Abstract
Background:
In amyloid-positive individuals at risk for Alzheimer’s disease (AD), high soluble 42-amino acid amyloid-β (Aβ42) levels are associated with normal cognition. It is unknown if this relationship applies longitudinally in a genetic cohort.
Objective:
To test the hypothesis that high Aβ42 preserves normal cognition in
amyloid-positive individuals with Alzheimer’s disease (AD)-causing mutations
(
Methods:
Cognitive progression was defined as any increase in Clinical Dementia Rating (CDR = 0,
normal cognition; 0.5, very mild dementia; 1, mild dementia) over 3 years.
Amyloid-positivity was defined as a standard uptake value ratio (SUVR) ≥1.42 by
Pittsburgh compound-B positron emission tomography (PiB-PET). We used modified Poisson
regression models to estimate relative risk (RR), adjusted for age at onset, sex,
education,
Results:
Of 232 mutation carriers, 108 were PiB-PET-positive at baseline, with 43 (39.8%)
meeting criteria for progression after 3.3±2.0 years. Soluble Aβ42 levels
were higher among CDR non-progressors than CDR progressors. Higher Aβ42
predicted a lower risk of progression (adjusted RR, 0.36; 95% confidence interval [CI],
0.19–0.67;
Conclusion:
High CSF Aβ42 levels predict normal cognition in amyloid-positive individuals with AD-causing genetic mutations.
INTRODUCTION
Key support for the toxic amyloid hypothesis comes from the observation that mutations in
any of three genes (
The hypothesis of Aβ toxicity has traditionally been supported by the notion that
AD-causing mutation carriers must have
We recently observed that among amyloid positron emission tomography (PET)-positive
individuals, higher levels of soluble Aβ42 were associated with normal cognition
and brain volumes in all tertiles of brain amyloidosis, with an effect size greater than
that of increases in brain amyloid burden [11]. We
here tested the hypothesis that in amyloid PET-positive individuals with AD-causing
METHODS
Overview
We conducted a retrospective longitudinal study among mutation carriers participating in the Dominantly Inherited Alzheimer Network (DIAN) cohort study. The DIAN study longitudinally evaluates families with dominantly inherited AD and non-carrier relatives of the probands. Details about the DIAN study design can be found in previous publications [1, 12]. We included all mutation carriers with at least two follow-up visits including baseline, without applying any other inclusion/exclusion criteria. All analyses were conducted in two cohorts: 1) amyloid PET-positive cohort, the most critical as these are individuals considered at greater risk for developing dementia; and 2) overall cohort, which included both amyloid PET-positive and amyloid PET-negative subjects.
Clinical assessment
Each assessment comprised a detailed medical history, neurological, and neuropsychological examination. Cognitive function was primarily quantified using the Clinical Dementia Rating scale (CDR = 0, indicates normal cognition; 0.5, very mild dementia; 1, mild dementia; 2, moderate dementia; and 3, severe dementia) [13]. Our primary endpoint was CDR progression, defined as any increase in CDR over the follow-up period. Secondary endpoints were progression to CDR ≥0.5 confirmed in two consecutive visits as per DIAN guidelines to minimize misclassification, progression to CDR ≥1, Mini-Mental State Examination (MMSE) score ≤24 at last visit (higher means better cognition) [14], and a CDR sum of boxes (CDR-SB) score ≥4.5 at last visit (lower means better cognition) [15]. CDR-SB = 4.5 corresponds to mild dementia [15].
CSF biomarkers
We evaluated CSF Aβ42, p-tau, and t-tau levels (INNO-BIA AlzBio3 from Fujirebio, Malvern, PA). In order to limit the variability due to measurement error, we considered only data from the same assay, as used in previous analyses [4]. While CSF Aβ40 was not available from this particular assay, precluding us from entering it into our analysis, it did not limit the testing of our hypothesis since Aβ40 exhibits lower fibrillogenicity and lesser depletion than Aβ42 [16], and is therefore less relevant to the process of protein aggregation than Aβ42.
Neuroimaging
Pittsburgh compound B PET (PiB-PET) was used to quantify the burden of insoluble brain amyloid plaques. To reduce partial volume effects, data were processed using a regional spread function (RSF), shown to enhance sensitivity [17]. We defined subjects as amyloid PiB-PET-positive if their standard uptake value ratio (SUVR) was ≥1.42 [18], validated by using +5% higher SUVR threshold (≥1.49) as a sensitivity analysis. At the last visit, we also used RSF-processed fluorodeoxyglucose (FDG)-PET to quantify the metabolism of the precuneus (average of both hemispheres) and brain magnetic resonance imaging (MRI) to quantify hippocampal volume (average of both hemispheres) adjusted for intracranial volume, using the DIAN protocol equation.
Aims and sample size calculation
Our primary aim was to test the hypothesis that higher CSF Aβ42 at baseline reduces the risk of CDR progression to a greater extent than lower SUVR, lower p-tau, and lower t-tau levels among PiB-PET-positive gene carriers. Secondarily, we tested whether compared with lower baseline SUVR, p-tau, and t-tau levels, higher baseline CSF Aβ42 predict lower risk of progression to CDR ≥0.5 in at least two consecutive visits, progression to CDR ≥1 at any visit, MMSE ≤24 at last visit, CDR-SB ≥4.5 at last visit, and higher FDG-PET metabolism and hippocampi volume at last visit. While these aims were focused on the PiB-PET-positive cohort, we conducted similar analyses on the full cohort, which included PiB-PET-negative participants.
Sample size calculation
In a previous cross-sectional study of PiB-PET-positive individuals, we found that
higher CSF Aβ42 levels were associated with greater odds of normal cognition
than AD (adjusted odds ratio [OR], 6.26;
Statistical analysis
Continuous standardized CSF and SUVR levels were considered primary predictors in the
analyses for a direct comparison across effects of CSF and SUVR levels. For the primary
outcome of CDR progression, we used a modified Poisson model regression [19] to determine the associations of baseline CSF
Aβ42, CSF t-tau and p-tau, and SUVR levels with CDR progression after
adjusting for age at onset (estimated mean age of symptom onset), sex, education,
Ethics approval
The study protocol for DIAN was approved by the local ethical committees of all participating institutions. The DIAN study was conducted in accordance with the Declaration of Helsinki and written informed consent was obtained from each participant.
RESULTS
Of 534 subjects participating in the DIAN study (mean age, 38±11.1 years), 232 mutation carriers met eligibility criteria (mean age, 38.3±11 years). Among them, 191 had available PiB-PET data at baseline (mean SUVR, 1.9±1.0), of whom 108 were PiB-PET-positive (mean SUVR, 2.5±1.0) (Table 1). These subjects were followed for a mean of 3.3±2.0 years (range = 1, 9). Because of missing data on some CSF and SUVR levels, multivariable analyses were based on 93 PiB-PET-positive samples, 85 PiB-PET-positive samples (SUVR ≥1.49), and 162 overall samples which include PiB-PET-positive and negative samples (Supplementary Figure 1). No differences in the baseline characteristics except CDR at baseline were observed between cohorts with missing versus without missing data (Supplementary Table 1).
Characteristics of study cohort
n, number of subjects;
Primary endpoint
Amyloid PiB-PET-positive cohort
A total of 43 (39.8%) subjects met criteria for CDR progression. In adjusted analyses,
the risk of progression was reduced to a greater extent by higher CSF Aβ42
(RR, 0.36; 95% CI, 0.19–0.67;
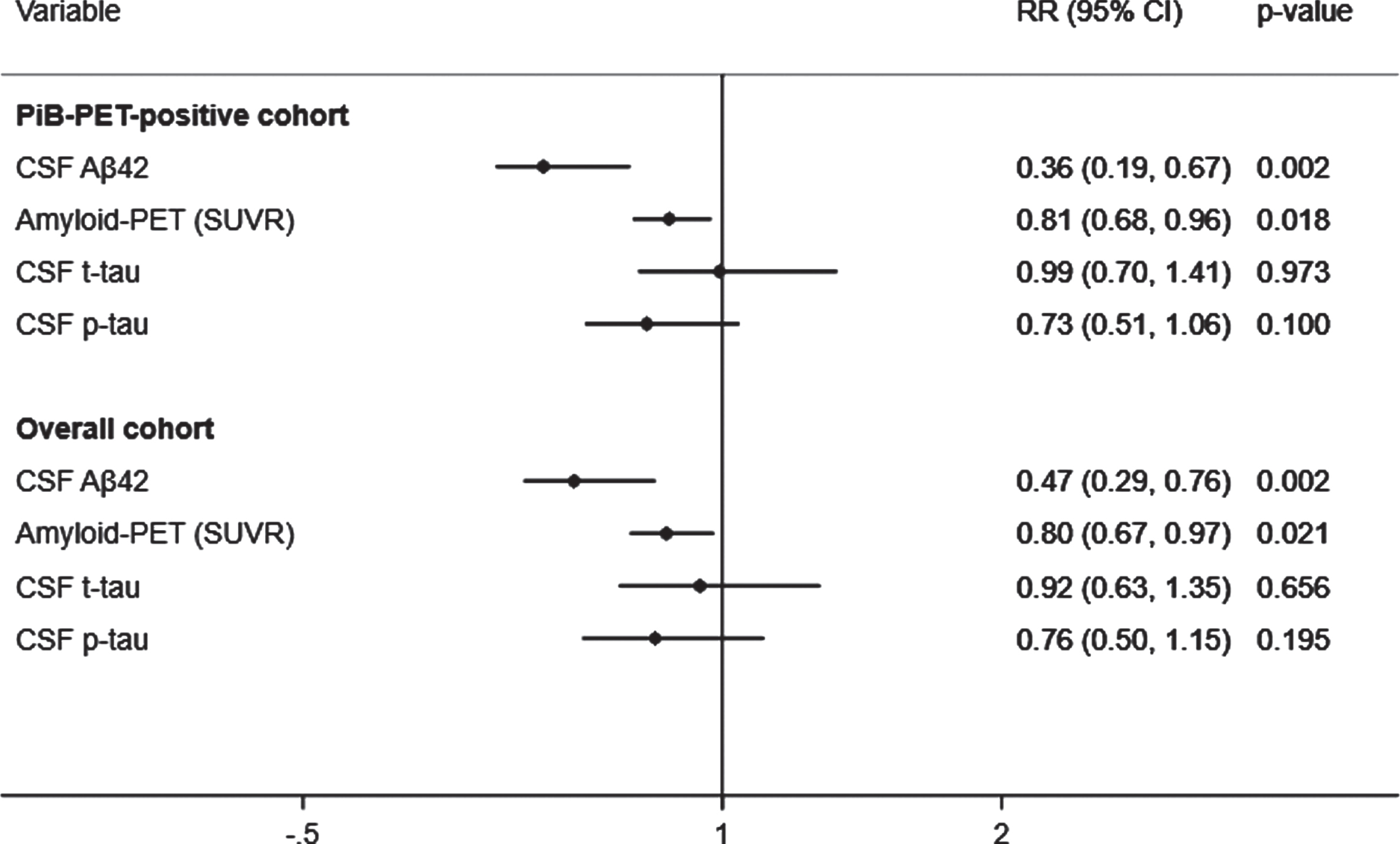
Adjusted prediction of CDR progression with baseline CSF Aβ42, p-tau, t-tau, and
amyloid (PiB)-PET SUVR levels. CSF, cerebrospinal fluid; PiB-PET, Pittsburgh
compound B positron emission tomography; SUVR, standardized uptake value ratio; CDR,
Clinical Dementia Rating; Aβ42, 42-amino acid amyloid-beta peptide;
p-tau, phospho-Tau; t-tau, total-Tau; HR, hazard ratio; CI, confidence interval. RR
reflects effect size for the association with a one standard deviation higher in CSF
Aβ42 levels and lower in SUVR, CSF t-tau and p-tau levels. Analyses
were adjusted for age at onset, sex, education,
Overall cohort
A total of 67 (28.9%) subjects met criteria for CDR progression in the overall cohort.
Higher CSF Aβ42 levels predicted a reduced risk of progression (RR, 0.47; 95%
CI, 0.29–0.76;
CSF Aβ42 and CDR progression
CSF Aβ42 levels were higher among CDR non-progressors than CDR progressors
(Fig. 2). CSF Aβ42
<270 pg/ml predicted progression (area under the curve, 80.5%; sensitivity, 72.2%;
specificity, 74.5%) regardless of increasing SUVR levels. The progression-free survival
was longer with CSF Aβ42 ≥270 pg/ml compared to CSF Aβ42 <270
pg/ml over the follow-up period in both PiB-PET-positive (
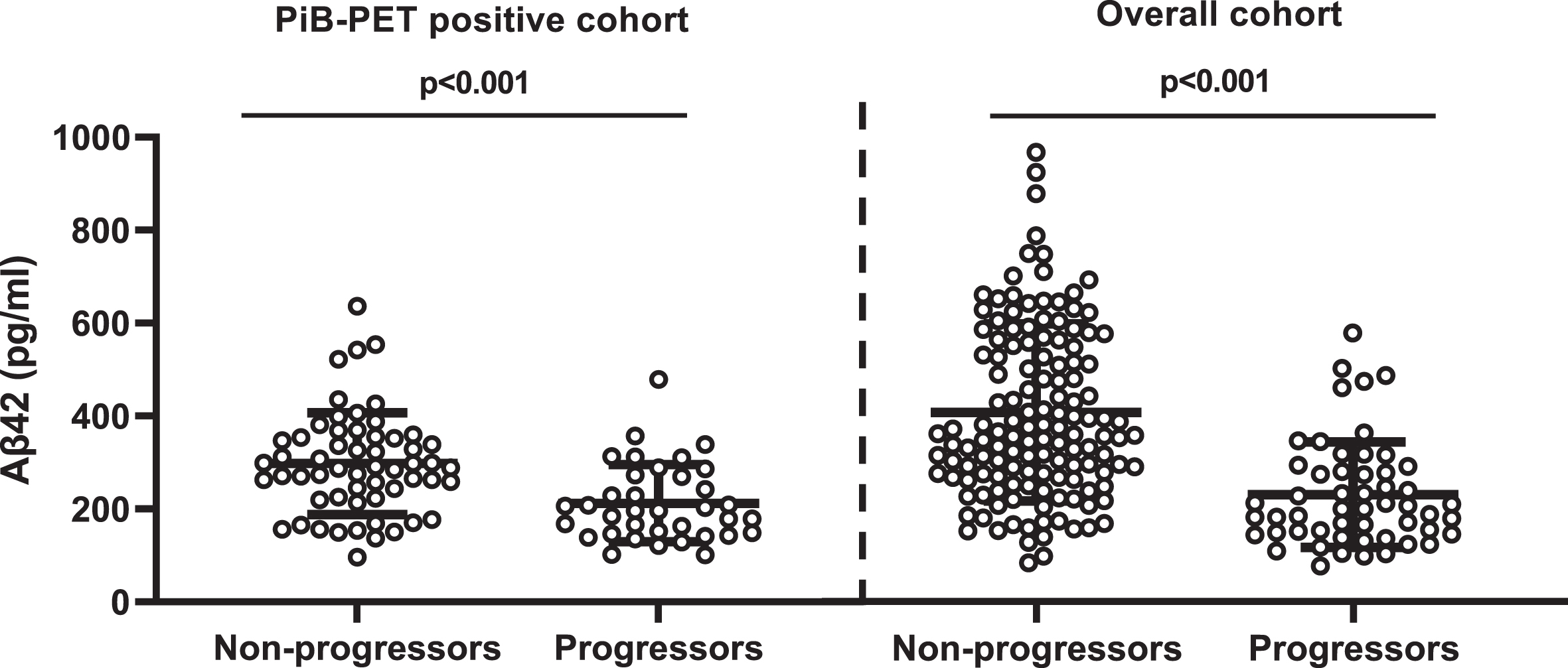
Comparison of CSF Aβ42 levels between non-progressors and progressors. PiB-PET-positive cohort: non-CDR progressors (297.73±13.66) versus CDR progressors (218.73±17.22); overall cohort: non-CDR progressors (380.83±14.5) versus CDR progressors (313.35±26.46). Error bar represents the standard error of mean. CSF, cerebrospinal fluid; PiB-PET, Pittsburgh compound B positron emission tomography; CDR, Clinical Dementia Rating; Aβ42, 42-amino acid amyloid-beta peptide. Overall cohort includes PiB-PET-positive and negative samples.
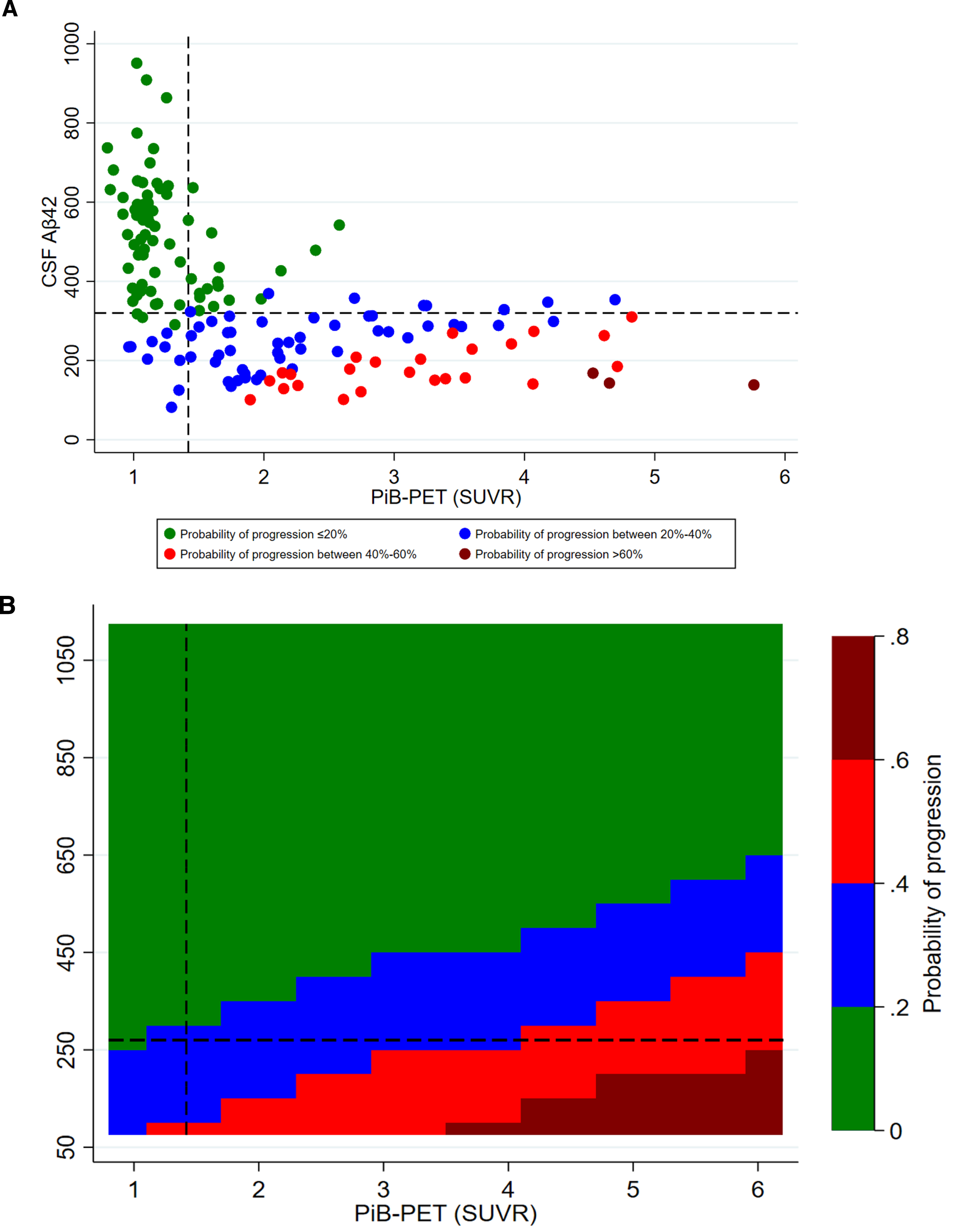
Adjusted probability of CDR progression. A) scatter plot of CSF Aβ42 and
PiB-PET SUVR levels; B) contour plot of CSF Aβ42 and PiB-PET SUVR levels.
CSF, cerebrospinal fluid; Aβ42, 42-amino acid amyloid-beta peptide;
PiB-PET, Pittsburgh compound B positron emission tomography; CDR, Clinical Dementia
Rating; p-tau, phospho-Tau; t-tau, total-Tau. All models were adjusted for age at
onset, sex, education,
Secondary endpoints
Amyloid PiB-PET-positive cohort
Higher baseline CSF Aβ42 predicted a reduced risk of progression to CDR ≥0.5 (RR, 0.55;
95% CI, 0.36–0.83;
Adjusted associations of baseline CSF and SUVR with secondary cognitive outcomes
*Relative risk (RR) is with a one standard deviation higher in CSF
Aβ42 levels and lower in CSF t-tau, CSF p-tau, and SUVR levels. CI,
confidence interval; CDR, clinical dementia rating; CSF, cerebrospinal fluid;
Aβ42, 42-amino acid amyloid-beta peptide; t-tau, total tau; p-tau,
phospho-Tau; CDR-SB, CDR sum of boxes; PiB-PET, Pittsburgh compound B positron
emission tomography; SUVR, standardized uptake value ratio; MMSE, Mini-Mental State
Examination; FDG, fluorodeoxyglucose. All CSF and SUVR values are standardized.
Analysis adjusted for mean mutation age of symptom onset, sex, education,
Overall cohort
Higher baseline CSF Aβ42 predicted a reduced risk of progression to CDR ≥0.5
(RR, 0.52; 95% CI, 0.35, 0.77;
DISCUSSION
This longitudinal analysis of a genetic cohort showed that in amyloid PiB-PET-positive individuals with autosomal dominant AD-causing mutations higher levels of CSF Aβ42 were associated with reduced risk of progression to cognitive impairment, as well as larger hippocampal volume and higher brain metabolism in the precuneus. Conversely, cognitive deterioration was strongly predicted by lower levels of soluble Aβ42 but not by increases in SUVR, t-tau, or p-tau. These data are in agreement with those of a cross-sectional analysis of a sporadic AD cohort (in participants of the Alzheimer’s Disease Neuroimaging Inventory cohort study), in which higher CSF Aβ42 levels were associated with normal cognition regardless of (and despite increasing) SUVR levels among PiB-PET-positive individuals [11], making a reduction in CSF Aβ42 levels a better marker of progression than higher levels of CSF t-tau, p-tau, or amyloid PiB-PET SUVR.
A threshold of compensation can tentatively be drawn from these data, with further studies
needed to validating it. We found that levels of CSF Aβ42 above 270 pg/ml
predicted a reduced risk of CDR progression across any levels of PiB-PET SUVR. The better
prediction of disease conversion by the reduction of CSF Aβ42 levels than by the
increase in brain amyloidosis is in line with its observed decline as early as 25 years
before the onset of cognitive impairment in genetic AD [4]. There is also support for a greater pathophysiologic importance of the loss of
soluble Aβ42 over the accrual of insoluble PiB-PET amyloid from knockout animal
models of the Aβ42 precursor, AβPP, which yields neurodegeneration without brain
amyloidosis [20], and in AD patients treated with
BACE-1 inhibitors, which reduced CSF Aβ42 levels and worsened cognitive symptoms
regardless of changes in brain amyloid [21].
Amyloids represent the end product of an irreversible phase transition process of proteins,
changing from a soluble to an insoluble state via a physicochemical process termed
nucleation [26]. Nucleation is the rate-limiting
step of the first stable amyloid cluster (nucleus), after which the phase transition
proceeds spontaneously until all the available soluble substrate is consumed. While
nucleation is not favorable under normal conditions, it becomes favorable at higher
concentrations, such as those attained via gene duplication, or when the protein is rendered
unstable due to structural mutations [27]. The net
outcome is decreased availability of soluble Aβ42. This explains why their
depletion is universal across all reported familial AD mutations, including
Major strengths of this analysis are its longitudinal design and the relatively large number of subjects with disease-causing mutations. The findings are supported by the use of several complementary analytic methods on cognitive assessments and neuroimaging data and confirmed by sensitivity analyses. Major limitations include the inability to adjust for the estimated year to symptom onset (highly correlated with CSF and SUVR levels and did not serve as a confounder), and the lack of data on CSF Aβ40 (and therefore of Aβ42/Aβ40 ratio; see below) and on oligomeric species. On the last point, subjects with normal cognition despite high PiB-PET plaque burden had high, not low CSF Aβ42 [11], opposite to the direction predicted by the hypothesis of oligomeric toxicity. Oligomers are very transient and the majority of them dissociate back to monomers rather than progress to aggregates [31]. Importantly, the reduction of soluble Aβ42 levels during the disease course reduces the substrate for the oligomers, further questioning any sustainable toxicity from them.
A note related to the issue of ratio versus absolute values is worth making. Early studies
argued that absolute levels of Aβ42 decrease but the
Aβ42/Aβ40 ratio
In conclusion, higher soluble Aβ42 levels are associated with reduced risk of CDR progression, normal cognition, normal hippocampal volume, and normal precuneus metabolism to a greater extent than lower brain amyloid, lower p-tau, and lower t-tau levels in amyloid PiB-PET-positive individuals with autosomal dominant AD-causing genetic mutations. Brain toxicity in AD may be predominantly mediated by a reduction of the soluble protein pool, its functional fraction, rather than its accrual into amyloids.
DATA SHARING
All data are available upon request from the Dominantly Inherited Alzheimer Network (DIAN) through the study website, https://dian.wustl.edu/.
Footnotes
ACKNOWLEDGMENTS
We thank the Knight Alzheimer Disease Research Center (Knight ADRC) of the Dominantly Inherited Alzheimer Network (DIAN). The authors thank all the participants and their families and the support of all the research staff involved at each of the participating sites. We particularly thank the DIAN consortia statisticians for independently confirming the accuracy of the data source and replicating the results of the analysis conducted for this study.
Data collection and sharing for this project was supported by The Dominantly Inherited Alzheimer Network (DIAN, U19AG032438) funded by the National Institute on Aging (NIA) and the Alzheimer’s Association (SG-20-690363-DIAN). Further funding came from the German Center for Neurodegenerative Diseases (DZNE), Raul Carrea Institute for Neurological Research (FLENI), Partial support by the Research and Development Grants for Dementia from Japan Agency for Medical Research and Development, AMED, and the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), Spanish Institute of Health Carlos III (ISCIII), Canadian Institutes of Health Research (CIHR), Canadian Consortium of Neurodegeneration and Aging, Brain Canada Foundation, and Fonds de Recherche du Québec – Santé. This manuscript has been reviewed by DIAN Study investigators for scientific content and consistency of data interpretation with previous DIAN Study publications. We acknowledge the altruism of the participants and their families and contributions of the DIAN research and support staff at each of the participating sites for their contributions to this study.
