and depression in the face of genetic risk contribute significantly to sex differences in Aβ pathology in an otherwise cognitively healthy population.
Preclinical risk for AD, including Aβ deposition, begins 10–15 years prior to clinical manifestation. 7 Recognizing the critical nature of the preclinical period of AD, in 2011 the National Institute on Aging and Alzheimer's Association (NIA-AA) updated the guidelines for AD, creating separate diagnostic recommendations for the preclinical, mild cognitive impairment (MCI), and dementia stages of AD. The guidelines were unified under a research framework defining AD by its underlying pathologic processing, focusing on the biomarkers within living people (Aβ deposition, pathologic tau, and neurodegeneration). 55 Bateman et al. 56 followed participants who were at risk for carrying a mutation for autosomal dominant AD over several years using comprehensive clinical, cognitive, imaging, and biochemical assessments. They found that concentrations of Aβ in cerebrospinal fluid may decline as early as 25 years before expected symptom onset, and Aβ deposition measured by PET was detected 15 years before expected symptom onset. 57 During the preclinical stages of the disease, risk of progression increases and is accompanied by subtle changes in cognitive function associated with AD biomarkers, likely representing the earliest symptomatic phase of AD prior to a diagnosis of MCI. 58 Therefore, the preclinical phase of AD is the critical time to target in intervention work. In the current study, we were able to successfully identify in midlife (ages of 52–71) those who may be at high-risk for AD before the onset of overt clinical symptoms, even though they were considered cognitively healthy. Results are consistent with studies underscoring the impact of chronic medical illnesses (e.g., HYP, T2D and depression) on memory circuitry aging.
In this study, one-third of HR participants overall were Aβ positive and the impact of HR status differed by sex. We found that HR women had significantly more Aβ pathology, worse memory performance, and altered memory circuitry function compared to LR women, while only minimal differences were found between HR and LR men. Previous studies have shown significant sex differences in the frequency and clinical manifestation of AD. Within APOE4 carriers, women have an increased risk of developing MCI between the ages of 55–70 and AD between 65–75 years compared to men, even though overall men have a higher rate of MCI than women. 59 55–70 is a significant age range in women as it coincides with the onset of menopause. Having one APOE4 allele is associated with significantly higher risk for AD in women compared to men. 6 Given the majority of our sample have the ε3/ε4 genotype, the differential impact of one APOE4 allele in men and women may explain why effects were primarily found in women. Women APOE4 carriers also have more tau pathology, 60 reduced connectivity in brain regions associated with healthy memory function, 61 increased brain hypometabolism and brain atrophy, 62 and accelerated cell aging in the form of shorter leukocyte telomere length 63 compared to men APOE4 carriers. In the face of Aβ pathology and MCI diagnosis, women have a faster rate of neurodegeneration64,65 and cognitive decline64,66,67 compared to men. Data from a meta-analysis reviewing studies examining sex differences in cognitive functioning in AD convincingly and consistently showed greater decline in women with AD than men across a broad range of cognitive domains including visuospatial, verbal, episodic memory, and semantic memory. Some of these domains typically reveal a sex-related processing advantage for healthy women. 68 Similarly, postmortem studies have found that women with AD had more severe tau pathology 24 and a stronger association between AD pathology and clinical symptoms compared to men. 25 Our results suggest that genetic and clinical risk in midlife may have a greater in impact in women compared to men. However, this will need to be confirmed with replication in a larger sample size. Taken together, these investigations emphasize the importance of taking into consideration sex differences when examining AD risk in midlife.
There is also extensive evidence demonstrating the role of estradiol in memory circuitry function and AD pathology. In rodents, estrogen treatment changes excitatory synaptic density in hippocampal CA1 neurons69,70 and enhances spatial memory.71–75 In rhesus monkeys, reproductive age, over chronological age, is associated with lower recognition memory and synaptic density in the hippocampus. 76 In clinical studies, estradiol levels across the menstrual cycle correlate with verbal memory performance,77–79 hippocampal volume,78,80 and hippocampal fractional anisotropy. 81 Some women also experience memory decline82–84 as well as functional changes in episodic and working memory networks27,28 across the menopausal transition.27,28,37,84–90 We and others have demonstrated direct associations between sex steroid hormone levels and memory performance28,83,91 as well as functional activity and connectivity in the memory circuitry.80,92 Comparing men and women in early midlife, women outperform men on verbal and associative memory tasks, 84 an effect which attenuates postmenopause due, in part, to estradiol decline. Together, these results underscore the impact of estradiol in shaping memory function in women and suggest that pathological changes occur over menopause. Additionally, ovariectomization in rodents leads to hippocampal degeneration and increased Aβ deposition, an effect that is reversed with estradiol treatment.34–36 In clinical studies, reduced grey and white matter volumes in AD-vulnerable regions 37 and Aβ accumulation are observed across the menopausal transition. These studies suggest that estradiol decline may increase vulnerability to AD pathology in women.
Hypertension, depression, and type 2 diabetes in midlife are important clinical risk factors for AD14,93–98 and there are significant sex differences in the development of these diseases and disorders. In midlife, hypertension and diabetes are more prevalent in men compared to women and depression is more prevalent in women.99–101 This greater risk for depression in women emerges at puberty102,103 and worsens during the menopausal transition.104,105 The clinical manifestation and comorbidity of these diseases and disorders also differ by sex. In women, cardiovascular disease is primarily associated with microvascular disease, while in men, the main contributor is obstructive coronary artery disease. 106 There is an overall increase in comorbidities among women with diabetes compared to men, 99 including physical and cognitive limitations, 107 anxiety and depression, 108 cardiovascular disease,109,110 and mortality. 99 Further, some studies suggest that these clinical risk factors may exacerbate the impact of genetic risk for AD. Studies looking at the relationship between hypertension and APOE status have found the negative impact of hypertension on neuronal damage and synaptic loss, 22 cerebral white matter lesions, 23 and Aβ burden 111 are all exacerbated by having the APOE ε4 allele. Similarly, the relationship between diabetes and lower cognitive performance 17 and AD pathology 18 was also found to be stronger in APOE ε4 carriers. Studies have also found that having multiple vascular risk factors in midlife is associated with greater Aβ deposition compared to having only one or none. 96 The association between MDD and APOE genotype remains unclear 112 ; however, the presence of the ε4 allele has been significantly associated with severe depression after adjusting for vascular diseases and lipid profiles in a southern Taiwanese population. 113 Depression is associated with alterations of the innate immune system, resulting in chronic inflammatory states.114,115 Other brain changes such as hippocampal volume changes may also explain how MDD emerges as a risk factor for dementia. 116 These studies are consistent with our current findings that genetic risk in conjunction with clinical risk factors are associated with increased AD risk in midlife. Further, having a greater number of clinical risk factors was associated with worse memory performance and greater Aβ pathology, suggesting a cumulative impact of comorbid conditions. This is especially relevant for women given that comorbidity between MDD and CVD is significantly higher in women than men.117,118
The goal of this study was to isolate as much as possible the impact of our genetic and clinical risk factors of interest. However, beyond genetic and clinical risk factors, we found HR participants overall had lower education, were less likely to be White, had higher BMI, and were older than the LR group. While these factors were unselected in the recruitment process and were controlled for in analyses, it is interesting to note that they are all independently related to increased risk of AD. Higher education contributes to cognitive reserve and can delay the onset of cognitive impairment and AD95,119; there is a higher risk of AD in African Americans and Hispanics compared to Whites 120 ; higher BMI and obesity in midlife significantly increase the risk for AD121,122; and older age is the biggest risk factor for AD. As with any study, it is not possible to control for everything that may impact AD risk without over-correcting. In fact, in unadjusted models, we found that overall effect sizes were larger, especially in men, suggesting that adjusting for these covariates may have a larger impact in men compared to women. These results demonstrate the complexity and comorbidity of AD risk factors, including genetic, clinical, and sociodemographic risk factors and highlight how a comprehensive approach to AD risk assessment, such as a clinical risk algorithm, would significantly improve our ability for early detection and prevention of the disease, analyses on which we are currently working.
The N-back paradigm evokes a robust response throughout the working memory circuitry, 27 including pronounced activation of the DLPFC which is typically accompanied by a robust deactivation of the HIPP.27,123–125 In our previous work, we found that deactivation of the hippocampus observed in pre/perimenopausal women was attenuated in postmenopausal women, suggesting a failure to disengage. Others have found similar effects in clinical populations and have hypothesized that a failure to disengage is related to a failure to suppress the default mode network during the task.126,127 Here, we found that HR postmenopausal women exhibited this failure to disengage the HIPP, while LR postmenopausal women showed similar patterns of deactivation to those previously reported for pre/perimenopausal women. 27 Similarly, we also found that HR women had higher activity in the HIPP during the verbal encoding task compared to LR women. These results suggest that altered fMRI activity in the HIPP previously reported in some postmenopausal women may primarily be driven by HR status and be clinically meaningful. To support this, we found that increased fMRI activity in the HIPP during working memory and verbal encoding tasks were related to both poor working memory and verbal encoding performance, respectively, and increased AD pathology. Alterations in fMRI activity related to AD pathology has been reported in several other studies8–11 which may reflect either a compensatory strategy or neural inefficiency in the early stages of the disease.10,12 These alterations differ by task, age, sex, and memory stages which may explain some of the inconsistencies in the literature in terms of directionality. 12 While some studies have reported hyperactivity in memory regions as a function of amyloid pathology,9–11 similar to our study, others found decreased activity in key memory circuitry regions such as the HIPP. 8 For example, Song et al. 11 found in younger elderly, higher cortical Aβ deposition in cognitively healthy adults was related to reduced task-related activity in the HIPP. In contrast, McDonough et al. 9 found a positive association between number of clinical risk factors and brain activity in the HIPP, default mode network, and entorhinal cortex during a memory retrieval task. In a separate cohort, we previously demonstrated that postmenopausal women with lower levels of brain-derived neurotrophic factor, a neuroprotective growth factor, similarly had higher fMRI BOLD activity in the HIPP during the same working memory task. 128 We also found that postmenopausal women with MDD that had lower levels of Dehydroepiandrosterone-sulfate (DHEAS), an adrenal androgen that is, in part, aromatized to estradiol and/or estrone, 129 also displayed this failure to disengage the HIPP. 130 Taken together, these studies suggest that at-risk women may show altered patterns of functional activity in postmenopause that may have clinical significance for risk of AD.
While task-based functional activity in the brain had a stronger association with Aβ pathology in women, cognitive function was more strongly related to Aβ pathology in men. In women, this relationship was only observed in women with poor memory performance (i.e., the bottom tertile of women). This supports work indicating a verbal memory advantage in women over men and suggests that Aβ accumulation may not be detected until there is more severe cognitive decline in women than would be necessitated for men. Women have a life-long advantage in verbal memory compared to men,82,131–134 which was also demonstrated in this study. Women performed significantly better than men on the verbal memory task. However, this advantage may mask underlying pathology that can hinder early detection in women. The same cut-offs on cognitive tests are used for men and women, which, as Maki et al. 135 have argued, may lead to underdiagnosis of cognitive impairment in women. This discrepancy may explain the higher rate of MCI in men versus women. However, as noted above, Aβ pathology had a stronger association with functional brain changes in women compared to men. As such, while cognitive changes may be more apparent in men, the underlying functional changes are greater in women and may be masked due to the female advantage in verbal memory. It may also explain why women with MCI show faster rates of cognitive decline 67 and brain atrophy 65 compared to men. If detection is delayed in women, progression may appear faster once they are diagnosed with MCI. Cognitive decline and brain atrophy progress faster in later stages of the disease. 56 Faster decline in women with MCI will have significant clinical implications for the individual and their families. As such, timing plays a key role in prevalence and manifestation such that sex differences in the clinical progression of AD indicate that women are protected relative to men at the prodromal phases but later exhibit steeper cognitive decline and higher rates of brain atrophy. 33
The current results should be viewed in light of certain limitations. The first is that the sample sizes when broken down by sex and risk status were relatively small, and thus we are currently aiming to replicate these findings in a larger sample. However, even with the relatively smaller sample size, we observed large differences between HR and LR participants, especially within the women, suggesting validity of the results. The small sample size may be why we did not find significant sex differences in the associations between risk status, cognitive function, and amyloid pathology. Secondly, our HR group had both genetic and clinical risk factors and therefore, it is not possible to assess the differential impact of each in this study. However, as noted above, we are currently conducting a follow-up study that is recruiting HR genetic only and HR clinical only groups, which will allow for the differentiation of the effects of clinical and genetic risks. In the same study, we are currently conducting a 7T MRI protocol which includes neurovascular structural and functional sequences. This will provide further information about peripheral cardiovascular measures on neurovascular integrity and risk for AD by sex. Peripheral measures of vascular function, such as measures of hypertension, can impact differences in brain activity and risk for AD 9 and we are not able to make this distinction with the current data. Further, our sample is primarily white and highly educated. As such, there are limitations to the generalizability of the results and caution is recommended when drawing comparisons with different demographics and cultures of genetic ancestry. The higher educated sample, however, may also provide some advantage for assessing clinical and genetic risk in the face of adequate social environment and resources and higher education. Finally, given that the current sample of women was all postmenopausal, it is not possible to assess the impact of menopause on AD risk, which we and others have previously demonstrated to be a critical transition period in women when early AD pathology emerges.27,28,37,84,88 Similarly, HRT was an exclusionary criteria and therefore, we are not able to assess the impact of HRT on AD risk. Five participants met HR status with only the APOE4 proxy. However, when we ran the analyses with and without these subjects, the results did not change.
Conclusions
Taken together, these findings contribute to our broader understanding of early risk factors for AD and the sex differences therein. Our results demonstrate genetic and clinical risk factors are associated with poorer memory performance, altered memory circuitry function, and higher Aβ deposition, which can be detected in midlife individuals who are considered cognitively healthy. Further, effects were primarily found in women, thus highlighting important sex differences in midlife. The ability to identify high-risk individuals before the onset of overt clinical symptoms will greatly improve early detection and prevention of the disease. It will also allow us to identify potential sex-dependent therapeutic targets associated with AD risk that could be deployed early in the trajectory of AD to maintain intact memory with aging.
Supplemental Material
sj-xlsx-1-alz-10.1177_13872877251366701 - Supplemental material for Sex differences in the impact of genetic and clinical risk factors for Alzheimer's disease in midlife
Supplemental material, sj-xlsx-1-alz-10.1177_13872877251366701 for Sex differences in the impact of genetic and clinical risk factors for Alzheimer's disease in midlife by Kyoko Konishi, Dylan S Spets, Paris Fisher, Sarah Aroner, Sarah M Sant, Dmitry Prokopenko, Hrishikesh Lokhande, Rohit Patel, Harlyn Aizley, Brianna Smith, Anne Remington, Emma Spooner, Alexandra Touroutoglou, Jonathan Rosand, Steven Arnold, Hang Lee, Bradford Dickerson, Rudolph E Tanzi, Tanuja Chitnis and Jill M Goldstein in Journal of Alzheimer's Disease
Supplemental Material
sj-xlsx-2-alz-10.1177_13872877251366701 - Supplemental material for Sex differences in the impact of genetic and clinical risk factors for Alzheimer's disease in midlife
Supplemental material, sj-xlsx-2-alz-10.1177_13872877251366701 for Sex differences in the impact of genetic and clinical risk factors for Alzheimer's disease in midlife by Kyoko Konishi, Dylan S Spets, Paris Fisher, Sarah Aroner, Sarah M Sant, Dmitry Prokopenko, Hrishikesh Lokhande, Rohit Patel, Harlyn Aizley, Brianna Smith, Anne Remington, Emma Spooner, Alexandra Touroutoglou, Jonathan Rosand, Steven Arnold, Hang Lee, Bradford Dickerson, Rudolph E Tanzi, Tanuja Chitnis and Jill M Goldstein in Journal of Alzheimer's Disease
Supplemental Material
sj-xlsx-3-alz-10.1177_13872877251366701 - Supplemental material for Sex differences in the impact of genetic and clinical risk factors for Alzheimer's disease in midlife
Supplemental material, sj-xlsx-3-alz-10.1177_13872877251366701 for Sex differences in the impact of genetic and clinical risk factors for Alzheimer's disease in midlife by Kyoko Konishi, Dylan S Spets, Paris Fisher, Sarah Aroner, Sarah M Sant, Dmitry Prokopenko, Hrishikesh Lokhande, Rohit Patel, Harlyn Aizley, Brianna Smith, Anne Remington, Emma Spooner, Alexandra Touroutoglou, Jonathan Rosand, Steven Arnold, Hang Lee, Bradford Dickerson, Rudolph E Tanzi, Tanuja Chitnis and Jill M Goldstein in Journal of Alzheimer's Disease
Supplemental Material
sj-xlsx-4-alz-10.1177_13872877251366701 - Supplemental material for Sex differences in the impact of genetic and clinical risk factors for Alzheimer's disease in midlife
Supplemental material, sj-xlsx-4-alz-10.1177_13872877251366701 for Sex differences in the impact of genetic and clinical risk factors for Alzheimer's disease in midlife by Kyoko Konishi, Dylan S Spets, Paris Fisher, Sarah Aroner, Sarah M Sant, Dmitry Prokopenko, Hrishikesh Lokhande, Rohit Patel, Harlyn Aizley, Brianna Smith, Anne Remington, Emma Spooner, Alexandra Touroutoglou, Jonathan Rosand, Steven Arnold, Hang Lee, Bradford Dickerson, Rudolph E Tanzi, Tanuja Chitnis and Jill M Goldstein in Journal of Alzheimer's Disease
Footnotes
Acknowledgements
We would like to thank Elizabeth Karlson, M.D. and her team for the initiation of the MGB Biobank and their help with the recruitment of HATCH participants.
Ethical considerations
The Partners (now Mass General Brigham (MGB)) Human Research Committee Institutional Review Board granted Human Studies participants’ approval.
Consent to participate
All volunteers gave written informed consent and were paid for their participation.
Author contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Institute on Aging (AG074008 (JMG, P.I.), and Dr Konishi's time on NIA K01AG081500 and ORWH-NICHD BIRCWH K12HD051959. Initial pilot work was funded by the Harvard NeuroDiscovery Center (PI: JMG) and BrightFocus Foundation (PI: JMG). Additional support was by Henry and Allison McCance Center for Brain Health, National Institute of Biomedical Imaging and Bioengineering (P41EB015896), and Harvard Catalyst | The Harvard Clinical and Translational Science Center (National Center for Advancing Translational Sciences, NIH Award UL 1TR002541) and financial contributions from Harvard University and affiliated academic healthcare centers. The content is solely the responsibility of the authors and does not necessarily represent the official views of Harvard Catalyst, Harvard University and its affiliated academic healthcare centers, or the National Institutes of Health.
National Institute of Biomedical Imaging and Bioengineering, BrightFocus Foundation, Harvard NeuroDiscovery Center, Office of Research on Women's Health, National Institute on Aging, National Center for Advancing Translational Sciences, (grant number P41EB015896, CA2018607, K12HD051959, AG074008, K01AG081500, UL 1TR002541).
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JMG is on the scientific advisory board for and has equity interest in Cala Health (a neuromodulation company). JMG’s interests are managed by MGH and MassGeneral Brigham HealthCare in accordance with the conflict of interest policies. However, the work in the study presented here is unrelated thus there is no conflict of interest. Dr. Chitnis has received compensation for speaking engagements from Prime Education, LLC. Dr. Chitnis has received research support from the National Institutes of Health, National MS Society, US Department of Defense, Sumaira Foundation, Brainstorm Cell Therapeutics, Bristol Myers Squibb, Genentech, EMD Serono, I-Mab Biopharma, Mallinckrodt ARD, Massachusetts Life Sciences Center, Novartis Pharmaceuticals, Octave Bioscience, Sanofi Genzyme, Tiziana Life Sciences, and Wesley Clover International. All activities and funding have occurred within the past 24 months and disclosures do not conflict with the work being presented.
Data availability statement
The deidentified data that support the findings of this study are available from the corresponding author, JMG, dependent on reasonable request, our human studies requirements, institutional restrictions, and signature of a formal data sharing agreement.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
