Abstract
Background
Intravenous hyper-immune anti-cytomegalovirus immunoglobulins (CMV-IG) therapy are licensed for prophylaxis, yet their therapeutic value in established CMV disease remains poorly defined. We aimed to evaluate the clinical and virological impact of adjunctive CMV-IG in combination with standard antiviral therapy in immunocompromised patients with CMV disease, compared with standard antiviral therapy alone.
Methods
We performed a single-center retrospective case-control study at A.O.U. “Federico II” University Hospital, Naples, Italy, including consecutive immunocompromised patients with CMV disease between March 2021 and November 2024. Cases received standard antiviral therapy plus CMV-IG (100 IU kg-1 day-1 for three consecutive days). Controls received antivirals only.
Results
Thirty-four patients were analyzed (15 cases, 19 controls). Thirty-day survival was 93% vs 95% (p=>0.9) and composite outcome (combination of 30-day mortality and clinical response) occurred in 93% vs 90% (p=>0.9) in cases and controls, respectively. Median time-to-virological response in days was achieved 4 vs 7 days (p=<0.001), as was median duration of antiviral therapy in days 12 vs 16 days (p=0.001) and hospitalization 14 vs 19 days (p=0.004) in cases and controls, respectively. Adverse-event rates were low and comparable in the 2 groups.
Conclusions
In this real-world cohort, adjunctive CMV-IG did not affect mortality, but was associated with faster virological clearance and shorter antiviral and hospital courses, without additional toxicity.
Keywords
Introduction
Human cytomegalovirus (CMV) remains a leading cause of morbidity in immunocompromised hosts, notably recipients of solid-organ transplantation (SOT), hematopoietic-stem-cell transplantation (HSCT), individuals with hematological malignancies, autoimmune diseases under immunosuppression, advanced HIV infection and pregnant women.1–5
Antiviral agents such as ganciclovir, valganciclovir, foscarnet, and cidofovir have markedly improved outcomes for cytomegalovirus (CMV) infection, but their use is constrained by drug resistance, nephrotoxicity, myelotoxicity, and potential teratogenicity.6–8 Recently, the novel antiviral maribavir became available for refractory or resistant cases; however, real-world data are still limited, and its efficacy may be curtailed by a relatively low genetic barrier to resistance and suboptimal penetration into certain anatomical sites.9,10
CMV-specific hyper-immune immunoglobulins (CMV-IG) provide high-titer neutralizing antibodies and may exert additional immunomodulatory effects. While meta-analyses support their prophylactic use in high-risk SOT and HSCT, evidence for therapeutic use in established CMV disease is scarce and scanty, largely confined to historical series of pneumonitis or colitis and isolated case reports of refractory infection.11–20 International guidelines therefore give only weak, low-quality recommendations for CMV-IG as adjunctive therapy in severe or resistant disease.13–16
Motivated by repeated clinical scenarios of severe CMV disease despite optimal antivirals, we implemented adjunctive CMV-IG in selected cases and designed the present study to compare real-life outcomes with those of matched controls receiving antivirals alone.
Methods
We conducted a retrospective, single-center, case-control study at the A.O.U. “Federico II” University Hospital in Naples, Italy. Eligible patients were those with laboratory-confirmed CMV infection accompanied by attributable clinical manifestations between 1 March 2021 and 30 November 2024. Therefore, all immunocompromised patients with CMV disease were recruited. CMV disease was diagnosed by quantitative real-time PCR on whole blood together with compatible symptoms or end-organ involvement as per international consensus definitions. 1 In particular, regarding CMV syndrome, CMV detected in whole blood < 7 days from symptom onset and at least two of the following: fever ≥ 38 °C × ≥2 days; new/worsening grade-2 malaise/fatigue; leukopenia/neutropenia (>20 % drop or WBC < 3.5 × 109/L); ≥5 % atypical lymphocytes; thrombocytopenia (>20 % drop or platelets < 100 × 109/L); ALT/AST > 2 × ULN (Upper Limit of Normal) (non-liver grafts). Regarding tissue-invasive (end-organ) disease, compatible symptoms/signs plus direct documentation of CMV in the affected organ by histopathology with inclusions, immunohistochemistry, in-situ hybridisation, culture or PCR. This is the criterion for “proven” disease; very high organ-specific viral load may be accepted as “possible/probable” when biopsy is not feasible. Sites include GI tract, lung, liver, retina, CNS, kidney, bladder, myocardium, pancreas, etc. 1
All patients received systemic antivirals tailored to renal function: intravenous ganciclovir (5 mg kg-1 q12h) or foscarnet (60 mg kg-1 q8h), oral valganciclovir (900 mg q12h), maribavir (400 mg q12h) for refractory infection, or valaciclovir (8 g day-1) in pregnancy.
Cases additionally received CMV-IG, 100 IU kg-1 day-1 infused i. v. over 2 h for three consecutive days (off-label therapeutic use). The decision to administer CMV-specific immunoglobulins was discretionary and based on clinical judgment.
Depending on the individual clinical context and degree of immunosuppression, immunosuppressive therapy was reassessed on a case-by-case basis whenever feasible.
We aimed to evaluate primary and secondary endpoints, in particular:
Demographic, clinical, laboratory and therapeutic data were extracted from electronic records. Viral load was quantified every 72–96 h. Renal impairment was defined as a ≥0.3 mg dL-1 or ≥50 % rise in serum creatinine.
Continuous variables are reported as medians with ranges (min-max) and compared by Mann–Whitney U test. Categorical variables are summarized as counts (percentages) and compared by χ2 or Fisher’s exact test, as appropriate. Survival was plotted with Kaplan–Meier curves and compared by log-rank test. A univariate log-linear regression was used for the two variables of hospitalization duration and antiviral therapy duration using the type of treatment as a predictor; variables with p<0.05 in univariable analyses entered the model. Two-tailed p<0.05 denoted significance. Analyses were performed using R software. The power analysis for sample size calculation was not done.
Verbal consent was obtained from each participant and the IRB (Institutional Review Board) waived the requirement for written consent. The IRB approved this study.
Results
Demographic and clinical characteristics of the study population.

SOT: Solid Organ Transplants, HSCT: hematopoietic Stem Cell Transplant, HIV/AIDS: Human Immunodeficiency Virus/Acquired Immunodeficiency Syndrome, Category C3: Classification Centers for Disease Control and Prevention (CDC) 1993, SLE: Systemic lupus erythematosus, IBD: Inflammatory Bowel Diseases. Two-tailed p<0.05 denoted significance.
CMV-related clinical manifestations and antiviral treatments.

awith fever and/or pancytopenia. Two-tailed p<0.05 denoted significance.
Baseline clinical characteristics of patients, as well as type of disease and antiviral did not differ significantly between cases and controls (Tables 1 and 2). However, median duration of antiviral therapy was shorter with CMV-IG (12 [8–20] vs 16 [12–26] days; p =0.001). All cases completed the planned three-day CMV-IG course; no repeat courses were given (Table 2).
Outcomes and adverse events: CMV-IG combination vs no combination.

adefined as resolution of clinical manifestations.
bdefined as negativization of CMV-DNA in plasma or blood.
Two-tailed p<0.05 denoted significance.
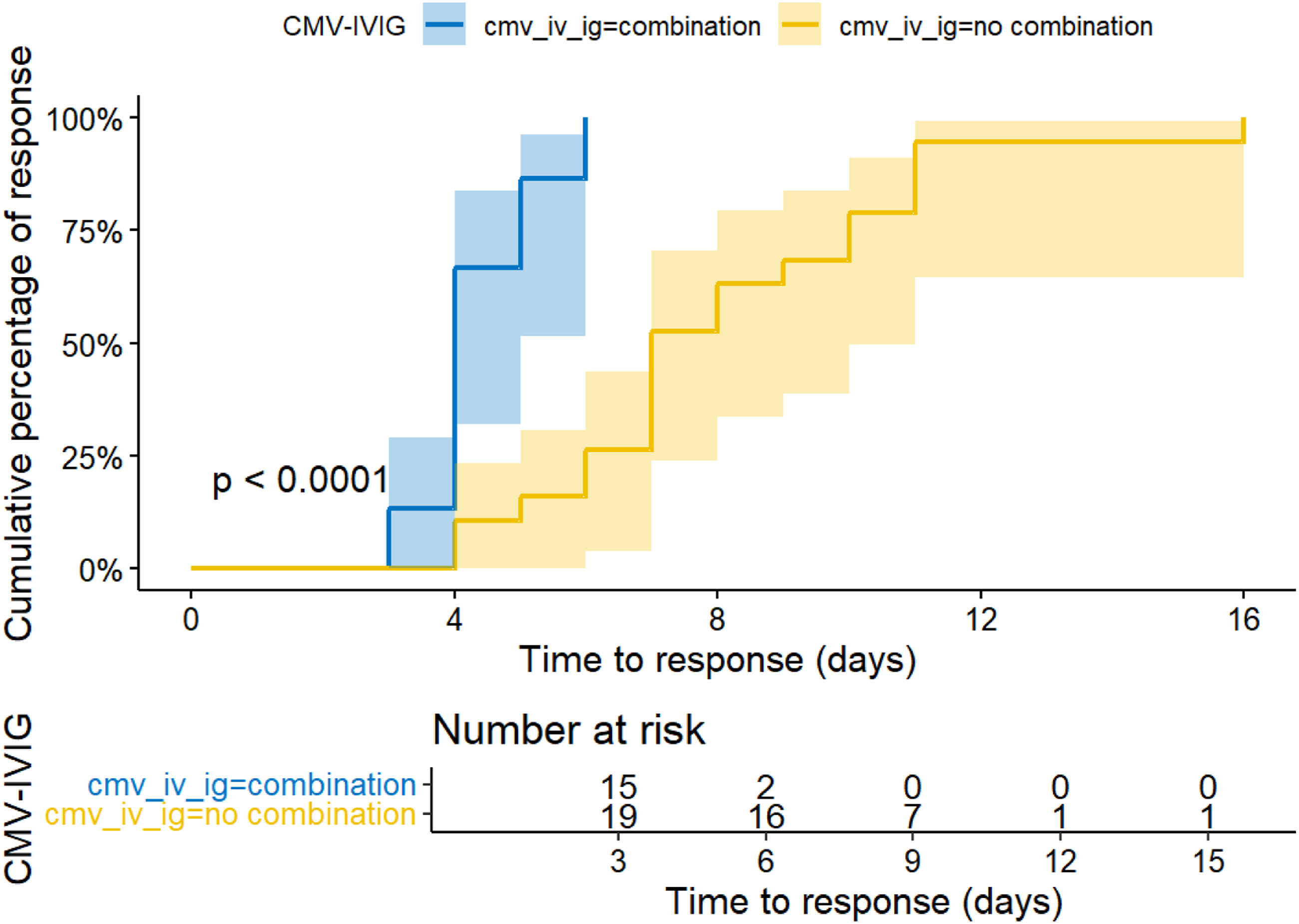
Timing of viral response: CMV-IV Ig combination treatment vs No CMV-IV Ig combination treatment.
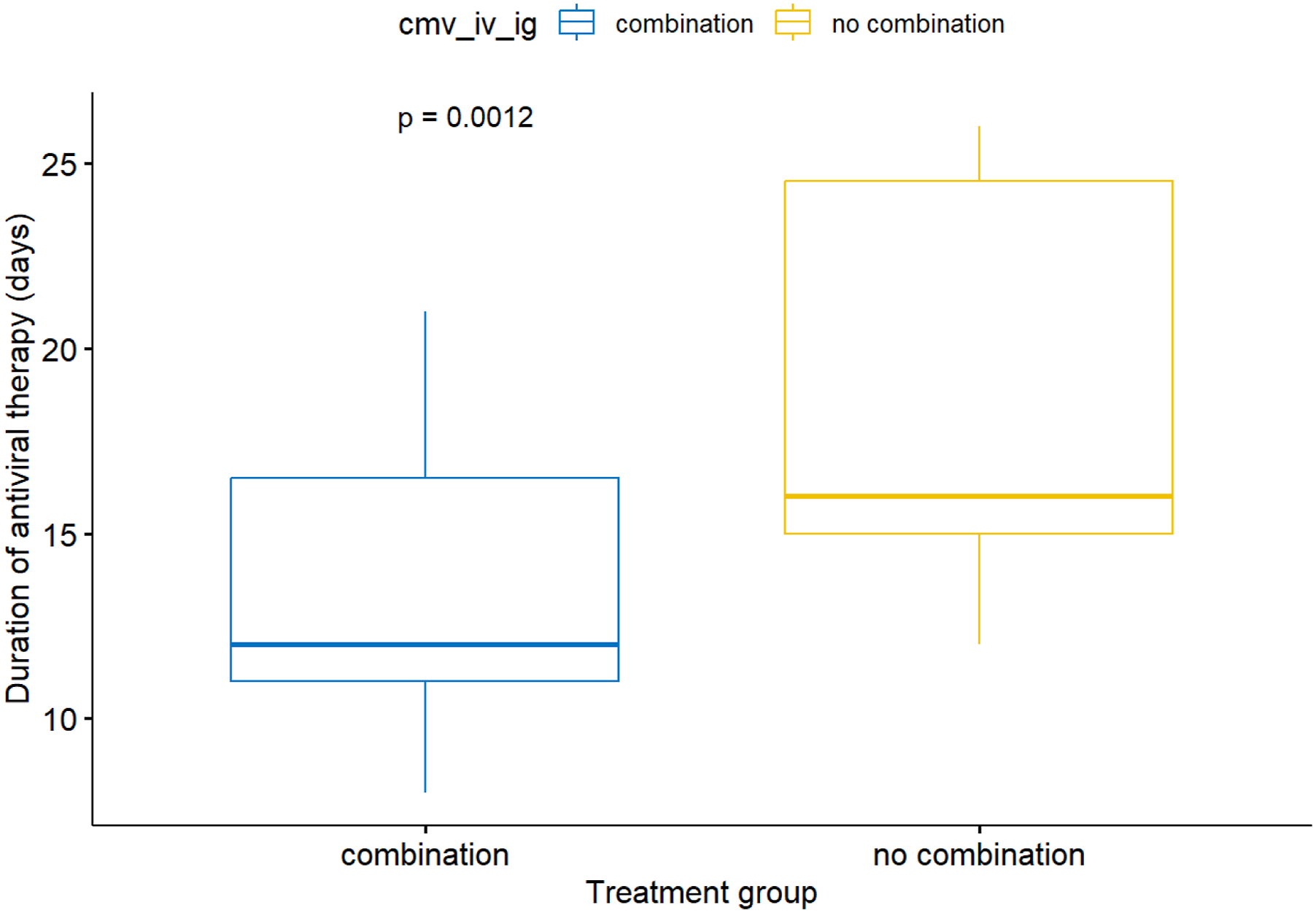
Duration in days of antiviral therapy: CMV-IV Ig combination treatment vs No CMV-IV Ig combination treatment.
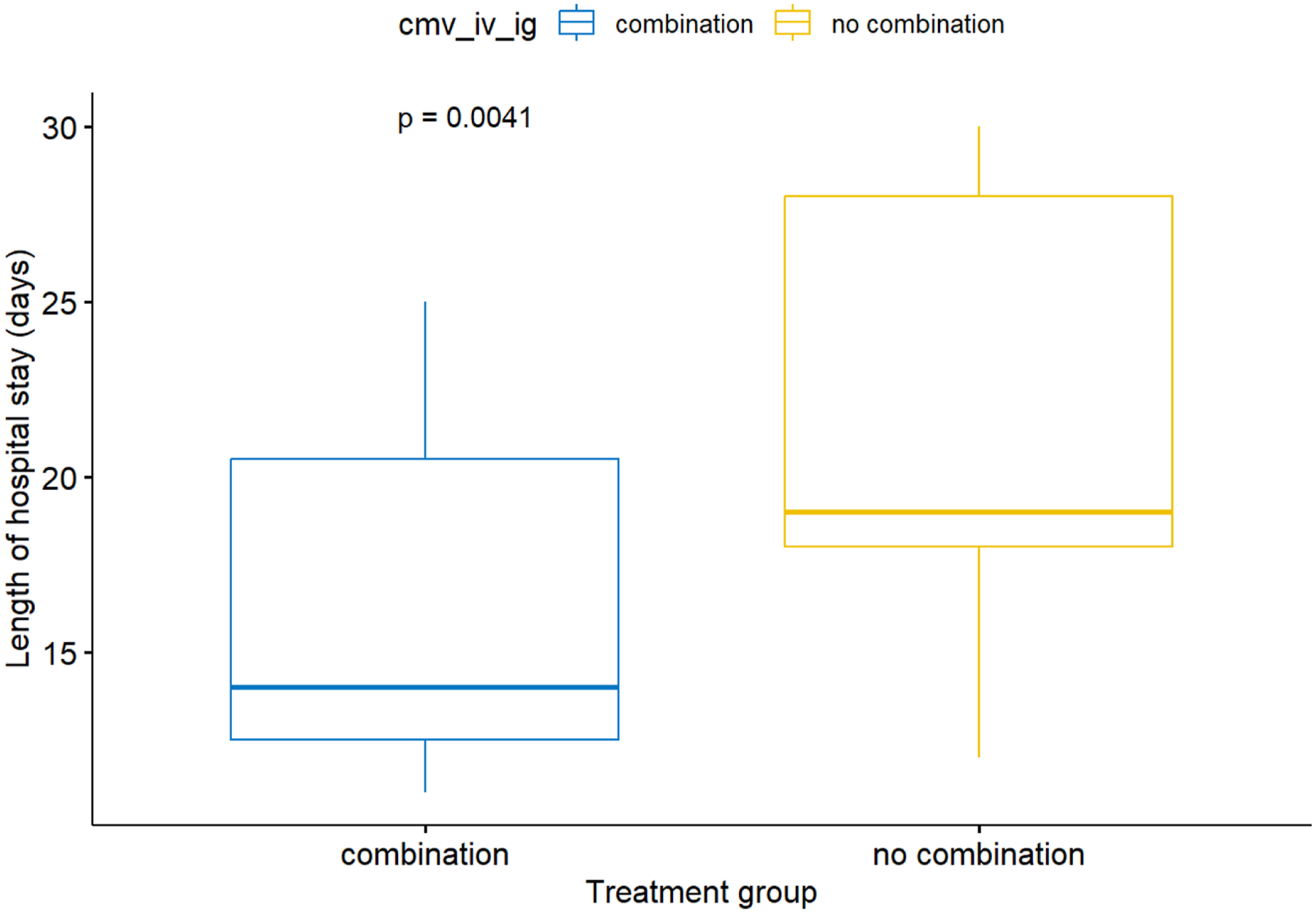
Duration of hospitalization: CMV-IV Ig combination treatment vs No CMV-IV Ig combination treatment.
Adverse-event profiles were comparable (Table 3). One case of reversible renal impairment (7 %) and one mild infusion-related rash (7 %) occurred in the CMV-IG group; two renal events (10 %) and one rash (5 %) occurred among controls. No thromboembolic events or volume-overload episodes were observed.
Discussion
In this real-world heterogeneous cohort of immunocompromised patients with CMV disease, adjunctive CMV-IG was associated with significantly faster virological clearance and shorter hospital and antiviral exposure, whereas mortality, overall clinical response, and safety outcomes did not differ from treatment with standard antivirals alone.
Our findings add new evidence to a field in which data have historically been limited —mostly restricted to CMV pneumonitis in HSCT or severe tissue-invasive disease in SOT—suggesting a potential benefit of CMV-IG when combined with standard antivirals.11–14 Whereas previous reports described survival rates of 31–78 %, we observed >90 % clinical response, possibly reflecting contemporary improved supportive care and treatment, inclusion of non-transplant populations and early CMV-IG administration.11–14 In this regard, it is important to emphasize that our study also included populations not undergoing transplantation, such as patients with hematologic malignancies, chronic inflammatory bowel diseases, autoimmune diseases, and pregnant women.
In all patient groups included in our real-world cohort, CMV-specific immunoglobulins did not appear to affect overall mortality, clinical or virological response, or safety. However, our findings suggest that they may help accelerate viral clearance, potentially reducing the duration of exposure to direct antivirals and shortening hospital stay. Therefore, despite the limitations of our study, CMV-specific immunoglobulins may represent a useful adjunct in selected clinical settings. It is noteworthy that, beyond passive neutralization, CMV-IG may enhance antibody-dependent cellular cytotoxicity, activate complement and modulate T-cell responses; these mechanisms could accelerate viral clearance and allow earlier discontinuation of antiviral therapy. Moreover, a shorter hospital stay might translate into lower nosocomial-infection risk and costs, partially offsetting the price of CMV-IG—an aspect warranting formal cost-effectiveness analysis.
In line with our findings, the most recent 2025 international guidelines on CMV management in solid organ transplantation acknowledge that CMV hyperimmune globulin (CMV-IG) may be considered as an adjunctive therapeutic option in selected clinical scenarios, including severe disease, resistant or refractory CMV infection, intolerance to antiviral agents, and/or hypogammaglobulinemia (IgG < 400 mg/dL), particularly in thoracic transplant recipients. At the same time, these guidelines emphasize that no recent randomized clinical trials have demonstrated a clear superiority of this strategy. 15
A similar position is reflected in the 2024 British guidelines, which identified only one randomized controlled trial, a small study conducted in heart transplant recipients, and considered the available evidence insufficient to support a strong recommendation.3,16,17
Notably, however, the trial by Vrtovec et al. did not assess CMV-IG in the setting of overt CMV disease or established CMV-related clinical manifestations, but rather compared intravenous ganciclovir with CMV hyperimmune globulin as a pre-emptive strategy in CMV-positive heart transplant recipients. 17 This distinction is clinically relevant, as evidence derived from pre-emptive treatment cannot be directly extrapolated to patients with active or severe CMV disease.
Additional support for a potential role of CMV-IG comes from case reports and small case series, suggesting that, in selected and difficult-to-treat cases, CMV-IG may serve as a useful component of combination therapy. In this regard, Di Cristanziano et al. reported sustained control of prolonged CMV viremia in three transplant recipients—one allogeneic hematopoietic stem cell transplant recipient and two kidney transplant recipients—treated with a combination of letermovir, CMV-specific intravenous immunoglobulins, and valganciclovir/ganciclovir. 18
In the hematopoietic stem cell transplantation (HSCT) setting, the evidence is even more limited. Recent reviews and guidance papers are largely centered on letermovir, maribavir, and adoptive cellular approaches, whereas CMV-IG is mentioned mainly in retrospective series or small observational studies. 19 Thus, the issue is not necessarily the absence of a potential clinical signal, but rather the persistently low and heterogeneous quality of the available evidence.
The evidence is even less compelling in the setting of pregnancy and the prevention of congenital CMV infection. Of the two pregnant women included in our cohort, one received combination therapy, whereas the other was treated with valaciclovir alone. No differences were observed in maternal outcomes or fetal transmission; however, these findings must be interpreted with caution given the extremely small sample size and the resulting lack of statistical power. Consistent with our observations, the randomized trial by Hughes et al. showed that administration of CMV hyperimmune globulin before 24 weeks of gestation did not reduce the composite outcome of congenital CMV infection or perinatal death compared with placebo. 5 In contrast, Nigro, in a narrative review, suggested that the available experimental and clinical evidence may support a potential role for hyperimmune globulin in pregnancy. 20
Taken together, these data suggest that CMV-IG should not be regarded as standard first-line therapy, but rather as a selective adjunctive/off-label option in carefully chosen patients, particularly those with thoracic solid organ transplants, severe invasive disease, resistant or refractory CMV infection, antiviral toxicity or intolerance, or hypogammaglobulinemia. Conversely, in pregnancy and congenital CMV infection, the current evidence does not support routine use: the available randomized trial was negative, however this therapeutic approach needs to be further investigated.5,20
Weakness of the present study include a retrospective, single-center design and a small sample size. A limitation was that the power analysis for sample size calculation was not performed. The heterogeneity of underlying conditions restricts generalizability and precludes a robust mortality analysis, as residual confounding cannot be excluded. In addition, CMV-IG dosing was empirical (100 IU kg-1 ×3 days), so the optimal schedule remains undefined. In more than 50% of subjects, there was no organ involvement requiring potentially prolonged therapy (e.g., retinitis). Finally, CMV-specific cellular immunity was not assessed.
Conclusion
Adjunctive CMV-IG, administered early and for three days, appears to be safe and may accelerate virological clearance, enabling shorter courses of antiviral therapy and reduced hospital stay in heterogeneous immunocompromised patients with CMV disease. Randomized controlled trials are warranted to confirm efficacy, define the optimal dosing regimen and assess cost-effectiveness.
Footnotes
Ethical considerations
The design of the work has been approved by the “Federico II” Ethics Committee with the code number 87/21. The study was conducted in compliance with the Declaration of Helsinki and the principles of good clinical practice.
Consent to participate
Yes.
Consent for publication
Yes.
Author contributions
Biagio Pinchera conceived idea and participated in substantial contributions to the conception and design of the work and the acquisition, analysis and interpretation data for the work. Ludovica Fusco participated in the acquisition and analysis of data for the work. and being accountable for the accuracy/integrity of the content. Vincenzo Fotticchia participated in analysis, interpretation of data for the work and being accountable for the accuracy/integrity of the content. Antonio Riccardo Buonomo participated in the acquisition of data for the work. Emanuela Zappulo participated in the acquisition of data for the work. Nicola Schiano Moriello participated in revising the initial draft of the article and approving the final version to be published. Ivan Gentile participated in substantial contributions to the conception and design of the work; the acquisition, analysis and interpretation of data for the work; approval for the final version to be published.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data are available upon request to the corresponding author via email
